Back to Journals » Drug Design, Development and Therapy » Volume 9
Gamma scintigraphic evaluation of floating gastroretentive tablets of metformin HCl using a combination of three natural polymers in rabbits
Authors Razavi M, Karimian H, Yeong CH, Chung LY , Nyamathulla S, Noordin MI
Received 8 April 2015
Accepted for publication 21 May 2015
Published 6 August 2015 Volume 2015:9 Pages 4373—4386
DOI https://doi.org/10.2147/DDDT.S86263
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Shu-Feng Zhou
Mahboubeh Razavi,1 Hamed Karimian,1 Chai Hong Yeong,2 Lip Yong Chung,1 Shaik Nyamathulla,1 Mohamed Ibrahim Noordin1,3
1Department of Pharmacy, Faculty of Medicine, University of Malaya, 2Department of Biomedical Imaging and University Malaya Research Imaging Centre, Faculty of Medicine, University of Malaya, 3Center for Natural Products and Drug Discovery (CENAR), Department of Chemistry, Faculty of Science, University of Malaya, Kuala Lumpur, Malaysia
Abstract: The present research was aimed at formulating a metformin HCl sustained-release formulation from a combination of polymers, using the wet granulation technique. A total of 16 formulations (F1–F16) were produced using different combinations of the gel-forming polymers: tamarind kernel powder, salep (palmate tubers of Orchis morio), and xanthan. Post-compression studies showed that there were no interactions between the active drug and the polymers. Results of in vitro drug-release studies indicated that the F10 formulation which contained 5 mg of tamarind kernel powder, 33.33 mg of xanthan, and 61.67 mg of salep could sustain a 95% release in 12 hours. The results also showed that F2 had a 55% similarity factor with the commercial formulation (C-ER), and the release kinetics were explained with zero order and Higuchi models. The in vivo study was performed in New Zealand White rabbits by gamma scintigraphy; the F10 formulation was radiolabeled using samarium (III) oxide (153Sm2O3) to trace transit of the tablets in the gastrointestinal tract. The in vivo data supported the retention of F10 formulation in the gastric region for 12 hours. In conclusion, the use of a combination of polymers in this study helped to develop an optimal gastroretentive drug-delivery system with improved bioavailability, swelling, and floating characteristics.
Keywords: gastroretentive drug delivery, gamma scintigraphy, sustain-release study, salep, tamarind seed, xanthan
Introduction
Drug delivery refers to different methods and approaches that are used to deliver a pharmaceutical dosage form to the body in order to achieve the desired therapeutic outcome.1 The concept of drug delivery is highly related to the dosage form and the route of administration. The oral route is the most accepted route of drug administration. Oral drug release, absorption, distribution, and elimination can be modified through drug-delivery technologies that help to achieve formulations with better efficacy and patient convenience.2 Controlled-release techniques can be used to decrease the frequency of drug intake by increasing drug-retention time and gradual release of the active ingredient in the stomach.3
Gastroretentive drug-delivery systems are a group of approaches that are used to prolong the gastric retention time of drugs and hence the absorption window in the upper part of the gastric region for better local or systemic effects. Gastroretentive dosage forms are formulated in such a way that they can remain in the gastric region for a prolonged period of time in order to achieve complete drug release and the optimum bioavailability.4 The floating gastroretentive drug-delivery system is designed based on a low-density system which has the ability to float within a short time and remain buoyant in the upper part of the gastric region for several hours, during which the drug is released gradually at a controlled rate.5
Pharmaceutical dosage forms contain the pharmacologically active drugs in combination with inert materials known as pharmaceutical excipients. Excipients provide a range of functions such as lubricity and flow ability enhancement which improve compressibility and compatibility of dosage forms.6 Natural polymers contain polysaccharides, which swell in the presence of aqueous media and form viscous solutions.7 Currently, pharmaceutical industries prefer to use plant material instead of chemical and synthetic excipients as many of the synthetic excipients are toxic, hence posing problems for drug approval by regulatory authorities.8 Natural polymers have advantages over the synthetic ones, as they are chemically inert, biodegradable, cheaper, sustainable, renewable resources, and have fewer adverse effects.9 Pharmaceutical polymers have different applications: they can be used as binders in tablet preparation; they can be used to control the viscosity of liquids, suspension, and emulsions; they can be used to disguise and mask the unpleasant taste of drugs; they can be used to improve the drug stability for longer periods; and they can be used to control and modify the release patterns in dosage forms.9
In the current study, metformin HCl was examined as a model drug. Metformin HCl is an antihyperglycemic agent which is widely used in the treatment of non-insulin dependent diabetes mellitus (type II). The oral delivery of metformin HCl has a low systemic bioavailability due to incomplete absorption of metformin HCl.10 Metformin HCl has an absorption window in the upper part of the gastrointestinal tract. It is mainly absorbed in the upper part of the intestine, but poorly absorbed in the stomach, jejunum, and ileum, and is excreted in an unchanged form through the feces.11 Moreover, frequent administration of metformin HCl leads to gastrointestinal side effects such as nausea, diarrhea, vomiting, and loss of appetite.12 Hence, in order to achieve a maximum therapeutic effect with this drug, it would be beneficial to develop a floating gastroretentive matrix tablet with a prolonged gastric retention time and gradual release of the drug at the absorption site.
To develop a formulation with the optimum floating and sustainability properties, different combinations of three polymers – tamarind kernel powder (TKP), salep, and xanthan – were used. These polymers were selected based on their good swelling and gel-forming properties. TKP is a biodegradable plant polysaccharide obtained from the seeds of Tamarindus indica Linn. (Leguminosae family).13 It is a hydrophilic polymer with gelling, thickening, suspending, and emulsifying properties.14 Salep is a hydrocolloid powder from dried roots/tubers of Orchis morio var. mascula, (Orchidaceae family). The palmate tubers of salep used in this study contained 48% mucilage. Salep is used as a gelling, stabilizing, and thickening agent in ice cream production.15 In this study, TKP and salep were used in combination with xanthan (another well-known polymer) in drug delivery.
The aim of the present study was to develop a new gastroretentive formulation with improved cohesion and gradual release of metformin HCl in the stomach and upper gastrointestinal tract. The main objective was to come up with matrix tablets which have short floating lag time (FLT), good floating and swelling abilities, and with close to 100% drug release within 12 hours. Matrix tablets were prepared using a combination of TKP, salep, and xanthan by the wet granulation method. This was followed by in vitro and in vivo studies. The effects of combining natural gums on gel-forming, floating properties, and release characteristics of metformin HCl were also evaluated.
Materials and methods
Materials
Metformin HCl BP 98 (crystalline white powder, batch number 2011MP0244 with purity of 99.61%) was provided by Euro Chemo-Pharma Sdn. Bhd (Selangor, Darul Ehsan, Malaysia). The palmate tubers of O. mascula were procured from the Agricultural Research Center, Boushehr, Iran with voucher number of 4310/266/1. The seeds of tamarinds were kindly offered by Herbs Research Laboratory of Amritum Bio-Botanica (Indore, India). Sodium bicarbonate (NaHCO3, M =84.01 g/mol), microcrystalline cellulose ((C6H10O5)n), pure xanthan, potassium chloride (M =74.56 g/mol), hydrochloric acid 37% (M =36.46 g/mol), magnesium stearate, and talc were provided from R&M Marketing, Essex, UK.
Collection and preparation of powders
The collected tamarind seeds contained both intact and broken portions. The intact seeds were separated from the broken ones and the pulp was removed. Then, the seeds were fed into a cutting mill machine (Retsch GmbH, Haan, Germany) and the machine was run several times. The dark cover of the size-reduced seeds was removed using a sharp knife and the dehusked size-reduced kernels were ground using a blender into a fine TKP. Well-dried palmate tubers of salep were fed into a cutting mill machine, then the size-reduced particles were blended to obtain a fine powder of salep. TKP and salep powders were then passed through a sieve (size number 100) and stored in well-closed containers until further use. In previous studies, the polymer was physicochemically characterized and the results indicated the excellent swelling, viscosity, and gel-forming properties of salep; this polymer also had the ability to sustain release, and TKP was able to form a matrix gel due to good swelling and gel-forming properties.14,15
Preparation of the floating gastroretentive matrix tablets of metformin HCl
In the previous studies, the physicochemical characterization of TKP and salep demonstrated the abilities of these powders to individually sustain drug release.14,15 In the present study, we compared different combinations of three natural polymers in sustaining drug release.
Gastroretentive matrix tablets of metformin HCl were prepared using the wet granulation method. Each tablet contained 500 mg of the active drug (metformin HCl) in combination with release-retarding polymers (such as TKP, xanthan, and salep), the gas generating agent (NaHCO3), microcrystalline cellulose as a diluent, magnesium stearate as a lubricant, and talc as a glidant.16 The compositions of the 16 formulations designed in this study are shown in Table 1. All the ingredients, except magnesium stearate and talc, were initially mixed using the geometrical method. The mixed powders were granulated using a binder solution, and a pestle and mortar were used to mix the ingredients well. The binder solution (1% w/v) had been prepared using 33.33 mg of each polymer (salep, xanthan, and TKP) dissolved in 10 mg distilled water. The wet granules were then passed through a number 10 sieve and dried at 45°C for 25 minutes. The dried granules were then passed through a number 18 sieve and then mixed with talc and magnesium stearate. Finally, the exact amount of each mixture was weighed and fed manually into the die of a single-punch tableting machine (MTCM-I Manual Tablet Compaction Machine; GlobePharma, North Brunswick, NJ, USA) with a round concave 12 mm punch and compressed at compression pressure of 2,200 psi to form tablets.
Physical evaluation of the prepared matrix tablets
Weight variation, friability, hardness, FLT, total floating time (TFT), and drug content uniformity of the prepared tablets were evaluated.
Weight variation test for prepared tablets
Twenty tablets were selected randomly and the individual weight of each tablet estimated using an analytical balance (Model MS-S/MS-L, Greifensee, Switzerland). The 20 tablets were then weighed together, and the average weight calculated. Individual tablet weights were compared with the average weight of the 20 tablets in order to determine the weight variation.17
Diameter and thickness of prepared tablets
A tablet hardness tester (Model 6D; Dr Schleuniger Pharmatron, Solothun, Switzerland) was used to measure the diameter and thickness of ten tablets from each formulation. The results were recorded and compared among the different formulations.18
Hardness of prepared tablets
The hardness of ten tablets from each formulation were measured using a tablet hardness tester (Model 6D; Dr Schleuniger Pharmatron, Solothun, Switzerland). The results were recorded and compared among the formulations.19
Friability of prepared tablets
Ten tablets were weighed and the weight recorded as W1. The tablets were then placed in a friability tester (Model Tar10; Erweka, Ensenstam, Germany) that was adjusted to rotate at 25 rpm. After 4 minutes (100 rotations), the tablets were de-dusted and weighed again (W2).20 The percentage loss in tablet weight (friability) was determined using the following formula:
Friability = (W1 - W2)/W1 ×100 | (1) |
The acceptable range is the percentage below 1% friability.
Drug content of prepared tablets
From each formulation, three tablets were randomly selected and a mortar and pestle were used to convert tablets into powder form. One hundred milligrams of the crushed tablets of each batch was weighed, added to a volumetric flask, and the volume brought up to 100 mL using distilled water. The solution was passed through filter paper and absorbance of the solution was determined at 233 nm using ultraviolet spectrophotometer (Lambda 35 UV Vis Spectrometer; PerkinElmer Inc., Waltham, MA, USA). The percentage of the drug content was calculated.21
FLT and TFT of the prepared tablets
FLT is the time duration taken by a tablet to emerge onto the surface of the medium, while TFT is the duration of time that a tablet remains continuously floating on the surface of the medium. These two parameters are used to measure a tablet’s buoyancy. Three randomly selected tablets from each formulation were placed in a beaker containing 500 mL of the buffer media at pH 1.2, and the FLT and TFT were determined.22
Evaluation of in vitro drug release from the prepared tablets
The release of the metformin HCl from the floating matrix tablets was evaluated using a United States Pharmacopeia dissolution apparatus II, paddle type (Copley Scientific Limited, Nottingham, UK). A tablet was placed inside the dissolution vessel containing 900 mL of 0.1 N HCl (pH 1.2) at 37°C±0.5°C and stirred at a speed of 50 rpm. Five milliliters of sample was withdrawn at 0, 0.5, 1, 1.5, 2, 3, 4, 5, 6, 7, 8, 9, 10 and 12 hour time intervals and each time the same amount of fresh media was used to maintain a constant volume. The amount of drug release was determined using a spectrophotometer (Lambda 35 UV–Vis Spectrometer; PerkinElmer Inc.) at a wavelength of 233 nm against 0.1 N HCl as a blank. The amount of drug in each sample was calculated using a standard calibration curve. The in vitro drug release was carried out in triplicate for each batch of tablets. Release profiles of the designed formulations were compared with the commercial formulation (C-ER). Similarity and difference factors were calculated using appropriate formulas. The in vitro release data were analyzed using mathematical models representing: 1) first order; 2) zero order; 3) Hixson- Crowell; and 4) Higuchi’s equations.15
Fourier transform infrared (FTIR) spectroscopy of the optimum formulation
To investigate the possible interactions between the tablet’s ingredients, the powder of crushed tablets, pure polymers, and the active drug were analyzed using a FTIR spectrophotometer (model Nicolet IS10; Thermo Fisher Scientific, Waltham, MA, USA). A small amount of each sample was mixed with potassium bromide and pressed to form a thin pellet. FTIR analysis was performed using the pellets.14
X-ray diffraction (XRD) analysis of the optimum formulation
The XRD spectra of the optimum formulation was performed using an XRD diffractometer (BTX324 model; Inxitu, Suite A Mountain View, CA, USA). Pellets were formed by pressing the dry sample powder, and their diffractograms were recorded using Cu-Kα radiation (40 kV, 60 mA). The speed of the machine was fixed at 2°/minute and a chart speed of 2°/2 cm per 2θ was used to measure the XRD of the optimum formulation tablets (powder), metformin HCl, and the polymer used in the optimum formulation.15
Differential scanning calorimetry (DSC) of the optimum formulation
Small amount of drugs, polymers, and powder of crushed tablets were loaded into an aluminum pan, which was then crimped to seal the pan. The sample was analyzed by using a differential scanning calorimeter (DSC-6; PerkinElmer Inc.) with a heating rate of 10°C/minute and nitrogen purge gas at a flow rate of 20 mL/minute.23
In vivo evaluation of gastric retention ability of optimum formulation
Radiolabeling of F10 formulation
Radioactive samarium-153 (III) oxide (153Sm2O3) with half-life of 46.3 hours was used to radiolabel the F10 formulation. Approximately 10 mg of stable 152Sm2O3 was weighed for each tablet and mixed with the tablet ingredients during tablet preparation. The tablets were then sent for neutron activation at a nuclear reactor facility. FLT and the in vitro drug release of each tablet was calculated before and after tablet neutron activation.
Neutron activation of F10 tablets for in vivo study
The neutron activation of the tablets was carried out at the nuclear reactor facility at Malaysian Nuclear Agency (Bangi, Selangor, Malaysia). This facility has a 250 kW open pool-type research reactor (Triga Mark II; General Atomics, San Diego, CA, USA), which utilizes uranium zirconium hydride assembly with low-enriched uranium (20 wt % 235U) fuel source. Each tablet was heat-sealed into an individual polyethylene vial and packed into a polyethylene ampoule (commonly known as a “rabbit”). The ampoule was then delivered to the reactor core by a pneumatic transport system. The tablet was irradiated in a neutron flux of 1×1013 cm−2·s−1 for 5 minutes to achieve nominal radioactivity of 5 MBq at 48 hours after neutron activation. Gamma spectroscopy was carried out 24 hours after neutron activation using a coaxial, p-type, 40% relative efficiency germanium detector (Canberra, Meriden, CT, USA) and gamma spectrum analysis software (Genie TM 2000 Ver. 3.2; Canberra) to detect any radioactive impurities. The tablets were then kept in a radioactive storage room until analysis to allow for the decay of the unwanted activated byproducts.24
Gamma imaging in rabbits
Approval for animal use was obtained from the Institutional Animal Care and Use Committee (IACUC) of the Faculty of medicine, University of Malaya (2013-12-03/PHAR/R/MR). The gamma scintigraphy of control and test formulations were performed in six healthy male New Zealand White rabbits weighing between 2.5 and 4 kg. The oral dose of metformin HCl was determined based on a previous study which showed that metformin is safe in rabbits at doses of up to 600 mg/kg/day.25 To prevent any hypoglycemic complication to the rabbits, the metformin HCl dose was fixed at 250 mg. The same ingredients used in in vitro F10 formulation were used in the in vivo formulation, and the quantity of each in vivo ingredient was half of the one used in the in vitro formulation (Table 2).
  | Table 2 Composition of test formulation and a control formulation of F10 prepared for in vivo study (mg) |
The rabbits used in this study were not under any medication and did not have history of any medical disorders. All gastroretentive matrix tablets were orally administered using a pill dispenser with the aid of 30 mL of drinking water. The rabbits were allowed to drink freely during the imaging period. Scintigrams of test preparation were recorded using a dual-detector single-photon emission computed tomography (SPECT)–computed tomography (CT) system (BrightView XCT; Philips, Eindhoven, The Netherlands) mounted with a low-energy high-resolution collimator. At predetermined time intervals of 0, 1, 2, 4, 6, 8, 10, and 12 hours after drug administration, the rabbits underwent anterior whole-body static scintigraphy for 15 minutes. During the imaging procedure, the rabbits were restrained in a towel to decrease movement.
The CT images were acquired using 120 kVp, 247 mAs, and 1 mm slice thickness at 512×512 matrix size, followed by SPECT imaging (128×128 matrix size, 64 angulations for 360°, 15 seconds per angulation).
Image analysis
A region of interest (ROI) (a circle with 50 mm diameter) was drawn manually around the 3D SPECT–CT images of the stomach. No correction for scatter was performed. The ROI position was fixed on every image at three different planes, ie, sagittal, coronal, and axial planes. Quantification of the ROI was performed with a dedicated software. The radioactivity in the ROI at 0 hour was taken as the control with 100% 153Sm activity and 0% drug release. Since during tablet preparation the 10 mg 153Sm was well mixed and granulated with the rest of the ingredients, the release of 153Sm was considered as the metformin HCl release.
Results and discussion
The wet granulation method was used to prepare floating matrix tablets of metformin HCl using a combination of natural polymers.
Post-compression parameters for floating matrix tablets
The tablets’ colour was dependent on the type and concentration of polymer used in the formulation. Tablets with a high concentration of salep were yellowish while those with a lower concentration of salep were whitish. All the tablet were smooth with round concave shapes on both sides and no cracks were observed on tablets from any formulation.
The mean diameter of the tablets was 11.7 mm while the mean thickness was 6.1 mm. The mean hardness ranged from 113–191 N. The friability percentage of the formulations were less than 1% and no capping, cracking, cleavage, or breakage of tablets was noticed after the friability test. The results of the hardness and friability tests indicated that the tablets had sufficient strength to withstand physical abrasion and mechanical resistance (Table 3).26,27
  | Table 3 Evaluation of floating matrix tablet prepared using different combinations of natural polymers9 |
The drug content analysis showed that the tablets had a drug content that ranged from 97.8% to 98.9% of the tablets. This range meets the standard pharmacopoeia requirements. The mean weight of tablets ranged between 758 and 760 mg, which is within the United States Pharmacopeia acceptable range of ±5% of tablets weighing 324 mg or more (Table 3).28
In vitro buoyancy test
In the present study, a floating system was achieved using NaHCO3 in an optimized concentration of 100 mg as a gas-forming agent to provide the shortest possible FLT and floating duration of up to 24 hours. Floating matrix tablets contained the sodium bicarbonate-induced carbon dioxide formation in the presence of dissolution medium. The generated carbon dioxide will be trapped in the strong matrix layer formed by gel-forming agents and this causes density reduction of the tablet. As the density of the tablet decrease below 1 g/mL, the tablet becomes buoyant.
The FLT for prepared formulations was between 3 and 31 minutes. The formulation containing the highest concentration of salep had the shortest FLT, followed by the formulation containing the highest concentration of xanthan and TKP, respectively. In the previous study, the high swelling index, viscosity, and gel-forming ability of salep was proved;15 since the hydrophilic and swellability properties of the polymer affects the FLT,29 we can conclude that the swelling and the gel-forming ability of salep was stronger when compared to that of the other two polymers. It was observed that the increase in amount of salep and xanthan leads to a decrease in the cumulative percentage of drug released. Hence, as the amount of salep was increased, the FLT decreased, indicating that a high concentration of salep is desirable to achieve short FLT. But high concentration of TKP was not successful in giving sufficient strength to the matrix to prolong drug release and decrease the FLT.
Figure 1 represents the FLT of the prepared tablets. The F8 formulation had the shortest FLT (3 minutes) and F4 had the longest FLT (31 minutes).
  | Figure 1 Buoyancy of formulated metformin HCl floating tablets (n=3). |
In vitro drug-release kinetics
The rate and mechanism of the drug release are dependent on the properties of the polymer which confers its ability to sustain the matrix of the tablet. The mechanism of drug release, which can be diffusion, swelling, matrix release, or drug leaching, is related to the polymer used in the formulation. The release pattern of a formulation prepared using a combination of polymers usually produce better results as it follows multiple mechanisms of release.
The effects of different amounts and combinations of polymers on drug-release properties are shown in Figure 2A and B. Formulations containing salep showed rapid hydration and higher release in the first 1 hour; however, the high swelling and gel-forming ability of salep formed strong and viscous gel gradually. This gel provides a strong protective matrix for controlling the release process. The matrix formed by salep could sustain the release for 12 hours in the F1, F5, F8, and F10 formulations. Formation of a relatively strong matrix in the F8 formulation, which contained the highest concentration of salep (90 mg) in combination with 5 mg of xanthan and 5 mg of TKP, decreased the porosity and drug release. As a result, only 89.9% of the drug from the F8 formulation was released in 12 hours. In the F3, F9, and F15 formulations, the high viscosity and gel-forming ability of xanthan prevented the initial burst effect of drug release, but the release was dependent on the other two polymers. In the F2 formulation which contained the highest concentration of TKP, drug release was faster than in the formulations which contained the highest amounts of salep (F9 and F15). The F3 formulation which contained 90 mg xanthan released 86.9% of the drug over 12 hours. This could be due to the viscosity of the gel matrix which could result in a reduction of the effective diffusion coefficient of the drug and hence a decreased drug release into the dissolution medium. As shown in Figure 2A and B, the F1, F2, F5, F9, F10, F11, and F15 formulations, which contained higher concentrations of salep and xanthan in combination with lower concentrations of TKP, exhibited constant drug release up to 12 hours. As the concentration of salep and xanthan gum increased, the drug-release rate decreased and high concentrations of TKP facilitated the drug release in the combination state. Results from a previous study showed that TKP alone cannot form a strong matrix, but can be beneficial in a sustained-release formulation if combined with other polymers.14 In this study, the F4 and F7 formulations, which contained the highest concentrations of TKP, achieved complete drug release at only 10 hours. The use of TKP in combination with salep and xanthan was found to be beneficial and useful in achieving a more than 90% release in 12 hours (Figure 3). In this study, the F10 formulation was found to be the optimum formulation as it had shortest FLT and could achieve a 95% drug release in 12 hours. When the result was compared with the other formulation, F10 also had the highest similarity (F2=55%) with the commercial formulation. The drug-release profiles of F1–F16 formulations are shown in Figure 2A and B.
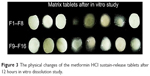  | Figure 3 The physical changes of the metformin HCl sustain-release tablets after 12 hours in vitro dissolution study. |
The correlation coefficient values (R) obtained from different kinetics models were also analyzed. When the highest correlation coefficient values were considered, the released data for most of the formulations fitted better with zero order and Higuchi models. The optimum formulation (F10) fit the zero order kinetic (R=0.993).
FTIR spectroscopy of the F10 formulation
The F10 formulation and its ingredients were characterized using FTIR spectra. The (C-O) functional group was measured at 950–1,200 cm−1 (finger print region). Salep’s peaks were obtained at 3,340, 1,670, and 1,060 cm−1, xanthan’s peaks were detected at 3,360, 2,910, 1,610, and 1,070 cm−1, and the peaks representing TKP were detected at 3,350, 2,930, 1,650, 1,040 cm−1, whereas metformin HCl peaks were detected at 3,370, 3,310, 3,170, 1,570, 1,480, 1,070, 938, and 737 cm−1. The peaks for the F10 formulation were detected at 3,370, 3,310, 3,160, 1,570, 1,070, and 938 cm−1 (Figure 4). No new peaks were detected in the F10 formulation; in comparison to the pure ingredients, this indicates that no changes occurred to the chemical structure of ingredients during tablet preparation and compression.
  | Figure 4 The FTIR spectra of optimized formulation, pure drug, and polymers. |
XRD analysis of the F10 formulation
The X-ray diffractograms of metformin HCl, salep, xanthan, TKP, and the F10 formulation are presented in Figure 5. The X-ray diffractogram of the pure drug displayed characteristic peaks at 12.7, 17.8, 22.35, 23.3, 24.55, 27.4, 28.5, 29.5, 31.4, 35.85, and 37.35 angles (°2θ). These sharp peaks represent the drug crystallinity. Salep and xanthan showed characteristic peaks at 20.4 and 20.2 angles (°2θ), respectively; the broad peaks of these polymers indicate their amorphous nature. In the X-ray diffractogram of the F10 formulation, drop in crystallinity of the metformin HCl was reflected by a decrease in the peak’s intensity. No missing peaks were detected in the F10 formulation. The insignificant differences detected in the XRD can be due to physical interactions that occurred while the tablet was prepared and compressed.
  | Figure 5 The X-ray diffraction analysis of optimum formulation, pure drug, and polymers. |
DSC of F10 formulation
DSC thermogram of metformin HCl indicated a sharp peak at 234.8°C, indicating the drug meting point and an anhydrous crystalline structure of molecule with high purity. Meanwhile, broad peaks were observed at 116.55°C, 112.15°C, and 115.00°C from the DSC thermogram of pure salep, TKP, and xanthan, respectively, representing that the melting transition of powders. The melting endotherm of polymers are a broad peak demonstrating a polymer with components of multiple thermal characteristics. From the DSC thermograms of crushed F10 tablet, peaks corresponding to the melting endotherm of drug and polymers were shown. There was no significant change in the melting point of the drug and powder in crushed tablets (Figure 6). The results shows no interaction between famotidine and the polymers. In additional, the drug and polymers were compatible.
  | Figure 6 DSC thermogram of pure metformin HCl, TKP, xanthan, salep, and powder from crushed tablet of F10. |
In vivo gamma scintigraphy study
The in vivo drug function behavior was monitored using gamma scintigraphy after labeling the dosage forms with radionuclide elements and feeding them into rabbits. The gamma scintigraphy technique is one of the most common methods for investigating the in vivo gastrointestinal performance of dosage forms. Radioactive 153Sm2O3 was found to be ideal for use in the controlled-release formulations.30 153Sm2O3 is a radionuclide with a 46.3-hour physical half-life, which decays by emitting beta and gamma radiations. In the last two decades, 153Sm2O3 has been used for assessing pharmacoscintigraphic study because of its availability, suitable gamma energies, easy production, and minimal radiation exposure.31 153Sm2O3 is chemically stable and it cannot be absorbed into the GI tract and blood plasma because it is water insoluble, and it will be excreted from the body through feces and eventually decays to a stable nuclide (153Eu), hence special radioactive waste management is not required.30
In controlled-release studies, it is important to use unstable isotopes instead of readily radioactive markers for the following reasons: safety during the preparation of tablets; minimizing the in vitro and in vivo error when using the equipment and during tablet preparation; and for the uniform distribution of the marker with the ingredients. By achieving a uniform distribution of 153Sm2O3 as the marker in the dosage form, the release of 153Sm2O3 can be considered as the release of the drug.
After samarium was activated in thermal neutron flux to 153Sm2O3 by neutron capture, emitted radiations from 153Sm2O3 were captured by gamma cameras.
The FLT and the in vitro drug release of the size-reduced F10 containing 153Sm2O3 was measured before and after tablet activation. The test was performed under the same conditions as in the in vitro studies. The results indicated that size reduction and 153Sm2O3 did not affect the FLT and there was more than an 80% similarity between the drug release result of tablets before and after size reducing and activating.
The dosage form was exposed to the neutron flux for a very short period of time (300 seconds). The short time is important for maintaining the tablet’s characteristics and integrity, and for preventing tablet degradation and polymer cross-linking due to bombardment. During the activation, the stable isotope of 152Sm was converted to the radioactive gamma-emitting isotope of 153Sm. In this stage, the tablet is activated and can be detected by the gamma camera. The gamma spectroscopy data showed that neutron activation of the tablets did not produce any unnecessary radioactive byproducts (Figure 7).
Hybrid imaging of SPECT and CT was chosen in this study as the SPECT to evaluate the results based on functional and metabolic information of organs and cells. Integration of X-ray CT into SPECT gives excellent results in medical imaging, by presenting anatomical details and explaining functional and metabolic information.
The location of the test (for the F10 formulation) and control formulations in the rabbits’ gastrointestinal tracts at different time points after their administration are shown in Figure 8. The test and control formulations containing a combination of three different polymers were evaluated to determine their floating and sustained-release ability. The gamma scintigraphy images clearly indicated that the F10 formulation remained buoyant and that the drug was released gradually over the 12-hour study period; however, the rabbits that were administered the control tablets did not show the ability to sustain drug release that was shown in rabbits that were administered the formulation. This clearly shows the effects of combining salep, xanthan, and TKP polymers in controlling drug release. The in vivo result obtained from gamma scintigraphy showed an agreement between the in vitro and in vivo results (Figures 9 and 10).
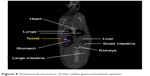  | Figure 8 Anatomical structure of the rabbit gastrointestinal system. |
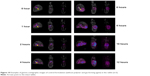  | Figure 10 Examples of gamma scintigraphic images of control formulation (without polymer and gas-forming agent) in the rabbit (n=3). |
The 153Sm2O3 activity decreased in the stomach region over time, while it was distributed gradually over the intestine region. The release profile showed a sustained release pattern and the ability of the formulations to control the drug release. The percentages of drug release from the F10 tablet in vivo were found to be 0%, 28.1%, 42.2%, 53%, 64.1%, 78%, 92.3%, and 95%, while that of the control formulation was 55.2%, 80.3%, 85.0%, 88.5%, 91.5%, 94.8%, and 95.0% from 0 to 12 hours (Figure 11). The results showed that more than 80% of drug was released in 2 hours in the control formulation, but it took 10 hours for the same amount of the drug to be released from the test formulation. The similarity factor between in vivo and in vitro formulations was 58% (Figure 11).
  | Figure 11 Quantitative gastrointestinal transit plots of 153Sm-labeled tablet (250 mg), and comparison of in vivo result of control and the test formulation with in vitro result of F10 (n=3). |
Conclusion
Floating gastroretentive matrix tablets of Metformin HCl were successfully formulated using a combination of natural polymers. Tablets containing TKP (5 mg), xanthan (33.33 mg), and salep (61.67 mg) as gel-forming polymers showed desirable in vitro and in vivo release properties. The optimized formulation showed a short FLT and total floating time of at least 24 hours, and could sustain drug release for 12 hours.
Acknowledgments
The authors gratefully acknowledge the financial support from the University Malaya for the research grants, numbers SF011-2014 Science Fund, MOSTI, RG044/11BIO, and PG052-2012B.
Disclosure
The authors report no conflicts of interest in this work.
References
Verma RK, Garg S. Current status of drug delivery technologies and future directions. Pharmaceutical Technology On-Line. 2001;25(2):1–14. | ||
Sastry SV, Nyshadham JR, Fix JA. Recent technological advances in oral drug delivery – a review. Pharm Sci Technolo Today. 2000;3(4): 138–145. | ||
Park K, Mrsny RJ. Controlled drug delivery: designing technologies for the future. Washington, DC: American Chemical Society; 2000. | ||
Surana AS, Kotecha RK. An overview on various approaches to oral controlled drug delivery system via gastroretention. Int J Pharm Sci Rev Res. 2010;2(2):68–72. | ||
Arora S, Ali J, Ahuja A, Khar RK, Baboota S. Floating drug delivery systems: a review. AAPS Pharm Sci Tech. 2005;6(3):E372–E390. | ||
Kalász H, Antal I. Drug excipients. Curr Med Chem. 2006;13(21):2535–2563. | ||
Kumar MNR. A review of chitin and chitosan applications. React Funct Polym. 2000;46(1):1–27. | ||
Tekade BW, Chauhadri YA. Gums and Mucilages: Excipients for modified Drug Delivery System. Journal of Advanced Pharmacy Education & Research. 2013;3(4):359–367. | ||
Pifferi G, Restani P. The safety of pharmaceutical excipients. Farmaco. 2003;58(8):541–550. | ||
Proctor WR, Bourdet DL, Thakker DR. Mechanisms underlying saturable intestinal absorption of metformin. Drug Metab Dispos. 2008;36(8):1650–1658. | ||
Sander C, Nielsen HM, Jacobsen J. Buccal delivery of metformin: TR146 cell culture model evaluating the use of bioadhesive chitosan discs for drug permeability enhancement. Int J Pharm. 2013;458(2):254–261. | ||
Rojas LB, Gomes MB. Metformin: an old but still the best treatment for type 2 diabetes. Diabetol Metab Syndr. 2013;5(1):6. | ||
Gupta V, Puri R, Gupta S, Jain S, Rao GK. Tamarind kernel gum: An upcoming natural polysaccharide. Systematic Reviews in Pharmacy. 2010;1:50–54. | ||
Razavi M, Nyamathulla S, Karimian H, Moghadamtousi SZ, Noordin MI. Hydrogel polysaccharides of tamarind and xanthan to formulate hydrodynamically balanced matrix tablets of famotidine. Molecules. 2014;19(9):13909–13931. | ||
Razavi M, Nyamathulla S, Karimian H, Noordin MI. Novel swellable polymer of orchidaceae family for gastroretentive drug delivery of famotidine. Drug Des Devel Ther. 2014;8:1315–1329. | ||
Faure A, York P, Rowe RC. Process control and scale–up of pharmaceutical wet granulation processes: a review. Eur J Pharm Biopharm. 2001;52(3):269–277. | ||
Weh FH, Razavi M, Erh CH, et al. Formulation and in vitro evaluation of hydrodynamically balanced matrix tablets of famotidine using pectin as controlled release polymer. Latin American Journal of Pharmacy. 2014;33(3):420–431. | ||
Jacob S, Shirwaikar AA, Joseph A, Srinivasan KK. Novel co–processed excipients of mannitol and microcrystalline cellulose for preparing fast dissolving tablets of glipizide. Indian J Pharm Sci. 2007;69(5):633–639. | ||
Patra CN, Kumar AB, Pandit HK, Singh SP, Devi MV. Design and evaluation of sustained release bilayer tablets of propranolol hydrochloride. Acta Pharm. 2007;57(4):479–489. | ||
Bomma R, Swamy Naidu RA, Yamsani MR, Veerabrahma K. Development and evaluation of gastroretentive norfloxacin floating tablets. Acta Pharm. 2009;59(2):211–221. | ||
Jaimini M, Rana AC, Tanwar YS. Formulation and evaluation of famotidine floating tablets. Curr Drug Deliv. 2007;4(1):51–55. | ||
Paul Y, Kumar M, Singh B. Formulation, evaluation and study of effect of hydrophilic polymers on release rate of cefixime floating tablets. Int J Pharma Bio Sci. 2011;2(4):472–488. | ||
Mura P, Gratteri P, Faucci MT. Compatibility studies of multicomponent tablet formulations. DSC and experimental mixture design. J Therm Anal Calorim. 2002;68(2):541–551. | ||
Yeong CH, Perkins AC. Sm–153 In Gastrointestinal Imaging: Extended Use of Sm–153 in Oral Drug Delivery and Diagnostic Imaging. LAP Lambert Academic Publishing; 2012. | ||
Seli E, Duleba AJ. Should patients with polycystic ovarian syndrome be treated with metformin? Hum Reprod. 2002;17(9):2230–2236. | ||
Manivannan R, Chakole V. Formulation and development of extended release floating tablet of atenolol. International Journal of Recent Advances in Pharmaceutical Research. 2011;3:25–30. | ||
Savic IM, Nikolic K, Nikolic G, Savic I, Agbaba D, Cakic M. Application of mathematical modeling for the development and optimization formulation with bioactive copper complex. Drug Dev Ind Pharm. 2013;39(7):1084–1090. | ||
Radhika PR, Pal TK, Sivakumar T. Formulation and evaluation of sustained release matrix tablets of glipizide. Iranian Journal of Pharmaceutical Sciences. 2009;5(4):205–214. | ||
Rathi M, Medhekar R, Pawar A, Yewale C, Gudsoorkar V. Floating and bioadhesive delivery system of metoprolol succinate: Formulation, development and in vitro evaluation. Asian Journal of Pharmaceutics. 2012;6(3):227–236. | ||
Yeong CH, Abdullah BJ, Ng KH, et al. Neutron–activated 153Sm–ion–exchange resin as a tracer for gastrointestinal scintigraphy. Nucl Med Commun. 2011;32(12):1256–1260. | ||
Yeong CH, Abdullah BJ, Ng KH, et al. Production and first use of 153SmCl3–ion exchange resin capsule formulation for assessing gastrointestinal motility. Appl Radiat Isot. 2012;70(3):450–455. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.