Back to Journals » Drug Design, Development and Therapy » Volume 13
Id-1 Promotes Reendothelialization In The Early Phase After Vascular Injury Through Activation Of NFkB/survivin Signaling Pathway
Received 14 March 2019
Accepted for publication 2 October 2019
Published 31 October 2019 Volume 2019:13 Pages 3799—3811
DOI https://doi.org/10.2147/DDDT.S208707
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Prof. Dr. Yan Zhu
Wei Li, Dayong Du, Yuntian Li
Department of Cardiology, Beijing 100017, People’s Republic of China
Correspondence: Yuntian Li
Department of Cardiology, 305 Hospital of PLA, Beijing 100017, People’s Republic of China
Tel +86-13811021786
Email [email protected]
Background: Percutaneous coronary intervention (PCI) treatment can benefit patients, but also cause irreversible mechanical damage to the vascular endothelium, ultimately leading to restenosis of the target vessel. Thus, achieving rapid re-endothelialization and restoring the integrity of the vascular endothelium and function plays an important role in inhibiting neointimal hyperplasia and preventing restenosis. Id1 (inhibitor of DNA binding/differentiation factor 1) plays an important role in promoting cell proliferation and angiogenesis.
Study objective: This study aims to investigate the relationship between Id1 and NFκB/survivin signaling pathways and their role in injured vascular repair by establishing a rat carotid balloon injury model.
Methods: The carotid artery model of rat balloon injury was established. The injured common carotid artery was obtained at different time points after vascular injury. RNA and protein were extracted and the mRNA and protein expression levels of Id1, NFκB and survivin were detected in vascular injury. The NFκB blocker BAY 11–7082 and survivin blocker YM155 were used and the effects of Id1, NFκB, survivin mRNA and protein expression, revascularization of blood vessels and neointimal responsiveness after vascular injury were observed in the vascular tissues of Ad-Id1 transfected balloon injury.
Results: Id1, NFκB and survivin were expressed in injured rat carotid arteries. Overexpression of Id1 promoted re-endothelialization of injured vessels through NFκB/survivin signaling pathway, inhibited early vascular endometrial reactive hyperplasia; blocked NFκB the/survivin signaling pathway attenuates the re-endothelialization of Ad-Id1 and the early endothelium of Ad-Id1. Blocking the NFκB/survivin signaling pathway attenuates the re-endothelialization and early reactive hyperplasia of vascular intima of Ad-Id1.
Conclusion: NF-kappa B/survivin signaling pathway may play an important role in Id1 promoting vascular re-endothelialization, inhibiting neointimal hyperplasia and preventing vascular restenosis.
Keywords: inhibitor of DNA binding/differentiation 1, survivin, vascular injury, intimal hyperplasia
Introduction
Coronary atherosclerotic heart disease (CAHD) is widely prevalent both in the middle-aged and elderly patients, as evidenced by the demonstration of coronary plaques in autopsy studies, and is responsible for about one-third or more of all deaths in people over the age of 35.1,2 Percutaneous coronary intervention (PCI) recently has been widely used for treatment of coronary heart disease (CAD) in patients because it can achieve good revascularization, significantly improve myocardial ischemia and prevent the occurrence of end-events such as myocardial infarction.3 However, during the process of PCI, the physical effects of the guide wire and balloon cause the endothelial damage, eventually leading to the atherosclerosis and restenosis after PCI stent implantation.4,5 Thus, accelerated re-endothelialization and restoration of vascular endothelium integrity play an important role in inhibiting neointimal hyperplasia and preventing restenosis.5,6 Endothelial progenitor cells (EPCs) have the ability of proliferation, circulation, and the development of functional progeny to mediate reparative processes in the cardiovascular system.7,8 These EPCs mobilize and differentiate into mature endothelial cell (ECs), and contribute to the neovascularization and re-endothelialization. Recent studies also suggest that in response to the injury of the endothelium caused by tissue ischemia or trauma, EPCs mobilize from the bone marrow into the peripheral blood.9–11
Inhibitor of differentiation or DNA binding 1 (Id1) is a member of the helix-loop-helix (HLH) family of transcription factor which regulates angiogenesis and inflammation. Increasing the expression of Id1 has been shown to promote the proliferation and differentiation of EPCs, promote the repair of damaged blood vessels, and achieve re-endothelialization.12–14 The exact mechanism by which Id1 promotes the repair of damaged blood vessels remains a subject of great debate. Moreover, studies have shown that Id1 is vital to EPCs population formation and angiogenesis.13,14 Inhibition of Id1 in the bone marrow has been linked with significant decreases EPCs.14
Recent studies have revealed that Id1 can inhibit cell apoptosis and promote angiogenesis by activating nuclear factor kappa B (NF-kappa B)/survivin-related signaling pathway, and ultimately promote the proliferation of a variety of cancer cells.15–19 NF-kappa B can regulate the expression of survivin, which is a new member of the inhibitor of apoptosis protein (IAP) family. Survivin is a downstream signal of NF-kappa B which inhibits cell apoptosis and promotes cell proliferation.20,21 It is not only expressed in tumor cells and tissues, but also in brain, myocardium, kidney and other tissues.21 Many studies22,23 have demonstrated that Id1 can activate the NF-κB/survivin signaling pathway, but the mechanisms involved in Id1-mediated activation of NF-κB/survivin signaling cascade are not clear. In this study, we used the rat carotid artery balloon-injury model to investigate the role of Id1 and NF-kappa B/survivin signaling pathways in the re-endothelialization of injured blood vessels.
Materials And Methods
Animals
One to 2 months old healthy male Sprague-Dawley rats were obtained from the Experimental Animal Centre of Third Military Medical University. All animal experiments were performed in accordance with the guidelines of the Animal Research Committee of the Third Military Medical University, Chongqing, People's Republic of China. All study protocols were reviewed and approved by the Animal Ethics Committee of the Third Military Medical University (the decision number:3172016018).
Reagents
The anti-Id1 antibody was purchased from Abcam; anti-survivin, and anti-NFκB/p65 antibodies were obtained from Cell Signaling Technology. BAY 11–7082 (the NFκB blocker), and YM155 (survivin blocker) were obtained from Santa Cruz Biotechnology.
Recombinant Adenoviral Vectors Expressing Id1
An adenovirus (Ad) vector expressing Id1 and Ad-encoding green fluorescent protein (GFP; Ad-GFP) were used as controls and were previously created by our group.24
Rat Carotid Artery Balloon Injury Model
Male Sprague-Dawley (SD) rats (1–2 months old, 180–220 g weight) were fed ad libitum and kept in standard cages under a 12 hr light/dark cycle. They were randomly divided into six groups: 1) sham operation (normal control) group, the rats in the sham group underwent the same operation without carotid artery balloon insertion; 2) simple operation group, using 1.5F catheter for carotid artery balloon injury; 3) Ad-Id1 group, Ad-Id1 was injected into the injured vessel, incubated for 20 mins; 4) Ad-GFP group, Ad-GFP was injected into the injured vessel, incubating for 20 mins. 5) Control group: Ad-Id1+BAY 11–7082 group, ad-Id1 was injected into the injured blood vessel, incubated for 20 mins, subcutaneously injected BAY 11–7082 for 5 consecutive days; 6) Ad-Id1+YM155 group, ad-Id1 was injected into the injured blood vessel locally, incubated for 20 mins, and subcutaneously injected YM155 for 5 consecutive days. The procedures for rat carotid artery balloon injury model and sham-operated rats (Sham) were carried out as described previously.24,25 In brief, SD rats were anesthetized with 60 mg/kg sodium pentobarbital solution. Middle cervical incision was made to completely expose the left common carotid artery, the internal carotid artery, and the external carotid artery. Firstly, the proximal end of the common carotid artery and the distal end of the internal carotid artery were ligated with silk suture to block the blood flow, and then the distal end of the external carotid artery was ligated. The common carotid artery and internal carotid artery were temporarily clamped using a small animal artery clip. Balloon injury was performed into the common carotid artery by a 1.5-French catheter (Edwards Lifesciences) three times as previously described. The viral infusion mixtures 5×108 plaque-forming units of adenoviral vectors, diluted to a total volume of 30 μL in L-DMEM were then instilled into the arterial segment and incubated for 20 mins after the balloon injury. The common carotid artery of the injured side was obtained at 0 hr, 6 hrs, 12 hrs, 24 hrs, 2 d, 7 d, 14 d and 21 d after carotid artery. The number of rats started the test in each of six groups and the number of rats ended the experiment (dropout rate) were listed in the experimental results.
Western Blot Analysis
Rat carotid artery was crushed into powder and resuspended in lysis buffer. Each group of SD rats was sacrificed, and the rat carotid artery related to the experiment was taken out, and the unrelated tissues such as the muscle attached thereto were removed. The carotid arteries were washed with PBS. The blood vessels required for extracting the protein were put into a mortar and liquid nitrogen was added. The mixture was grinded together until the blood vessel became fine powder. The ground tissues were transferred into a 1.5 mL EP tube (pre-added RIPA lysate, Phenylmethanesulfonyl fluoride (PMSF, Sigma), and protease), mixed, and, placed on ice for 30 min. Samples were then centrifuged at high speed (1,2000g) for 30 mins at 4°C, and then transferred the clear supernatant to a new pre-chilled 1.5 mL EP tube for further analysis. The Proteins concentration of cell lysates was estimated by using the Bradford method. An equal amount of cell lysate was separated by a 10–15% SDS-polyacrylamide gel electrophoresis (SDS-PAGE) and electrophoretically transferred onto polyvinylidene difluoride membranes using the iBot Gel Transfer System (Invitrogen). The membranes were blocked with 5% non-fat milk. The membranes were allowed to react with a primary antibody, and the protein bands were visualized via enhanced chemiluminescence (Pierce, USA) as per the manufacturer’s instructions. Quantity One software was used to quantify densitometric signals.
Reverse Transcription-PCR (RT-PCR)
Total RNA from injured arteries was extracted with TRIzol reagent (Invitrogen) following the manufacturer’s instruction. RNA extracted above was reversely transcribed into cDNA using a RT kit (Takara). The primer sequences for PCR were as following:
Id1: Upstream: 5ʹ- GGGCGAAGTGGTGCTTGGTCT −3ʹ
Downstream: 5ʹ- TGCGGTTCTGAGGCAGGGTAGG −3ʹ
NFκB: Upstream: 5ʹ- TCTGTTTCCCCTCATCTTTCC −3ʹ
Downstream: 5ʹ- GCGTCTTAGTGGTATCTGTGCTT −3ʹ
Survivin: Upstream:5ʹ- GACCACCGGATCTACACCTTC −3ʹ
Downstream: 5ʹ- GAGTGCTTCCTATGCTCCTCTAT −3ʹ
Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as the internal control and cDNA was synthesized. The intensity of the bands was quantified by Quantity One.
Evaluation Of Re-Endothelialization In Injured Rat Carotid Artery
Re-endothelialization was quantified in Evans blue-stained carotid arteries at 7, 14 and 21 days after vascular injury. Evans blue was injected into the injured common carotid artery of rats at the dosage of 25 mg/kg through tail vein. Thirty minutesafter the Evans blue injection, the rats were anesthetized with 2% pentobarbital sodium solution at the dosage of 75 mg/kg.
Rats were sacrificed by removing the common carotid artery and cultured in PBS. The common carotid arteries were placed into a petri dish containing PBS and were cut longitudinally by removing excess muscles and connective tissue. These arteries were rinsed again with PBS and then plated on glass slide. Images were captured using a micro-camera to determine the re-endothelialization of the injured vessels. The total area of the vessels and the area of the Evans blue stained area were measured with Lecia Qwin image analysis system. The percentage of re-endothelialization of the injured vessels was calculated by the following equation: (the total area of injured blood vessel - the area of the Evans blue stained area)/the total area of the injured blood vessel.
Detection Of Neointimal Thickness In Injured Vessel By HE Method
Hematoxylin-eosin staining (H-E) was used to detect the changes of neointimal thickness of injured common carotid artery in rats 7, 14 and 21 days after operation. The experimental procedure was as follows: Experimental rats were anesthetized with 2% pentobarbital sodium solution at the dose of 60mg/kg; the injured blood vessels of the rats were surgically removed, and cut into small sections according to requirements. The injured carotid arteries were fixed in 4% formaldehyde for 24 hrs and then embedded in paraffin. Using a slicing machine, transverse slices of the carotid arteries were obtained and stained with hematoxylin-eosin (HE). Inverted microscope was used to analyze of neointimal hyperplasia. The intimal and medial cross-sectional areas were measured by Lecia Qwin image analysis software and the intima/media ratios (I/M) were calculated.
Statistical Analysis
All data are reported as the mean ± standard deviation (SD). Statistical analyses were performed by using SPSS 18.0 statistical software (IBM). Independent-sample Student’s t-test was used to compare the samples between the two groups. One-way analysis of variance (ANOVA) was used for multiple comparisons followed by Tukey’s multiple comparison tests. A P-value ≤0.05 was considered significant.
Result
Establishment Of A Rat Model Of Carotid Artery Balloon Injury
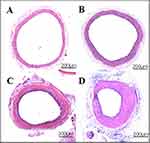
In order to study the injury of artery caused by balloon during PCI and to clarify the mechanism of vascular repair, a rat model of carotid artery balloon injury was established in our laboratory on the basis of long-term experiments, research and advanced experience at home and abroad. The method of establishing the model is simple, and has been widely recognized. Moreover, after long-term test, the experimental model is stable, and there were basically no infections and deaths. In this experiment, the common carotid artery (about 1–2 cm) was punctured by 2F catheter and filled with balloon at 2–3 atm pressure for about 30 s. Then, the balloon was repeatedly inflated three times to achieve complete injury of vascular endothelium. Paraffin sections and hematoxylin-eosin staining were performed on the injured vessels at 7, 14 and 21 days after operation and images were obtained under inverted microscope.
Through observation and analysis, it was found that there was no neointimal hyperplasia in the control side of the carotid artery (Figure1A), and only a single layer of endothelial cells covered the internal elastic membrane. However, in the injured vessels, there were obvious inflammatory reactions, disordered arrangement of cells and increased extracellular matrix. Overall, the lumen showed concentric or eccentric stenosis, with obvious intimal hyperplasia. For simple balloon injured vessels, localized intimal hyperplasia began on the 7th day, and then gradually became apparent (Figure 1B). Obvious inflammatory reaction and unusual arrangement of cells was observed on 14th day (Figure 1C). On the 21st day, obvious stenosis of vascular lumen caused by intimal hyperplasia was observed (Figure 1D).
Injury Of Arteries Can Induce Up-Regulation Of Id1, NF-κB And Survivin Expression
Injury Can Induce Up-Regulation Of Id1, NF-κB And Survivin mRNA Expression
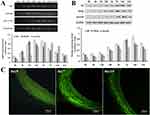
In order to confirm the effect of balloon injury on the relevant signaling factors related to NFkB/survivin signaling pathway, we used RT-PCR to detect the expression of Id1, NFkB and Survivin mRNA in injured blood vessels. Thirty rats started the test in this group and twenty eight ended the experiment (dropout rate 6.67%). The results showed that after 6 hrs of vascular injury, the expression of Id1 began to increase, peaked at 24 hrs, then gradually decreased to normal level. Similarly, NFkB began to increase at 6 hrs, peaked at 12 hrs, and then decreased gradually and survivin began to increase at 6 hrs, peaked at 2 days, and then decreased gradually (Figure 2A).
Injury Can Up-Regulate The Expression Of Id1, NF-κB And Survivin Protein Expression
To confirm the effect of balloon injury on signaling factors related to NFkB/survivin signaling pathway, we used Western blot to detect the expression of Id1, NFkB and survivin protein in injured blood vessels. Thirty rats started the test in this group and twenty nine ended the experiment (dropout rate 3.33%). The results demonstrated that after 12 hrs of vascular injury, the expression of Id1 protein began to increase, peaked at 14 days, and then gradually decreased to normal level. NF-κB protein began to increase after 24 hrs of injury, peaked at 7 days, then gradually decreased. Survivin protein began to increase after 24 hrs of injury, peaked at 14 days, and then gradually decreased (Figure 2B). Then, the accumulation pattern of vascular Id1 was analyzed by immunohistochemistry before and after vascular injury. While weak staining for Id1 was detected in the intima and media of uninjured vessels, enhanced Id1 staining in the media and adventitia was detectable 7 days after balloon injury, and by day 14 Id1 was mainly detected in the media and neointima (Day 0, 7, 14 in Figure 2C). No staining was observed with unspecific IgG, demonstrating the specificity of the Id1 staining. The observed dynamic expression pattern during vascular injury implied a potential role of Id1 in vascular regeneration.
BAY 11-7082 And YM155 Can Down-Regulate The Expression Of NF-ΚB And Survivin In Injured Vessels
BAY 11-7082 And YM155 Can Down-Regulate The Expression Of NF-Kappa B And Survivin mRNA Expression In Injured Vessels
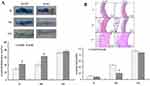
We analyzed the effect BAY 11–7082 and YM155 on the expression NFkB and survivin signaling pathway, respectively, in balloon-injured rats (ad-Id1 was injected into the injured blood vessel, incubated for 20 mins, subcutaneously injected BAY 11–7082 or YM155 for 5 consecutive days), and then on the 7th day of balloon injury, RT-PCR was used to detect the expression of Id1, NFkB and Survivin mRNA in injured vessels. Ten rats started the test in Ad-GFP group and ten ended the experiment (dropout rate 0%). Ten rats started the test in Ad-Id1 group and ten ended the experiment (dropout rate 0%). Ten rats started the test in Ad-Id1+BAY 11–7082 group and eight ended the experiment (dropout rate 20%). Ten rats started the test in Ad-Id1+YM155 group and nine ended the experiment (dropout rate 10%). The results revealed that Id1 is overexpressed after ad-Id1 injection and after BAY 11–7082 intervention, the expression of NF-kappa B and Survivin was down-regulated, while the expression of Id1 remained unchanged. After intervention with survivin blocker YM155, the expression of Survivin mRNA was down-regulated, but the expression of Id1 and NF-kappa B was not affected (Figure 3A).
BAY 11-7082 And YM155 Can Down-Regulate The Expression Of NF-Kappa B And Survivin Proteins In Injured Vessels
To further clarify the effects of balloon injury on the relevant signaling factors in the NFκB/survivin signaling pathway, we used NFκB blocker BAY 11–7082, survivin blocker YM155 to intervene in balloon-injured rats (ad-Id1 was injected into the injured blood vessel, incubated for 20 mins, subcutaneously injected BAY 11–7082 or YM155 for 5 consecutive days), and then on the 14th day of the balloon injury, Western blot was used to detect the expression of Id1, NFkB and survivin protein in injured blood vessels. Ten rats started the test in Ad-GFP group and ten ended the experiment (dropout rate 0%). Ten rats started the test in Ad-Id1 group and nine ended the experiment (dropout rate 10%). Ten rats started the test in Ad-Id1+BAY 11–7082 group and nine ended the experiment (dropout rate 10%). Ten rats started the test in Ad-Id1+YM155 group and ten ended the experiment (dropout rate 0%). The results showed that that Id1 is overexpressed after ad-Id1 injection. And after BAY 11–7082 intervention, the expression of NF-kappa B and Survivin in injured blood vessels was down-regulated, while the expression of Id1 protein remained unchanged. After intervention with survivin blocker YM155, the expression of survivin protein in injured blood vessels was down-regulated, but the expression of Id1 and NF-kappa B protein was not affected (Figure 3B).
Overexpression Of Id1 Can Inhibit Restenosis Of Injured Vessels
Overexpression Of Id1 Promotes Rapid Re-Endothelialization Of Injured Blood Vessels
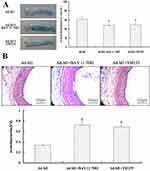
Impaired vascular endothelial integrity and the loss of endothelial protective function is an important aspect of vascular restenosis caused by vascular injury. Therefore, promoting the rapid re-endothelialization of injured blood vessels and maintaining the integrity of vascular endothelium are of great significance to inhibit the intimal hyperplasia and achieve the repair of injured blood vessels. In this experiment, Evans blue staining was used to detect the degree of re-endothelialization of the injured common carotid artery in rats 7, 14 and 21 days after Ad-Id1 transfection. Lecia Qwin image analysis system was used to measure and analyze the effect of over-expression of Id1 on rapid re-endothelialization of injured vessels. Thirty rats started the test in Ad-GFP group and twenty nine ended the experiment (dropout rate 3.33%). Thirty rats started the test in Ad-Id1 group and twenty eight ended the experiment (dropout rate 6.67%). Our results showed that at the 7th day of injury, that the re-endothelialization rate of Ad-GFP group and Ad-Id1 group was 28.35±3.74% and 41.76±5.52%, respectively (P<0.05). At the 14th day of injury, the re-endothelialization rates of Ad-GFP group and Ad-Id1 group were 39.57±4.03% and 62.31±5.19%, respectively (P<0.05). On day 21 after injury the re-endothelialization rates of Ad-GFP group and Ad-Id1 group were 71.68 ± 5.24% and 75.8 ± 4.33%, respectively (P>0.05) (Figure 4A). These results suggest that overexpression of Id1 may promote rapid re-endothelialization of injured blood vessels.
Overexpression Of Id1 Inhibits Early Reactive Intimal Hyperplasia
According to previous studies,26,27 promoting rapid endothelialization of blood vessels is conducive to inhibiting early reactive intimal hyperplasia. In this experiment, we first used hematoxylin-eosin staining (H-E) method to detect the changes of neointimal thickness of injured common carotid artery in rats 7, 14 and 21 days after Ad-Id1 transfection, and then used Lecia Qwin image analysis system to measure and analyze effect of Id1 over-expression on the intimal hyperplasia of injured vessels. Thirty rats started the test in Ad-GFP group and thirty ended the experiment (dropout rate 0%). Thirty rats started the test in Ad-Id1 group and twenty nine ended the experiment (dropout rate 3.33%). The results showed that the ratio of intima to media (I/M) in Ad-GFP group and Ad-Id1 group were 0.157±0.031 and 0.126±0.027, respectively at day 7 (P>0.05) (Figure 4B). At 14 days after injury, the ratio of intimal and media (I/M) in Ad-GFP group and Ad-Id1 group was 0.693±0.032 and 0.299±0.035, respectively (P<0.05) (Figure 4B). At 21 days after injury, the ratio of intimal and medial ratio (I/M) in Ad-GFP group and Ad-Id1 group was 1.365±0.068 and 1.294±0.052, respectively, and there was no significant difference (P>0.05) (Figure 4B). These results suggest that over-expression of Id1 may promote the inhibition of early reactive intimal hyperplasia.
Blocking κB And Survivin Attenuates The Inhibitory Effect Of Ad-Id1 On Restenosis Of Injured Vessel
BAY 11-7082 And YM155 Can Attenuate The Rapid Re-Endothelialization Of Injured Vessels With Id1 Over-Expression
To further clarify the relationship between Id1 over-expression promoting rapid re-endothelialization of injured vessels and the signal pathway related to NFkB/survivin, ad-Id1 was injected into the injured blood vessel, incubated for 20 mins, subcutaneously injected BAY 11–7082 or YM155 for 5 consecutive days. On the 14th day of balloon injury, the Lecia Qwin image analysis system was used to measure the area ratio of Evans blue staining in injured vessels, and the effect of NFκB/survivin signal blockade on the rapid re-endothelialization of Ad-Id1-induced vascular injury was examined. Ten rats started the test in Ad-Id1 group and ten ended the experiment (dropout rate 0%). Ten rats started the test in Ad-Id1+BAY 11–7082 group and nine ended the experiment (dropout rate 10%). Ten rats started the test in Ad-Id1+YM155 group and nine ended the experiment (dropout rate 10%). The results showed that the re-endothelialization rates of Ad-Id1 group, Ad-Id1+BAY 11–7082 group and Ad-Id1+YM155 group were 68.35+5.76%, 47.9+4.17% and 49.7+4.85%, respectively, on the 14th day of injury (P<0.05) (Figure 5A). These results suggest that blockade of NF-kappa B/survivin signaling may attenuate the effect of Ad-Id1 in promoting rapid re-endothelialization of injured vessels.
Blockade Of NF-Kappa B/survivin Signaling Attenuates Early Reactive Intimal Hyperplasia Of Ad-Id1
To further clarify the relationship between the inhibition of early reactive intimal hyperplasia by Ad-Id1 transfection and the signal factors related to NF-kappa B and survivin, we used subcutaneous injection of NF-kappa B blocker BAY 11–7082 and survivin blocker YM155, respectively for 5 consecutive days. On the 14th day of balloon injury, the Lecia Qwin image analysis system was used to measure the thickness of injured intima and to detect the effect of NFkB/survivin signal blockade on the inhibition of early reactive proliferation of injured intima by Ad-Id1. Ten rats started the test in Ad-Id1 group and ten ended the experiment (dropout rate 0%). Ten rats started the test in Ad-Id1+BAY 11–7082 group and ten ended the experiment (dropout rate 0%). Ten rats started the test in Ad-Id1+YM155 group and ten ended the experiment (dropout rate 0%). The results showed that the ratio of intima to media (I/M) in Ad-Id1 group, Ad-Id1 + BAY 11–7082 group and Ad-Id1 + YM155 group were 0.337±0.016, 0.725±0.054, 0.683±0.041, respectively (P<0.05) (Figure 5B). These results suggest that blocking NF-kappa B and survivin-related signaling factors can attenuate the early reactive hyperplasia of the intima of Ad-Id1.
Discussion
Percutaneous coronary stent implantation is one of the most widely accepted diagnostic and therapeutic methods for patients with coronary heart disease. However, due to balloon dilatation and mechanical injury of stent implantation during PCI, vascular endothelial injury will inevitably occur, which will eventually lead to in-stent restenosis (ISR). Although the introduction of drug-eluting stents reduces the incidence of early restenosis to a certain extent, long-term restenosis still occurs because of its special anti-smooth muscle cells (SMCs) proliferation effect which interferes with vascular endothelialization.28 It can be seen from this that restenosis after PCI has become an urgent problem to be solved in interventional therapy of coronary heart disease. Therefore, more in-depth and comprehensive studies on its mechanism and prevention strategies are needed.
In this study, we found that: 1) Id1, NFκB and Survivin were dynamically expressed in injured blood vessels, while BAY 11–7082 and YM155 could down-regulate the expression of NFκB and Survivin in injured blood vessels; 2) Id1 over-expression could promote rapid re-endothelialization of injured blood vessels and inhibit early reactive hyperplasia of intima; 3) BAY 11–7082 and YM155 can attenuate the rapid re-endothelialization and early reactive hyperplasia of vascular intima caused by Id1 over-expression. These results suggest that Id1 may promote the repair of injured vessels through the NFκB/survivin signaling pathway, which is consistent with previous studies.29,30
Id1 widely distributed in mammalian tissue cells, which can bind with basic helix-loop-helix (bHLH) protein, to inhibit the binding of bHLH to DNA, thereby promoting cell proliferation, inhibiting cell apoptosis and differentiation. In this study, we found that the protein expression showed a dynamic change, and the expression in the normal vascular tissue was low. After 12 hrs of vascular injury, the expression began to rise, peaked at 14 days, and gradually decreased to normal level at 21 day. The expression peak of Id1 in injured vessels is different from that of smooth muscle cells, suggesting that Id1 may regulate the proliferation of EPCs and ECs and their roles in the repair of injured vessels before regulating the proliferation of SMCs. Combining with the results of the previous experiments,31,32 Id1 promotes EPCs proliferation through NFκB/survivin signaling pathway, the expression of Id1 in injured blood vessels suggests that Id1 may indeed play an important role in the repair of injured blood vessels.
As a multifunctional nuclear transcription factor widely existing in mammals, NFκB participates in various physiological and pathological processes, such as immunity, inflammation and so on. It plays a central role in signal transduction mediated by various intracellular and extracellular stimuli. Studies have confirmed33,34 that NF-kappa B plays an important role in inflammation and cell proliferation, and can affect cell proliferation and apoptosis through various pathways. In this experiment, we demonstrated that the expression of NFκB protein began to increase 24 hrs after injury, reached its peak at day 7, and then gradually decreased to normal level at 21 days. The expression peak was inconsistent with the proliferation peak of SMCs, suggesting that NF-kappa B is not only involved in the regulation of SMCs proliferation, but also possibly regulated the role of EPCs and ECs in vascular repair after injury.
Survivin, a new member of the inhibitor of apoptosis protein (IAP) family, is a downstream signal of NFκB, which can inhibit cell apoptosis and promote cell proliferation. It is not only expressed in tumor cells and tissues, but also in injured brain, myocardium, kidney and other tissues.21 Our study confirmed that survivin protein began to increase after 24 hrs of injury, peaked at 14 days and gradually decreased to normal level after 21 days, suggesting that survivin not only participated in the regulation of SMCs proliferation, but also possibly regulated the role of EPCs and ECs in the repair of injured blood vessels. The dynamic expression of Id1, NFκB and survivin, and their role in the proliferation of EPCs and the repair of damaged blood vessels can confirm that Id1 may promote the repair of injured vessel through NFκB/survivin signaling pathway before the proliferation effect of SMCs appears.
To further clarify the role of Id1, NFκB and survivin in injured vascular repair, we conducted an in-depth study using the Evans blue staining method and the hematoxylin-eosin staining (H-E) method. The results showed that Ad-Id1 transfected for 7 days, 14 days and 21 days could significantly increase the degree of re-endothelialization of the injured common carotid artery in rats. NFκB blocker BAY 11–7082 and survivin blocker YM155 could attenuate the over-expression of Id1 and promote rapid re-endothelialization of injured vessels. After 14 days of transfection, Ad-Id1 significantly inhibited neointimal proliferation of the injured common carotid artery in rats. NF-kappa B blocker BAY 11–7082 and Survivin blocker YM155 could attenuate Id1 overexpression and promote early reactive intimal hyperplasia. Moreover, the expression trend of Id1, NFκB and Survivin in injured blood vessels is basically consistent with the time-kinetics curve of EPCs mobilization, proliferation and migration in vivo.35,36 It has been unveiled by a copious body of studies37,38 that the expression of circulating EPCs immediately increased after PCI, and at 24 hrs, the level of EPCs increased 2–3 times, reaching peak.
EPCs isolated and cultured in vitro return to the site of vascular injury about 5 days after transplantation and participate in the repair of vascular injury, suggesting that Id1, NFκB and survivin have a certain relationship with the function of mobilization, proliferation and migration of EPCs. It can be preliminarily confirmed that in the early stage of vascular injury, Id1 can affect the mobilization and proliferation of EPCs through NF-kappa B/survivin signaling pathway, accelerate the re-endothelialization of injured vessels, inhibit the proliferation of neointima of common carotid artery on the injured side of rats, and then promote the repair of injured vessels, and the time point of action is different from that of promoting the proliferation of SMCs. This pathway may be a potential molecular mechanism for vascular repair. However, Autieri et al39 used NFκB antisense oligonucleotides that significantly inhibit the proliferation of SMCs in vitro and the proliferation of reactive intima of carotid artery after balloon injury in rats.20
As a key molecule in the regulation of transcriptional genes, NFκB can promote the proliferation of SMCs after being stimulated by stimuli and inflammatory factors, and can promote the secretion of growth factors and adhesion molecules by SMCs, ECs and macrophages (Macrophage, Mφ).40,41 Cytokines, which in turn promote the migration of SMCs to the intima, lead to intimal hyperplasia, which is contrary to our findings. This can be understood and explained in many ways. 1) NF-kappa B not only promotes the proliferation of EPCs, but also promotes the proliferation and migration of SMCs in injured blood vessels. According to the dynamic expression of NFκB in injured blood vessels, it can be seen that in the early stage of vascular injury, NF-kappa B mainly affects the proliferation of EPCs, promotes rapid re-endothelialization of injured blood vessels and inhibits reactive intimal hyperplasia. However, in the middle and late stage of vascular injury, the more prominent role of NF-kappa B is reflected in the influence on the proliferation and migration of SMCs, vascular remodeling and aggravation of intimal hyperplasia. In the early stage of injury, the re-endothelialization of injured vessels was obvious and the intimal hyperplasia was not significant. However, in the late stage, the proliferation of SMCs was dominant and the intimal hyperplasia was obvious due to the poor re-endothelialization. 2) The mechanism of Id1 promoting vascular repair is complex. There must be more than one signaling pathway. There can be a possibility that multiple signaling pathways interact with each other to form a signaling network to promote vascular repair. NF-kappa B is only one of the downstream signaling pathways that Id1 to promotes vascular repair. Its role in promoting the proliferation and migration of SMCs may be interfered and inhibited by other signaling molecules. Therefore, its role in vascular repair is limited and may not play a leading role, but its role cannot be ignored.
Although our experiment preliminarily confirmed that Id1 can affect the mobilization and proliferation of EPCs through NFkB/survivin signaling pathway, accelerate the re-endothelialization of injured vessels, inhibit the proliferation of neointima of injured common carotid artery in rats, and promote the repair of injured vessels, there are still some imitations of our study. 1) Our animal model is only a rat model of balloon injury without atherosclerotic basal lesions. The model is too simple, and the results obtained from this model cannot fully represent the study of vascular injury repair after PCI for coronary heart disease. 2) The experiment lacks the study of Id1 gene silencing, and there is no gene silencing of NFκB and survivin. The role of Id1/NFκB/survivin signaling pathway in injured vascular repair is further studied, so the current results cannot fully confirm the role of Id1/NFκB/survivin signaling pathway in the repair of injured vessels. 3) Current experiments only focus on the Id1/NFkappa B/survivin signaling pathway. Because of the diversity of Id1’s biological functions in vivo, the mechanism of Id1 promoting vascular repair is complex, and cannot be achieved only by Id1/NFκB/survivin signaling pathway. Therefore, more research on signaling molecules is needed to fully understand the mechanism by which Id1 promotes vascular repair in vivo.
Conclusion
Through the current experimental study, we preliminarily confirmed the role of NFκB/Survivin signaling pathway in Id1-induced injury in intimal repair, which provides a new idea and theoretical basis for damaged vascular repair. However, it is still necessary to further study the role and mechanism of Id1 in promoting injured vascular repair through more complete and reasonable experiments.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Benjamin EJ, Virani SS, Callaway CW, et al. Heart disease and stroke statistics-2018 update: a report from the American Heart Association. Circulation. 2018;137:e67. doi:10.1161/CIRCULATIONAHA.117.031422
2. Nemetz PN, Smith CY, Bailey KR, Roger VL, Edwards WD, Leibson CL. Trends in coronary atherosclerosis: a tale of two population subgroups. Am J Med. 2015;129(3):307–314. doi:10.1016/j.amjmed.2015.10.032
3. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40(2):87–165. doi:10.1093/eurheartj/ehy394
4. Buccheri D, Piraino D, Andolina G, Cortese B. Understanding and managing in-stent restenosis: a review of clinical data, from pathogenesis to treatment. J Thorac Dis. 2016;8(10):E1150–E1162. doi:10.21037/jtd.2016.10.93
5. Inoue T, Croce K, Morooka T, Sakuma M, Node K, Simon DI. Vascular inflammation and repair: implications for re-endothelialization, restenosis, and stent thrombosis. JACC Cardiovasc Interv. 2011;4(10):1057–1066. doi:10.1016/j.jcin.2011.05.025
6. Bedair TM, ElNaggar MA, Joung YK, Han DK. Recent advances to accelerate re-endothelialization for vascular stents. J Tissue Eng. 2017;8:2041731417731546. doi:10.1177/2041731417731546
7. Urbich C, Dimmeler S. Endothelial progenitor cells: characterization and role in vascular biology. Circ Res. 2004;95:343–353. doi:10.1161/01.RES.0000137877.89448.78
8. Du F, Zhou J, Gong R, et al. Endothelial progenitor cells in atherosclerosis. Front Biosci. 2012;17:2327–2349. doi:10.2741/4055
9. Lin HH, Chen YH, Yet SF, Chau LY. After vascular injury, heme oxygenase-1/carbon monoxide enhances re-endothelialization via promoting mobilization of circulating endothelial progenitor cells. J Thromb Haemost. 2009;7:1401–1408. doi:10.1111/j.1538-7836.2009.03478.x
10. Siavashi V, Asadian S, Taheri-Asl M, Keshavarz S, Zamani-Ahmadmahmudi M, Nassiri SM. Endothelial progenitor cell mobilization in preterm infants with sepsis is associated with improved survival. J Cell Biochem. 2017;118:3299–3307. doi:10.1002/jcb.25981
11. Briasoulis A, Tousoulis D, Antoniades C, Papageorgiou N, Stefanadis C. The role of endothelial progenitor cells in vascular repair after arterial injury and atherosclerotic plaque development. Cardiovasc Ther. 2011;29:125–139. doi:10.1111/j.1755-5922.2009.00131.x
12. Huang L, Wang H, Yu Y, et al. Inhibitor of DNA binding-1 promotes the migration and proliferation of endothelial progenitor cells in vitro. Mol Cell Biochem. 2010;335:19–27. doi:10.1007/s11010-009-0236-9
13. Pellegrino S, Ferri N, Colombo N, et al. Synthetic peptides containing a conserved sequence motif of the Id protein family modulate vascular smooth muscle cell phenotype. Bioorg Med Chem Lett. 2009;19(22):6298–6302. doi:10.1016/j.bmcl.2009.09.105
14. Mellick AS, Plummer PN, Nolan DJ, et al. Using the transcription factor inhibitor of DNA binding 1 to selectively target endothelial progenitor cells offers novel strategies to inhibit tumor angiogenesis and growth. Cancer Res. 2010;70(18):7273–7282. doi:10.1158/0008-5472.CAN-10-1142
15. Healey MA, Deaton SL, Alder JK, Winnepenninckx V, Casero RA
16. Yap WN, Zaiden N, Tan YL, et al. Id1, inhibitor of differentiation, is a key protein mediating anti-tumor responses of gamma-tocotrienol in breast cancer cells. Cancer Lett. 2010;291:187–199. doi:10.1016/j.canlet.2009.10.012
17. Chen Y, Yang G, Yang ZH. Effect of DNA binding protein inhibitor Id1 on the proliferation of osteosarcoma cells. J Trop Med. 2014;14:71–74.
18. Rothschild SI, Kappeler A, Ratschiller D, et al. The stem cell gene ‘inhibitor of differentiation 1ʹ (ID1) is frequently expressed in non-small cell lung cancer. Lung Cancer. 2011;71:306–311. doi:10.1016/j.lungcan.2010.06.018
19. Hu XM, Lin T, Huang XY, et al. ID1 contributes to cell growth invasion and migration in salivary adenoid cystic carcinoma. Mol Med Rep. 2017;16(6):8907–8915. doi:10.3892/mmr.2017.7744
20. Cui X, Shen D, Kong C, et al. NF-κB suppresses apoptosis and promotes bladder cancer cell proliferation by upregulating survivin expression in vitro and in vivo. Sci Rep. 2017;7:40723. doi:10.1038/srep40723
21. Ghosh G, Wang VY, Huang DB, Fusco A. NF-κB regulation: lessons from structures. Immunol Rev. 2012;246(1):36–58. doi:10.1111/j.1600-065X.2012.01097.x
22. Yang Y, Liou HC, Sun XH. Id1 potentiates NF-kappaB activation upon T cell receptor signaling. J Biol Chem. 2006;281(46):34989–34996. doi:10.1074/jbc.M608078200
23. Peng X, Wang Y, Kolli S, et al. Physical and functional interaction between the ID1 and p65 for activation of NF-κB. Am J Physiol Cell Physiol. 2012;303(3):C267–C77. doi:10.1152/ajpcell.00365.2011
24. Yu Y, Gao Y, Qin J, et al. CCN1 promotes the differentiation of endothelial progenitor cells and reendothelialization in the early phase after vascular injury. Basic Res Cardiol. 2010;105:713–724. doi:10.1007/s00395-010-0117-0
25. Chen SJ, Li H, Durand J, Oparil S, Chen YF. Estrogen reduces myointimal proliferation after balloon injury of rat carotid artery. Circulation. 1996;93(3):577–584. doi:10.1161/01.cir.93.3.577
26. Hristov M, Weber C. Ambivalence of progenitor cells in vascular repair and plaque stability. Curr Opin Lipidol. 2008;19(5):491–497. doi:10.1097/MOL.0b013e32830dfe33
27. Patel SD, Waltham M, Wadoodi A, Burnand KG, Smith A. The role of endothelial cells and their progenitors in intimal hyperplasia. Ther Adv Cardiovasc Dis. 2010;4:129–141. doi:10.1177/1753944710362903
28. Denes L, Entz L, Jancsik V. Restenosis and therapy. Int J Vasc Med. 2012;2012:406236.
29. Flamini V, Jiang WG, Lane J, Cui YX. Significance and therapeutic implications of endothelial progenitor cells in angiogenic-mediated tumour metastasis. Crit Rev Oncol Hematol. 2016;100:177–189. doi:10.1016/j.critrevonc.2016.02.010
30. Li W, Wang H, Kuang CY, et al. An essential role for the Id1/PI3K/Akt/NFkB/survivin signalling pathway in promoting the proliferation of endothelial progenitor cells in vitro. Mol Cell Biochem. 2012;363(1–2):135–145. doi:10.1007/s11010-011-1166-x
31. Wang H, Yu Y, Guo RW, et al. Inhibitor of DNA binding-1 promotes the migration and proliferation of endothelial progenitor cells in vitro. Mol Cell Biochem. 2010;335(1–2):19–27. doi:10.1007/s11010-009-0236-9
32. Yu Y, Liang Y, Yin C, et al. Inhibitor of DNA-binding 1 promotes endothelial progenitor cell proliferation and migration by suppressing E2-2 through the helix-loop-helix domain. Int J Mol Med. 2016;38(5):1549–1557. doi:10.3892/ijmm.2016.2734
33. Lawrence T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb Perspect Biol. 2009;1:a001651. doi:10.1101/cshperspect.a001651
34. Liu T, Zhang L, Joo D, Sun SC. NF-κB signaling in inflammation. Signal Transduct Target Ther. 2017;2:17023. doi:10.1038/sigtrans.2017.23
35. Bonello L, Basire A, Sabatier F, Paganelli F, Dignat-George F. Endothelial injury induced by coronary angioplasty triggers mobilization of endothelial progenitor cells in patients with stable coronary artery disease. J Thromb Haemost. 2006;4:979–981. doi:10.1111/j.1538-7836.2006.01858.x
36. Marboeuf P, Corseaux D, Mouquet F, Van Belle E, Jude B, Susen S. Inflammation triggers colony forming endothelial cell mobilization after angioplasty in chronic lower limb ischemia. J Thromb Haemost. 2008;6:195–197. doi:10.1111/j.1538-7836.2007.02783.x
37. Gao M, Yao Q, Liu Y, Sun F, Ma Y, Sun G. Association between mobilization of circulating endothelial progenitor cells and time or degree of injury from angioplasty in patients with exertional angina: a prospective study. Exp Ther Med. 2015;10(2):809–815. doi:10.3892/etm.2015.2571
38. Koutroumpi M, Dimopoulos S, Psarra K, Kyprianou T, Nanas S. Circulating endothelial and progenitor cells: evidence from acute and long-term exercise effects. World J Cardiol. 2012;4(12):312–326. doi:10.4330/wjc.v4.i12.312
39. Autieri MV, Yue TL, Ferstein GZ, Ohlstein E. Antisense oligonucleotides to the p65 subunit of NF-kB inhibit human vascular smooth muscle cell adherence and proliferation and prevent neointima formation in rat carotid arteries. Biochem Biophys Res Commun. 1995;213:827–836. doi:10.1006/bbrc.1995.2204
40. Bentzon JF, Weile C, Sondergaard CS, Hindkjaer J, Kassem M, Falk E. Smooth muscle cells in atherosclerosis originate from the local vessel wall and not circulating progenitor cells in ApoE knockout mice. Arterioscler Thromb Vasc Biol. 2006;26:2696–2702. doi:10.1161/01.ATV.0000247243.48542.9d
41. Doran AC, Meller N, McNamara CA. Role of smooth muscle cells in the initiation and early progression of atherosclerosis. Arterioscler Thromb Vasc Biol. 2008;28:812–819. doi:10.1161/ATVBAHA.107.159327
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.