Back to Journals » Drug Design, Development and Therapy » Volume 9
Edible bird’s nest attenuates procoagulation effects of high-fat diet in rats
Authors Yida Z, Imam M , Ismail M, Ismail N, Hou Z
Received 1 May 2015
Accepted for publication 2 June 2015
Published 29 July 2015 Volume 2015:9 Pages 3951—3959
DOI https://doi.org/10.2147/DDDT.S87772
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Prof. Dr. Wei Duan
Zhang Yida,1,2 Mustapha Umar Imam,1 Maznah Ismail,1,3 Norsharina Ismail,1 Zhiping Hou1
1Laboratory of Molecular Biomedicine, Institute of Bioscience, Universiti Putra Malaysia, Serdang, Selangor, Malaysia; 2Cardiology Department, Affiliated Hospital of Chengde Medical University, Chengde, Hebei, People’s Republic of China; 3Faculty of Medicine and Health Sciences, Department of Nutrition and Dietetics, Universiti Putra Malaysia, Serdang, Selangor, Malaysia
Abstract: Edible bird’s nest (EBN) is popular in Asia, and has long been used traditionally as a supplement. There are, however, limited evidence-based studies on its efficacy. EBN has been reported to improve dyslipidemia, which is closely linked to hypercoagulation states. In the present study, the effects of EBN on high-fat diet- (HFD-) induced coagulation in rats were evaluated. Rats were fed for 12 weeks with HFD alone or in combination with simvastatin or EBN. Food intake was estimated, and weight measurements were made during the experimental period. After sacrifice, serum oxidized low-density lipoprotein (oxLDL), adiponectin, leptin, von willibrand factor, prostacyclin, thromboxane and lipid profile, and whole blood coagulation indices (bleeding time, prothrombin time, activated partial thromboplastin time, red blood count count, and platelet count) were estimated. Furthermore, hepatic expression of coagulation-related genes was evaluated using multiplex polymerase chain reaction. The results indicated that EBN could attenuate HFD-induced hypercholesterolemia and coagulation similar to simvastatin, partly through transcriptional regulation of coagulation-related genes. The results suggested that EBN has the potential for lowering the risk of cardiovascular disease-related hypercoagulation due to hypercholesterolemia.
Keywords: edible bird’s nest, coagulation, high-fat diet, hypercholesterolemia, nutrigenomics
Introduction
Cardiovascular diseases (CVD) cause huge morbidity and mortality burdens, and continue to grow despite advances in the management of the diseases.1 Several variables are involved in the pathogenesis and progression of CVD including blood coagulation status. Effective control of CVD will therefore entail controlling multiple variables like hypercholesterolemia and coagulation.2,3 There is heightened interest in the use of alternative therapies for CVD management due to concerns of side effects with synthetic drugs.4 Edible bird’s nest (EBN), popular among Asians, especially in the People’s Republic of China, has a long history of traditional use. EBN has been a highly valuable food tonic among the Chinese people for thousands of years, and it was believed to treat diseases such as tuberculosis, cough, dyspnea, and stomach ulcers. It has also been used to improve general well-being, especially among the old and in postpartum women, as a result of its perceived abilities to enhance renal function, strengthen the immune system, increase energy and metabolism, and regulate circulation. Despite its traditional uses over the years, evidence-based studies on its functional effects are limited.5,6 EBN is antioxidant-rich and can attenuate inflammation and oxidative stress,7,8 which are disease states that have been closely linked with hypercoagulation states.9 Accordingly, coagulation can be potentiated by inflammation and oxidative stress, which can be alleviated by antioxidants.10 Based on the rich antioxidant nature of EBN and its ability to improve inflammation and lipid profiles, we hypothesized that it may have beneficial effects on CVD-related hypercoagulation.
Experience over the years with traditional use of EBN suggests that it may be free from any toxic effects since it has been used for generations without any documented deleterious effects. This means EBN may have potential use as a safe supplement or nutraceutical. In the present study, therefore, we aimed to study the effects of EBN on high-fat diet- (HFD-) induced hypercholesterolemia and hypercoagulation, including a possible mechanistic basis for observed effects.
Materials and methods
Materials
The Adiponectin ELISA (enzyme-linked immunosorbent assay) kit was purchased from Millipore (Billerica, MA, USA), while those of oxLDL (oxidized low-density lipoprotein), thromboxane, prostacyclin, leptin, and vWF (von Willebrand factor) were purchased from Elabscience Biotechnology Co., Ltd (Wuhan, Hubei, People’s Republic of China). The RNA extraction kit was purchased from RBC Bioscience Corp. (Taipei, Taiwan), and the GenomeLab™ GeXP Start Kit was purchased from Beckman Coulter Inc (Miami, FL, USA). Simvastatin was purchased from Pfizer (New York, NY, USA), while Cholesterol and Cholic acid were purchased from Amresco (Solon, OH, USA) and Santa Cruz Biotechnology (Santa Cruz, CA, USA), respectively. RCL2 Solution was purchased from Alphelys (Toulouse, Plaisir, France), and analytical grade ethanol was purchased from Merck (Darmstadt, Hesse, Germany). Palm oil and Standard rat pellet were from Yee Lee Edible oils Sdn. Bhd. (Ipoh, Perak, Malaysia) and Specialty feeds (Glen Forrest, WA, Australia), respectively. Lipid profile kits were from Randox Laboratories Ltd (Crumlin, County Antrim, UK).
EBN
EBN was supplied by Blossom View Sdn. Bhd (Terrengganu, Malaysia). It was cleaned under tap water for 5 minutes, dried at room temperature and ground into powder manually using mortar and pestle. The ground EBN was stored at 4°C, and was used to prepare fresh pellets 1 day prior to being fed to rats. Only freshly prepared pellet was given to rats, while any leftover pellet on the rat cages from the previous day was discarded.
Animal study
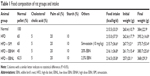
The animal study was approved by the Animal Care and Use Committee of the Faculty of Medicine and Health Sciences, Universiti Putra Malaysia (Project approval number: UPM/IACUC/AUP-R011/2014). Sprague Dawley rats (10-week old, 230–280 g, n=30) were handled as stipulated by standard guidelines for handling animals. They were housed at 25°C±2°C, 12/12 hours light/dark cycle and allowed to acclimatize for 2 weeks with free access to normal pellet and water. Rats were then divided into five groups: the normal group were fed with normal pellet, the HFD group were fed with HFD containing 4.5% cholesterol and 0.5% cholic acid, the HFD + simvastatin (SIM) group were fed with HFD and SIM (10 mg/kg/day), and two EBN groups were fed with low- or high-dose EBN with HFD (Table 1). Intervention lasted 12 weeks, and body weights were measured weekly, while food intake was calculated every day by subtracting the leftovers from what was added the previous day. Rats were sacrificed at the end, and their blood and tissue samples collected for further analyses.
Activated partial thromboplatin time, prothrombin time, bleeding time, platelet count, and red blood cell (RBC) count
For prothrombin time (PT) and APTT (activated partial thromboplatin time) assays, blood was collected in the vacutainer tubes (buffered sodium citrate), and centrifuged to get the serum, which was analyzed using an automated coagulation analyzer (START-4, Diagnostica Stago, Parsippany, NJ, USA). Rat tail bleeding time (BT) was measured after anesthesia on a hotplate. The tail was heated at 37°C, and 2 mm of its tip was amputated with a blade. The blood was blotted onto a filter paper every 30 seconds until the paper was no longer stained with blood. The period between amputation and the cessation of bleeding was considered the bleeding time (second). For hematologic cell counts, whole blood was collected in vacutainer tubes (K3EDTA) and analyzed using the MEDONIC CA530 hematology analyzer (Medonic, Sweden) within 2 hours after collection.
Ex vivo platelet aggregation
Ex vivo platelet aggregation was performed using baseline and final blood samples, with adenosine diphosphate (ADP) as agonist. Rat whole blood was collected in a vacutainer containing 3.2% sodium citrate (9:1, v/v), and thoroughly mixed by inverting the vacutainer several times. The blood was spun at 100× g for 20 minutes to get the platelet-rich plasma. Then, 50 ng/mL of prostacyclin was added in to prevent platelet activation. Platelet-rich plasma was again spun at 240× g for 10 minutes to obtain the platelet pellet. The platelet pellet was resuspended in Tyrode-HEPES buffer (134 mM NaCl, 2.9 mM KCl, 0.34 mM Na2HPO4, 12 mM NaHCO3, 20 mM HEPES, and 5 mM glucose, pH 7.3) to a cell density of 108 cells/mL. The solution (100 μL) was then mixed with 1 μL of ADP in a 96-well plate, and the optical density changes read at 405 nm for 20 minutes in a microplate reader. Readings were taken every minute, and the plate was shaken in double orbital mode.
Biochemical analyses and serum ELISA analysis
Serum samples were analyzed for lipid profile using Randox analytical kits on a Selectra XL instrument (Vita Scientific, Dieren, Rheden, the Netherlands). Serum ELISA analyses were performed using oxLDL, prostacyclin, thromboxane, Adiponectin, Leptin, vWF, and PAI-1 ELISA kits according to manufacturers’ recommendations. Absorbances were read on a BioTeK Synergy H1 Hybrid Reader (BioTek Instruments Inc., Winooski, VT, USA) at the appropriate wavelengths, and results were expressed as fold changes in relation to the values for the normal control group.
Gene expression
Primer design
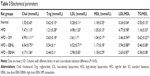
The primers were designed by GenomeLab eXpress Profiler software using input sequences from the National Center for Biotechnology Information website (http://www.ncbi.nlm.nih.gov/nucleotide/). Primers were tagged with an 18-nucleotide universal forward and 19-nucleotide universal reverse sequence, respectively (Table 1), and synthesized by Integrated DNA Technologies (Singapore).
RNA extraction, reverse transcription, and polymerase chain reaction
RNA was extracted using a Total RNA isolation kit (RBC Biotech Corp., Taipei, Taiwan), and diluted to 20 ng/mL. Reverse transcription and polymerase chain reaction (PCR) program were performed according to the GenomeLab™ GeXP Start Kit protocol (Beckman Coulter, Inc, Miami, FL, USA), as shown in Table 2.
GeXP genetic analysis system and multiplex data analysis
Sample loading solution (38.5 μL) and DNA size standard 400 (0.5 μL) (GenomeLab GeXP Start Kit; Beckman Coulter, Inc) were mixed with 1 μL PCR products, and the mixture was loaded onto a 96-well sample plate for analysis on the GeXP genomelab genetic analysis system (Beckman Coulter, Inc). Gene expression results were analyzed with the Fragment Analysis module of the GeXP system software, and the data exported and normalized on the eXpress Profiler software.
Data analysis
Data are presented as mean ± standard deviation, and comparisons of means was done using one-way analysis of variance on SPSS 17.0 software (SPSS Inc., Chicago, IL, USA). The significance of the difference between comparisons was determined by Tukey’s range test. P<0.05 was considered significantly different.
Results and discussions
Body weight changes
Table 2 shows the food intakes (calories) and body weights for different groups during the intervention period. No significant differences were observed in terms of food intake and body weights between the groups after 12 weeks of intervention, although the percentage body weight changes were different. HFD, HFD + SIM, lose dose EBN with HFD, and high dose EBN with HFD groups had 50%, 40%, 45%, and 43% changes in body weights, respectively.
Lipid profile
Table 3 shows the lipid profiles of the different groups. The normal group had significantly lower cholesterol levels compared with the other groups. The cholesterol levels of the EBN groups were lower than that of the HFD group, although it was only significantly so for the HFD + EBNH group when compared with the HFD group. Typically, simvastatin is expected to reduce the cholesterol level, as seen with the present data.11,12 Based on the results of the EBN group, it can be suggested that EBN can prevent worsening of lipid profiles, which may contribute toward attenuating the risk of coagulation since hypercholesterolemia is associated with hypercoagulatory states.13
APPT, PT, BT, platelet count, platelet aggregation, and RBC count
APTT and PT are important indices of the endogenous and exogenous coagulation systems, respectively.14 Figure 1A shows that APTT was significantly shorter in the HFD group compared with the other groups (P<0.05). A similar pattern was observed for PT which was also shorter in the HFD group compared with the other groups (Figure 1B), although only the HFD + EBNH and normal groups were significantly different from the HFD group (P<0.05). These results indicated that HFD promoted hypercoagulation, while EBN attenuated this pathological change. Additionally, BT showed a similar trend with APTT; the HFD group had shorter bleeding time compared with the other groups, while EBN attenuated the HFD-induced changes (Figure 1C). With regard to RBC cell count, no differences were observed between all the groups (Figure 2A). However, the platelet count and platelet aggregation were significantly higher in the HFD group in comparison with the other groups except the HFD + SIM group (Figure 2B and C, respectively). Moreover, increased platelet count and higher platelet aggregation have been linked to increased risk of hypercoagulation.15 Overall, these results indicated that HFD could promote hypercoagulation partly as a result of the hypercholesterolemia it induces,16 and EBN may have prevented HFD-induced hypercoagulation partly through regulating cholesterol levels.
Serum oxLDL, adiponectin, leptin, thromboxane, vWF, and prostaglandin F1α
oxLDL is an important predictor of CVD risk, which is a product of LDL oxidation. In fact, higher levels have been associated with a higher risk of coagulation.16 Figure 3A shows that EBN and simvastatin reduced oxLDL significantly in comparison with the HFD group. Moreover, simvastatin has been reported to lower oxLDL.17 Additionally, adiponectin is an important regulator of lipid metabolism and coagulation.18,19 As shown in Figure 3B, the HFD + EBNH group, but not the HFD + EBNL group, had a significantly higher adiponectin level in comparison with the HFD and HFD + SIM groups. Simvastatin, however, did not show improvement in adiponectin levels similar to previous reports.20 Furthermore, leptin is a regulator of lipid metabolism and has implications for coagulation.21 As shown in Figure 3C, the HFD + SIM and HFD + EBNH groups significantly lowered leptin levels, while the HFD + EBNL group did not produce as much change as with the former groups. Thromboxane levels reflect the state of thrombosis, and increased thromboxane levels are reported to denote a higher risk of CVD.22 Figure 4A shows that the HFD group had significantly higher thromboxane levels than the other groups, suggesting that the group had a higher risk of thrombosis-related CVD, while EBN and simvastatin had lower risks. vWF, like thromboxane, is indicative of the state of thrombosis,23 and just as with the thromboxane levels in the present study, vWF was significantly higher in the HFD group (Figure 4B). The results again suggest an increased risk of thrombosis-related events in this group, and lower risks of such events in the EBN and simvastatin groups. Prostacyclin is a potent vasodilator,24 and the present study showed that the HFD group had lower levels (Figure 4C) in comparison with the EBN groups, suggesting that HFD promoted vasoconstriction, while EBN promoted vasodilatation.
In aggregate, the data demonstrated that HFD will promote hypercholesterolemia and hypercoagulation, while EBN attenuates the HFD-induced changes and may therefore lower the risk of CVD.
Hepatic tissue mRNA levels of coagulation genes
In order to provide insights into the mechanistic basis for the effects of EBN, mRNA levels of related hepatic genes were measured. Figure 5 shows the mRNA levels of hepatic leptin, adiponectin, and NOS genes. Similar to the serum markers, adiponectin and leptin mRNA levels were equally modulated by the interventions. The HFD group showed leptin and adiponectin mRNA changes that reflected the serum changes and were in keeping with worsening of metabolic control, while EBN prevented the HFD-induced changes. NOS is a potent mediator of vasodilatation,25 and its increased expression by EBN may have underlined the changes in serum prostacyclin levels observed. Moreover, NOS has been reported to modulate prostacyclin levels as part of the basis for its vasodilatory effects.26 Additionally, in Figure 6, vWF gene expression was reflective of the serum changes observed in this study, indicating that the transcriptional changes in vWF induced by EBN similar to those of adiponectin and leptin may have been the basis for the changes in serum markers. PAI-1, another marker of coagulation,27 was also upregulated in the livers of the HFD-fed rats, while EBN and simvastatin attenuated such effects. Overall, the gene expression data are in keeping with the biochemical changes observed, and suggest that EBN is able to prevent the HFD-induced procoagulatory hypercholesterolemic condition partly through transcriptional regulation of genes related to coagulation and regulation of metabolism.
The effects of EBN may not be explained by the presence of a single bioactive in view of the wide variety of its components. Moreover, the concept of food synergy has been proposed to suggest that food is the functional unit of nutrition, since the effects of food are not normally explained by the presence of a single compound.28 Accordingly, the multiple bioactive compounds in EBN may have produced their effects through synergism.
Conclusion
The results of this study showed that HFD induced hypercholesterolemia and hypercoagulation by worsening lipid profile and elevating procoagulation factors. Transcriptional modulation of related genes seems to be involved in EBN-associated attenuation of the HFD-induced changes. EBN showed effects similar to those of simvastatin, although the results suggested that EBN may be better because it did not show a tendency for worsening of metabolism. The present data demonstrate an important health benefit of EBN, the implications of which may mean more targeted patronage of EBN because of the increasing scientific evidence for its health benefits. The translational implications of these findings, however, need to be confirmed through clinical trials, which have the potential to establish EBN as a functional food for lowering the risk of CVD-related hypercoagulation due to hypercholesterolemia.
Acknowledgments
The authors thank the Ministry of Science, Technology and Innovation (MOSTI), Malaysia, for sponsoring this research (e-Sciencefund 02-01-04-SF1453), and the staff of the Laboratory of Molecular Biomedicine for their assistance during the study.
Disclosure
The authors report no conflicts of interest in this work.
References
Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics – 2013 update. Circulation. 2013;127:e6–e245. | ||
Levine GN, Keaney JF Jr, Vita JA. Cholesterol reduction in cardiovascular disease – clinical benefits and possible mechanisms. N Engl J Med. 1995;332(8):512–521. | ||
Pike RN, Buckle AM, le Bonniec BF, Church FC. Control of the coagulation system by serpins. FEBS J. 2005;272(19):4842–4851. | ||
Calixto BJ. Efficacy, safety, quality control, marketing and regulatory guidelines for herbal medicines (phytotherapeutic agents). Braz J Med Biol Res. 2000;33(2):179–189. | ||
Yida Z, Imam MU, Ismail M, et al. Edible bird’s nest prevents high fat diet-induced insulin resistance in rats. J Diabetes Res. 2015;501: 760535. | ||
Chua KH, Lee TH, Nagandran K, et al. Edible bird’s nest extract as a chondro-protective agent for human chondrocytes isolated from osteoarthritic knee: in vitro study. BMC Complement Altern Med. 2013; 13(1):19. | ||
Aswir AR, Wan Nazaimoon WM. Effect of edible bird’s nest on cell proliferation and tumor necrosis factor-alpha (TNF-α) release in vitro. Int Food Res J. 2011;18(3):1073–1077. | ||
Yida Z, Imam MU, Ismail M. In vitro bioaccessibility and antioxidant properties of edible bird’s nest following simulated human gastro-intestinal digestion. BMC Complement Altern Med. 2014;14(1):468. | ||
Strukova S. Blood coagulation-dependent inflammation. Coagulation-dependent inflammation and inflammation-dependent thrombosis. Front Biosci. 2005;11:59–80. | ||
Pashkow FJ. Oxidative stress and inflammation in heart disease: do antioxidants have a role in treatment and/or prevention? Int J Inflamm. 2011;2011:514623. | ||
Scandinavian Simvastatin Survival Study Group. Randomised trial of cholesterol lowering in 4,444 patients with coronary heart disease: the Scandinavian Simvastatin Survival Study (4S). Lancet. 1994; 344(8934):1383–1389. | ||
Heart Protection Study Collaborative Group. MRC/BHF heart protection study of cholesterol lowering with simvastatin in 20 536 high-risk individuals: a randomised placebo-controlled trial. Lancet. 2002; 360(9326):7–22. | ||
Wada H, Mori Y, Kaneko T, et al. Hypercoagulable state in patients with hypercholesterolemia: effects of pravastatin. Clin Ther. 1991;14(6):829–834. | ||
Koutts J. Clinching the diagnosis: assessment of hemostatic function. Pathology. 1985;17(4):643–647. | ||
Simanek RR, Vormittag C, Ay G, et al. High platelet count associated with venous thromboembolism in cancer patients: results from the Vienna Cancer and Thrombosis Study (CATS). J Throm Haemost. 2010;8(1):114–120. | ||
Owens AP, Byrnes JR, Mackman N. Hyperlipidemia, tissue factor, coagulation, and simvastatin. Trends Cardiovasc Med. 2014;24(3):95–98. | ||
Imam MU, Ishaka A, Der Juin O, et al. Germinated brown rice regulates hepatic cholesterol metabolism and cardiovascular disease risk in hypercholesterolemic rats. J Funct Foods. 2014;8:193–203. | ||
Rothenbacher D, Brenner H, März W, Koenig W. Adiponectin, risk of coronary heart disease and correlations with cardiovascular risk markers. Eur Heart J. 2005;26(16):1640–1646. | ||
Faber DR, De Groot PG, Visseren FLJ. Role of adipose tissue in haemostasis, coagulation and fibrinolysis. Obes Rev. 2009;10(5):554–563. | ||
Koh KK, Quon MJ, Han SH, et al. Simvastatin improves flow-mediated dilation but reduces adiponectin levels and insulin sensitivity in hypercholesterolemic patients. Diabetes Care. 2008;31(4):776–782. | ||
Wannamethee SG, Tchernova J, Whincup P, et al. Plasma leptin: associations with metabolic, inflammatory and haemostatic risk factors for cardiovascular disease. Atherosclerosis. 2007;191(2):418–426. | ||
Eikelboom JW, Hankey GJ, Thom J, et al. Incomplete inhibition of thromboxane biosynthesis by acetylsalicylic acid determinants and effect on cardiovascular risk. Circulation. 2008;118(17):1705–1712. | ||
Brill A, Fuchs TA, Chauhan AK, et al. von Willebrand factor–mediated platelet adhesion is critical for deep vein thrombosis in mouse models. Blood. 2011;117(4):1400–1407. | ||
Wilson JR, Kapoor SC. Contribution of prostaglandins to exercise-induced vasodilation in humans. Am J Physiol Heart Circ Physiol. 1993;265(1):H171–H175. | ||
Förstermann U, Sessa WC. Nitric oxide synthases: regulation and function. Eur Heart J. 2012;33(7):829–837. | ||
Mollace V, Muscoli C, Masini E, Cuzzocrea S, Salvemini D. Modulation of prostaglandin biosynthesis by nitric oxide and nitric oxide donors. Pharmacol Rev. 2005;57(2):217–252. | ||
Wiman B. Plasminogen activator inhibitor 1 (PAI-1) in plasma: its role in thrombotic disease. Thromb Haemost. 1995;74(1):71–76. | ||
Jacobs DR, Tapsell LC. Food, not nutrients, is the fundamental unit in nutrition. Nutr Rev. 2007;65:439–450. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.