Back to Journals » Drug Design, Development and Therapy » Volume 9
Liraglutide reduces lipogenetic signals in visceral adipose of db/db mice with AMPK activation and Akt suppression
Authors Shao Y, Yuan G, Zhang J, Guo X
Received 12 December 2014
Accepted for publication 19 January 2015
Published 18 February 2015 Volume 2015:9 Pages 1177—1184
DOI https://doi.org/10.2147/DDDT.S79175
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Shu-Feng Zhou
Yimin Shao, Geheng Yuan, Junqing Zhang, Xiaohui Guo
Department of Endocrinology, Peking University First Hospital, Beijing, People’s Republic of China
Abstract: Liraglutide, a glucagon-like peptide-1 analog, has been proved to reduce body weight and visceral adipose tissue (VAT) in human studies. In this study, we aimed at examining lipogenetic signal changes in VAT after weight-loss with liraglutide in db/db mice. The mice were divided into two groups: liraglutide-treated group (n=14, 8-week-old, fasting glucose. >10 mmol/L, liraglutide 300 µg/kg twice a day for 4 weeks) and control group (n=14, saline). We found body weight gain and food intake were reduced after liraglutide treatment (P<0.05). Compared to the control group, the VAT weights were significantly lower in the treated group (2.32±0.37 g versus 3.20±0.30 g, P<0.01) than that in control group. In VAT, compared with control group, the lipogenetic transcription factors PPARγ and C/EBPα expressions were both reduced with pAMPK and pACC increased 3.5-fold and 2.31-fold respectively, while pAkt and pP38MAPK were reduced 0.38-fold and 0.62-fold respectively (P<0.01). In conclusion, VAT was reduced after weight loss with AMPK activation and Akt suppression with liraglutide treatment, which was associated with reduction of lipogenetic process in VAT.
Keywords: liraglutide, visceral adipose tissue, AMP-activated protein kinase, lipogenesis
Introduction
The growing prevalence of obesity constitutes a major health problem worldwide.1 Associated with obesity, particularly abdominal obesity, metabolic disorders including hyperinsulinemia, impaired glucose tolerance, and dyslipidemia are often observed, which increase the risk for type 2 diabetes mellitus, cancer, and heart disease.2–6 Indeed, visceral and subcutaneous depots differ considerably from the histological, physiological, and metabolic points of view.7 Abdominal fat accumulation represents a risk factor per se.8 To reduce visceral adipose tissue (VAT) is crucial to type 2 diabetes mellitus and cardiovascular disease therapy.
Lipid accumulation increases throughout the adipogenic process, and it is regulated by genetic and growth factors.9 PPARγ and C/EBPα are two major lipogenetic transcription factors.10,11 AMPK is a serine/threonine heterotrimeric kinase that acts as an intracellular energy sensor12,13 or “fuel gauge”.14 In keeping with its energy sensor role, starvation activates AMPK in adipose tissue,15,16 and AMPK exerts antilipolytic effects,15–17 as well as inhibiting adipocyte fatty acid synthesis, by phosphorylating ACC16 and inhibiting insulin-induced glucose uptake.17 The overall effect of AMPK is to convert adipocytes into lipid oxidizing cells with suppressed lipogenesis and lipolysis.16
GLP-1, an insulinotropic gastrointestinal peptide produced mainly from intestinal endocrine L-cells in response to food intake, lowers blood glucose, delays gastric emptying, and increases satiety as well as reduces body weight.18–21 Liraglutide is a GLP-1 analog with 97% amino acid sequence identity to native human GLP-1 and an acyl side-chain attachment, which makes it bind to albumin. These small structural differences prolong the half-life of the analog to 13 hours, making it suitable for once daily administration.22 Large phase III clinical studies have consistently shown that liraglutide improves glycemic control, blood pressure, and lipid profiles with weight loss.23–28 In clinical trials, body weight index and the waist/hip ratio are significantly reduced after liraglutide treatment.29 The liraglutide effect and action in diabetes for 26 weeks (LEAD-2) and 52 weeks (LEAD-3) studies have shown that reductions in body weight with liraglutide primarily come from reductions in fat mass rather than lean tissue mass.30 In addition, the computed tomography (CT) assessment from within the LEAD-2 study showed that the mean reductions in tissue area from baseline were greater for VAT (−16.4%) than abdominal subcutaneous adipose tissue (−8.5%).31 Now, liraglutide 3.0 mg per day has already been approved by the US Food and Drug Administration (FDA) in obesity treatment.
In our study, we aimed at examining the effects of liraglutide on lipogenetic signal changes in VAT.
Materials and methods
Animals and procedures
All experiments were carried out with permits from the Animal Experiments Ethical Committee of Peking University First Hospital. Six-week-old male db/db mice (C57BL/KsJ-db/db) were purchased from Peking University Laboratory Animal Center. All the mice were housed (seven mice/cage) in an air-conditioned room at 22°C±2°C with controlled ambient conditions following a 12-hour light:12-hour dark cycle, with lights on at 8 am. Drinking water and high fat rodent diet with 23% fat content (HFK Bioscience, Beijing, People’s Republic of China) were supplied ad libitum.
After a week of adjustable feeding, mice were randomly assigned into the liraglutide-treated group (n=14) and the control group (n=14). When fasting blood glucose was above 10 mmol/L, the nearly 8-week-old mice received subcutaneous injections of liraglutide (300 μg/kg) or 0.9% saline of the equal volume twice a day for 4 weeks. Fasting blood glucose, food intake, and body weight were measured every week.
At the end of the experimental period, all mice were fasted overnight with free access to water and then killed by decapitation. Plasma was collected and frozen and VAT were quickly dissected and weighed. The perirenal, epididymal, and omental adipose tissue, as representative of visceral fat, were pooled.
Ribonucleic acid (RNA) isolation and cDNA synthesis
Total RNA was isolated from mouse islets using TRI reagent. First-strand cDNA was synthesized from total RNA using a reverse transcriptase synthesis system (SuperScript III; Invitrogen, Carlsbad, CA, USA) and random hexamers. Real-time polymerase chain reaction was performed with a sequence detection system (ABI Prism 7500; Thermo Fisher Scientific). The messenger RNA (mRNA) levels were calculated by comparative CT methods (XTest/XGAPDH=2−ΔΔCT) with GAPDH as the endogenous reference gene. Primers and probes used in quantitative real-time polymerase chain reaction were designed by Primer Express 3.0 as listed in Table 1.
  | Table 1 Primer pairs for real-time PCR of genes associated with lipogenesis |
Western blot analysis
Total protein was extracted and then quantified using a BCA protein quantification kit (Beyotime, Shanghai, People’s Republic of China). A total of 50 μg protein from each sample was separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (12%) and transferred to a nitrocellulose membrane (0.45 μm pore size; PALL Corporation, Port Washington, NY, USA). The membranes were blocked in Tris-buffered saline containing 0.1% Tween-20 (TBST) and 5% bull serum albumin for 1 hour at room temperature. After incubation with the primary antibodies at 4°C overnight, the membranes were washed in TBST for 15 minutes three times prior to incubation with the secondary anti-rabbit (Abgent Company, San Diego, CA, USA) or anti-mouse (Beijing Zhongshan Golden Bridge Biotechnology Co., Beijing, People’s Republic of China) horseradish peroxidase-conjugated antibody for 1 hour at room temperature. After the membranes were washed again with TBST for 15 minutes three times, the bands were visualized with enhanced chemiluminescence reagents (EMD Millipore, Billerica, MA, USA). Primary antibodies PPAR-γ, pAkt, Akt, pP38 MAPK, P38 MAPK, pERK1/2, ERK1/2, FoxO1, pAMPK, AMPK, pACC, ACC (Cell Signaling Technology, Danvers, MA, USA), and β-actin (Beijing Zhongshan Gold Bridge Biotechnology Co.) were used.
Statistical analysis
Data were expressed as mean ± standard error. Significant differences among groups were evaluated by one-way analysis of variance and Tukey’s multiple comparisons test or by unpaired two-tailed Student’s t-test by using PRISM software. Groups of data from both were considered to be significantly different if P<0.05.
Results
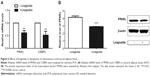
Liraglutide reduced body weight gain and food intake
There was an insignificant difference on baseline body weights and food intake between the treated and control groups. Compared to baseline, the body weights of liraglutide-treated group were significantly decreased after 1-week treatment (P<0.001, Figure 1A), whereas body weight of the control group was steadily increased (Figure 1A). An average relative body weight gain was calculated by subtracting the baseline body weight from the body weight measured each week. We found that the body weight gain was markedly lower in liraglutide-treated group than the control group (P=0.02, Figure 1B).
After 4-week treatment, food intake of the liraglutide-treated group was decreased from baseline of 5.21±0.56 g/d per mouse to 4.47±0.32 g/d per mouse, while that of the control group was increased from 4.62±0.47 g/d per mouse to 6.56±0.45 g/d per mouse (P<0.01, Figure 1C). The food intake decrease of the treated group was significantly lower than that of the control group (P=0.002, Figure 1D).
VAT contents were reduced after liraglutide treatment
We took all the perirenal, epididymal, and omental adipose tissue as VAT. After liraglutide treatment, VAT weights were reduced significantly compared with the control group respectively (2.32±0.37 g versus 3.20±0.30 g, P<0.01, Figure 2).
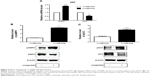
Liraglutide reduced lipogenetic transcription factors expressions in VAT
The mRNA expressions of specific lipogenetic transcription factor PPARγ and C/EBPα were all reduced in VAT compared with the control group (P<0.01, Figure 3A). In addition, the protein levels of PPARγ were reduced by 0.703-fold in VAT as assessed by Western blot analysis (P<0.0001, Figure 3B).
Effect of liraglutide on Akt signaling pathways
Further, we evaluated the protein expressions of pAkt, pP38 MAPK, and pERK1/2. Protein levels of pAkt, pP38 MAPK were reduced by 0.38-fold and 0.62-fold in VAT compared to the control group (P<0.0001, Figure 4A–C). There was no significant difference for pERK1/2 in VAT between the treated and control group. Since a previous study has shown that Akt-mediated inhibition of FoxO1 induces PPARγ and subsequent adipocyte differentiation32 and PI3K is in the upstream of Akt signaling pathway, we further found that the protein levels of FoxO1 were increased in VAT (P<0.0001, Figure 4D). In VAT, the mRNA expression of FoxO1 in the treated group was increased by 1.7-fold and PI3K was reduced by 0.56-fold compared to the control group (P<0.0001, Figure 4E).
Effect of liraglutide on AMPK signaling pathways
AMPK has been demonstrated to mediate inactivation of lipogenic enzymes by inducing phosphorylation of ACC.33 We found that the mRNA levels of AMPK in the treated group were increased by 1.83-fold and that of ACC was reduced by 0.56-fold in VAT compared to the control group (P<0.01, Figure 5A). After liraglutide treatment, AMPK signaling pathways were activated in VAT with protein levels of pAMPK increased by 3.5-fold (P<0.0001, Figure 5B) and those of pACC increased by 2.31-fold (P<0.0001, Figure 5C) compared with that in control group.
Discussion
In this study, we have shown that liraglutide reduced body weight gain and VAT. The expressions of lipogenetic transcription factors PPARγ and C/EBPα in VAT were reduced with activation of AMPK signaling pathway and suppression of Akt signaling pathway.
It is known that activated AMPK is a master regulator of cellular responses to low energy states, which suppresses lipogenesis pathways.34 It has been reported that the expression of AMPK was reduced in VAT both in obese mice35 and in morbidly obese patients.36 Gauthier et al reported that AMPK activity is lower in VAT of morbidly obese individuals who are insulin resistant than in comparably obese people who are insulin sensitive.37 In the present study, the activation of AMPK in VAT suggested liraglutide partly reversed pathophysiology of visceral obesity.
AMPKs are activated by two distinct signals: a Ca2+-dependent pathway mediated by CaMKKb and an AMP-dependent pathway mediated by LKB1.38,39 The calcium concentration could be increased either by direct stimulation of sympathetic nerves or the GLP-1 receptor-cAMP-PKA signaling pathway.40 Central nervous system GLP-1 directly controls adipocyte lipid metabolism and this action seems to be partially modulated through effects on sympathetic outflow, and more specifically by β1- and β2-adrenergic receptor.41 In adipose tissue, fasting and exercise activate AMPK.15,17,42 Since both situations are concomitant with adrenergic stimulation, it could be anticipated that β-adrenergic agonists and their second messenger cAMP would stimulate AMPK activity. Nogueiras et al found chronic central infusion of GLP-1 reduced body weight and fat mass with up sympathetic nerve activity independent of food intake.41 It was indicated that the actions of GLP-1 on VAT should be modulated by sympathetic tone to activate AMPK, leading to decreased lipogenesis and reduced triglyceride content (Figure 6).
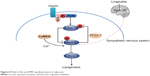  | Figure 6 Model of Akt and AMPK signaling network in adipocyte. |
Akt is known to mediate a number of biological responses to insulin. Inhibition of Akt activity resulted in a reduction in de novo lipid synthesis. In our study, AMPK activation and Akt suppression led to increased pACC in VAT after weight loss with GLP-1. Consequently, the lipogenesis in VAT was down-regulated. But how Akt was inactivated and the effects of insulin on it were not clear. Kim et al found AMPK stimulates dephosphorylation of Akt/PKB through PP2A activation in MDA-MB-231 cells.43 We supposed there were mutual effects between Akt and AMPK, just as shown in Figure 6.
One of the limitations of the present study is that we could not conclude that these were direct effects of liraglutide on VAT because of the present study design. We are going to design an AMPK knockdown experiment to confirm the present results in the future. The other is that the evaluation of body fat content was not conducted by imaging methods due to experimental conditions. Even though the adipose tissue was removed as much as possible, it still could not reflect the whole condition of the body fat. Besides, as an animal study, caution is advised when applying the results to people.
In summary, we found that VAT was reduced after weight loss with AMPK activation and Akt suppression with liraglutide. It was associated with the reduction of lipogenetic process in VAT.
Acknowledgments
We would like to thank technicians from pathology department of Peking University First Hospital for histological assistance and Novo Nordisk for providing liraglutide.
Disclosure
The authors declare that they have no conflicts of interest.
References
Hossain P, Kawar B, El Nahas M. Obesity and diabetes in the developing world – a growing challenge. N Engl J Med. 2007;356(3):213–215. | ||
Kahn BB, Flier JS. Obesity and insulin resistance. J Clin Invest. 2000;106(4):473–481. | ||
Kopelman PG. Obesity as a medical problem. Nature. 2000;404(6778):635–643. | ||
Mokdad AH, Ford ES, Bowman BA, et al. Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001. JAMA. 2003;289(1):76–79. | ||
Bray GA. Medical consequences of obesity. J Clin Endocrinol Metab. 2004;89(6):2583–2589. | ||
Buse JB, Ginsberg HN, Bakris GL, et al. Primary prevention of cardiovascular diseases in people with diabetes mellitus: a scientific statement from the American Heart Association and the American Diabetes Association. Diabetes Care. 2007;30(1):162–172. | ||
Tarui S, Tokunaga K, Fujioka S, Matsuzawa Y. Visceral fat obesity: anthropological and pathophysiological aspects. Int J Obes. 1991;15 Suppl 2:1–8. | ||
Piernas C, Hernández-Morante JJ, Canteras M, Zamora S, Garaulet M. New computed tomography-derived indices to predict cardiovascular and insulin-resistance risks in overweight/obese patients. Eur J Clin Nutr. 2009;63(7):887–897. | ||
Cristancho AG, Lazar MA. Forming functional fat: a growing understanding of adipocyte differentiation. Nat Rev Mol Cell Biol. 2011;12(11):722–734. | ||
Farmer SR. Regulation of PPARgamma activity during adipogenesis. Int J Obes (Lond). 2005;29 Suppl 1:S13–S16. | ||
Farmer SR. Transcriptional control of adipocyte formation. Cell Metab. 2006;4(4):263–273. | ||
Carling D. The AMP-activated protein kinase cascade – a unifying system for energy control. Trends Biochem Sci. 2004;29(1):18–24. | ||
Hardie DG, Hawley SA, Scott JW. AMP-activated protein kinase – development of the energy sensor concept. J Physiol. 2006;574({t 1):7–15. | ||
Hardie DG, Carling D. The AMP-activated protein kinase – fuel gauge of the mammalian cell? Eur J Biochem. 1997;246(2):259–273. | ||
Sponarova J, Mustard KJ, Horakova O, et al. Involvement of AMP-activated protein kinase in fat depot-specific metabolic changes during starvation. FEBS Lett. 2005;579(27):6105–6110. | ||
Daval M, Foufelle F, Ferre P. Functions of AMP-activated protein kinase in adipose tissue. J Physiol. 2006;574(Pt 1):55–62. | ||
Daval M, Diot-Dupuy F, Bazin R, et al. Anti-lipolytic action of AMP-activated protein kinase in rodent adipocytes. J Biol Chem. 2005;280(26):25250–25257. | ||
Flint A, Raben A, Astrup A, Holst JJ. Glucagon-like peptide 1 promotes satiety and suppresses energy intake in humans. J Clin Invest. 1998;101(3):515–520. | ||
Herrmann C, Göke R, Richter G, Fehmann HC, Arnold R, Göke B. Glucagon-like peptide-1 and glucose-dependent insulin-releasing polypeptide plasma levels in response to nutrients. Digestion. 1995;56(2):117–126. | ||
Imeryüz N, Yeğen BC, Bozkurt A, Coşkun T, Villanueva-Peñacarrillo ML, Ulusoy NB. Glucagon-like peptide-1 inhibits gastric emptying via vagal afferent-mediated central mechanisms. Am J Physiol. 1997;273(4 Pt 1):G920–G927. | ||
Meier JJ, Nauck MA. The potential role of glucagon-like peptide 1 in diabetes. Curr Opin Investig Drugs. 2004;5(4):402–410. | ||
Agersø H, Jensen LB, Elbrønd B, Rolan P, Zdravkovic M. The pharmacokinetics, pharmacodynamics, safety and tolerability of NN2211, a new long-acting GLP-1 derivative, in healthy men. Diabetologia. 2002;45(2):195–202. | ||
Marre M, Shaw J, Brändle M, et al. Liraglutide, a once-daily human GLP-1 analogue, added to a sulphonylurea over 26 weeks produces greater improvements in glycaemic and weight control compared with adding rosiglitazone or placebo in subjects with Type 2 diabetes (LEAD-1 SU). Diabet Med. 2009;26(3):268–278. | ||
Nauck M, Frid A, Hermansen K, et al. Efficacy and safety comparison of liraglutide, glimepiride, and placebo, all in combination with metformin, in type 2 diabetes: the LEAD (liraglutide effect and action in diabetes)-2 study. Diabetes Care. 2009;32(1):84–90. | ||
Garber A, Henry R, Ratner R, et al. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 Mono): a randomised, 52-week, phase III, double-blind, parallel-treatment trial. Lancet. 2009;373(9662):473–481. | ||
Zinman B, Gerich J, Buse JB, et al. Efficacy and safety of the human glucagon-like peptide-1 analog liraglutide in combination with metformin and thiazolidinedione in patients with type 2 diabetes (LEAD-4 Met+TZD). Diabetes Care. 2009;32(7):1224–1230. | ||
Russell-Jones D, Vaag A, Schmitz O, et al. Liraglutide vs insulin glargine and placebo in combination with metformin and sulfonylurea therapy in type 2 diabetes mellitus (LEAD-5 met+SU): a randomised controlled trial. Diabetologia. 2009;52(10):2046–2055. | ||
Buse JB, Rosenstock J, Sesti G, et al. Liraglutide once a day versus exenatide twice a day for type 2 diabetes: a 26-week randomised, parallel-group, multinational, open-label trial (LEAD-6). Lancet. 2009;374(9683):39–47. | ||
Inoue K, Maeda N, Kashine S, et al. Short-term effects of liraglutide on visceral fat adiposity, appetite, and food preference: a pilot study of obese Japanese patients with type 2 diabetes. Cardiovasc Diabetol. 2011;10:109. | ||
Jendle J, Nauck MA, Matthews DR, et al. Weight loss with liraglutide, a once-daily human glucagon-like peptide-1 analogue for type 2 diabetes treatment as monotherapy or added to metformin, is primarily as a result of a reduction in fat tissue. Diabetes Obes Metab. 2009;11(12):1163–1172. | ||
Nauck M, Frid A, Hermansen K, et al. Long-term efficacy and safety comparison of liraglutide, glimepiride and placebo, all in combination with metformin in type 2 diabetes: 2-year results from the LEAD-2 study. Diabetes Obes Metab. 2013;15(3):204–212. | ||
Nakae J, Kitamura T, Kitamura Y, Biggs WH 3rd, Arden KC, Accili D. The forkhead transcription factor FoxO1 regulates adipocyte differentiation. Dev Cell. 2003;4(1):119–129. | ||
Zhang BB, Zhou G, Li C. AMPK: an emerging drug target for diabetes and the metabolic syndrome. Cell Metab. 2009;9(5):407–416. | ||
Postic C, Girard J. Contribution of de novo fatty acid synthesis to hepatic steatosis and insulin resistance: lessons from genetically engineered mice. J Clin Invest. 2008;118(3):829–838. | ||
Gaidhu MP, Anthony NM, Patel P, Hawke TJ, Ceddia RB. Dysregulation of lipolysis and lipid metabolism in visceral and subcutaneous adipocytes by high-fat diet: role of ATGL, HSL, and AMPK. Am J Physiol Cell Physiol. 2010;298(4):C961–C971. | ||
Martínez-Agustin O, Hernández-Morante JJ, Martínez-Plata E, Sánchez de Medina F, Garaulet M. Differences in AMPK expression between subcutaneous and visceral adipose tissue in morbid obesity. Regul Pept. 2010;163(1–3):31–36. | ||
Gauthier MS, O’Brien EL, Bigornia S, et al. Decreased AMP-activated protein kinase activity is associated with increased inflammation in visceral adipose tissue and with whole-body insulin resistance in morbidly obese humans. Biochem Biophys Res Commun. 2011;404(1):382–387. | ||
Sanders MJ, Grondin PO, Hegarty BD, Snowden MA, Carling D. Investigating the mechanism for AMP activation of the AMP-activated protein kinase cascade. Biochem J. 2007;403(1):139–148. | ||
Lin F, Ribar TJ, Means AR. The Ca2+/calmodulin-dependent protein kinase kinase, CaMKK2, inhibits preadipocyte differentiation. Endocrinology. 2011;152(10):3668–3679. | ||
Hayes MR, De Jonghe BC, Kanoski SE. Role of the glucagon-like-peptide-1 receptor in the control of energy balance. Physiol Behav. 2010;100(5):503–510. | ||
Nogueiras R, Pérez-Tilve D, Veyrat-Durebex C, et al. Direct control of peripheral lipid deposition by CNS GLP-1 receptor signaling is mediated by the sympathetic nervous system and blunted in diet-induced obesity. J Neurosci. 2009;29(18):5916–5925. | ||
Park H, Kaushik VK, Constant S, et al. Coordinate regulation of malonyl-CoA decarboxylase, sn-glycerol-3-phosphate acyltransferase, and acetyl-CoA carboxylase by AMP-activated protein kinase in rat tissues in response to exercise. J Biol Chem. 2002;277(36):32571–32577. | ||
Kim KY, Baek A, Hwang JE, et al. Adiponectin-activated AMPK stimulates dephosphorylation of AKT through protein phosphatase 2A activation. Cancer Res. 2009;69(9):4018–4026. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.