Back to Journals » Drug Design, Development and Therapy » Volume 9
Plumbagin induces cell cycle arrest and autophagy and suppresses epithelial to mesenchymal transition involving PI3K/Akt/mTOR-mediated pathway in human pancreatic cancer cells
Authors Wang F, Wang Q, Zhou Z, Yu S, Pan S, He Z, Zhang X, Wang D, Yang Y, Yang T, Sun T, Li M, Qiu J, Zhou S
Received 3 September 2014
Accepted for publication 24 September 2014
Published 17 January 2015 Volume 2015:9 Pages 537—560
DOI https://doi.org/10.2147/DDDT.S73689
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Prof. Dr. Wei Duan
Feng Wang,1,2,* Qi Wang,1,* Zhi-Wei Zhou,2,3 Song-Ning Yu,1 Shu-Ting Pan,4 Zhi-Xu He,3 Xueji Zhang,5 Dong Wang,6 Yin-Xue Yang,7 Tianxing Yang,8 Tao Sun,9 Min Li,10 Jia-Xuan Qiu,4 Shu-Feng Zhou2
1Department of Hepatobiliary Surgery, General Hospital of Ningxia Medical University, Yinchuan, Ningxia, People’s Republic of China; 2Department of Pharmaceutical Science, College of Pharmacy, University of South Florida, Tampa, FL, USA; 3Guizhou Provincial Key Laboratory for Regenerative Medicine, Stem Cell and Tissue Engineering Research Center and Sino-US Joint Laboratory for Medical Sciences, Guiyang Medical University, Guiyang, People’s Republic of China; 4Department of Oral and Maxillofacial Surgery, The First Affiliated Hospital of Nanchang University, Nanchang, People’s Republic of China; 5Research Center for Bioengineering and Sensing Technology, University of Science and Technology Beijing, Beijing, People’s Republic of China; 6Cancer Center, Daping Hospital and Research Institute of Surgery, Third Military Medical University, Chongqing, People’s Republic of China; 7Department of Colorectal Surgery, General Hospital, Ningxia Medical University, Yinchuan, Ningxia, People’s Republic of China; 8Department of Internal Medicine, University of Utah and Salt Lake Veterans Affairs Medical Center, Salt Lake City, UT, USA; 9Key Lab of Craniocerebral Diseases of Ningxia, Ningxia Medical University, Yinchuan, People’s Republic of China; 10Department of Medicine and Department of Surgery, The University of Oklahoma Health Sciences Center, Stanton L Young Biomedical Research Center, Oklahoma City, OK, USA
*These authors contributed equally to this work
Abstract: Plumbagin (PLB), an active naphthoquinone compound, has shown potent anticancer effects in preclinical studies; however, the effect and underlying mechanism of PLB for the treatment of pancreatic cancer is unclear. This study aimed to examine the pancreatic cancer cell killing effect of PLB and investigate the underlying mechanism in human pancreatic cancer PANC-1 and BxPC-3 cells. The results showed that PLB exhibited potent inducing effects on cell cycle arrest in PANC-1 and BxPC-3 cells via the modulation of cell cycle regulators including CDK1/CDC2, cyclin B1, cyclin D1, p21 Waf1/Cip1, p27 Kip1, and p53. PLB treatment concentration- and time-dependently increased the percentage of autophagic cells and significantly increased the expression level of phosphatase and tensin homolog, beclin 1, and the ratio of LC3-II over LC3-I in both PANC-1 and BxPC-3 cells. PLB induced inhibition of phosphatidylinositol 3-kinase (PI3K)/protein kinase B/mammalian target of rapamycin and p38 mitogen-activated protein kinase (p38 MAPK) pathways and activation of 5'-AMP-dependent kinase as indicated by their altered phosphorylation, contributing to the proautophagic activities of PLB in both cell lines. Furthermore, SB202190, a selective inhibitor of p38 MAPK, and wortmannin, a potent, irreversible, and selective PI3K inhibitor, remarkably enhanced PLB-induced autophagy in PANC-1 and BxPC-3 cells, indicating the roles of PI3K and p38 MAPK mediated signaling pathways in PLB-induced autophagic cell death in both cell lines. In addition, PLB significantly inhibited epithelial to mesenchymal transition phenotype in both cell lines with an increase in the expression level of E-cadherin and a decrease in N-cadherin. Moreover, PLB treatment significantly suppressed the expression of Sirt1 in both cell lines. These findings show that PLB promotes cell cycle arrest and autophagy but inhibits epithelial to mesenchymal transition phenotype in pancreatic cancer cells with the involvement of PI3K/protein kinase B/ mammalian target of rapamycin and p38 MAPK mediated pathways.
Keywords: Plumbagin, pancreatic cancer, cell cycle, autophagy, EMT, Sirt1
Introduction
Pancreatic cancer is the 12th most common and the most aggressive cancer in the world, and the incidence and death rates rise with advancing age.1 There were 338,000 cases diagnosed with pancreatic cancer in 2012, and the 5-year survival rate is 6.7% worldwide.1,2 In the United States, there were 45,220 cases of pancreatic cancer diagnosed in 2013, and it has been estimated that there will be 46,420 new cases (23,530 men and 22,890 women) diagnosed and 39,590 patients (20,170 men and 19,420 women) will die from pancreatic cancer in 2014.2 In China, pancreatic cancer is the sixth leading cause of cancer death, and the overall 5-year survival rate is 1%–3%.1,3,4 The incidence of pancreatic cancer is 2.70% and 2.55% in urban and rural areas, respectively, and the mortality due to pancreatic cancer is 4.08% and 3.66% in urban and rural areas, respectively, in 2009.5 The current therapies for the treatment of pancreatic cancer include surgery, chemotherapy, radiation therapy, immunotherapy, and vaccine therapy.1 However, due to the de novo chemoresistant behavior of pancreatic cancer cells to cytotoxic chemotherapeutic agents and/or radiotherapy, the therapeutic outcome of pancreatic cancer is disappointing in the clinic. Therefore, new anticancer drugs that can kill pancreatic cancer cells with improved efficacy and reduced side effects are certainly needed.
Targeting programmed cell death has become a promising strategy to fighting against pancreatic cancer, which mainly includes manipulation of apoptotic and autophagic cell death.6 It is well known that apoptosis and autophagy are often observed in pancreatic cancer,7 but the therapeutic and clinical implications are unclear. In particular, autophagy, the mammalian target of rapamycin (mTOR)-mediated cell death, involves a number of Atg proteins and other regulating molecules with an inconclusive role in the regulation of cancer cell survival and cell death.6,8 Recently, it has been reported that autophagy has emerged as a promising target for the treatment of pancreatic cancer, although the underlying mechanism has not been fully elucidated.9,10 On the other hand, emerging evidence suggests that the epithelial to mesenchymal transition (EMT) is activated during pancreatic cancer development, growth, progression, and metastasis.11,12 It has been proposed that EMT facilitates metastatic dissemination of pancreatic cancer cells from a primary organ to secondary sites,13 and thus intervention of this process may represent a novel strategy to prevent pancreatic cancer metastasis.
Plumbagin (PLB) (5-hydroxy-2-methyl-1,4-naphthoquinone) (Figure 1A) is a natural naphthoquinone constituent occurring in Plumbago zeylanica L., Juglans regia, Juglans cinerea, and Juglans nigra, with a wide spectrum of pharmacological effects including anti-inflammatory, neuroprotective, anticancer, hypolipidemic, antiatherosclerotic, antibacterial, and antifungal in preclinical models.14 Recently, the anticancer effect of PLB has drawn a great deal of interest, and accumulating evidence shows that the anticancer effects of PLB are mainly attributed to induction of intracellular reactive oxygen species generation, apoptosis and autophagy, and cell cycle arrest;14,15 although, the underlying mechanism is not fully understood. In vitro and in vivo studies by our laboratory and other groups have showed that PLB induced cancer cell apoptosis and autophagy via modulation of cellular redox status, inhibition of nuclear factor kappa B (NF-κB) activation, upregulation of p53 via c-Jun N-terminal kinase phosphorylation, inhibition of phosphatidylinositol 3-kinase (PI3K)/protein kinase B (Akt)/mTOR pathway, and activation of 5′-AMP-dependent kinase (AMPK) pathway.16–22 However, the cancer cell killing effect and underlying mechanism of PLB in pancreatic cancer PANC-1 and BxPC-3 cells are unclear. In this regard, we aimed to investigate the effects of PLB on the cell cycle, autophagy, and EMT in human pancreatic cancer PANC-1 and BxPC-3 cells and underlying mechanism.
Materials and methods
Chemicals and reagents
Dulbecco’s Modified Eagle’s Medium (DMEM) and RPMI-1640 medium were obtained from Corning Cellgro Inc. (Herndon, VA, USA). Fetal bovine serum (FBS), Dulbecco’s phosphate buffered saline (PBS), PLB, thiazolyl blue tetrazolium bromide (MTT), RNase A, and propidium iodide (PI) were purchased from Sigma-Aldrich Inc. (St Louis, MO, USA). Phenol red-free culture medium was obtained from Invitrogen Inc. (Carlsbad, CA, USA). SB202190 (4-[4-fluorophenyl]-2-[4-hydroxyphenyl]-5-[4-pyridyl]1H-imidazole), a selective inhibitor of p38 mitogen-activated protein kinase (p38 MAPK), used as an autophagy inducer, and wortmannin (WM), a potent, irreversible, and selective PI3K inhibitor and a blocker of autophagosome formation were bought from InvivoGen Inc. (San Diego, CA, USA). The Cyto-ID® autophagy detection kit was obtained from Enzo Life Sciences Inc. (Farmingdale, NY, USA). The Pierce BCA protein assay kit and skim milk were bought from Thermo Scientific (Waltham, MA, USA), and polyvinylidene difluoride (PVDF) membrane was purchased from Bio-Rad (Hercules, CA, USA). Western blotting substrate was obtained from Thermo Scientific Inc. Primary antibodies against human p38 MAPK, phosphorylated (p-) p38 MAPK at Thr180/Tyr182, AMPK, p-AMPK at Thr172, Akt, p-Akt at Ser473, mTOR, p-mTOR at Ser2448, PI3K, p-PI3K/p85 at Tyr458, phosphatase and tensin homolog (PTEN), beclin 1, microtubule-associated protein 1A/1B-light chain 3 (LC3-I), LC3-II, and the EMT antibody sampler kit were all purchased from Cell Signaling Technology Inc. (Beverly, MA, USA). The EMT antibody sampler kit contains primary antibodies to N-cadherin, E-cadherin, zona occludens protein 1 (ZO-1), vimentin, slug, snail, zinc finger E-box-binding homeobox 1 (TCF-8/ZEB1), and β-catenin. The antibody against human β-actin was obtained from Santa Cruz Biotechnology Inc. (Santa Cruz, CA, USA).
Cell lines and cell culture
Two pancreatic cancer cell lines PANC-1 and BxPC-3 cells were obtained from the American Type Culture Collection (Manassas, VA, USA) and cultured in DMEM (PANC-1 cells) and RPMI-1640 (BxPC-3 cells) media containing L-glutamine, phenol red, L-cysteine, L-methionine, sodium bicarbonate, and sodium pyruvate supplemented with 10% heat-inactivated FBS, and 1% antibiotics (penicillin and streptomycin). The cells were maintained in a 5% CO2/95% air humidified incubator at 37°C. PLB was dissolved in dimethyl sulfoxide (DMSO) with a stock concentration of 100 mM and was freshly diluted to predetermined concentrations with culture medium. The final concentration of DMSO was at 0.05% (v/v). The control cells received the vehicle only.
Cell viability assay
The MTT assay was performed to examine the effect of PLB on cell viability of PANC-1 and BxPC-3 cells. Briefly, PANC-1 and BxPC-3 cells were seeded into a 96-well culture plate at a density of 8,000 cells/well. After cells were seeded for 24 hours at a volume of 100 μL, the PANC-1 and BxPC-3 cells were treated with PLB at 0.1–20 μM for 24 and 48 hours. Following the PLB treatment, 10 μL of MTT stock solution (5 mg/mL) was added to each well and incubated for 4 hours. Following that, the solution was carefully aspirated, and 150 μL DMSO was added into each well to disolve the crystal. The absorbance was measured after 10-minute incubation at 37°C using a Synergy H4 Hybrid microplate reader (BioTek Inc., Winooski, VT, USA) at a wavelength of 450 nm. The half maximal inhibitory concentration (IC50) values were determined using the relative viability over PLB concentration curve.
Cell cycle distribution analysis
The effect of PLB on cell cycle of PANC-1 and BxPC-3 cells was determined using PI as the DNA stain by flow cytometry as described previously.23 Briefly, PANC-1 and BxPC-3 cells were seeded into 60 mm Petri dishes. After cells were seeded for 24 hours, the cells reached ~75% confluence and were then treated with PLB at concentrations of 0.1, 1, 5, and 10 μM for 24 hours. In separate experiments, PANC-1 and BxPC-3 cells were treated with 5 μM PLB for 4, 8, 12, 24, 48, and 72 hours. Following the treatment, cells were detached and fixed with 70% ethanol at -20°C overnight. Subsequently, the cells were incubated with 25 μg/mL RNase A and 50 μg/mL PI for 30 minutes in the dark. A total number of 1×10 4 cells was subject to cell cycle analysis using a flow cytometer (Becton Dickinson Immunocytometry Systems, San Jose, CA, USA).
Quantification of cellular autophagy
To examine the effect of PLB on autophagy in PANC-1 and BxPC-3 cells, cellular autophagy was detected using flow cytometry as described previously.21 Briefly, PANC-1 and BxPC-3 cells were seeded in 60 mm Petri dishes. After cells were seeded for 24 hours, the cells reached ~75% confluence and then treated with fresh medium alone, control vehicle alone (0.05% DMSO, v/v), or PLB (0.1, 1, and 5 μM) for 24 hours. In separate experiments, PANC-1 and BxPC-3 cells were treated with 5 μM PLB for 4, 8, 12, 24, 48, and 72 hours. Following the PLB treatment, cells were resuspended in 250 μL of phenol red-free culture medium containing 5% FBS, and 250 μL of the diluted Cyto-ID®Green stain solution was added to each sample and mixed well. Cells were incubated for 30 minutes at 37°C in the dark and then collected by centrifugation at 250×g . The cell pellet was washed with 1× assay buffer in the Cyto-ID® autophagy detection kit and resuspended in 500 μL fresh 1× assay buffer. Cells were analyzed using the green (FL1) channel of a flow cytometer (Becton Dickinson Immunocytometry Systems).
Confocal fluorescence microscopy
In order to further detect the cellular autophagy level, the cellular autophagy level was examined using confocal fluorescence microscopy. Briefly, PANC-1 and BxPC-3 cells were seeded into an 8-well chamber slide. The cells were treated with PLB at 0.1, 1, and 5 μM for 24 hours. In separate experiments, cells were treated with 5 μM of PLB for 4, 8, 12, 24, 48, and 72 hours. After the PLB treatment, the cells were washed with 1× assay buffer in the Cyto-ID® autophagy detection kit, following by incubation with 100 μL of microscopy dual detection reagent for 30 minutes at 37°C in the dark. After the incubation, the cells were washed with 1× assay buffer to remove detection reagent, and the cells were then examined using a Leica TCS SP2 laser scanning confocal microscopy (Wetzlar, Germany) using a standard fluorescein isothiocyanate filter set for imaging the autophagic signal at wavelengths of 405/488 nm.
Western blotting analysis
The levels of various cellular proteins were determined using Western blotting assays as described previously.21 The PANC-1 and BxPC-3 cells were incubated with PLB at 0.1, 1, and 5 μM for 24 hours. After PLB treatment, cells were washed with precooled PBS and lysed with the RIPA buffer (50 mmol 4-[2-hydroxyethyl]piperazine-1-ethanesulfonic acid [HEPES] at pH 7.5, 150 mmol sodium chloride [NaCl], 10% glycerol, 1.5 mmol magnesium chloride [MgCl2], 1% Triton-X 100, 1 mmol ethylenediaminetetraacetic acid [EDTA] at pH 8.0, 10 mmol sodium pyrophosphate, and 10 mmol sodium fluoride) containing the protease inhibitor and phosphatase inhibitor cocktails. Protein concentrations were measured by Pierce BCA protein assay kit. Equal amount of protein sample at 20 μg was electrophoresed on 7%–12% sodium dodecyl sulfate polyacrylamide gel electrophoresis minigel after thermal denaturation for 5 minutes at 95°C. Proteins were transferred onto Immobilon® PVDF membrane at 100 V for 2 hours at 4°C. Subsequently, membranes were blocked with 5% skim milk and probed with indicated primary antibody overnight at 4°C and then blotted with respective secondary antibody. Visualization was performed using the Bio-Rad system. Protein level was normalized to the matching densitometric value of β-actin.
Statistical analysis
Data are presented as the mean ± standard deviation. Multiple comparisons were evaluated by one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison. A value of <0.05 was considered statistically significant. Assays were performed in triplicate.
Results
PLB inhibits the viability of PANC-1 and BxPC-3 cells
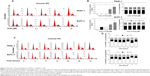
First, we tested the effect of PLB on cell viability in PANC-1 and BxPC-3 cells using the MTT assay. Incubation of both cell lines with PLB at concentrations ranging from 0.1 to 20 μM for 24 and 48 hours significantly decreased cell viability. In comparison to the control cells, the values of cell viability were 83.1%, 77.3%, 69.0%, 46.1%, and 38.5% when PANC-1 cells were treated with PLB at 0.1, 1, 5, 10, and 20 μM for 24 hours, respectively. There was a similar effect of PLB on the cell viability of PANC-1 cells after 48 hours PLB treatment. The values of cell viability of PANC-1 cells were 80.7%, 72.0%, 60.0%, 40.7%, and 34.6% when cells were incubated with PLB at 0.1, 1, 5, 10, and 20 μM for 48 hours, respectively (Figure 1B). For BXPC-3 cells, the values of cellular viability were 92.3%, 79.9%, 43.5%, 14.1%, and 8.7% compared to control cells when cells were treated with PLB at 0.1, 1, 5, 10, and 20 μM for 24 hours, respectively; the cellular viability was 87.6%, 69.0%, 39.8%, 3.4%, and 2.6% when cells were exposed to PLB at 0.1, 1, 5, 10, and 20 μM for 48 hours, respectively (Figure 1C). The IC50 values were 9.9 and 7.0 μM for PANC-1 cells after 24 and 48 hour incubation with PLB, respectively. For BXPC-3 cells, the IC50 values were 8.4 and 5.9 μM after 24 and 48 hour treatment with PLB, respectively (Figure 1). The results show that PLB has a potent inhibitory effect on cell proliferation in PANC-1 and BxPC-3 cells.
PLB regulates the cell cycle distribution of PANC-1 and BxPC-3 cells
We next examined the effect of PLB on cell cycle distribution in PANC-1 and BxPC-3 cells using flow cytometry. PLB showed similar effects on the cell cycle distribution in PANC-1 and BxPC-3 cells. Treatment of cells with PLB at 0.1, 1, 5, and 10 μM did not significantly alter the cell cycle distribution in G1, S, and G2 phases in both cell lines, except for a significant decrease in S phase in PANC-1 cells treated with 5 μM PLB. However, PLB treatment concentration-dependently increased the cell population in sub-G1 in PANC-1 and BxPC-3 cells (Figure 2A and B). In separate experiments, we examined the effect of PLB on cell cycle distribution over 72 hours. Incubation of cells with 5 μM PLB led to varying effects on cell cycle distribution in PANC-1 and BxPC-3 cells (Figure 2C and D). In PANC-1 cells, there was a 12.8%, 17.7%, 18.9%, and 29.9% reduction in G1 phase when cells were treated with 5 μM PLB for 4, 8, 12, and 48 hours, respectively (<0.05), and there was a 1.4-, 1.6-, 1.5-, 1.7-, and 1.3-fold increase in S phase after cells were incubated with 5 μM PLB for 4, 8, 12, 48, and 72 hours, respectively (<0.05). Treating PANC-1 cells with 5 μM PLB for 48 hours significantly increased G2 phase 1.4-fold (Figure 2C and D). For BxPC-3 cells, incubation with 5 μM PLB for 48 hours increased G1 phase 1.3-fold (<0.001) and treatment of cells with 5 μM PLB for 48 and 72 hours decreased S phase 25.6% and 21.8%, respectively (<0.001) (Figure 2C and D). There was no significant change in G2 phase when BxPC-3 cells were treated with 5 μM PLB over 72 hours.
PLB modulates key cell cycle regulators in PANC-1 and BxPC-3 cells
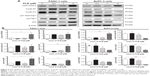
Since we have observed the regulatory effect of PLB on cell cycle distribution in PANC-1 and BxPC-3 cells, we further examined the effect of PLB on the expression levels of cell cycle regulators, including CDK1/CDC2, cyclin B1, cyclin D1, p21 Waf1/Cip1, p27 Kip1, and p53 in PANC-1 and BxPC-3 cells using Western blotting assay. Incubation of PANC-1 cells with PLB at 0.1, 1, and 5 μM resulted in varying alterations in the expression levels of cell cycle regulators (Figure 3A and B). Incubation of PANC-1 cells with PLB at 5 μM resulted in a 4.6-, 4.2-, 3.1-, and 3.1-fold increase in the expression levels of CDK1/CDC2, p21 Waf1/Cip1, p27 Kip1, and p53, respectively. In BxPC-3 cells, there was a differential response to PLB treatment (Figure 3A and B). There was a 3.1-fold increase in the expression level of CDK1/CDC2, but there was a 63.6% and 94.2% reduction in the expression levels of p21 Waf1/Cip1 and p53 in BxPC-3 with the treatment of 5 μM PLB, respectively. The results show that PLB treatment differentially alters the expression levels of cell cycle regulators, which may be associated with the differential regulatory effects of PLB on cell cycle distribution in PANC-1 and BxPC-3 cells.
PLB induces autophagy in both PANC-1 and BxPC-3 cells
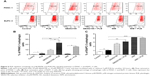
Autophagy is one of the predominant programmed cell death routes, and its role in cancer cell survival and death is controversial.8,24 Recently, it has been reported that induction of autophagy is an emerging strategy for the treatment of pancreatic cancer.10,24 As such, we examined the effect of PLB on autophagy in PANC-1 and BxPC-3 cells using flow cytometry and confocal microscopy. As shown in Figure 4A and B, the percentage of autophagic cells at basal level was 2.9% and 2.4% for PANC-1 and BxPC-3 cells, respectively. Incubation of PANC-1 and BxPC-3 cells with PLB for 24 hours significantly increased the percentage of autophagic cells. In PANC-1 cells, there was a 4.2-fold increase in the percentage of autophagic cells when treated with 5 μM PLB for 24 hours compared to the control cells. Treatment of BxPC-3 cells with 1 and 5 μM PLB for 24 hours resulted in a 1.4- and 2.1-fold increase in the percentage of autophagic cells, respectively. Incubation of PANC-1 and BxPC-3 cells with 0.1 μM PLB for 24 hours did not significantly induce autophagic death.
In separate experiments, the autophagic effect of PLB in PANC-1 and BxPC-3 cells was examined when cells were treated for over 72 hours (Figure 4C and D). In PANC-1 cells, the percentage of autophagic cells was increased from 0.6% at basal level (zero time) to 2.1%, 1.6%, 1.4%, 2.6%, 13.3%, and 20.6% when the cells were treated for 4, 8, 12, 24, 48, and 72 hours, respectively. Treatment of BxPC-3 cells with 5 μM PLB increased the percentage of autophagic cells from 1.3% at basal level (zero time) to 4.2%, 4.8%, 4.9%, 1.7%, 8.2%, and 10.5% when the cells were treated for 4, 8, 12, 24, 48, and 72 hours, respectively.
We further determined the autophagy-inducing effects of PLB in PANC-1 and BxPC-3 cells using confocal microscopic examination. In comparison to the control cells, PLB treatment caused a significant concentration-dependent increase in autophagic cells in PANC-1 and BxPC-3 cells (Figure 5A and B). There was a 1.02- and 1.11-fold increase in the autophagic death of PANC-1 cells when treated with PLB at 1 and 5 μM for 24 hours, respectively. In BxPC-3 cells, there was a 1.1-fold increase in autophagy when treated with 1 and 5 μM PLB. Treatment of cells with PLB at 0.1 μM did not significantly affect the autophagy in PANC-1 and BxPC-3 cells.
In addition, the autophagy-inducing effects of PLB on PANC-1 and BxPC-3 cells over 72 hours were examined (Figure 5C and D). There was a time-dependent increase in autophagy when PANC-1 cells were treated with 5 μM PLB. Compared to the control cells, 5 μM PLB resulted in 1.05-, 1.04-, 1.05-, and 1.06-fold increase in the autophagic death of PANC-1 cells after 12, 24, 48, and 72 hour incubation, respectively. In BxPC-3 cells, 5 μM PLB time-dependently increased the autophagy over 72 hours (<0.001). These results demonstrate that PLB induces autophagy in both PANC-1 and BxPC-3 cells. Both PANC-1 and BxPC-3 cells are largely sensitive to high concentrations of PLB.
WM and SB202190 enhance PLB-induced autophagy in PANC-1 and BxPC-3 cells
Following the examination of autophagic effect of PLB in PANC-1 and BxPC-3 cells, we also tested the possible involved signaling pathways. We employed WM, a potent, irreversible, and selective PI3K inhibitor and a blocker of autophagosome formation, and SB202190, a selective inhibitor of p38 MAPK used as an autophagy inducer. As shown in Figure 6A and B, both WM and SB202190 significantly enhanced the autophagic effect of PLB in PANC-1 and BxPC-3 cells. Compared to the control, incubation of cells with SB202190 and WM increased 13.2- and 2.5-fold in the percentage of autophagic PANC-1 cells and 3.7- and 6.0-fold in the autophagic BxPC-3 cells, respectively. Treatment of PANC-1 cells with SB202190 and WM increased 10.4- and 7.0-fold in autophagic cells, respectively, compared to PLB treated cells. In BxPC-3 cells, incubation with SB202190 and WM led to a 3.9-fold increase in the autophagic cells compared to PLB treated cells (Figure 6A and B). Taken together, the results suggest that PLB-induced autophagy may be, at least in part, ascribed to the regulation of PI3K- and p38 MAPK-mediated signaling pathways.
PLB suppresses PI3K/Akt/mTOR axis in PANC-1 and BxPC-3 cells
Since we have observed an involvement of PI3K in PLB-induced autophagy in PANC-1 and BxPC-3 cells, we next investigated the effect of PLB on PI3K-mediated signaling pathway in PANC-1 and BxPC-3 cells. First, we examined the phosphorylation levels of PI3K at Tyr199 that is the upstream signaling molecule of Akt/mTOR pathway and plays an important role in the regulation of cell proliferation and autophagic cell death.6,8,25 PI3K catalyzes the formation of phosphatidylinositol-3,4,5-triphosphate via phosphorylation of phosphatidylinositol, phosphatidylinositol-4-phosphate, and phosphatidylinositol-4,5-bisphosphate, initiating a number of signaling pathways related to cell proliferation, cell metabolism, cell survival, and cell death.26 Treatment of PANC-1 and BxPC-3 cells with PLB resulted in significant alterations in the expression and phosphorylation level of key functional proteins that regulate autophagy signaling pathway (Figures 7, 8A–H, 9A–H, S1A–L, S2A–L). There was a concentration-dependent decrease in the phosphorylation level of PI3K, Akt, and mTOR after cells were incubated with PLB at 0.1, 1, and 5 μM. There was a 51.5%, 44.9%, and 49.0% reduction in the ratio of p-PI3K over PI3K; 22.9%, 29.8%, and 64.8% decline in the ratio of p-Akt over Akt; and 22.2%, 45.4%, and 72.9% decrease in the ratio of p-mTOR over mTOR in PANC-1 cells treated with PLB at 0.1, 1, and 5 μM, respectively. Similarly, there was a 2.4%, 67.1%, and 89.8% reduction in the ratio of p-PI3K over PI3K; 9.7%, 11.7%, and 53.0% decrease in the ratio of p-Akt over Akt; and 7.8%, 13.3%, and 43.4% decline in the ratio of p-mTOR over mTOR in BxPC-3 cells treated with PLB at 0.1, 1, and 5 μM, respectively.
The effect of PLB on the expression levels of PTEN, beclin 1, LC3-I, and LC3-II was examined in PANC-1 and BxPC-3 cells (Figures 7, 8F–H, 9F–H, S1K and L, S2K and L). PLB treatment concentration-dependently increased the expression level of PTEN and beclin 1 in PANC-1 cells. In comparison to the control cells, incubation of PANC-1 cells with PLB at 1 and 5 μM PLB led to a 1.4- and 3.0-fold increase in the expression of PTEN and 1.1- and 2.3-fold increase in the expression of beclin 1, respectively. The ratio of LC3-II over LC3-I was increased 1.4-, 2.1-, and 5.9-fold when PANC-1 cells were incubated with PLB at 0.1, 1, and 5 μM. There was a similar regulatory effect of PLB on the expression levels of PTEN, beclin 1, LC3-I, and LC3-II in BxPC-3 cells. Treatment of cells with PLB at 0.1, 1, and 5 μM increased the expression level of PTEN 9.1-, 10.3-, and 17.3-fold and elevated the expression level of beclin 1 3.4-, 3.4-, and 3.9-fold compared to the control cells, respectively. The ratio of LC3-II over LC3-I was increased 5.1-fold when BxPC-3 cells were treated with 5 μM PLB. Taken together, the regulatory effect of PLB on the PI3K/Akt/mTOR signaling pathway contributes to its antiproliferative effect on pancreatic cancer cells.
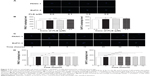
PLB activates AMPK signaling pathway but inhibits p38 MAPK signaling pathway in PANC-1 and BxPC-3 cells
AMPK is highly conserved from yeast to plants and animals and plays a key role in the regulation of energy homeostasis, cell survival, and cell death.27 p38 MAPK regulates cellular responses to cytokines and stress and thus controls cell differentiation, cell death, cell migration, and invasion.28 We speculated that PLB can regulate the AMPK and p38 MAPK signaling pathways in PANC-1 and BxPC-3 cells. We treated both cell lines with PLB at 0.1, 1, and 5 μM for 24 hours. There were differential effects of PLB on the phosphorylation and expression levels of AMPK and p38 MAPK (Figures 7, 8B and C, 9B andC). In comparison to the control cells, PLB concentration-dependently increased 2.2-, 2.2-, and 3.9-fold the ratio of p-AMPK over AMPK but decreased 28.1%, 39.8%, and 42.4% the ratio of p-p38 MAPK over p38 MAPK in PANC-1 cells treated with PLB at 0.1, 1, and 5 μM, respectively. In comparison to the control cells, incubation of BxPC-3 cells with PLB at 1 and 5 μM increased the ratio of p-AMPK over AMPK 1.5- and 16.6-fold, and treatment of BxPC-3 cells with PLB at 0.1 and 1 μM reduced the ratio of p-p38 MAPK over p38 MAPK 20.6% and 34.0%, respectively. The results show that PLB can activate AMPK signaling but inactivate p38 MAPK signaling pathways in PANC-1 and BxPC-3 cells.
PLB inhibits EMT in PANC-1 and BxPC-3 cells
EMT is a critical process playing an important role in cancer invasion and metastasis.11,12,29 EMT depends on a reduction in expression of cell adhesion molecules. E-cadherin is considered to be an active suppressor of invasion and growth of many epithelial cancers. Therefore, the effect of PLB on the expression of E-cadherin and other key EMT markers and regulators was examined in PANC-1 and BxPC-3 cells. Cells were treated with PLB at 0.1, 1, and 5 μM for 24 hours, resulting in varying changes in the expression level of EMT related proteins (Figures 10, 11A–H, 12A–H). Incubation of PANC-1 cells with PLB at 1 and 5 μM increased the expression of E-cadherin 1.4- and 2.1-fold, respectively. Treatment of PANC-1 cells with PLB at 0.1, 1, and 5 μM decreased the expression of N-cadherin 40.0%, 53.2%, and 59.0%, respectively. Treating PANC-1 cells with PLB at 0.1, 1, and 5 μM significantly suppressed the expression of slug by 40.9%, 54.2%, and 50.7%, respectively. There was a 3.0-fold increase in the expression of ZO-1 and 88.9% decrease in the expression level of vimentin when PANC-1 cells were treated with 5 μM PLB.
In BxPC-3 cells, there was a similar modulating effect of PLB on the key functional proteins that regulate EMT (Figures 10, 11A–H, 12A–H). The expression level of E-cadherin was increased 2.1-, 1.9-, and 2.4-fold, and the expression level of N-cadherin was reduced 30.5%, 32.0%, and 57.4% when cells were treated with PLB at 0.1, 1, and 5 μM, respectively. The expression level of slug, the E-cadherin suppressor, was downregulated 22.1% and 55.7% when treated with 1 and 5 μM of PLB, respectively. In addition, incubation of BxPC-3 cells with PLB at 0.1, 1, and 5 μM decreased the expression level of β-catenin 31.9%, 42.3%, and 50.6% but increased the expression level of ZO-1 1.2-, 1.4-, and 3.3-fold, respectively. Taken together, the results show that PLB can inhibit key functional proteins regulating the EMT process, which may contribute to the anticancer effect of PLB in PANC-1 and BxPC-3 cells.
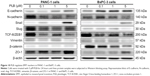
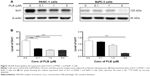
PLB downregulates the expression of Sirt1 in PANC-1 and BxPC-3 cells
Recently, accumulating evidence shows that Sirt1 plays an important role in the regulation of cellular autophagy.30,31 Sirt1 can directly regulate autophagy via deacetylation of key components of the autophagy induction network, such as Atg proteins 5, 7, and 8.31 Nuclear Sirt1 can induce the expression of key components of the autophagy pathway by activating FoxO transcription factor family members.31 We speculate that PLB may regulate Sirt1 expression in PANC-1 and BxPC-3 cells, and thus we examined the effect of PLB on the expression level of Sirt1 in both cell lines. There was a significant decrease in the expression level of Sirt1 in PANC-1 and BxPC-3 cells treated with PLB for 24 hours (Figure 13A and B). In PANC-1 cells, there was a concentration-dependent decrease in the expression level of Sirt1 by 50.0%, 59.3%, and 59.6% when treated with PLB at 0.1, 1, and 5 μM, respectively. Incubation of BxPC-3 cells with 5 μM PLB led to a 79.2% reduction in the expression level of Sirt1. It shows that PLB can remarkably inhibit the expression of Sirt 1 in PANC-1 and BxPC-3 cells.
Discussion
Treatment of pancreatic cancer remains a major challenge due to the lack of progress in primary prevention, early diagnosis, and poor efficacy of current therapies. Natural compounds emerge as an alternative resource for new anticancer drug discovery, and there is an increased interest in seeking new effective drugs for the treatment of pancreatic cancer from natural compounds. PLB, a naturally occurring compound, has been found to exhibit potent anticancer effects in both in vitro and in vivo models, which are mainly attributed to its effects on multiple signaling pathways related to cell cycle arrest, apoptosis, autophagy, EMT inhibition, and reactive oxygen species generation.14–19,21,22,32–34 In the present study, we have found that PLB promoted cell cycle arrest and autophagic cell death but inhibited EMT with the involvement of PI3K/Akt/mTOR- and p38 MAPK-mediated pathways in PANC-1 and BxPC-3 cells.
Cell proliferation is an essential process for growth, development, and regeneration of eukaryotic organisms, and abnormalities in cell cycle have a close relationship with tumorigenesis; induction of cell cycle arrest can prevent cell proliferation by modulation of a number of cell cycle regulators, which can eventually stop tumor growth.35–37 It has been reported that targeting cell cycle may be a promising strategy to fight against cancer through the regulation of CDK-cyclin complexes and related molecular targets.37 In particular, development of cell cycle inhibitors has emerged for the treatment of pancreatic cancer.38 In the present study, we have observed an inhibitory effect of PLB on cell proliferation in PANC-1 and BxPC-3 cells. Furthermore, we have found that PLB treatment can significantly increase the expression levels of p21 Waf1/Cip1, a tumor suppressor protein which functions as an inhibitor of cell cycle progression, and it serves to inhibit kinase activity and block progression through G1/S in association with CDK2 complexes.39 Thus, increased expression levels of p21 Waf1/Cip1 will lead to cell cycle arrest in PANC-1 and BxPC-3 cells. Treating PANC-1 and BxPC-3 cells with PLB also resulted in a significant increase in the expression levels of p27 Kip1 and p53 in the present study. It has been reported that p53 is phosphorylated and upregulates p21 Waf1/Cip1 transcription via a p53-responsive element, and CDC2/cyclin B or CDK2/cyclin A are active during cell cycle stages. p27 Kip1 is a member of the Cip/Kip family of cyclin-dependent kinase inhibitors, enforcing the G1 restriction point via its inhibitory binding to CDK2/cyclin E and other CDK/cyclin complexes and resulting in cell cycle arrest.40 Moreover, p53 is a tumor suppressor protein that plays a major role in cellular response to DNA damage and other genomic aberrations.41 PLB-induced activation of p53 can lead to either cell cycle arrest and DNA repair or apoptosis in PANC-1 and BxPC-3 cells. Notably, the differential response to PLB treatment with regard to the cell cycle regulators may be due to the difference in the origin of the cell lines, different cytogenetics, and other possible factors.
Autophagy is a primary morphologically distinctive mode of programmed cell death via a lysosomal catabolic pathway for engulfment, degradation, and recycling of nonessential or abnormal cellular organelles and proteins in living cells.6,8 This complicated process is executed through multiple steps from intracellular membrane/vesicle reorganization to form double-membraned autophagosomes that fuse with lysosomes to form autophagolysosomes, which degrade the contents via acidic lysosomal hydrolases.6,8,42 Recently, the role of autophagy in cancer cell death is still controversial. Genetic evidence indicates that autophagy can act as a tumor suppressor, but it also can promote the survival of established tumors under stress conditions and in response to chemotherapy.6,24 However, increasing evidence shows that targeting autophagy is a promising strategy for the treatment of pancreatic cancer.24,43,44 Our results have shown that PLB treatment significantly increases the percentage of autophagic cells in both PANC-1 and BxPC-3 cells. It suggests that PLB may be a promising agent used to induce autophagic cell death in pancreatic cancer.
The PI3K/Akt/mTOR signaling pathway is a central pathway related to autophagic cell death.45–47 PI3K activates the downstream serine/threonine kinase Akt, which in turn, through a cascade of regulators, triggers the phosphorylation and activation of the serine/threonine kinase mTOR.47 In the present study, PLB showed a significant inhibitory effect on PI3K/Akt/mTOR in both PANC-1 and BxPC-3 cells, and WM, a potent, irreversible, and selective PI3K inhibitor and a blocker of autophagosome formation, remarkably enhanced the autophagy-inducing effect of PLB in PANC-1 and PxPC-3 cells, which may contribute to its anticancer activity. As a matter of fact, a number of previous studies have showed the autophagy-inducing effect of PLB in various cancer cell lines through the negative modulation of the PI3K/Akt/mTOR axis.21,48 Our previous study has also revealed that PLB predominantly induced autophagy through the inhibition of the PI3K/Akt/mTOR pathway in non-small-cell lung cancer cells.21 Thus, the inhibition of the PI3K/Akt/mTOR signaling pathway contributes, at least in part, to the autophagy-inducing effect of PLB in PANC-1 and BxPC-3 cells.
AMPK, the master energy sensor, is an important regulator of cell death through the activation of p53, inhibition of mTOR, and other related signaling pathways.6 In the present study, there was a significant activation of AMPK with PLB treatment, which may contribute to the inhibition of mTOR, which in turn can exacerbate mTOR-mediated autophagy. In fact, previous studies have reported that a number of natural compounds including vincristine, taxol, temozolomide, doxorubicin, ursolic acid, honokiol, quercetin, and fisetin can activate AMPK to promote autophagy in various types of cancer cells.49,50 Furthermore, we have observed that PLB significantly suppressed the phosphorylation of p38 MAPK and that SB202190, a p38 MAPK inhibitor, remarkably increased the PLB-induced autophagic cell death in both PANC-1 and BxPC-3 cells. It has been reported that p38 MAPK is responsive to stress stimuli such as cytokines, ultraviolet irradiation, heat shock, and osmotic shock and regulates cell differentiation, apoptosis, and autophagy.6,45 Collectively, both AMPK and p38 MAPK play important roles in PLB-induced pancreatic cancer cell autophagy.
EMT has been implicated in cancer initiation, invasion, migration, metastasis, and drug resistance.12,51 A number of previous studies have shown that regulation of EMT may represent an emerging therapeutic approach to fight against pancreatic cancer.12,29 EMT depends on a reduction in expression of cell adhesion molecules and loss of tight junctions. E-cadherin can act as an active suppressor of invasion and growth of many epithelial cancers, and tight junctions or zonula occludens are able to form a continuous barrier to fluids across the epithelium and endothelium.11,12,29 In the present study, our findings have demonstrated that PLB treatment significantly increased the expression level of E-cadherin and ZO-1 in both PANC-1 and BxPC-3 cells. In fact, E-cadherin mediates calcium-dependent cell–cell adhesion, which also is suppressed by snail/slug, and our data have shown that PLB treatment inhibited the expression of slug, which in turn may result in the increased expression of E-cadherin. Furthermore, β-catenin can act as an integral component of a protein complex in adherent junctions, which helps cells maintain epithelial layers, and β-catenin participates in the Wnt signaling pathway as a downstream target.13,29 PLB treatment decreased the expression of β-catenin in BxPC-3 cells but not in PANC-1 cells, which may be due to the difference in cell type.
In addition, Sirt1 is a NAD+-dependent DNA repair enzyme originally discovered in yeast (Sir2) and is involved in a myriad of cellular processes.52 Recently, studies have shown that Sirt1 has an important role in the regulation of autophagy and that it deacetylates both histone and nonhistone proteins, such as p53 and FoxOs.52 Sirt1 also can affect autophagy via direct regulation of autophagy machinery, including deacetylation of Atg5, Atg7, and Atg8.30 In the present study, we observed a significant reduction in the expression level of Sirt1 in PANC-1 and BxPC-3 cells when cells were treated with PLB, suggesting that PLB-induced autophagy in pancreatic cancer cells may involve Sirt1-mediated pathway.
In summary, we investigated the potential molecular mechanisms of PLB for its anticancer effect in human pancreatic cancer PANC-1 and BxPC-3 cells. The mechanisms of action of PLB were mainly related to cell cycle arrest, induction of autophagy, and inhibition of EMT and their associated signaling pathways. PLB induced the inhibition of PI3K/Akt/mTOR and p38 MAPK pathways and the activation of AMPK, contributing to the autophagy-inducing activities of PLB. PLB may represent a new anticancer drug that can kill pancreatic cancer cells. More studies are needed to reveal the underlying mechanisms and other potential targets of PLB in the treatment of pancreatic cancer.
Acknowledgments
The authors appreciate the financial support from the Startup Funds of the College of Pharmacy, University of South Florida, Tampa, FL, USA. Dr Zhi-Wei Zhou holds a postdoctoral scholarship from the College of Pharmacy, University of South Florida, Tampa, FL, USA.
Disclosure
The authors report no conflicts of interest in this work.
References
Ferlay J, Soerjomataram I, Ervik M, et al. GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 [webpage on the Internet]. Lyon, France: International Agency for Research on Cancer; 2013. Available from: http://globocan.iarc.fr. Accessed September 1, 2014. | ||
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014 CA Cancer J Clin. 2014;64(1):9–29. | ||
Guo X, Cui Z. Current diagnosis and treatment of pancreatic cancer in China Pancreas. 2005;31(1):13–22. | ||
Long J, Luo GP, Xiao ZW, et al. Cancer statistics: current diagnosis and treatment of pancreatic cancer in Shanghai, China Cancer Lett. 2014;346(2):273–277. | ||
Chen W, Zheng R, Zhang S, et al. Report of incidence and mortality in China cancer registries, 2009 Chin J Cancer Res. 2013;25(1):10–21. | ||
Denton D, Nicolson S, Kumar S. Cell death by autophagy: facts and apparent artefacts Cell Death Differ. 2012;19(1):87–95. | ||
Giatromanolaki A, Sivridis E, Mendrinos S, Koutsopoulos AV, Koukourakis MI. Autophagy proteins in prostate cancer: relation with anaerobic metabolism and Gleason score Urol Oncol. 2014;32(1):39.e11–e18. | ||
Klionsky DJ, Emr SD. Autophagy as a regulated pathway of cellular degradation Science. 2000;290(5497):1717–1721. | ||
Kang R, Tang D. Autophagy in pancreatic cancer pathogenesis and treatment Am J Cancer Res. 2012;2(4):383–396. | ||
Mujumdar N, Saluja AK. Autophagy in pancreatic cancer: an emerging mechanism of cell death Autophagy. 2010;6(7):997–998. | ||
Zheng H, Kang Y. Multilayer control of the EMT master regulators Oncogene. 2014;33(14):1755–1763. | ||
Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial-mesenchymal transition Nat Rev Mol Cell Biol. 2014;15(3):178–196. | ||
Nauseef JT, Henry MD. Epithelial-to-mesenchymal transition in prostate cancer: paradigm or puzzle? Nat Rev Urol. 2011;8(8):428–439. | ||
Padhye S, Dandawate P, Yusufi M, Ahmad A, Sarkar FH. Perspectives on medicinal properties of plumbagin and its analogs Med Res Rev. 2012;32(6):1131–1158. | ||
Hafeez BB, Jamal MS, Fischer JW, Mustafa A, Verma AK. Plumbagin, a plant derived natural agent inhibits the growth of pancreatic cancer cells in in vitro and in vivo via targeting EGFR, Stat3 and NF-κB signaling pathways Int J Cancer. 2012;131(9):2175–2186. | ||
Subramaniya BR, Srinivasan G, Sadullah SS, et al. Apoptosis inducing effect of plumbagin on colonic cancer cells depends on expression of COX-2 PLoS One. 2011;6(4):e18695. | ||
Nazeem S, Azmi AS, Hanif S, et al. Plumbagin induces cell death through a copper-redox cycle mechanism in human cancer cells Mutagenesis. 2009;24(5):413–418. | ||
Xu KH, Lu DP. Plumbagin induces ROS-mediated apoptosis in human promyelocytic leukemia cells in vivo Leuk Res. 2010;34(5):658–665. | ||
Sun J, McKallip RJ. Plumbagin treatment leads to apoptosis in human K562 leukemia cells through increased ROS and elevated TRAIL receptor expression Leuk Res. 2011;35(10):1402–1408. | ||
Shieh JM, Chiang TA, Chang WT, et al. Plumbagin inhibits TPA-induced MMP-2 and u-PA expressions by reducing binding activities of NF-kappaB and AP-1 via ERK signaling pathway in A549 human lung cancer cells Mol Cell Biochem. 2010;335(1–2):181–193. | ||
Li YC, He SM, He ZX, et al. Plumbagin induces apoptotic and autophagic cell death through inhibition of the PI3K/Akt/mTOR pathway in human non-small cell lung cancer cells Cancer Lett. 2014; 344(2):239–259. | ||
Sinha S, Pal K, Elkhanany A, et al. Plumbagin inhibits tumorigenesis and angiogenesis of ovarian cancer cells in vivo Int J Cancer. 2013; 132(5):1201–1212. | ||
Lecoeur H. Nuclear apoptosis detection by flow cytometry: influence of endogenous endonucleases Exp Cell Res. 2002;277(1):1–14. | ||
Yang S, Kimmelman AC. A critical role for autophagy in pancreatic cancer Autophagy. 2011;7(8):912–913. | ||
Muilenburg D, Parsons C, Coates J, Virudachalam S, Bold RJ. Role of autophagy in apoptotic regulation by Akt in pancreatic cancer Anticancer Res. 2014;34(2):631–637. | ||
Cantley LC. The phosphoinositide 3-kinase pathway Science. 2002; 296(5573):1655–1657. | ||
Dunlop EA, Tee AR. The kinase triad, AMPK, mTORC1 and ULK1, maintains energy and nutrient homoeostasis Biochem Soc Trans. 2013; 41(4):939–943. | ||
Arthur JS, Ley SC. Mitogen-activated protein kinases in innate immunity Nat Rev Immunol. 2013;13(9):679–692. | ||
Cannito S, Novo E, di Bonzo LV, Busletta C, Colombatto S, Parola M. Epithelial-mesenchymal transition: from molecular mechanisms, redox regulation to implications in human health and disease Antioxid Redox Signal. 2010;12(12):1383–1430. | ||
Lee IH, Cao L, Mostoslavsky R, et al. A role for the NAD-dependent deacetylase Sirt1 in the regulation of autophagy Proc Natl Acad Sci U S A. 2008;105(9):3374–3379. | ||
Ng F, Tang BL. Sirtuins’ modulation of autophagy J Cell Physiol. 2013;228(12):2262–2270. | ||
Wang CC, Chiang YM, Sung SC, Hsu YL, Chang JK, Kuo PL. Plumbagin induces cell cycle arrest and apoptosis through reactive oxygen species/c-Jun N-terminal kinase pathways in human melanoma A375.S2 cells Cancer Lett. 2008;259(1):82–98. | ||
Hsu YL, Cho CY, Kuo PL, Huang YT, Lin CC. Plumbagin (5-hydroxy-2-methyl-1,4-naphthoquinone) induces apoptosis and cell cycle arrest in A549 cells through p53 accumulation via c-Jun NH2-terminal kinase-mediated phosphorylation at serine 15 in vitro and in vivo. J Pharmacol Exp Ther. 2006;318(2):484–494. | ||
Yang SJ, Chang SC, Wen HC, Chen CY, Liao JF, Chang CH. Plumbagin activates ERK1/2 and Akt via superoxide, Src and PI3-kinase in 3T3-L1 cells Eur J Pharmacol. 2010;638(1–3):21–28. | ||
Williams GH, Stoeber K. The cell cycle and cancer J Pathol. 2012; 226(2):352–364. | ||
Rengarajan T, Rajendran P, Nandakumar N, Balasubramanian MP, Nishigaki I. Cancer preventive efficacy of marine carotenoid fucoxanthin: cell cycle arrest and apoptosis Nutrients. 2013;5(12):4978–4989. | ||
Diaz-Moralli S, Tarrado-Castellarnau M, Miranda A, Cascante M. Targeting cell cycle regulation in cancer therapy Pharmacol Ther. 2013;138(2):255–271. | ||
Bayraktar S, Rocha Lima CM. Emerging cell-cycle inhibitors for pancreatic cancer therapy Expert Opin Emerg Drugs. 2012;17(4):571–582. | ||
Warfel NA, El-Deiry WS. p21WAF1 and tumourigenesis: 20 years after Curr Opin Oncol. 2013;25(1):52–58. | ||
Yoon MK, Mitrea DM, Ou L, Kriwacki RW. Cell cycle regulation by the intrinsically disordered proteins p21 and p27 Biochem Soc Trans. 2012;40(5):981–988. | ||
Carvajal LA, Manfredi JJ. Another fork in the road – life or death decisions by the tumour suppressor p53 EMBO Rep. 2013;14(5):414–421. | ||
Chen Y, Yu L. Autophagic lysosome reformation Exp Cell Res. 2013; 319(2):142–146. | ||
Yang A, Rajeshkumar NV, Wang X, et al. Autophagy is critical for pancreatic tumor growth and progression in tumors with p53 alterations Cancer Discov. 2014;4(8):905–913. | ||
Iacobuzio-Donahue CA, Herman JM. Autophagy, p53, and pancreatic cancer N Engl J Med. 2014;370(14):1352–1353. | ||
Taylor RC, Cullen SP, Martin SJ. Apoptosis: controlled demolition at the cellular level Nat Rev Mol Cell Biol. 2008;9(3):231–241. | ||
Estaquier J, Vallette F, Vayssiere JL, Mignotte B. The mitochondrial pathways of apoptosis Adv Exp Med Biol. 2012;942:157–183. | ||
Rodon J, Dienstmann R, Serra V, Tabernero J. Development of PI3K inhibitors: lessons learned from early clinical trials Nat Rev Clin Oncol. 2013;10(3):143–153. | ||
Kuo PL, Hsu YL, Cho CY. Plumbagin induces G2-M arrest and autophagy by inhibiting the AKT/mammalian target of rapamycin pathway in breast cancer cells Mol Cancer Ther. 2006;5(12):3209–3221. | ||
Mihaylova MM, Shaw RJ. The AMPK signalling pathway coordinates cell growth, autophagy and metabolism Nat Cell Biol. 2011;13(9):1016–1023. | ||
Law BY, Chan WK, Xu SW, et al. Natural small-molecule enhancers of autophagy induce autophagic cell death in apoptosis-defective cells Sci Rep. 2014;4:5510. | ||
Yamada S, Fuchs BC, Fujii T, et al. Epithelial-to-mesenchymal transition predicts prognosis of pancreatic cancer Surgery. 2013;154(5):946–954. | ||
Preyat N, Leo O. Sirtuin deacylases: a molecular link between metabolism and immunity J Leukoc Biol. 2013;93(5):669–680. |
Supplementary materials
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.