Back to Journals » Drug Design, Development and Therapy » Volume 9
Selective oropharyngeal decontamination versus selective digestive decontamination in critically ill patients: a meta-analysis of randomized controlled trials
Authors Zhao D, Song J, Gao X, Gao F, Wu Y, Lu Y, Hou K
Received 14 March 2015
Accepted for publication 21 May 2015
Published 14 July 2015 Volume 2015:9 Pages 3617—3624
DOI https://doi.org/10.2147/DDDT.S84587
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Wei Duan
Di Zhao,1,* Jian Song,2,* Xuan Gao,3 Fei Gao,4 Yupeng Wu,2 Yingying Lu,5 Kai Hou1
1Department of Neurosurgery, The First Hospital of Hebei Medical University, 2Department of Neurosurgery, 3Department of Neurology, The Second Hospital of Hebei Medical University, 4Hebei Provincial Procurement Centers for Medical Drugs and Devices, 5Department of Neurosurgery, The Second Hospital of Hebei Medical University, Shijiazhuang People’s Republic of China
*These authors contributed equally to this work
Background: Selective digestive decontamination (SDD) and selective oropharyngeal decontamination (SOD) are associated with reduced mortality and infection rates among patients in intensive care units (ICUs); however, whether SOD has a superior effect than SDD remains uncertain. Hence, we conducted a meta-analysis of randomized controlled trials (RCTs) to compare SOD with SDD in terms of clinical outcomes and antimicrobial resistance rates in patients who were critically ill.
Methods: RCTs published in PubMed, Embase, and Web of Science were systematically reviewed to compare the effects of SOD and SDD in patients who were critically ill. Outcomes included day-28 mortality, length of ICU stay, length of hospital stay, duration of mechanical ventilation, ICU-acquired bacteremia, and prevalence of antibiotic-resistant Gram-negative bacteria. Results were expressed as risk ratio (RR) with 95% confidence intervals (CIs), and weighted mean differences (WMDs) with 95% CIs. Pooled estimates were performed using a fixed-effects model or random-effects model, depending on the heterogeneity among studies.
Results: A total of four RCTs involving 23,822 patients met the inclusion criteria and were included in this meta-analysis. Among patients whose admitting specialty was surgery, cardiothoracic surgery (57.3%) and neurosurgery (29.7%) were the two main types of surgery being performed. Pooled results showed that SOD had similar effects as SDD in day-28 mortality (RR =1.03; 95% CI: 0.98, 1.08; P=0.253), length of ICU stay (WMD =0.00 days; 95% CI: -0.2, 0.2; P=1.00), length of hospital stay (WMD =0.00 days; 95% CI: -0.65, 0.65; P=1.00), and duration of mechanical ventilation (WMD =1.01 days; 95% CI: -0.01, 2.02; P=0.053). On the other hand, compared with SOD, SDD had a lower day-28 mortality in surgical patients (RR =1.11; 95% CI: 1.00, 1.22; P=0.050), lower incidence of ICU-acquired bacteremia (RR =1.38; 95% CI: 1.24, 1.54; P=0.000), and lower rectal carriage of aminoglycosides (RR =2.08; 95% CI: 1.68, 2.58; P=0.000), ciprofloxacin-resistant Gram-negative bacteria (RR =1.84; 95% CI: 1.48, 2.29; P=0.000), and respiratory carriage of third-generation cephalosporin-resistant Gram-negative bacteria (RR =2.50; 95% CI: 1.78, 3.5; P=0.000).
Conclusion: SOD has similar effects as SDD in clinical outcomes, but has higher incidence of ICU-acquired bacteremia, and higher carriage of antibiotic-resistant Gram-negative bacteria. However, due to the high cost of SDD and the increased risk of development of antibiotic resistance with the widespread use of cephalosporins in SDD, we would recommend SOD as prophylactic antibiotic regimens in patients in the ICU. More well-designed, large-scale RCTs are needed to confirm our findings.
Keywords: selective digestive decontamination, selective oropharyngeal decontamination, intensive care units, meta-analysis
Introduction
Respiratory tract infections are the major complications of the treatment of critically ill patients in intensive care units (ICUs), which have been associated with increased morbidity, mortality, and health care costs.1–3 For patients on ICU admission or during the ICU stay, the infections are mainly caused by potentially pathogenic microorganisms carried in the throat and gut. Prophylactic antibiotic regimens, including selective decontamination of the digestive tract, selective digestive decontamination (SDD), and selective oropharyngeal decontamination (SOD),4,5 have been proved to reduce the prevalence of respiratory tract infections. However, one disadvantage, which is brought about from these interventions, is the increased risk of selection for antibiotic-resistant pathogens.
The aim of the SDD approach is to eradicate potentially pathogenic microorganisms by means of prophylactic application of nonabsorbable antibiotics in the oropharynx and gastrointestinal tract. It also preemptively treats possible commensal infections in the respiratory tract through systemic administration of cephalosporins during the patient’s first 4 days in the ICU, and preserves anaerobic intestinal flora through selective use of antibiotics.6,7 SOD, in which the same topical antibiotics are used in the oropharynx only, is considered as an effective measure in the prevention of ventilator-associated pneumonia.7
Multiple clinical trials and meta-analysis have assessed the effects of SDD or SOD on mortality and antimicrobial resistance in critically ill patients.8,9 In one meta-analysis from 29 randomized controlled trials (RCTs),8 Price et al detected that SDD significantly reduced the mortality of patients in general ICUs, whereas the effect of SOD remained less certain. In another meta-analysis from 64 studies,9 Daneman et al assessed the effect of SDD or SOD on antimicrobial-resistant pathogens. They found that there was no relation between the application of SDD or SOD and the development of antimicrobial-resistant pathogens in ICU patients.9
While the above-mentioned meta-analyses evaluate the effect of SDD or SOD on mortality, infection, and antimicrobial resistance, no meta-analysis has been conducted to compare the effects of SOD and SDD on clinical outcomes. Therefore, we performed this meta-analysis of related studies to compare the effects of SDD versus SOD on clinical outcomes and antibiotic resistance.
Methods and materials
Literature search strategy
We conducted this meta-analysis of RCTs in accordance with the preferred reported items for systematic reviews and meta-analyses guidelines.10 PubMed, Embase, and Web of Science were searched using the following search items: Selective [All Fields] AND (“decontamination” [MeSH Terms] OR “decontamination” [All Fields]) AND (“gastrointestinal tract” [MeSH Terms] OR (“gastrointestinal” [All Fields] AND “tract” [All Fields]) OR “gastrointestinal tract” [All Fields] OR (“digestive” [All Fields] AND “tract” [All Fields]) OR “digestive tract” [All Fields]) AND selective [All Fields] AND (“oropharynx” [MeSH Terms] OR “oropharynx” [All Fields] OR “oropharyngeal” [All Fields]) AND (“decontamination” [MeSH Terms] OR “decontamination” [All Fields]) AND (“intensive care” [MeSH Terms] OR (“intensive” [All Fields] AND “care” [All Fields]) OR “intensive care” [All Fields]). This search had no language limitation, but was restricted to human subjects. Additionally, we also searched the reference lists of the included studies and related publications until no potential citations could be found. This search was conducted on November 10, 2014. In cases where the same clinical trial appeared in diverse publications, we only included the most recent or complete-information study.
Inclusion and exclusion criteria
Studies were included in this meta-analysis if they met the following inclusion criteria: 1) study design: RCT; 2) study population: critically ill patients; 3) interventions: SOD, and SDD; 4) outcomes: day-28 mortality, length of ICU stay, length of hospital stay, incidence of ICU-acquired bacteremia, prevalence of antibiotic-resistant Gram-negative bacteria. We excluded studies in which the trial was not randomized controlled design, or in which the information was not usable. Additionally, articles were also excluded from the final analysis if they were comments, reviews, abstracts, letters, editorials, and systematic reviews.
Data extraction
Two researchers (Di Zhao and Jian Song) independently performed the data extraction from the eligible studies. The following information was extracted: first author, year of publication, patients’ age and sex, total number of patients in the SOD and SDD groups, and number of interest events in the SOD and SDD groups. Disagreements between the two reviewers were resolved by discussion and consensus.
Quality assessment
We used the Jadad scale11 to assess the methodological quality of each trial. The Jadad scale consists of three items, including randomization (0–2 points), blinding (0–2 points), and dropouts and withdrawals (0–1 point), to report a RCT. A score of 1 is obtained when the above description is met. A further point is given when the method of randomization and/or blinding is given and is appropriate. The highest score in the scale is 5 points, and a higher score indicates better reporting. The study is considered to be of high quality if the Jadad scale score is ≥3 points.12
Statistical analysis
We compared the effects of SOD and SDD in critically ill patients by analyzing the data from the included RCTs. Day-28 mortality, incidence of ICU-acquired bacteremia, and prevalence of antibiotic-resistant Gram-negative bacteria were treated as dichotomous variables and were expressed as risk ratio (RR) with 95% confidence intervals (CIs). Length of ICU stay, length of hospital stay, and duration of mechanical ventilation were treated as continuous variables; thus, they were expressed as weighted mean difference (WMD) with 95% CIs.
Pooled estimates were generated by using a fixed-effects model (Mantel–Haenszel method)13 or random-effect model (DerSimonian–Laird method),14 depending on the heterogeneity among the included studies. Before the data syntheses, Q statistic was used to detect the degree of heterogeneity among the studies, in which a P-value <0.10 was defined as significant heterogeneity. I2 statistic was estimated to describe the percentage of the variability that attributed to the heterogeneity across the studies rather than the chance. Studies with I2<25%, ~50%, ~75%, and ~100% were considered to have no, low, moderate, and high heterogeneity, respectively.15 Since the number of included studies was less than ten, the assessment of publication bias was not performed. A P-value <0.05 was judged as statistically significant, except where otherwise specified. All statistical analyses were conducted by using STATA software version 12.0 (Stata Corp LP, College Station, TX, USA).
Results
Literature research and selection of studies
Initially, a total of 374 potentially relevant studies were identified from PubMed, Embase, and Web of Science databases, of which 83 were excluded because of duplication. In the process of titles/abstracts screening, 238 studies were excluded because they were reviews, comments, letters, non-RCTs, or single-arm studies, leaving 53 studies for the full-text review. Among the 53 potentially relevant studies, 49 were excluded because they were unrelated to our topics, or had no outcomes of interest,16–19 or contained overlapping data.20 Finally, four studies, with a total number of 23,822 patients, were included in this meta-analysis21–24 (Figure 1).
  | Figure 1 Search strategy and flow chart for this meta-analysis. |
Study characteristics and quality assessment
The main characteristics of the included studies are summarized in Table 1. These studies were published between 2009 and 2014. All of these patients received mechanical ventilation for a duration of >48 hours or an ICU stay of >72 hours. The baseline characteristics of patients, including age, sex, and need for mechanical ventilation, were comparable between the two groups, except in one trial,21 in which patients in the SOD group had higher acute physiology and chronic health evaluation IV scores than those in the SDD group 75 (interquartile range [IQR], 55–99) vs 73 (IQR, 54–96).21 The median Jadad score of the studies included was 3.5 (range of 3–4).
Day-28 mortality
Day-28 mortality data were reported in three trials,21,22,24 and two of them presented the data in surgical and nonsurgical patients.21,22 Pooling these data using a fixed-effects model showed that SOD had a similar effect as SDD on day-28 mortality (RR =1.03; 95% CI: 0.98, 1.08; P=0.253) (Figure 2). The test for heterogeneity was not significant (heterogeneity P=0.351, I2=8.4%).
We also performed a subgroup analysis based on surgical status, namely, surgical and nonsurgical patients. The pooled results revealed that compared with SDD, SOD had a higher day-28 mortality in surgical patients (RR =1.11; 95% CI: 1.00, 1.22; P=0.050) (Figure 3), but a similar effect in nonsurgical patients (RR =1.01; 95% CI: 0.95, 1.07; P=0.790) (Figure 3).
Length of ICU stay, length of hospital stay, and duration of mechanical ventilation
The pooled analysis showed that compared with SDD, SOD had comparable effects on the length of ICU stay (WMD =0.00 days; 95% CI: −0.2, 0.2; P=1.00), length of hospital stay (WMD =0.00 days; 95% CI: −0.65, 0.65; P=1.00), and duration of mechanical ventilation (WMD =1.01 days; 95% CI: −0.01, 2.02; P=0.053) (Figure 4). There were no significant heterogeneity for the length of ICU stay (heterogeneity P=1.000, I2=0.0%) and length of hospital stay (heterogeneity P=1.000, I2=0.0%), but significant heterogeneity existed for the duration of mechanical ventilation (heterogeneity P=0.001, I2=86.8%).
ICU-acquired bacteremia
Three studies presented the data on ICU-acquired bacteremia.21,23,24 All these studies showed that SOD was associated with a higher incidence of ICU-acquired bacteremia when compared with SDD. Pooled estimates using a fixed-effects model showed that SDD significantly reduced the incidence of ICU-acquired bacteremia, as compared with SOD (RR =1.38; 95% CI: 1.24, 1.54; P=0.000) (Figure 5). There was no evidence of heterogeneity among the included studies (heterogeneity P=0.755, I2=0.0%).
Prevalence of antibiotic-resistant Gram-negative bacteria
Three studies reported the data on prevalence of antibiotic-resistant Gram-negative bacteria.21,23,24 Pooled data suggested that the prevalence of Gram-negative bacteria resistant to aminoglycosides (RR =2.08; 95% CI: 1.68, 2.58; P=0.000) and ciprofloxacin (RR =1.84; 95% CI: 1.48, 2.29; P=0.000) in rectal sample were lower in the SDD group than that in the SOD group (Table 2). In addition, the prevalence of Gram-negative bacteria resistant to third-generation cephalosporin (RR =2.50; 95% CI: 1.78, 3.5; P=0.000) in respiratory sample was lower in the SDD group than that in the SOD group (Table 2).
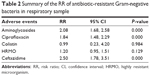  | Table 2 Summary of the RR of antibiotic-resistant Gram-negative bacteria in respiratory sample |
Discussion
Our meta-analysis suggests that the application of SOD or SDD in ICU patients caused no significant differences in day-28 mortality, length of ICU stay, length of hospital stay, and duration of mechanical ventilation, but SDD seemed to have lower day-28 mortality in surgical patients than SOD. Moreover, SDD was associated with a lower incidence of ICU-acquired bacteremia, and a lower rectal carriage of aminoglycosides, ciprofloxacin-resistant Gram-negative bacteria, and respiratory carriage of third-generation cephalosporin-resistant Gram-negative bacteria.
To the best of our knowledge, this is the first comprehensive meta-analysis to compare the effects of SOD and SDD in the patients who were critically ill. Our results suggest that, compared with SOD, SDD significantly decreased day-28 mortality in surgical patients. Our result was consistent with a recently published meta-analysis, in which SDD was compared with standard care in surgical patients.25 In that study, Nathens and Marshall25 found that mortality was significantly reduced with the application of SDD in critically ill surgical patients (odds ratio [OR] =0.7; 95% CI: 0.52, 0.93), but not in critically ill medical patients (OR =0.91; 95% CI: 0.71, 1.18).25
Numerous studies have assessed the effects of SDD or SOD on day-28 mortality; however, their results remained controversial. In one trial conducted by de Smet et al24 patients were randomly assigned to standard care, SOD, and SDD. The crude day-28 mortality for those people were 27.5%, 26.6%, and 26.9%, respectively,24 and the difference between them was not statistically significant. However, when the authors used a random-effects logistic regression model to calculate the ORs for death at day 28, the OR values became 0.86 (95% CI: 0.74, 0.99) in the comparison of SOD with standard care, and 0.83 (95% CI: 0.72, 0.97) in the comparison of SDD with standard care.24 The day-28 mortality rate was reduced by 3.5% with SDD, and by 2.9% with SOD, as compared with standard care.24 In another meta-analysis,8 Price et al reported that, compared with control, SDD significantly reduced the risk of mortality (OR =0.74; 95% CI: 0.63, 0.86), but SOD did not (OR =0.82; 95% CI: 0.62, 1.02). Therefore, the authors raised the possibility that the application of SOD might increase the risk of mortality in critically ill patients.
With regards to other important clinical outcomes, including length of ICU stay, length of hospital stay, and duration of mechanical ventilation, the two procedures seemed to have similar effects. In the NTR1780 trial,21 the lengths of ICU stay for patients in the SOD group and SDD group were both 6 days (IQR: 4–11), and lengths of hospital stay were both 19 days (IQR: 11–35). This indicated that SOD and SDD had no statistically significant differences in the lengths of ICU and hospital stays.
Although the application of SOD and SDD significantly reduced the mortality and infection rates, these interventions have not been widely used because their use may lead to the development of antimicrobial-resistant pathogens. However, a recently published meta-analysis considers this problem.9 In that study, Daneman et al9 pooled 47 RCTs to assess the effect of SOD or SDD on antimicrobial resistance rates in patients in ICUs. They detected that there was no significant difference in the incidence of colonization with antimicrobial-resistant pathogens between patients who received selection decontamination and those who did not.9 Moreover, patients treated with selection decontamination had a lower incidence in polymyxin-resistant Gram-negative bacteria (OR =0.58; 95% CI: 0.46, 0.72), and third-generation cephalosporin-resistant Gram-negative bacteria (OR =0.33; 95% CI: 0.20, 0.52), as compared with those who received no intervention.9
There were several limitations in this meta-analysis that should be considered when interpreting our results. First, our data were abstracted from publications rather than collected from individual patients; thus the effect of SOD and SDD may not been defined clearly.26 Second, our meta-analysis was performed based on only four RCTs, and some of the analyses were performed only on two or three RCTs. Although all the studies included were prospective, large-scale, well-designed RCTs, caution should be taken when interpreting our results. Third, because the studies included were less than ten, the assessment of publication bias was not performed. Thus, we could not exclude the possibility that our final estimates may be influenced by missing and unpublished data. Fourth, we know that, at the time of our writing, a cluster-randomized study, which assess the effects of SDD, SOD, and other decontamination strategies, is in progression.27 This ongoing trial might have a potential impact on our final results.
In summary, our study suggests that SDD has similar effects as SOD in terms of patient outcomes, including day-28 mortality, length of ICU stay, length of hospital stay, and duration of mechanical ventilation. However, SDD has a better effect on day-28 mortality of surgical patients, and has lower prevalence of ICU-acquired bacteremia, rectal carriage of aminoglycosides, ciprofloxacin-resistant Gram-negative bacteria, and respiratory carriage of third-generation cephalosporin-resistant Gram-negative bacteria. Overall, we consider that SDD has comparable effects to that of SOD in clinical outcomes, and is better, in some indices such as ICU-acquired bacteremia, and the carriage of antibiotic-resistant Gram-negative bacteria, than SOD. However, due to the high cost of SDD and the increased risk of development of antibiotic with the widespread use of cephalosporins in SDD, we would recommend SOD as prophylactic antibiotic regimens in patients in the ICU. Further well-designed, large-scale RCTs are needed to confirm our findings.
Disclosure
The authors report no conflicts of interest in this work.
References
Roberts RR, Hota B, Ahmad I, et al. Hospital and societal costs of antimicrobial-resistant infections in a Chicago teaching hospital: implications for antibiotic stewardship. Clin Infect Dis. 2009;49:1175–1184. | ||
Vincent JL. Nosocomial infections in adult intensive-care units. Lancet. 2003;361:2068–2077. | ||
Vincent JL, Rello J, Marshall J, et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA. 2009;302:2323–2329. | ||
Bergmans DC, Bonten MJ, Gaillard CA, et al. Prevention of ventilator-associated pneumonia by oral decontamination: a prospective, randomized, double-blind, placebo-controlled study. Am J Respir Crit Care Med. 2001;164:382–388. | ||
de Jonge E, Schultz MJ, Spanjaard L, et al. Effects of selective decontamination of digestive tract on mortality and acquisition of resistant bacteria in intensive care: a randomised controlled trial. Lancet. 2003;362:1011–1016. | ||
van der Waaij D, Berghuis-de Vries JM, Lekkerkerk L-V. Colonization resistance of the digestive tract in conventional and antibiotic-treated mice. J Hyg (Lond). 1971;69:405–411. | ||
Stoutenbeek CP, van Saene HK, Miranda DR, Zandstra DF. The effect of selective decontamination of the digestive tract on colonisation and infection rate in multiple trauma patients. Intensive Care Med. 1984;10:185–192. | ||
Price R, MacLennan G, Glen J; SuDDICU Collaboration. Selective digestive or oropharyngeal decontamination and topical oropharyngeal chlorhexidine for prevention of death in general intensive care: systematic review and network meta-analysis. BMJ. 2014;348:g2197. | ||
Daneman N, Sarwar S, Fowler RA, Cuthbertson BH. Effect of selective decontamination on antimicrobial resistance in intensive care units: a systematic review and meta-analysis. Lancet Infect Dis. 2013;13:328–341. | ||
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097. | ||
Jadad AR, Moore RA, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17:1–12. | ||
Kjaergard LL, Villumsen J, Gluud C. Reported methodologic quality and discrepancies between large and small randomized trials in meta-analyses. Ann Intern Med. 2001;135:982–989. | ||
WG C. The combination of estimates from different experiments. Biometrics. 1954;10:101–129. | ||
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–188. | ||
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560. | ||
Houben AJ, Oostdijk EA, van der Voort PH, Monen JC, Bonten MJ, van der Bij AK. Selective decontamination of the oropharynx and the digestive tract, and antimicrobial resistance: a 4 year ecological study in 38 intensive care units in the Netherlands. J Antimicrob Chemother. 2014;69:797–804. | ||
Benus RF, Harmsen HJ, Welling GW, et al. Impact of digestive and oropharyngeal decontamination on the intestinal microbiota in ICU patients. Intensive Care Med. 2010;36:1394–1402. | ||
Oostdijk EA, de Wit GA, Bakker M, de Smet AM, Bonten MJ; Dutch SOD-SDD Trialists Group. Selective decontamination of the digestive tract and selective oropharyngeal decontamination in intensive care unit patients: a cost-effectiveness analysis. BMJ Open. 2013;3(3):e002529. | ||
Oostdijk EA, Smits L, de Smet AM, Leverstein-van Hall MA, Kesecioglu J, Bonten MJ. Colistin resistance in gram-negative bacteria during prophylactic topical colistin use in intensive care units. Intensive Care Med. 2013;39:653–660. | ||
Oostdijk EA, de Smet AM, Blok HE, et al. Ecological effects of selective decontamination on resistant gram-negative bacterial colonization. Am J Respir Crit Care Med. 2010;181:452–457. | ||
Oostdijk EA, Kesecioglu J, Schultz MJ, et al. Effects of decontamination of the oropharynx and intestinal tract on antibiotic resistance in ICUs: a randomized clinical trial. JAMA. 2014;312:1429–1437. | ||
Melsen WG, de Smet AM, Kluytmans JA, Bonten MJ. Selective decontamination of the oral and digestive tract in surgical versus non-surgical patients in intensive care in a cluster-randomized trial. Br J Surg. 2012;99:232–237. | ||
de Smet AM, Kluytmans JA, Blok HE, et al. Selective digestive tract decontamination and selective oropharyngeal decontamination and antibiotic resistance in patients in intensive-care units: an open-label, clustered group-randomised, crossover study. Lancet Infect Dis. 2011;11:372–380. | ||
de Smet AM, Kluytmans JA, Cooper BS, et al. Decontamination of the digestive tract and oropharynx in ICU patients. N Engl J Med. 2009;360:20–31. | ||
Nathens AB, Marshall JC. Selective decontamination of the digestive tract in surgical patients: a systematic review of the evidence. Arch Surg. 1999;134:170–176. | ||
Stewart LA, Parmar MK. Meta-analysis of the literature or of individual patient data: is there a difference? Lancet. 1993;341:418–422. | ||
Resistance in Gram-Negative Organisms: Studying Intervention Strategies [webpage on the Internet]. R-GNOSIS. Available from: http://www.r-gnosis.eu/. Accessed July 29, 2014. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.





