Back to Journals » Drug Design, Development and Therapy » Volume 9
Effects of histamine and its antagonists on murine T-cells and bone marrow-derived dendritic cells
Authors Hu X, Zafar MI , Gao F
Received 3 June 2015
Accepted for publication 8 July 2015
Published 21 August 2015 Volume 2015:9 Pages 4847—4860
DOI https://doi.org/10.2147/DDDT.S89792
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Wei Duan
Xiufen Hu,1,* Mohammad Ishraq Zafar,2,* Feng Gao2
1Department of Paediatrics, Tongji Hospital, 2Department of Endocrinology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, People’s Republic of China
*These authors contributed equally to this work
Abstract: We determined the effects of histamine and its antagonists on the surface marker expression of dendritic cells (DCs) and the influence of lipopolysaccharide (LPS), histamine, and histamine receptor antagonists on DCs and T-cells. The bone marrow was extracted from the femurs and tibiae of 6- to 8-week-old female Balb/c mice and cultured in medium containing penicillin, streptomycin, L-glutamine, fetal calf serum, or granulocyte macrophage colony-stimulating factor (GM-CSF) alone or with interleukin (IL)-4. The cells received three different doses of LPS and histamine, plus three different doses of descarboethoxyloratadine (DCL). We assayed the supernatant for various cytokines. The spleen cells of DO11.10 mice were examined by flow cytometry, which included labeling and sorting CD4+ T-cells, as well as coculture of DCs and T-cells with ovalbumin (OVA)323–339 peptide. Histamine or histamine plus DCL did not affect the expression of major histocompatibility complex class II, CD11c, CD11b, CD86, and CD80. However, GM-CSF increased the expression of all markers except CD80. Histamine increased interferon-γ production in GM-CSF + IL-4-cultured cells; it also enhanced IL-10 production, but suppressed IL-12 production in LPS-stimulated DCs with no DCL. Cimetidine inhibited IL-10 production and restored IL-12 secretion in LPS-treated DCs. LPS increased IL-10 and decreased IL-12 levels. GM-CSF + IL-4-generated DCs had a stronger stimulatory effect on DO11.10 T-cell proliferation than GM-CSF-generated DCs. Inducible costimulator ligand expression was higher in GM-CSF + IL-4- than in GM-CSF-generated DC groups after 2 days of coculture, but decreased 4 days later. IL-13 production was higher in bone marrow DCs generated with GM-CSF than in those generated with GM-CSF + IL-4. OVA-pulsed DCs and OVA-plus-DCL DCs showed increased IL-12 levels. OVA plus LPS increased both IL-10 and interferon-α. Although histamine or histamine receptor-1 antagonists did not influence DC LPS-driven maturation, they influenced cytokine production. LPS and GM-CSF influenced surface marker expression and cytokine production.
Keywords: dendritic cells, Th1, Th2, histamine, histamine receptor antagonist, costimulatory molecule
Introduction
Histamine is one of the most important mediators of various physiological and pathological conditions, including neurotransmission and numerous brain functions, neuroendocrine functions, cardiovascular functions, and gastric acid secretion regulation.1,2 In addition to its well-characterized effects in acute inflammatory and allergic responses, histamine has been shown to potently influence the immune response.3,4 It regulates several aspects of antigen-specific immune response development, including the maturation of DCs and alterations in their T-cell-polarizing capacity. Histamine also regulates antigen-specific T helper 1 and T helper 2 cells, as well as related antibody isotype responses. Recent studies have focused on the immune-regulatory mechanisms triggered by histamine. T-cells, monocytes, and dendritic cells (DCs) all possess histamine receptors on their surfaces and are capable of producing and secreting histamine. It has been recently documented that histamine induces a Th2 shift at the level of antigen-presenting cells (monocytes and DCs), Th1 and Th2 cells, or antibody isotypes. Histamine also affects both the Th1/Th2 balance through differential regulation of T helper lymphocytes.5 Several studies have shown that histamine enhances Th2-type cytokine production and inhibits Th1-type cytokine production by binding to histamine receptors on the surface of T helper lymphocytes.3,4
In order to understand the effects of histamine and its antagonists on murine T-cells and bone marrow-derived DCs (BMDCs), we investigated the generation of DCs and characterized these cells. We also examined whether lipopolysaccharide (LPS), histamine, and histamine receptor antagonists influence DCs and T-cells. Our results provide a basis for the potential use of antihistamines in current therapeutics.
Materials and methods
DC culture
All experiments were performed using 6- to 8-week-old female Balb/c mice, they were euthanized using anesthesia (3% sodium pentobarbital; 50 mg/kg). All experimental procedures were approved by the Ethics committee of Tongji Medical College, Huazhong University of Science and Technology. The femurs and tibiae of mice were removed and purified from the surrounding muscle tissue by rubbing them with Kleenex tissues. Intact bones were placed in 70% ethanol for 30 seconds and then washed with phosphate-buffered saline (PBS). Both ends of the samples were cut with scissors and the bone marrow was flushed with RPMI-1640 containing 5% fetal calf serum (FCS, Biochrom, Berlin, Germany) using a syringe needle (0.45 mm). Clusters in the marrow suspension were disintegrated by vigorous pipetting and filtered through a 100 μm cell strainer.
Bone marrow cells were cultured in six-well culture plates in Dulbecco’s Modified Eagle Medium (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 100 U/mL penicillin, 100 μg/mL streptomycin, 2 mM L-glutamine, 10% FCS, and 10 ng/mL granulocyte macrophage colony-stimulating factor (GM-CSF) alone or together with 10 ng/mL interleukin (IL)-4. Every 2 days, the culture plates were gently swirled and half of the medium in the well was replaced with fresh culture medium containing an equivalent amount of cytokines(s) that had been removed. Non-adherent granulocytes and clusters of developing DCs remained loosely attached on a bed of firmly adherent macrophages. After 6 days, loosely adherent clusters were collected. Purified BMDCs were further seeded at a density of 1×105/well in 96-well plates containing 10% FCS-supplemented Dulbecco’s Modified Eagle Medium. The cells were treated with 1, 10, and 100 ng/mL LPS and 10−7, 10−5, and 10−3 M histamine as well as 10−7, 10−5, and 10−3 M descarboethoxyloratadine (DCL, Essex Pharma, Munich, Germany) or cimetidine (Sigma-Aldrich, St Louis, MO, USA), respectively. The supernatant was collected from each well for cytokine measurement 24 and 48 hours later.
Measurement of cytokines
Cell culture supernatants were assayed for various cytokines, including IL-10, IL-12, IL-4, IL-5, IL-13, and interferon (IFN)-γ using a standard enzyme-linked immunosorbent assay.
Flow-cytometric analysis of murine spleen cells
For in vitro stimulation, 2×106 spleen cells were mixed with 10 ng/mL of 4beta-phorbol 12-myristate 13-acetate (Sigma-Aldrich) and 1 μg/mL of ionomycin (Sigma-Aldrich) and incubated in a 24-well plate at 37°C and 5% CO2. After 2 hours, 5 μg/mL of the protein transport inhibitor brefeldin A (Sigma-Aldrich) was added. Four hours later, we resuspended the cells in a 15 mL tube and washed the cells two times with PBS for 10 minutes at 1,800× g and 4°C (Biochrom).
After stimulation, we collected the cells by centrifugation. A 50 μL 10× FC-block (BD Pharmingen, Heidelberg, Germany) and 4 μL antibody were added. Next, we incubated the cells for 20 minutes at 4°C in the dark, followed by washing twice in PBS for 10 minutes at 1,800× g and at 4°C.
To perform cell fixing, we resuspended the cells in PBS (Biochrom), added an equal volume of 4% formaldehyde (EMD Millipore, Billerica, MA, USA) and PBS, and incubated the cells for 20 minutes at room temperature. The cells were washed once with PBS, and then resuspended in fluorescence-activated cell sorting (FACS)-PBS (EMD Millipore). Cells were stored at 4°C in the dark for measurements of cell surface markers at a later point.
We resuspended the cells in 50 μL saponin buffer (Sigma-Aldrich) and incubated them with the primary antibody for 15–30 minutes at room temperature. After adding 1 mL of saponin buffer and spinning cells at 300× g for 5 minutes at 4°C–23°C, we washed the cells a second time with 1 mL saponin buffer. Cell concentration was adjusted using FACS buffer.
CD4+ cells were suspended at 1×107/mL in PBS with no protein. A 5 mM stock solution of 5-(and -6)-carboxyfluorescein diacetate succinimidyl ester in dimethylsulfoxide was added to achieve a final concentration of 5 μM and incubated at room temperature for 4 minutes. Next, the cells were immediately washed once with RPMI-1640 containing 20% FCS and then twice with FACS-PBS; the cells were resuspended in RPMI-1640 containing 10% FCS. We cocultured the cells with DCs in 24-well plates (ratio of DCs to CD4 positive cells =1:10).
Cell sorting by MIDI-magnetic cell sorting
Murine spleens were extracted from DO11.10 mice and remnants of fat were removed. We placed a 212 μm sieve into a petri dish and filled the dish with 50 mL FCS-free RPMI-1640. We transferred the spleens to the sieves and mashed them with the sterile piston of a 1 mL syringe. After rinsing the sieve and collecting the cell suspension in a 50 mL centrifuge tube, we rinsed the petri dishes with RPMI-1640 and filled the tube to 50 mL. The cells were centrifuged at 1,800× g for 10 minutes at 4°C. The pellet was resuspended in 4 mL PBS, and the cell suspension was filtered through a 100 μm nylon strainer (BD Biosciences, Franklin Lakes, NJ, USA). We rinsed the nylon strainer and filled the tube to 50 mL. After centrifuging the cells at 1,800× g for 10 minutes at 4°C, we resuspended the splenocytes in a 15 mL tube and counted the cells.
CD4+ cells were separated by high-gradient magnetic sorting using magnetic cell sorting (MACS) (Miltenyi Biotec, Gladbach, Germany). Spleen cells were incubated with saturating concentrations of CD4 Micro Beads for 15 minutes at 6°C and washed with MACS buffer (PAA Laboratories, Linz, Austria). Labeled and positively enriched cells were eluted after removing the columns from the magnetic device. After adding 10 mL MACS buffer and centrifuging the cells at 1,500× g for 10 minutes at 4°C, the supernatant was discarded, and MACS buffer, FC-Block, and CD4 Micro Beads were added.
We calculated the dosage of the MACS buffer, FC-Block, and CD4 Micro Beads based on the cell number. For each 107 cells, we used (in microliters) 40× MACS buffer, 15× FC-Block, and 10× CD4 microbeads. We incubated the cells at 6°C–12°C for 15 minutes and shook them by hand for 5 minutes.
After adding 10 mL of MACS buffer and centrifuging it at 1,500 revolutions per minute for 10 minutes at 4°C, the supernatant was removed and 4 mL of MACS buffer was added. We washed the MACS tube with 3 mL of MACS buffer, added cells into the tube, and washed the cells with 3 mL of MACS buffer three times (CD4- cells). After the tube was removed from MACS, 5 mL of MACS buffer was added into the tube and a syringe piston (CD4+ cells) was pressed into the tube. We then counted the CD4+ cells. We performed the MACS control using FACS analysis of the original, CD4+, and CD4- fractions.
To perform in vitro coculture of DCs and DO11.10 T-cells, we seeded the mixture of both cells (DCs:DO11.10 T-cells =1:10) into a 24-well plate at a density of 2×106 per well per mL. Ovalbumin (OVA)323–339 peptide was added to each well at a final concentration of 1 μg/mL. We added 1 mL of fresh medium to each well on day 2 or day 3 and on day 4 or day 5 if necessary.
Six days later, the cells were harvested, counted, and seeded them into a new 24-well plate at a density of 2×106/well. The cells were restimulated with 10 ng/mL of 4beta-phorbol 12-myristate 13-acetate and 1 μg/mL of ionomycin. The supernatant was collected for cytokine determination after 24 and 48 hours.
BMDCs were generated using GM-CSF only or GM-CSF + IL-4 for 6 days. On day 6, the cells were pulsed for 16 hours with OVA, OVA/LPS, OVA/histamine, OVA/cimetidine, or OVA/DCL. These cells were then cocultivated with CD4+ sorted cells and CD11c-depleted cells from DO11.10 mice for another 6 days. After 6 days of coculture, the cells were restimulated for 5 hours with 4beta-phorbol 12-myristate 13-acetate/ionomycin in the presence of brefeldin A (last 3 hours). The cells were then fixed and stained, and intracellular cytokine levels were measured using FACS analysis. For some samples, no brefeldin A was added, and cytokine production was measured using an enzyme-linked immunosorbent assay in the cell culture supernatant 24 and 48 hours later. Each experiment was conducted in triplicate, and the data shown are representative of three independent experiments.
Results
Effect of LPS, histamine, and its antagonists on surface marker expression in DCs
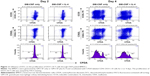
The surface markers of these myeloid DCs were analyzed by flow cytometry on days 0, 3, and 6 or 7. Figure 1 shows the simultaneous flow-cytometric analysis of CD11c+/CD11b+ cells (row 1), major histocompatibility complex (MHC) class II+/CD11c+ cells (row 2), and MHC class II+/CD11b+ cells (row 3). The mean percentages of CD11c+/CD11b+ cells were found to be 3%, 70%, and 96% on days 0, 3, and 7, respectively. The average percentages of MHC class II+/CD11c+ cells were 0.1%, 43%, and 17% on days 0, 3, and 7, respectively. The mean percentages of MHC class II+/CD11b+ cells were 50% and 16% on days 3 and 7, respectively. Furthermore, the expression of MHC class II, CD11c, CD11b, and CD86 was also investigated with histograms by FACS on days 0, 3, and 6 of culture (Figure 2). The expression of these surface markers was increased during the development of myeloid DCs.
The surface markers MHC class II, CD11c, CD11b, CD86, and CD80 were characterized by flow cytometry (Figure 3). Incubation of immature DCs with LPS, a strong inducer of maturation, did not significantly enhance the expression of the costimulatory molecules CD86, CD80, MHC class II, CD11c, or CD11b on DCs.
The influence of histamine and DCL on the expression of surface markers in LPS-stimulated myeloid DCs is shown in Figure 4. Flow cytometry revealed no differences in the expression of common myeloid surface markers (MHC class II, CD11c, and CD11b) and costimulatory molecules (CD86 and CD80) upon the addition of histamine or histamine plus DCL, indicating that neither histamine nor histamine receptor-1 antagonist influenced DC maturation driven by LPS.
Effect of LPS, histamine, and its antagonists on phenotypic characterization of OVA-pulsed BMDCs
The expression of surface markers MHC class II, CD11c, and inducible costimulator ligand (ICOS-L) was determined using FACS. Based on the expression of these markers, the cells were classified into three types, including CD11c+/MHC class II low/ICOS-L-, CD11c+/MHC class II high/ICOS-L+, and CD11c+/MHC class II high/ICOS-L- (Figure 5). There was no significant change in any of these markers, regardless of the type of stimulus applied, except that DCL treatment alone significantly decreased ICOS-L expression.
Effect of LPS, histamine, and histamine antagonists on cytokine production from DCs
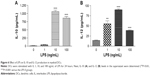
LPS-stimulated DCs produced high levels of IL-10 and low levels of IL-12 (Figure 6A and B) because 10 ng/mL LPS had the strongest stimulatory effect on IL-10 and IL-12 production.
Histamine appeared to have a paradoxal effect on IL-10 and IL-12 production in maturing DCs. Histamine greatly and most efficiently enhanced IL-10 production at a concentration of 10−5 M (P<0.001) (Figure 7A) and dose-dependently suppressed the secretion of the Th1 cytokine IL-12 in LPS-stimulated DCs (P<0.001) (Figure 7B). This indicates that histamine induced the Th2 response.
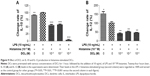
Cimetidine inhibited histamine-enhanced IL-10 production (Figure 8A) but restored histamine-reduced IL-12 secretion (Figure 8B) in LPS-treated DCs (P<0.01). Since IL-10 and IL-12 levels were brought to a histamine-free state by cimetidine, which completely blocked the effect of histamine, suggesting that histamine stimulates IL-10 and suppresses IL-12 production in DCs.
In the absence of DCL, histamine enhanced IL-10 production (Figure 9A) but inhibited IL-12 secretion (Figure 9B) in LPS-treated DCs. However, DCL potently suppressed IL-10 production to a lower level than in the histamine-free group (P<0.001) (Figure 9A). Moreover, DCL failed to restore histamine-reduced IL-12 secretion (P<0.001) (Figure 9B). In contrast, DCL further decreased IL-12 production.
After overnight culture with high-dose OVA (50 μg/mL) followed by the addition of 10 ng/mL of LPS, 10−5 M histamine, 10−5 M cimetidine, or 10−5 M DCL for another 24 hours, OVA-pulsed DCs produced high levels of IL-12, favoring a Th1 response. However, OVA plus LPS increased both IL-10 and INF-α, favoring a Th2 response. OVA plus histamine or cimetidine did not change IL-12, IL-10, and INF-α levels, but OVA plus DCL increased IL-12 production, favoring a Th1 response (Figure 10).
Effects of LPS, histamine, cimetidine, and DCL on the proliferation, surface marker expression, and cytokine production of DO11.10 T-cells
The Th2 cytokine IL-4 is commonly used for culturing DCs from bone marrow and is considered to be a maturation signal for DCs.6 Primary bone marrow cells were cultured in GM-CSF only or GM-CSF + IL-4-supplemented medium for 6 days. The freshly purified myeloid DCs were cultured overnight with a high dose of OVA (50 μg/mL), followed by the addition of 10 ng/mL of LPS, 10−5 M histamine, 10−5 M cimetidine, or 10−5 M DCL for another 24 hours. DO11.10 transgenic mice expressing the OVA-specific T-cell receptor have been used as a model of antigen-specific T-cell activation to study the role of Th cells in allergic diseases.7 Therefore, carboxyfluocein diacetate (CFDA)-labeled DO11.10 Th0 (CD4+) cells were added to the culture for an additional 2 or 4 days.
To examine the effects of LPS, histamine, and histamine antagonists on the capability of DCs to induce proliferation of antigen-specific CD4+ T-cells, a T-cell proliferation assay was performed by using CD4+ DO11.10 T-cells as responder cells in the presence of the OVA323–339 peptide. FACS analysis showed that DCL had no effect on the proliferation of DO11.10 T-cells that had been cocultured with OVA-pulsed BMDCs for 2 days, but DCL stimulated their proliferation after coculture for 4 days (Figure 11). Moreover, GM-CSF + IL-4 caused DCs to exhibit a stronger stimulatory capacity in DO.11.10 T-cell proliferation than GM-CSF-generated DCs. Similarly, LPS, histamine, and cimetidine (Figure 12) all generated antigen-specific T-cell proliferation to an extent similar to DCL on day 4. The M1 fraction (cells that did not proliferate at the time point of measurement) was reduced after DCL stimulation compared with that in cells pulsed with OVA/LPS.
The phenotypic characterization of CD4+ DO11.10 cells cocultured with pulsed BMDCs was studied with FACS staining as shown in Figure 13. This figure shows ICOS (early activation marker) and CD62L (highly expressed on naïve cells as well as late activation marker) expression in cocultured CD4+ cells. On day 2 of cocultivation, ICOS expression was approximately 30% on CFDA and CD4+ cells cocultivated with GM-CSF only, whereas ICOS expression was approximately 40% on CFDA and CD4+ cells cocultured with GM-CSF + IL-4. These data indicate stronger activation of CD4 cells through DCs generated with GM-CSF and IL-4. On day 4 of analysis, ICOS expression on CFDA and CD4+ cells cocultivated with GM-CSF only was approximately 61%, whereas CFDA cells cocultivated with GM-CSF + IL-4 showed ICOS expression of approximately 53%.
The expression of CD62L, which indicates naïve T-cells, was very similar; expression was approximately 90% for CFDA and CD4+ cells cocultivated with GM-CSF only and for CD4+ CFDA cells and CD4+ cells cocultivated with GM-CSF + IL-4 for 2 days. On day 4, approximately 45% of CD4+ and CFDA cells cocultivated only with GM-CSF expressed CD62L and approximately 60% of CD4+ and CFDA cells cocultivated with GM-CSF + IL-4 expressed CD62L. The different pulse treatments of BMDCs with OVA/LPS, OVA/histamine, OVA/cimetidine, and OVA/DCL did not significantly change the expression of ICOS and CD62L.
Figure 14 shows the intracellular levels of IL-4 and IL-13 in CD4+ cocultured cells. BMDCs generated with GM-CSF and no IL-4 showed higher production of IL-13 compared with BMDCs generated with GM-CSF and additional IL-4, regardless of the stimulus was applied (lower panel of Figure 14). CD4+ cells cocultivated with OVA/DCLpulsed BMDCs generated with GM-CSF alone showed the highest IL-4 levels (upper panel of Figure 14). These data together with intracellular IFN-γ and IL-10 detection as well as extracellular (supernatant) IFN-γ, IL-10, IL-5, and IFN-α determination are summarized in Table 1.
Discussion
In this study, we demonstrated that the timing of DC cocultivation with T-cells influences T-cell proliferation, regardless of whether the cells were pulsed with OVA and LPS, histamine, cimetidine, or DCL. The expression of CD62L was equal in GM-CSF + IL-4- and GM-CSF-generated DC groups after 2 days of coculture, but the expression was higher in the GM-CSF + IL-4-generated group versus the GM-CSF-generated DC group after 4 days (Figure 13). ICOS expression was higher in GM-CSF + IL-4- versus GM-CSF-generated DC groups after 2 days of coculture but the opposite was observed after 4 days.
Moreover, DC generation methods affect proliferation rate. Greater amounts of IL-13 were produced when IL-4 was omitted from BMDCs generated with GM-CSF versus when IL-4 was included. This change occurred regardless of whether LPS, histamine, cimetidine, or DCL was added. Similarly, for CD4+ DO11.10 cells cocultivated with pulsed BMDCs, IL-4 production was highest when GM-CSF was used alone. Among the cells in which GM-CSF was used without IL-4, the levels of IL-4 were highest when BMDCs were pulsed with OVA and DCL.
GM-CSF + IL-4-generated DCs exhibited a stronger stimulatory capacity on DO11.10 T-cell proliferation than GM-CSF-generated DCs, which is consistent with the results of a previous study by Jiang et al.8 This is because GM-CSF + IL-4-generated DCs were more mature in both phenotype and function. IL-4 in the culture enhanced DC proliferation and cluster formation and promoted DC maturation with CD11c+ and higher expression of MHC class II, CD86, and CD40.8
Effect of histamine
Our findings regarding the effect of histamine on common myeloid surface markers (MHC class II, CD11c, and CD11b) and costimulatory molecules (CD86 and CD80) are similar to those observed in other studies. In one study, histamine alone did not induce CD86, CD80, and CD11c expression. Histamine appeared to slightly increase the expression of these surface markers and costimulatory molecules when LPS was administered, but the increases were not statistically significant.9
In our study, histamine 10−5 M and 10−7 M significantly enhanced IL-10 production, with the greatest changes observed at a dose of 10−5 M. Histamine also significantly suppressed the secretion of the Th1 cytokine IL-12 in a dose-dependent manner. This is similar to studies in human DCs and murine spleen cells in which histamine significantly decreased IL-12 and IL-10 production by LPS-stimulated DCs in a dose- and concentration-dependent manner.9–11 This indicates that histamine favors a Th2 response.
Effect of LPS
Our findings regarding the effect of LPS on MHC class II, CD11c, CD11b, and the costimulatory molecules CD86 and CD80 are also similar to those in other studies. In one study, LPS treatment resulted in increased expression of MHC class II and CD86, and CD11c expression was closely correlated with the MHC class II expression, while CD11b expression varied.12 However, our results differ from those of the previous study in one respect. In that study, LPS treatment upregulated MHC class II expression in addition to CD86 and CD80 expression.12 Such differences may be attributed to differences in cultivation methods.
In our study, LPS-stimulated DCs showed significantly higher levels of IL-10 and lower levels of IL-12. These stimulatory effects were strongest at a concentration of 10 ng/mL. Our findings are in contrast to those of Thoma-Uszynski et al13 who concluded that LPS-stimulated DCs produced high IL-12 levels and low IL-10 levels, favoring Th1 polarization. This discrepancy may be because of differences in the DC subsets used in the experiments. It was reported that early-maturing DCs and late-maturing DCs gave rise to a completely different pattern of IL-12 and IL-10 secretion upon LPS stimulation. LPS-stimulated early-maturing DCs produced high levels of IL-10 and low levels of IL-12, whereas LPS-stimulated late-maturing DCs secreted high levels of IL-12 and low levels of IL-10.8 Despite this divergent cytokine response, both DC populations exhibited similar levels of MHC class II and costimulatory CD86 and CD40 expression.
Effect of cimetidine, histamine, and DCL
In our experiment, cimetidine stabilized the levels of IL-10 and IL-12 by inhibiting IL-10 production enhanced by histamine and restored IL-12 secretion in LPS-stimulated DCs. These results are similar to those of studies in monocytes in which cimetidine blocked the effect of histamine on LPS-induced IL-12 and IL-10 production.14 Although histamine and cimetidine did not significantly affect the expression MHC class II, CD11c, and ICOS-L, DCL treatment significantly decreased ICOS-L expression by suppressing IL-10 production to a lower level versus that in the histamine-free group. Rather than restoring histamine-reduced IL-12 secretion, DCL significantly decreased IL-12 production. Based on these data and the data indicating that cimetidine completely inhibits the effects of histamine on the production of IL-10 and IL-12, the DCL may not suppress IL-10 and IL-12 secretion by inhibiting the histamine pathway. Similarly, DCL did not appear to change the effect of OVA on IL-10 and IL-12. As such, the mechanism underlying DCL’s effect requires further analysis.
Effect of OVA and LPS doses in relation to cimetidine and histamine
In our experiment, changes in IL-12 and IL-10 levels that favor a Th2 response occurred when high-dose OVA or LPS was added. These results are similar to those found in another study in which OVA stimulation did not significantly induce secretion of IL-10 and IL-12, but simultaneous stimulation with OVA and LPS increased IL-10 production by three-fold, favoring a Th2 response.15 These results indicate that LPS is a stronger stimulator of BMDCs than OVA. Decreases in IL-12 and IL-10 levels resulting from the addition of histamine or cimetidine indicate that histamine or cimetidine may counteract OVA. Although a mechanism in the context of humoral immunity has been proposed, the mechanism underlying the interaction between OVA and histamine on DCs remains unclear.16
Conclusion
The timing used for cocultivation of DCs with T-cells influences the proliferation of T-cells, regardless of the stimulus applied. Histamine or histamine receptor-1 antagonists do not influence DC maturation driven by LPS. However, both histamine and histamine receptor-1 antagonists influence the production and secretion of cytokines such as IFN-γ, IL-10, and IL-12. Other factors involved in the expression of surface markers and cytokine production include LPS and GM-CSF. Additional research is necessary to determine the mechanism of the effect of DCL and the mechanism underlying the interaction between OVA and histamine on DCs.
Acknowledgment
This study was supported by The Scientific Research Grants by Ministry of Education, People’s Republic of China.
Disclosure
The authors report no conflicts of interest in this work.
References
Mathan TSM, Figdor CG, Buschow SI. Human plasmacytoid dendritic cells: from molecules to intercellular communication network. Front Immunol. 2013;4:372. | ||
Pino-Ángeles A, Reyes-Palomares A, Melgarejo E, Sánchez-Jiméneza F. Histamine: an undercover agent in multiple rare diseases? Cell Mol Med. 2012;16:1947–1960. | ||
Zampeli E, Tiligada E. The role of histamine H4 receptor in immune and inflammatory disorders. Br J Pharmacol. 2009;157:24–33. | ||
O’Mahony L, Akdis M, Akdis CA. Regulation of the immune response and inflammation by histamine and histamine receptors. J Allergy Clin Immunol. 2011;128:1153–1162. | ||
Fujimoto S, Komine M, Karakawa M, et al. Histamine differentially regulates the production of Th1 and Th2 chemokines by keratinocytes through histamine H1 receptor. Cytokine. 2011;54:191–199. | ||
Lutz MB, Kukutsch NA, Menges M, Rössner S, Schuler G. Culture of bone marrow cells in GM-CSF plus high doses of lipopolysaccharide generates exclusively immature dendritic cells which induce alloantigen-specific CD4 T cell anergy in vitro. Eur J Immunol. 2000;30:1048–1052. | ||
Kanaizumi E, Shirasaki H, Sato J, Watanabe K, Himi T. Establishment of animal model of antigen-specific T lymphocyte recruitment into nasal mucosa. Scand J Immunol. 2002;56(4):376–382. | ||
Jiang HR, Muckersie E, Robertson M, Xu H, Liversidge J, Forrester JV. Secretion of interleukin-10 or interleukin-12 by LPS-activated dendritic cells is critically dependent on time of stimulus relative to initiation of purified DC culture. J Leukoc Biol. 2002;72:978–985. | ||
Pavlinkova G, Yanagawa Y, Kikuchi K, Iwabuchi K, Onoé K. Effects of histamine on functional maturation of dendritic cells. Immunobiology. 2003;207(5):315–325. | ||
Idzko M, la Sala A, Ferrari D, et al. Expression and function of histamine receptors in human monocyte-derived dendritic cells. J Allergy Clin Immunol. 2002;109(5):839–846. | ||
Mazzoni A, Young HA, Spitzer JH, Visintin A, Segal DM. Histamine regulates cytokine production in maturing dendritic cells, resulting in altered T cell polarization. J Clin Invest. 2001;108(12):1865–1873. | ||
Brasel K, De Smedt T, Smith JL, Maliszewski CR. Generation of murine dendritic cells from flt3-ligand-supplemented bone marrow cultures. Blood. 2000;96(9):3029–3039. | ||
Thoma-Uszynski S, Kiertscher SM, Ochoa MT, et al. Activation of toll-like receptor 2 on human dendritic cells triggers induction of IL-12, but not IL-10. J Immunol. 2000;165(7):3804–3810. | ||
Elenkov IJ, Webster E, Papanicolaou DA, Fleisher TA, Chrousos GP, Wilder RL. Histamine potently suppresses human IL-12 and stimulates IL-10 production via H2 receptors. J Immunol. 1998;161(5):2586–2593. | ||
Ahrens B, Freund T, Rha RD, et al. Lipopolysaccharide stimulation of dendritic cells induces interleukin-10 producing allergen-specific T cells in vitro but fails to prevent allergic airway disease. Exp Lung Res. 2009;35(4):307–323. | ||
Jutel M, Watanabe T, Akdis M, Blaser K, Akdis CA. Immune regulation by histamine. Curr Opin Immunol. 2002;14:735–740. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.