Back to Journals » Drug Design, Development and Therapy » Volume 9
Ceftobiprole for the treatment of pneumonia: a European perspective
Authors Liapikou A, Cillóniz C, Torres A
Received 2 December 2014
Accepted for publication 31 December 2014
Published 18 August 2015 Volume 2015:9 Pages 4565—4572
DOI https://doi.org/10.2147/DDDT.S56616
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Shu-Feng Zhou
Adamantia Liapikou,1 Catia Cillóniz,2 Antonio Torres2
16th Respiratory Department, Sotiria Chest Diseases Hospital, Athens, Greece; 2Pulmonology Department, Clinic Institute of Thorax (ICT), Hospital Clinic of Barcelona, Spain Insitut d’Investigacions Biomèdiques August Pi i Sunyer (IDIBAPS), Barcelona, Spain
Abstract: Ceftobiprole, a new broad spectrum, parenteral cephalosporin, exhibits potent in vitro activity against a number of Gram-positive pathogens, including methicillin-resistant Staphylococcus aureus and penicillin-resistant Streptococcus pneumoniae, and Gram-negative pathogens associated with hospital-acquired pneumonia (HAP) and community-acquired pneumonia (CAP). Ceftobiprole has demonstrated noninferiority in two large-scale pivotal studies comparing it to ceftriaxone with or without linezolid in CAP, with clinical cure rates 86.6% versus 87.4%, or ceftazidime in HAP, with clinical cure rates of 77% versus 76%, respectively. However, ceftobiprole was inferior in the subgroup of patients undergoing mechanical ventilation. Ceftobiprole has so far demonstrated a good safety profile in preliminary studies, with similar tolerability to comparators. The most commonly observed adverse events of ceftobiprole included headache and gastrointestinal upset. It is the first cephalosporin monotherapy approved in the EU for the treatment of both CAP and HAP (excluding ventilator-associated pneumonia).
Keywords: antibiotic resistance, methicillin-resistant staphylococci, community-acquired pneumonia, hospital-acquired pneumonia, cephalosporins
Introduction
Community-acquired pneumonia (CAP) is a significant cause of morbidity and mortality in developed countries, accounting for a considerable number of hospital admissions, especially in the elderly.1,2 Hospital-acquired pneumonia (HAP) is associated with significant mortality and has been reported to account for >25% of all infections in intensive care units.3 HAPs dramatically increase both the hospital length of stay and the cost of care, and are associated with an overall mortality of 27%–51%,4 the poorer prognosis being reported in the elderly.
The most common bacteria causing CAP are Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis,1,2 and the most common bacteria causing HAP are Enterobacteriaceae (such as Klebsiella spp., Enterobacter spp., Serratia spp.), Staphylococcus aureus, Pseudomonas aeruginosa, and Acinetobacter baumannii.4,5 S. aureus is the predominant Gram-positive pathogen in HAP and ventilator-associated pneumonia (VAP).
Pathogens that are resistant to antibacterials, particularly methicillin-resistant S. aureus (MRSA) and multidrug-resistant (MDR) S. pneumoniae, are associated with poor outcomes and higher treatment cost.6 Presently, half of the deaths from clinical infection in Europe are associated with MDR bacteria.7,8 Despite a decrease in the incidence of MRSA infections in recent years, the proportion of S. aureus isolates reported as MRSA in 2012 was ≥25% in seven of 30 European countries that provided surveillance reports.9 Koulenti et al10 reported that MRSA was isolated in 16% of patients with nosocomial pneumonia (21.4% in HAP and 14.6% in VAP). A large, prospective study reporting 474 patients with VAP in Spain found that patients with MRSA VAP had significantly higher in-hospital mortality than patients with VAP caused by other microorganisms (59.5% versus 46.8%; P=0.02).11
The adequacy of empirical antimicrobial therapy is strongly predictive of hospital survival,3–5 making the definition of patients at risk for MDR pathogens a pivotal challenge.
Now with ceftaroline-fosamil and ceftobiprole, a new subclass of antimicrobials, cephalosporins with anti-MRSA activity has been introduced. Ceftobiprole medocaril is a newly approved drug in Europe for the treatment of CAP and HAP (excluding patients with VAP) in adults. This review focuses on the pharmacological properties of ceftobiprole and its clinical efficacy and tolerability in adult patients with CAP and HAP.
Mechanism of action and antimicrobial activity
Ceftobiprole medocaril is the prodrug form of ceftobiprole, which is an extended-spectrum cephalosporin that encompasses activity against both Gram-positive and most Gram-negative bacteria, including MRSA. It has bactericidal effects against community-acquired MRSA, vancomycin-resistant S. aureus, methicillin-resistant coagulase-negative staphylococci (CoNS), penicillin-resistant S. pneumoniae, Enterococcus faecalis, Clostridium perfringens, P. aeruginosa, and 85% of Enterobacteriaceae.12,13 (Figure 1)
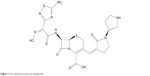  | Figure 1 The chemical structure of ceftobiprole. |
Like other cephalosporins, the binding of ceftobiprole to penicillin-binding proteins (PBPs) interferes with cell wall synthesis, inhibiting cell growth and ultimately leading to bacterial cell death.
Ceftobiprole was refractory to hydrolysis by the common staphylococcal PC1 β-lactamase, the class A TEM-1 β-lactamase, but labile to hydrolysis by class B, class D, and class A extended-spectrum β-lactamases, thus similar to cefepime and ceftazidime patterns of action.14 Ceftobiprole is relatively stable toward AmpC β-lactamases and has a strong affinity for PBPs, including PBP 2A, which mediates resistance to β-lactams in MRSA and methicillin (oxacillin)-resistant CoNS.15
It has high affinity against S. pneumoniae PBP 2× in penicillin-resistant and ceftriaxone-resistant strains and retains good in vitro activity against them.16,17 In a study by Kosowska et al18 testing its in vitro activity, Ceftobiprole minimum inhibitory concentrations (MICs) were similar to those of carbapenems against nearly all pneumococcal strains tested. Ceftobiprole was especially active against 299 drug-susceptible and -resistant pneumococci, with MIC of 50% (MIC50) and 90% (MIC90) values of 0.016 and 0.016 μg/mL (penicillin-susceptible isolates), 0.06 and 0.5 μg/mL (penicillin-intermediate isolates), and 0.5 and 1.0 μg/mL (penicillin-resistant isolates), respectively. Interestingly, during 50 serial passages in the presence of subinhibitory concentrations of antibiotics, the highest MIC achieved for ceftobiprole by a panel of ten pneumococcal isolates was 1 μg/mL.18
From the ceftobiprole SENTRY Antibiotic Surveillance Program in Europe (2005–2010), ceftobiprole continues to demonstrate high potency against the causative agents of CAP, with 99.3% of 4,443 S. pneumoniae isolates testing susceptible, as well as 2,052 strains of H. influenzae inhibited at MIC values of <0.5 μg/mL and 200 strains of M. catarrhalis inhibited at MIC values of <0.5 μg/mL.15 A 2008 US surveillance study on S. pneumoniae called, TRUST 12, showed that ceftobiprole was the most potent cephalosporin tested against S. pneumoniae with MIC50 (0.015 μg/mL) and MIC90 (0.5 μg/mL), with values two-fold lower than ceftriaxone.16
However, it is the bactericidal activity against MRSA that sets ceftobiprole apart from other cephalosporins.12 In time-kill analysis, ceftobiprole was bactericidal against community-acquired and hospital-acquired MRSA strains.19,20 In the SENTRY study, 4,147 of 15,426 (26.9%) S. aureus clinical isolates collected from Europe, Turkey, and Israel during 2005–2010 were MRSA, and 98.3% of these strains were susceptible to ceftobiprole.15 Moreover, ceftobiprole demonstrates in vitro activity against S. aureus resistant to linezolid, vancomycin, and daptomycin and ceftriaxone-resistant S. pneumonia.16,17,21 The CLASS study, evaluating the in vitro activity of ceftobiprole, published by Rossolini et al22 across 19 countries, further demonstrates the activity of ceftobiprole against all linezolid nonsusceptible staphylococcal isolates; a significant finding in light of recent reports of linezolid-resistant MRSA and CoNS strains.
Ceftobiprole also displayed bactericidal activity against Gram-negative pathogens comparable to cefepime, ceftazidime, or piperacillin-tazobactam in early studies.23,24 The SENTRY surveillance study,15 published in 2014, showed activity against P. aeruginosa (64.6% susceptible by the European Committee on Antimicrobial Susceptibility Testing nonspecies-specific susceptibility breakpoint of 4 μg/mL) that was lower than but similar to those of cefepime (78.6% susceptible) and ceftazidime (75.4% susceptible).
It has only moderate activity against Acinetobacter spp. and is not active against Proteus vulgaris, ceftazidime-nonsusceptible P. aeruginosa, Bacteroides spp., and some extended-spectrum β-lactamase-producing isolates.
Ceftobiprole has limited activity against Gram-negative anaerobes such as Bacteroides fragilis and non-fragilis Bacteroides spp. but is generally active against Clostridium spp. and Fusobacterium spp. (MIC90 8 mg/L).25,26
Pharmacokinetic/pharmacodynamic (PK/PD) profile
Like other β-lactams, ceftobiprole exhibits concentration-independent killing; studies in animals and of in vitro PK models have shown that the duration that the unbound concentration of an antibiotic remains above the MIC as a percentage of the dosing interval (%fT > MIC) is the PK/PD index that best correlates with drug-related response.
Muller et al27 using data from a randomized double-blind Phase III clinical trial, explored the relationship of ceftobiprole exposure with microbiological and clinical outcomes in patients with nosocomial pneumonia. According to regression analyses, a significant (P=0.0029) correlation was seen between %fT > MIC and clinical cure when %fT > MIC was >51.1%; a significant (P>0.0001) correlation was also observed for microbiological eradication when %fT > MIC was >62.2%.
Single- and multiple-dose PKs of ceftobiprole have been evaluated in healthy volunteers.28 With ceftobiprole 500 mg every 8 hours infused over 2 hours, the probability of attaining a 50% fT > MIC target was 80% for an MIC of 4 mg/L in subjects with normal renal function and 98.8% and 99.9% for MRSA and methicillin-sensitive S. aureus, respectively, with MIC90 1 and 0.5 mg/L, respectively.29
Utilizing Monte Carlo simulations, the probability of target attainment corresponding to 60% fT/MIC for ceftobiprole 500 mg every 8 hours (2-hour infusion) is 87.8% for AmpC-producing bacilli, with an MIC90 of 16 μg/mL and 94.1% for non-AmpC-producing bacilli for an MIC90 of <0.25 μg/mL.28,29
Peak levels of the active drug in plasma were achieved at the end of the 30-minute infusion. The apparent volume of distribution was similar to those reported to other β-lactams (18–20/L). Ceftobiprole demonstrates a low percentage of protein binding (16%)13 and is independent of the drug concentration across the range of 0.5–100 μg/mL. Renal clearance and urinary excretion of the free drug decreased while the elimination half-life increased with decreasing renal function. Patients with a creatinine clearance of 30<50 mL/min should have the dosing interval adjusted to every 12 hours. In patients with a creatinine clearance <30 mL/min, the dosing regimen should be 250 mg every 12 hours as a 2-hour intravenous infusion.13,19,29,30
Ceftobiprole exhibits a modest postantibiotic effect (30 minutes) for MRSA and a more prolonged postantibiotic effect (2 hours) for penicillin-resistant pneumococci.31
It does not inhibit the cytochrome P-450 isoenzyme system, so the possibility of drug–drug interactions is low.
Clinical efficacy in pneumonia
Because of its broad-spectrum activity against lower respiratory tract pathogens, ceftobiprole was evaluated in the treatment of CAP and HAP.
Clinical trials in CAP
Firstly, in a study conducted to compare the in vivo activity of ceftobiprole with that of ceftriaxone in a mouse model of acute pneumococcal pneumonia there was no significant difference in cumulative survival rates between the two antibiotics, but 100% survival was obtained at a five-fold lower total daily ceftobiprole dose than the dose of ceftriaxone used.32
The clinical efficacy of ceftobiprole in CAP demonstrated by the results of a randomized, double-blind, comparative study of ceftobiprole medocaril (500 mg every 8 hours infused over 2 hours) versus ceftriaxone (2 g once daily as 30-minute infusions) with or without linezolid (600 mg every 12 hours as 1-hour infusions, 7–14 days) in the treatment of subjects hospitalized with CAP.33 By the study design, linezolid was allowed for subjects with proven or suspected MRSA or ceftriaxone-resistant S. pneumoniae. Patients were stratified before randomization based on the Pneumonia Patient Outcomes Research Team (PORT); Pneumonia Severity Index (PSI), corresponding to class IV and V severity, and need for addition of linezolid versus placebo. If after day 3, patients met stability criteria, they were eligible to switch to oral cefuroxime 500 mg every 12 hours.
A typical bacterial pathogen was isolated at baseline in 28.8% of the intention-to-treat (ITT) population, with S. pneumoniae (n=68) and H. influenzae (n=26) being the most common. The ceftobiprole group had more polymicrobial infection than did the comparator group (20% versus 8%, P=0.016).
The severity of pneumonia at baseline had no effect on clinical cure rates at the test of cure visit between the two groups.
For the 469 clinically evaluable (CE) patients, cure rates were 86.6% versus 87.4% for ceftobiprole and comparator, respectively; in the ITT analysis of 638 CAP patients, these cure rates were 76.4% versus 79.3%, respectively, demonstrating the noninferiority of ceftobiprole compared to ceftriaxone in CAP therapy.
For the secondary end-point of clinical cure in patients with a pneumonia severity index ≥91, the cure rates for the above regimens were 90.2% and 84.5% compared with 85.6% and 88.3% for those with lower scores, respectively. In patients with CAP complicated by bacteremia, cure rates were 85.7% versus 85.7%; and with presence of systemic inflammatory response syndrome 84.6% versus 86.7% for ceftobiprole and ceftriafone linezolid therapy, respectively.
Referring to the secondary efficacy endpoint of microbiological eradication, noninferiority between the ceftobiprole and comparator groups was observed also. Subgroup analyses showed that among those who switched to oral cefuroxime, microbiological eradication rates were significantly lower with ceftobiprole medocaril than with ceftriaxone ± linezolid (89% versus 100%; n=37 and n=41, respectively).
Clinical trials in HAP
A Phase III randomized double-blind study was designed to compare the ceftobiprole medocaril (500 mg every 8 hours, infused over 2 hours) versus linezolid (600 mg every 12 hours as a 1-hour intravenous infusion) plus ceftazidime (2 g every 8 hours as a 2-hour intravenous infusion) for 7–14 days in 781 recruited subjects with clinical diagnosis of HAP, including VAP.34
The overall cure rates in the ITT population were 50% for ceftobiprole and 52.8% for combination therapy, and in the CE population, 69.3% in the ceftobiprole group and 71.6% demonstrating the noninferiority of ceftobiprole compared to the comparator group.34
Among patients without VAP, the cure rate in the ITT population was 77.4% in the ceftobiprole group and 76.3% in the comparator group, and in the CE population, it was 38.5% and 56.7%, respectively.
In the CE analysis set, a higher proportion of HAP (excluding VAP) patients in the ceftobiprole group than in the ceftazidime/linezolid group (CE, 86.9% versus 78.4%, respectively) showed early improvement (4 days after onset of therapy) (Table 1). The largest difference was for patients with a baseline culture positive for MRSA, with 94.7% in the ceftobiprole group having early improvement versus 52.6% in the ceftazidime/linezolid group (difference, 42.1%).35
The overall microbiological eradication rate in the ceftobiprole group was 62, 9% (73/116) compared to 67, 5% (81/120) in the ceftazidime/linezolid group. However, subjects with clinical cure 73/86 (85%) in the ceftobiprole group and 81/94 (86%) in the other group, had an outcome of microbiological eradication.
Of note, in patients with non-VAP, clinical cure rates were similar between ceftobiprole medocaril and ceftazidime plus linezolid recipients who had P. aeruginosa at baseline. Only for A. baumannii and Haemophilus spp. were numerically lower clinical cure and microbiological eradication rates observed in the ceftobiprole group.
Thirty-day pneumonia-specific mortality was similar between the two treatment groups, 5.9% and 5.6%, respectively (difference, 0.3%).
However, ceftobiprole was inferior in the subgroup of patients with VAP.13,34 The clinical cure rate in the CE population with VAP favored the linezolid/ceftazidime arm over the ceftobiprole arm, 56.7% versus 38.5%, respectively (P<0.05). Interestingly, in mechanically ventilated patients with non-VAP, clinical outcomes were in favor of ceftobiprole medocaril, suggesting that mechanical ventilation by itself is not associated with poor outcomes. In fact, of the 16 VAP patients ≤45 years with head trauma that were randomized to ceftobiprole, 12 were characterized as treatment failures compared to two out of the four assigned to the linezolid/ceftazidime group.
There are different explanations for this result: 1) It is thought by the authors that the small sample size and the substantial heterogeneity in baseline characteristics in the VAP subgroup may have contributed to the differential outcomes,34 and 2) the inferior outcome may have been the consequence of suboptimal concentrations of ceftobiprole at the site of infection, as a result of a change in the volume distribution owing to capillary leak from mechanical ventilation.36
In a more recent review,37 it was reported that in patients with normal PK and non-VAP, ceftobiprole is effective for the treatment of HAP in the recommended doses, but it is unlikely to achieve the desired PD targets when PK parameters are altered in VAP (eg, increased volume of distribution and clearance). Consequently, ceftobiprole medocaril is not approved for patients with VAP.
Tolerability
Ceftobiprole medocaril is generally well tolerated in patients with HAP or CAP in Phase III trials.33,34 The incidence of serious adverse events (AEs) in ceftobiprole medocaril versus comparator groups were 36.3% versus 31.9% in the HAP trial and 11.3% versus 11.5% in the CAP trial, respectively.38
The most common treatment-related AEs occurring in ceftobiprole recipients in the trials in patients with HAP or CAP included nausea, dysgeusia, diarrhea, infusion site reactions, vomiting, hepatic enzyme elevations, and hyponatremia (Table 2).
Particularly in the Nicholson et al trial,33 in patients with CAP the overall incidence of treatment-related AEs was higher in the ceftobiprole group, primarily owing to differences in rates of self-limited nausea and vomiting. There were no differences between groups for injection-site AEs, hyponatremia, or hepatic AEs.
In the Awad et al HAP study,34 ceftobiprole patients had fewer treatment-related events of diarrhea than patients treated with ceftazidime/linezolid (3.1% and 6.5%, respectively), whereas hyponatremia was observed more frequently with ceftobiprole than with ceftazidime/linezolid (4.4% and 2.6%, respectively).
One study found that the most common patient complaint was “caramel-like taste disturbances” (dysgeusia) when the medication was administered at higher doses. This was found to be most likely a result of the metabolism of ceftobiprole medocaril to diacetyl.28
The drug is contraindicated in patients with severe hypersensitivity (eg, anaphylactic reaction) to any other type of β-lactam antibacterial agent.
Regulatory affairs
Following approval under the European decentralized procedure, ceftobiprole has received national licenses in Austria, Belgium, Denmark, Finland, France, Germany, Norway, Spain, Sweden, and the United Kingdom; national authorization in Italy and Luxembourg; and reimbursement and pricing authorization in several countries including Spain is ongoing.39 It is also in preregistration stage in Switzerland for these indications.
Basilea Pharmaceutica Ltd. entered into an agreement with Quintiles in July 2014 for commercialization in key European countries of Zevtera®/Mabelio® (ceftobiprole medocaril) for patients with HAP and CAP.
In November 2008, the US Food and Drug Administration (FDA) declined to approve ceftobiprole, citing data integrity concerns with two of the supporting studies. The FDA determined that data from the two mentioned studies (BAP00154 and BAP00414) could not be relied on; inspections and audits of approximately one third of the clinical trial sites found the data from a large proportion of these sites to be unreliable or unverifiable.40
Expert opinion
One of the main challenges in the management of HAP and CAP is to overcome the resistance issues, which have become so important and common over the past several years. Management of pulmonary infections caused by resistant Gram-positive pathogens, particularly MRSA, remains a significant challenge. An analysis of pneumococcal resistance rates in the US spanning the years 1998 to 2009 demonstrated remarkable increases in nonsusceptibility to commonly used β-lactam agents.41
Treatment guidelines for HAP recommend rapid empiric therapy with combinations of antibiotics based on local resistance patterns and patient risk factors.4 It has been recommended that initial empirical monotherapy should be used whenever possible to reduce the risk of MDR development and adverse outcomes.
Cephalosporins are effective first-line therapies for many bacterial infections. Ceftobiprole, a new parenteral cephalosporin has notable activity against Gram-positive pathogens, including MRSA, and some major Gram-negative pathogens, particularly P. aeruginosa. It was shown to be noninferior to ceftazidime plus linezolid for the treatment of HAP and ceftriaxone ± linezolid for the treatment of CAP (in hospitalized patients) in two randomized clinical trials.33,34
Regarding, clinical efficacy in CAP treatment, further studies are needed to determine the efficacy of ceftobiprole in more severely ill CAP patients.
Alternatively, the inferiority of ceftobiprole to linezolid plus ceftazidime in the treatment of VAP raises serious concern about the efficacy of the drug in achieving adequate concentrations in the alveolar compartment of ventilated patients. We believe that if a larger dose of ceftobiprole had been used, the results might have been different. After all, a dose of 1,000 mg every 8 hours has been well tolerated in healthy volunteers.42 Therefore, further investigation assessing the efficacy of ceftobiprole medocaril in patients with VAP may be warranted.
Although ceftobiprole was not shown to induce resistance in clinical trials, recent studies demonstrate that low- and high-level ceftobiprole resistance develop when isolates are exposed to sub-MIC concentrations.43 As with other cephalosporins, ceftobiprole may lose activity as the degree of expression of efflux pumps increases by bacteria such as Pseudomonas spp. Although it is impossible to know how quickly resistance to ceftobiprole will emerge in a clinical setting, the emergence of ceftobiprole resistance via chromosomal mutation was not readily observable in the laboratory.
Ceftobiprole medocaril is generally well tolerated in clinical trials, and 10% of patients discontinued the treatment because of AEs. Importantly, it has no significant impact on the normal human intestinal flora, and Clostridium difficile colitis is uncommon with ceftobiprole medocaril.44
Ceftobiprole offers a number of advantages in potency, spectrum, and β-lactamase stability compared to currently marketed third-generation carbapenems and other β-lactams. It may be a powerful antibiotic prescribed as monotherapy in serious CAP and HAP (excluding VAP) as initial empirical treatment.
Conclusion
Ceftobiprole is a new parenteral cephalosporin with bactericidal activity against Gram-positive pathogens including MRSA, responsible for pneumonia, while preserving the anti-Gram-negative activity of existing broad-spectrum cephalosporins. Clinical data from two large Phase III studies have demonstrated that ceftobiprole medocaril is noninferior to the combination of high-dose ceftazidime and linezolid for the treatment of HAP (excluding VAP) and noninferior to high-dose ceftriaxone with or without linezolid for the treatment of CAP requiring hospitalization. Therefore ceftobiprole represents a safe and effective treatment of hospitalized CAP and HAP (excluding VAP).
Acknowledgment
Catia Cillóniz and Antonio Torres are supported by: SGR 2014/661, Ciber de Enfermedades Respiratorias (Ciberes CB06/06/0028), Pneumonia Corporate Research Program (CRP). The Ciberes is an initiative of the ISCIII. SGR: Support to research groups of Catalunya.
Disclosure
The authors report no conflicts of interest in this work.
References
Liapikou A, Rosales-Mayor E, Torres A. The management of severe community acquired pneumonia in the intensive care unit. Expert Rev Respir Med. 2014;8(3):293–303. | ||
Welte T, Torres A, Nathwani D. Clinical and economic burden of community-acquired pneumonia among adults in Europe. Thorax. 2012;67(1):71–79. | ||
Torres A, Ewig S, Lode H, Carlet J; European HAP working group. Defining, treating and preventing hospital acquired pneumonia: European perspective. Intensive Care Med. 2009;35(1):9–29. | ||
American Thoracic Society; Infectious Disease Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005;171(4):388–416. | ||
Rello J, Ulldemolins M, Lisboa T, et al; EU-VAP/CAP Study Group. Determinants of prescription and choice of empirical therapy for hospital-acquired and ventilator-associated pneumonia. Eur Respir J. 2011;37(6):1332–1339. | ||
Chastre J, Blasi F, Masterton RG, Rello J, Torres A, Welte T. European perspective and update on the management of nosocomial pneumonia due to methicillin-resistant Staphylococcus aureus after more than 10 years of experience with linezolid. Clin Microbiol Infect. 2014;20(Suppl 4):19–36. | ||
Watson R. Multidrug resistance responsible for half of deaths from healthcare associated infections in Europe. BMJ. 2008;336(7656):1266–1267. | ||
Vincent JL, Rello J, Marshall J, et al; EPIC II Group of Investigators. International study of the prevalence and outcomes of infection in intensive care units. JAMA. 2009;302(21):2323–2329. | ||
European Centre for Disease Prevention and Control [webpage on the Internet]. Surveillance of invasive bacterial diseases in Europe 2010 and 2011 [updated December 20, 2013]. Available from http://www.ecdc.europa.eu/en/press/news/_layouts/forms/News_DispForm.aspx?List=8db7286c-fe2d-476c-9133-18ff4cb1b568&ID=932. Accessed December 1, 2013. | ||
Koulenti D, Lisboa T, Brun-Buisson C, et al; EU-VAP/CAP Study Group. Spectrum of practice in the diagnosis of nosocomial pneumonia in patients requiring mechanical ventilation in European intensive care units. Crit Care Med. 2009;37(8):2360–2368. | ||
Bouza E, Giannella M, Bunsow E, et al; Gregorio Marañón Task Force for Pneumonia (GANG). Ventilator-associated pneumonia due to methicillin-resistant Staphylococcus aureus: risk factors and outcome in a large general hospital. J Hosp Infect. 2012;80(2):150–155. | ||
Pillar CM, Aranza MK, Shah D, Sahm DF. In vitro activity profile of ceftobiprole, an anti-MRSA cephalosporin, against recent gram-positive and gram-negative isolates of European origin. J Antimicrob Chemother. 2008;61(3):595–602. | ||
El Solh A. Ceftobiprole: a new broad spectrum cephalosporin. Expert Opin Pharmacother. 2009;10(10):1675–1686. | ||
Queenan AM, Shang W, Kania M, Page MG, Bush K. Interactions of ceftobiprole with beta-lactamases from molecular classes A to D. Antimicrob Agents Chemother. 2007;51(9):3089–3095. | ||
Farrell DJ, Flamm RK, Sader HS, Jones RN. Ceftobiprole activity against over 60,000 clinical bacterial pathogens isolated in Europe, Turkey, and Israel from 2005 to 2010. Antimicrob Agents Chemother. 2014;58(7):3882–3888. | ||
Davies TA, Flamm RK, Lynch AS. Activity of ceftobiprole against Streptococcus pneumoniae isolates exhibiting high-level resistance to ceftriaxone. Int J Antimicrob Agents. 2012;39(6):534–538. | ||
Davies TA, Page MG, Shang W, Andrew T, Kania M, Bush K. Binding of ceftobiprole and comparators to the penicillin-binding proteins of Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus, and Streptococcus pneumoniae. Antimicrob Agents Chemother. 2007;51(7):2621–2624. | ||
Kosowska K, Hoellman DB, Lin G, et al. Antipneumococcal activity of ceftobiprole, a novel broad-spectrum cephalosporin. Antimicrob Agents Chemother. 2005;49(5):1932–1942. | ||
Dauner DG, Nelson RE, Taketa DC. Ceftobiprole: a novel, broad-spectrum cephalosporin with activity against methicillin-resistant Staphylococcus aureus. Am J Health Syst Pharm. 2010;67(12): 983–993. | ||
Lin G, Appelbaum PC. Activity of ceftobiprole compared with those of other agents against Staphylococcus aureus strains with different resistotypes by time-kill analysis. Diagn Microbiol Infect Dis. 2008;60(2):233–235. | ||
Leonard SN, Cheung CM, Rybak MJ. Activities of ceftobiprole, linezolid, vancomycin, and daptomycin against community-associated and hospital-associated methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 2008;52(8):2974–2976. | ||
Rossolini GM, Dryden MS, Kozlov RS, et al. Comparative activity of ceftobiprole against Gram-positive and Gram-negative isolates from Europe and the Middle East: the CLASS study. J Antimicrob Chemother. 2011;66(1):151–159. | ||
Bogdanovich T, Clark C, Ednie L, et al. Activities of ceftobiprole, a novel broad-spectrum cephalosporin, against Haemophilus influenzae and Moraxella catarrhalis. Antimicrob Agents Chemother. 2006;50(6):2050–2057. | ||
Rouse MS, Hein MM, Anguita-Alonso P, Steckelberg JM, Patel R. Ceftobiprole medocaril (BAL5788) treatment of experimental Haemophilus influenzae, Enterobacter cloacae, and Klebsiella pneumoniae murine pneumonia. Diagn Microbiol Infect Dis. 2006;55(4):333–336. | ||
Arias CA, Singh KV, Panesso D, Murray BE. Time-kill and synergism studies of ceftobiprole against Enterococcus faecalis, including beta-lactamase-producing and vancomycin-resistant isolates. Antimicrob Agents Chemother. 2007;51(6):2043–2047. | ||
Ednie L, Shapiro S, Appelbaum PC. Antianaerobe activity of ceftobiprole, a new broad-spectrum cephalosporin. Diagn Microbiol Infect Dis. 2007;58(1):133–136. | ||
Muller AE, Punt N, Mouton JW. Exposure to ceftobiprole is associated with microbiological eradication and clinical cure in patients with nosocomial pneumonia. Antimicrob Agents Chemother. 2014;58(5):2512–2519. | ||
Schmitt-Hoffmann A, Nyman L, Roos B, et al. Multiple-dose pharmacokinetics and safety of a novel broad-spectrum cephalosporin (BAL5788) in healthy volunteers. Antimicrob Agents Chemother. 2004;48(7):2576–2580. | ||
Lodise TP, Pypstra R, Kahn JB, et al. Probability of target attainment for ceftobiprole as derived from a population pharmacokinetic analysis of 150 subjects. Antimicrob Agents Chemother. 2007;51(7):2378–2387. | ||
Hoffman-Roberts HL, C Babcock E, Mitropoulos IF. Investigational new drugs for the treatment of resistant pneumococcal infections. Expert Opin Investig Drugs. 2005;14(8):973–995. | ||
Pankuch GA, Appelbaum PC. Postantibiotic effect of ceftobiprole against 12 Gram-positive organisms. Antimicrob Agents Chemother. 2006;50(11):3956–3958. | ||
Azoulay-Dupuis E, Bédos JP, Mohler J, Schmitt-Hoffmann A, Schleimer M, Shapiro S. Efficacy of BAL5788, a prodrug of cephalosporin BAL9141, in a mouse model of acute pneumococcal pneumonia. Antimicrob Agents Chemother. 2004;48(4):1105–1111. | ||
Nicholson SC, Welte T, File TM, et al. A randomised, double-blind trial comparing ceftobiprole medocaril with ceftriaxone with or without linezolid for the treatment of patients with community-acquired pneumonia requiring hospitalisation. Int J Antimicrob Agents. 2012;39(3):240–246. | ||
Awad SS, Rodriguez AH, Chuang YC, et al. A Phase 3 randomized double-blind comparison of ceftobiprole medocaril versus ceftazidime plus linezolid for the treatment of hospital-acquired pneumonia. Clin Infect Dis. Epub 2014 Apr 9. | ||
Welte et al. Early clinical response in a randomized controlled phase 3 study of ceftobiprole versus ceftriaxone with or without linezolid in patients with community-acquired pneumonia requiring hospitalization. Poster presented at: 24th European Congress of Clinical Microbiology and Infectious Diseases (ECCMID); May 10–13, 2014; Barcelona, Spain. Poster eP431. | ||
Roberts JA, Lipman J. Pharmacokinetic issues for antibiotics in the critically ill patient. Crit Care Med. 2009;37(3):840–851; quiz 859. | ||
Lagacé-Wiens PR, Rubinstein E. Pharmacokinetic and pharmacodynamics evaluation of ceftobiprole medocaril for the treatment of hospital-acquired pneumonia. Expert Opin Drug Metab Toxicol. 2013;9(6):789–799. | ||
Basilea Pharmaceutica Ltd. Public Assessment Report: Zevtera 500 mg Powder for Concentrate for Solution for Infusion. London: Medicines and Healthcare Products Regulatory Agency; 2013. Available from: http://www.mhra.gov.uk/home/groups/par/documents/websiteresources/con369256.pdf. Accessed 15 July, 2014. | ||
Basilea Pharmaceutica Ltd. Available from: http://www.basilea.com/News-and-Media/Basilea-reports-2014-half-year-financials-ceftobiprole-launch-in-Germany-planned-for-second-half-of-2014/6bef004a-03b7-6db4-bccd-7c8db23eb16c. Accessed March 15, 2010. | ||
Johnson & Johnson. FDA issues complete response letter for ceftobiprole [webpage on the Internet]. New Brunswick, NJ: Johnson & Johnson Services Inc; 2009. Available from: www.jnj.com/connect/news/all/20091230_090000. Accessed March 15, 2010. | ||
Jones RN, Sader HS, Moet GJ, Farrell DJ. Declining antimicrobial susceptibility of Streptococcus pneumoniae in the United States: report from the SENTRY Antimicrobial Surveillance Program (1998–2009). Diagn Microbiol Infect Dis. 2010;68(3):334–336. | ||
Schmitt-Hoffmann A, Roos B, Schleimer M, et al. Single-dose phaimacokinelics and safety of a novel broad-spectrum cephalosporin (BAL5788) in healthy volunteers. Antimicrob Agents Chemother. 2004;48(7):2570–2575. | ||
Basilea Pharmaceutica Ltd. FDA issues ceftobiprole complete response letter [webpage on the Internet]. Basel: Basilea Pharmaceutica Ltd.; 2009. Available from: http://www.basilea.com/News-and-Media/FDA-issues-ceftobiprole-Complete-Response-Letter/317. Accessed March 15, 2010. | ||
Nerandzic MM, Donskey CJ. Effect of ceftobiprole treatment on growth of and toxin production by Clostridium difficile in cecal contents of mice. Antimicrob Agents Chemother. 2011;55(5):2174–2177. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


