Back to Journals » Drug Design, Development and Therapy » Volume 9
Alisertib induces cell cycle arrest and autophagy and suppresses epithelial-to-mesenchymal transition involving PI3K/Akt/mTOR and sirtuin 1-mediated signaling pathways in human pancreatic cancer cells
Authors Wang F, Li H, Yan X, Zhou Z, Yi Z, He Z, Pan S, Yang Y, Wang Z, Zhang X, Yang T, Qiu J, Zhou S
Received 1 October 2014
Accepted for publication 21 October 2014
Published 17 January 2015 Volume 2015:9 Pages 575—601
DOI https://doi.org/10.2147/DDDT.S75221
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Prof. Dr. Wei Duan
Feng Wang,1,2 Hai Li,3 Xiao-Gang Yan,4 Zhi-Wei Zhou,2 Zhi-Gang Yi,5 Zhi-Xu He,6 Shu-Ting Pan,7 Yin-Xue Yang,3 Zuo-Zheng Wang,1 Xueji Zhang,8 Tianxing Yang,9 Jia-Xuan Qiu,7 Shu-Feng Zhou2
1Department of Hepatobiliary Surgery, General Hospital, Ningxia Medical University, Yinchuan, People’s Republic of China; 2Department of Pharmaceutical Sciences, College of Pharmacy, University of South Florida, Tampa, FL, USA; 3Department of Colorectal Surgery, General Hospital, Ningxia Medical University, 4Department of Oncological Surgery, The First People’s Hospital of Yinchuan, 5Department of General Surgery, Changqing Yangehu Hospital, Yinchuan, 6Guizhou Provincial Key Laboratory for Regenerative Medicine, Stem Cell and Tissue Engineering Research Center and Sino-US Joint Laboratory for Medical Sciences, Guiyang Medical University, Guiyang, 7Department of Oral and Maxillofacial Surgery, The First Affiliated Hospital of Nanchang University, Nanchang, 8Research Center for Bioengineering and Sensing Technology, University of Science and Technology Beijing, Beijing, People’s Republic of China; 9Department of Internal Medicine, University of Utah and Salt Lake Veterans Affairs Medical Center, Salt Lake City, UT, USA
Abstract: Pancreatic cancer is the most aggressive cancer worldwide with poor response to current therapeutics. Alisertib (ALS), a potent and selective Aurora kinase A inhibitor, exhibits potent anticancer effects in preclinical and clinical studies; however, the effect and underlying mechanism of ALS in the pancreatic cancer treatment remain elusive. This study aimed to examine the effects of ALS on cell growth, autophagy, and epithelial-to-mesenchymal transition (EMT) and to delineate the possible molecular mechanisms in human pancreatic cancer PANC-1 and BxPC-3 cells. The results showed that ALS exerted potent cell growth inhibitory, pro-autophagic, and EMT-suppressing effects in PANC-1 and BxPC-3 cells. ALS remarkably arrested PANC-1 and BxPC-3 cells in G2/M phase via regulating the expression of cyclin-dependent kinases 1 and 2, cyclin B1, cyclin D1, p21 Waf1/Cip1, p27 Kip1, and p53. ALS concentration-dependently induced autophagy in PANC-1 and BxPC-3 cells, which may be attributed to the inhibition of phosphatidylinositol 3-kinase (PI3K)/protein kinase B (Akt)/mammalian target of rapamycin (mTOR), p38 mitogen-activated protein kinase (p38 MAPK), and extracellular signal-regulated kinases 1 and 2 (Erk1/2) but activation of 5'-AMP-dependent kinase signaling pathways. ALS significantly inhibited EMT in PANC-1 and BxPC-3 cells with an increase in the expression of E-cadherin and a decrease in N-cadherin. In addition, ALS suppressed the expression of sirtuin 1 (Sirt1) and pre-B cell colony-enhancing factor/visfatin in both cell lines with a rise in the level of acetylated p53. These findings show that ALS induces cell cycle arrest and promotes autophagic cell death but inhibits EMT in pancreatic cancer cells with the involvement of PI3K/Akt/mTOR, p38 MAPK, Erk1/2, and Sirt1-mediated signaling pathways. Taken together, ALS may represent a promising anticancer drug for pancreatic cancer treatment. More studies are warranted to investigate other molecular targets and mechanisms and verify the efficacy and safety of ALS in the treatment of pancreatic cancer.
Keywords: alisertib, pancreatic cancer, cell cycle, autophagy, EMT, Sirt1
Introduction
Pancreatic cancer is the most aggressive cancer worldwide with a 5-year survival rate of less than 6%.1,2 Pancreatic cancer is the 12th most common cancer, and there were 338,000 cases diagnosed with pancreatic cancer in 2012.1,2 In US, pancreatic cancer accounts for approximately 3% of all cancers and approximately 7% of cancer-related deaths.1,2 In 2013, there were 45,220 cases diagnosed with pancreatic cancer, and there will be 46,420 new cases of pancreatic cancer in 2014.2 In People’s Republic of China, pancreatic cancer is the sixth leading cause of cancer-related death with a lower 5-year survival rate of 1%–3%.1,3,4 Currently, the main therapies for pancreatic cancer treatment include surgery, chemotherapy, radiotherapy, immunotherapy, and vaccine therapy.1 However, the therapeutic outcome of pancreatic cancer is disappointing in clinic. It is mainly due to the resistance to cytotoxic chemotherapeutic agents and/or radiotherapy. Thus, there is an urgent need for new anticancer agents that can kill pancreatic cancer cells with improved clinical therapeutic outcomes.
Recently, accumulating evidence demonstrates that targeting programmed cell death is a promising strategy to treat pancreatic cancer, mainly including apoptosis and autophagy,5–9 although the therapeutic and clinical implications are not fully understood. In particular, autophagy, the mammalian target of rapamycin (mTOR)-mediated cell death, has an inconclusive role in the regulation of cancer cell survival and cell death with the involvement of a number of Atg proteins and other regulating molecules, such as 5′-AMP-dependent kinase (AMPK), p38 mitogen-activated protein kinase (p38 MAPK), and sirtuin 1 (Sirt1).5,10 On the other hand, emerging evidence suggests that the epithelial-to-mesenchymal transition (EMT) process has been implicated in the initiation, development, progression, metastasis, and drug resistance of pancreatic cancer with a dramatic loss in the epithelial features but gain in the mesenchymal characteristics.11,12 Consequently, EMT facilitates metastatic dissemination of pancreatic cancer cells and instigates drug resistance.13,14 Therefore, halt of EMT may represent a novel strategy for the treatment of pancreatic cancer via inhibition of metastasis and promotion of sensitivity to chemotherapeutics.
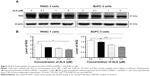
Aurora kinases, a family of serine/threonine kinases, consist of three members including Aurora kinase A/B/C (AURKA/B/C). Aurora kinases have a pivotal role in the regulation of mitosis, and the expression and activity of Aurora kinases are tightly regulated.15 Abnormalities in the expression and/or activity of Aurora kinases can cause genetic instability, aneuploidy, turmorigenesis, and cell death.15 AURKA is a key cell cycle regulator critical for mitotic events.16 Aberrant expression and activity of AURKA often occur in various malignancies, including the bladder, breast, colon, liver, ovaries, pancreas, stomach, and esophagus cancer.17 Thus, targeting AURKA has been proposed to be a very promising strategy for cancer treatment and a number of AURKA inhibitors have been developed.18 Alisertib (ALS, MLN8237, Figure 1A), an investigational small-molecule inhibitor, selectively inhibits AURKA.19 Currently, ALS is under various Phase I and Phase II clinical trials for advanced solid tumors and hematologic malignancies, although the underlying mechanism is not fully understood.20 To date, there is only one report on the anticancer effect of ALS in the treatment of pancreatic ductal adenocarcinoma.21 The effect of ALS on anchorage-independent growth in pancreatic ductal adenocarcinoma cell lines and growth of patient-derived xenografts was variable. RalA Ser194 phosphorylation levels in pancreatic ductal adenocarcinoma cell lines or patient-derived xenografted tumors did not correlate with the response to ALS. Notably, Ki67 (the cell proliferation-related antigen) was identified as a possible early biomarker for predicting the response to ALS in patient-derived xenografts.21 These findings indicate that ALS is an effective anticancer drug for a subgroup of patients with pancreatic ductal adenocarcinoma via signaling pathways independent of RalA Ser194 phosphorylation. However, this study did not investigate how ALS kills pancreatic cancer cells via inhibition of AURKA and why RalA Ser194 phosphorylation is not required for the ALS’s killing effect. There are only few data on the anticancer effect and underlying mechanisms of ALS in the treatment of pancreatic cancer. In this regard, we hypothesized that ALS inhibited the growth of pancreatic cancer cells via phosphatidylinositol 3-kinase (PI3K)/protein kinase B (Akt)/mTOR and Sirt1-mediated signaling pathways. To test this hypothesis, we investigated the anticancer effects and possible mechanisms of ALS in human pancreatic cancer PANC-1 and BxPC-3 cells with a focus on the effects of ALS on cell cycle, autophagy, EMT, and redox homeostasis.
Materials and methods
Chemicals and reagents
ALS (also known as MLN8237) was purchased from Selleckchem Inc (Houston, TX, USA). Dulbecco’s Modified Eagle’s Medium (DMEM) and RPMI-1640 medium were obtained from Corning Cellgro Inc (Herndon, VA, USA). Dimethyl sulfoxide (DMSO), Dulbecco’s phosphate-buffered saline (PBS), heat-inactivated fetal bovine serum (FBS), propidium iodide (PI), RNase A, and thiazolyl blue tetrazolium bromide (MTT) were purchased from Sigma-Aldrich Inc (St Louis, MO, USA). Phenol red-free culture medium was obtained from Invitrogen Inc (Carlsbad, CA, USA). Cyto-ID® Autophagy detection kit was obtained from Enzo Life Sciences Inc (Farmingdale, NY, USA). Polyvinylidene difluoride (PVDF) membrane was purchased from Bio-Rad (Hercules, CA, USA). Pierce BCA protein assay kit, skim milk, and Western blotting substrate were obtained from Thermo Scientific Inc (Hudson, NH, USA). Primary antibodies against human cyclin-dependent kinase (CDK) 1/CDC2, CDK2, cyclin B1, cyclin D1, p21 Waf1/Cip1, p27 Kip1, p53, Sirt1, p38 MAPK, phosphorylated (p-) p38 MAPK at Thr180/Tyr182, AMPK, p-AMPK at Thr172, PI3K, p-PI3K/p85 at Tyr458, Akt, p-Akt at Ser473, mTOR, p-mTOR at Ser2448, p-p44/42 MAPK (Erk1/2) at Thr202/204, p-53 at Ser392, Ac-p53 at Thr288, phosphatase and tensin homolog (PTEN), beclin 1, microtubule-associated protein 1A/1B-light chain 3 (LC3)-I, LC3-II, and nuclear factor (erythroid-derived 2)-like 2 (Nrf2), and Aurora (#3875) and EMT (#9782) antibody sampler kits were all purchased from Cell Signaling Technology Inc (Beverly, MA, USA). The antibodies against human β-actin and pre-B cell colony-enhancing factor (PBEF/visfatin), also called nicotinamide phosphoribosyltransferase, were obtained from Santa Cruz Biotechnology Inc (Santa Cruz, CA, USA).
Cell lines and cell culture
Two human pancreatic cancer cell lines PANC-1 and BxPC-3 were obtained from the American Type Culture Collection (Manassas, VA, USA) and cultured in DMEM (PANC-1 cells) and RPMI-1640 (BxPC-3 cells) medium supplemented with 10% heat-inactivated FBS and 1% penicillin/streptomycin. The cells were maintained in a 5% CO2/95% air-humidified incubator at 37°C. ALS was dissolved in DMSO with a stock concentration of 100 mM and the stock solution was stored at −20°C. ALS was freshly diluted to the predetermined concentrations with culture medium. The final concentration of DMSO was at 0.05% (v/v). The control cells received the vehicle only.
Cell viability assay
The effect of ALS on the viability of PANC-1 and BxPC-3 cells was examined using MTT assay. Briefly, PANC-1 and BxPC-3 cells were seeded into a 96-well plate at a density of 8,000 cells/well. After cells were seeded for 24 hours at a volume of 100 μL medium, the PANC-1 and BxPC-3 cells were treated with ALS at concentration ranging from 0.1 μM to 50 μM for 24 hours. Following the ALS treatment, 10 μL of MTT stock solution (5 mg/mL) was added to each well and incubated for 4 hours. Then the medium was carefully aspirated and 100 μL of DMSO was added into each well. The cell viability was determined by reduction of MTT after 10-minute incubation at 37°C. The absorbance was measured using a Synergy H4 Hybrid microplate reader (BioTek Inc, Winooski, VT, USA) at a wavelength of 450 nm. The IC50 values were determined using the relative viability over ALS concentration curve.
Cell cycle distribution analysis
The effect of ALS on the cell cycle distribution of PANC-1 and BxPC-3 cells was determined using flow cytometry as described previously.22 Briefly, PANC-1 and BxPC-3 cells were seeded into 60 mm Petri dishes. After cells were seeded for 24 hours, the cells reached ~75% confluence and were then treated with ALS at concentrations of 0.1 μM, 1 μM, and 5 μM for 24 hours. In separate experiments, PANC-1 and BxPC-3 cells were treated with 5 μM ALS for 4 hours, 8 hours, 12 hours, 24 hours, 48 hours, and 72 hours. Following the treatment, cells were detached and fixed with 70% ethanol at −20°C overnight. Subsequently, the cells were collected by centrifugation at 250× g for 5 minutes. Then, the cells were washed with PBS and incubated with 25 μg/mL RNase A and 50 μg/mL PI for 30 minutes in the dark. A total number of 1×104 cells were subject to cell cycle analysis using a flow cytometer (Becton Dickinson Immunocytometry Systems, San Jose, CA, USA).
Quantification of cellular autophagy
To examine the effect of ALS on autophagy in PANC-1 and BxPC-3 cells, cellular autophagy was first detected using flow cytometry as described previously.23 Briefly, PANC-1 and BxPC-3 cells were seeded into 60 mm Petri dishes. After cells were seeded for 24 hours, the cells reached ~75% confluence and were then treated with fresh medium alone and ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours. Following the ALS treatment, cells were detached and resuspended in 250 μL of phenol red-free culture medium containing 5% FBS. Following that, 250 μL of the diluted Cyto-ID® Green stain solution was added to each sample. Cells were incubated for 30 minutes at 37°C in the dark and then collected by centrifugation at 250× g. The cell pellet was washed with 1× assay buffer given in Cyto-ID® Autophagy detection kit and resuspended in 500 μL fresh 1× assay buffer. Cells were analyzed using the green (FL1) channel of a flow cytometer.
Confocal fluorescence microscopy examination
The cellular autophagy level was further detected by examining using confocal fluorescence microscopy. Briefly, PANC-1 and BxPC-3 cells were seeded into eight-well chamber slide. The cells were treated with ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours. After the ALS treatment, the cells were washed with 1× assay buffer given in Cyto-ID® Autophagy detection kit, followed by incubation with 100 μL of microscopy dual detection reagent for 30 minutes at 37°C in the dark. After the incubation, the cells were washed with 1× assay buffer to remove detection reagent and then the cells were examined using a Leica TCS SP2 laser scanning confocal microscopy (Leica Microsystems, Wetzlar, Germany) using a standard FITC filter set for imaging the autophagic signal at wavelengths of 405/488 nm.
Western blotting analysis
To examine the effect of ALS on the expression of various cellular proteins, the Western blotting assays were performed as described previously.23 The PANC-1 and BxPC-3 cells were incubated with ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours. After ALS treatment, cells were washed with precold PBS and lysed with the RIPA buffer containing the protease inhibitor and phosphatase inhibitor cocktails. Protein concentrations were measured by Pierce BCA protein assay kit. Equal amount of protein sample (20 μg) was electrophoresed on 7% or 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis mini-gel after thermal denaturation for 5 minutes at 95°C. Following that, proteins were transferred onto methonal-activated PVDF membrane at 100 V for 2 hours at 4°C. Subsequently, membranes were blocked with 5% skim milk and probed with indicated primary antibody overnight at 4°C and then blotted with respective secondary antibody. Visualization was performed using Bio-Rad system. The blots were analyzed using ImageLab 3.0 (Bio-Rad) and protein level was normalized to the matching densitometric value of β-actin.
Statistical analysis
Data are expressed as the mean ± standard deviation. Multiple comparisons were evaluated by one-way analysis of variance followed by Tukey’s multiple comparison. A value of P<0.05 was considered statistically significant. Assays were performed at least three times independently.
Results
ALS decreases cell viability of PANC-1 and BxPC-3 cells
First, we tested the effect of ALS on the viability of PANC-1 and BxPC-3 cells using MTT assay. Incubation of both cell lines with ALS at concentrations ranging from 0.1 μM to 50 μM for 24 hours significantly decreased the cell viability (Figure 1B). In comparison to the control cells, the percentage of the viability was 86.7%, 74.7%, 59.6%, 46.4%, and 25.4% when PANC-1 cells were treated with ALS at 0.1 μM, 1 μM, 5 μM, 10 μM, and 50 μM for 24 hours, respectively (Figure 1B). For BxPC-3 cells, the percentage of the viability of BxPC-3 cells was 83.5%, 71.4%, 42.1%, 6.9%, and 3.2% compared to control, when cells were treated with ALS at 0.1 μM, 1 μM, 5 μM, 10 μM, and 50 μM for 24 hours, respectively (Figure 1B). The IC50 values were 7.1 μM and 6.8 μM for PANC-1 and BxPC-3 cells after 24-hour incubation with ALS, respectively (Figure 1B). The results show that ALS exerts a potent inhibitory effect on cell proliferation in PANC-1 and BxPC-3 cells.
ALS inhibits the phosphorylation of AURKA
ALS has been demonstrated as a potent AURKA inhibitor; herein, we first tested the effect of ALS on the phosphorylation of AURKA and its downstream target p53 in PANC-1 and BxPC-3 cells. As shown in Figure 2, treatment of PANC-1 and BxPC-3 cells with ALS significantly inhibited the phosphorylation of AURKA at Thr288 and p53 at Ser392 in a concentration-dependent manner. There was a 13.3%, 29.7%, and 63.3% reduction in the level of p-AURKA at Thr288, and 9.9%, 18.0%, and 20.0% decrease in the expression level of p-53 at Ser392 in PANC-1 cells when treated with ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours, respectively (Figure 2A and B). Similarly, there was a 31.6%, 41.3%, and 50.9% decline in the level of p-AURKA, and 12.3%, 19.8%, and 24.2% reduction in the level of p-p53 in BxPC-3 cells when treated with ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours, respectively (Figure 2A and B). Furthermore, in comparison to the control, incubation of PANC-1 cells with ALS at 0.1 μM, 1 μM, and 5 μM resulted in a 33.7%, 70.6%, and 90.2% reduction in the ratio of p-AURKA over AURKA, and 13.1%, 20.6%, and 26.5% decrease in the ratio of p-p53 over p53, respectively (Figure 2A and B). Similarly, there was a 31.6%, 41.3%, and 50.9% decline in the ratio of p-AURKA over AURKA, and 5.5%, 22.9%, and 33.0% decline in the ratio of p-p53 over p53 in BxPC-3 cells when treated with ALS at 0.1 μM, 1 μM, and 5 μM, respectively (Figure 2A and B).
ALS regulates cell cycle distribution of PANC-1 and BxPC-3 cells
We next examined the effect of ALS on cell cycle distribution in PANC-1 and BxPC-3 cells using flow cytometry. ALS significantly induced G2/M phase arrest in both cell lines (Figure 3). Treatment of cells with ALS concentration-dependently increased the percentage of PANC-1 and BxPC-3 cells in G2/M phase (Figure 3A and B). The percentage of PANC-1 in G2/M phase was increased from 30.3% at basal level to 46.0%, 77.5%, and 80.9% when treated with ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours, respectively, and the percentage of BxPC-3 cells in G2/M phase was raised from 29.9% at basal level to 41.2%, 84.9%, and 85.0% when treated with ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours, respectively (Figure 3A and B). In separate experiments, we also examined the effect of ALS on cell cycle distribution over 72 hours. In comparison to the control cells, the percentage of PANC-1 cells in G2/M phase was increased from 28.1% at basal level to 36.7%, 49.9%, 51.8%, 68.7%, 67.7%, and 59.1% after 4-hour, 8-hour, 12-hour, 24-hour, 48-hour, and 72-hour treatment with 5 μM ALS, respectively (Figure 3C and D). Similarly, the percentage of BxPC-3 cells in G2/M phase was increased from 31.1% at basal level to 45.6%, 59.0%, 65.9%, 82.0%, 88.6%, and 85.4% after 4-hour, 8-hour, 12-hour, 24-hour, 48-hour, and 72-hour treatment with 5 μM ALS compared to the control cells, respectively (Figure 3C and D). The results show that ALS treatment remarkably induces PANC-1 and BxPC-3 cells arrest in G2/M phase, which may be attributable, at least in part, to the inhibition of the AURKA activation.
ALS modulates key cell cycle regulators in PANC-1 and BxPC-3 cells
Since we have observed the inducing effect of ALS on cell cycle arrest in PANC-1 and BxPC-3 cells, we further examined the effect of ALS on the expression of cell cycle regulators, including CDK1/CDC2, CDK2, cyclin B1, cyclin D1, p21 Waf1/Cip1, p27 Kip1, and p53 in PANC-1 and BxPC-3 cells using Western blotting assay. Incubation of PANC-1 cells with ALS at 0.1 μM, 1 μM, and 5 μM resulted in varying alterations in the expression levels of cell cycle regulators (Figure 4). CDC2 and cyclin B1 are two key regulators for G2-to-M phase transition.24 The expression of CDC2, cyclin B1, and CDK2 was significantly suppressed in both cell lines with the treatment of ALS at concentrations of 0.1 μM, 1 μM, and 5 μM for 24 hours. There was a 18.3%, 26.6%, and 51.1% reduction in the expression level of CDC2, 7.50%, 12.9%, and 48.4% decline in the expression level of cyclin B1, and 9.60%, 17.6%, and 23.3% decrease in the expression level of CDK2 in PANC-1 cells when treated with ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours, respectively (Figure 4A and B). Similarly, there was a 18.3%, 26.6%, and 51.1% reduction in the expression level of CDC2, 12.4%, 29.6%, and 60.9% decrease in the expression level of cyclin B1, and 12.0%, 21.3%, and 31.7% decline in the expression level of CDK2 in BxPC-3 cells when treated with ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours, respectively (Figure 4A and B). For cyclin D1, there was a 16.3%, 22.7%, and 30.8% reduction in PANC-1 cells, but there was a 1.25-fold, 1.32-fold, and 1.41-fold increase in BxPC-3 cells with the treatment of 0.1 μM, 1 μM, and 5 μM ALS, respectively (Figure 4A and B).
Furthermore, p21 Waf1/Cip1, p27 Kip1, and p53 play an important role in the regulation of cell cycle. The tumor suppressor protein p21 Waf1/Cip1 acts as an inhibitor of cell cycle progression, and it serves to inhibit kinase activity and block progression through G1/S in association with CDK2 complexes.25 Phosphorylated p53 upregulates p21 Waf1/Cip1 transcription via a p53-responsive element, and activation of p53 leads to either cell cycle arrest and DNA repair or apoptosis. The expression of p21 Waf1/Cip1, p27 Kip1, and p53 was significantly increased in both cell lines with the treatment of ALS at concentration of 0.1 μM, 1 μM, and 5 μM for 24 hours. In comparison to the control cells, incubation of PANC-1 cells with ALS at 0.1 μM, 1 μM, and 5 μM led to a 1.6-fold, 1.8-fold, and 2.4-fold increase in the expression of p21 Waf1/Cip1, 1.1-fold, 1.6-fold, and 1.8-fold rise in the expression of p27 Kip1, and 1.2-fold, 1.3-fold, and 1.7-fold elevation in the expression of p53, respectively (Figure 4A and B). There was a similar regulatory effect of ALS on the expression levels of p21 Waf1/Cip1, p27 Kip1, and p53 in BxPC-3 cells. Treating cells with ALS at 0.1 μM, 1 μM, and 5 μM increased the expression level of p21 Waf1/Cip1 by 1.3-fold, 1.5-fold, and 1.9-fold, elevated the expression level of p27 Kip1 by 1.2-fold, 1.3-fold, and 1.4-fold, and increased the expression level of p53 by 5.6-fold, 6.6-fold, and 7.1-fold compared to the control cells, respectively (Figure 4A and B). These results indicate that ALS significantly downregulates the expression of CDC2, cyclin B1, and CDK2 but upregulates p21 Waf1/Cip1, p27 Kip1, and p53 in both PANC-1 and BxPC-3 cells, which contributes, at least in part, to the observed cell cycle arrest phenomenon in both cell lines.
ALS induces autophagy in PANC-1 and BxPC-3 cells
Autophagy is one of the predominant programmed cell death routes, and its role in cancer cell survival and death is controversial.10,26 Recently, it has been reported that induction of autophagy is an emerging strategy for the treatment of pancreatic cancer.26,27 As such, we examined the effect of ALS on autophagy in PANC-1 and BxPC-3 cells using flow cytometry and confocal microscopy. As shown in Figure 5, the percentage of autophagic cells at basal level is 3.7% and 1.5% for PANC-1 and BxPC-3 cells, respectively. Treating cells with ALS significantly increased autophagy in a concentration-dependent manner. In PANC-1 cells, there was a 2.0-fold, 3.1-fold, and 4.3-fold increase in autophagy when treated with 0.1 μM, 1 μM, and 5 μM ALS for 24 hours compared to the control cells (Figure 5A and B). In BxPC-3 cells, incubation of cells with 1 μM and 5 μM ALS for 24 hours gave a 5.2-fold and 6.1-fold rise in the percentage of autophagic cells, respectively (Figure 5A and B). Treating BxPC-3 cells with 0.1 μM ALS for 24 hours did not significantly induce autophagy. We further examined the autophagy-inducing effect of ALS in PANC-1 and BxPC-3 cells using confocal microscopy. As shown in Figure 5C and D, ALS treatment results in a significant increase in autophagy in PANC-1 and BxPC-3 cells, compared to the control. There was a 1.1-fold and 1.1-fold increase in the autophagic death of PANC-1 cells when treated with ALS at 1 μM and 5 μM for 24 hours, respectively. In BxPC-3 cells, there was a 1.2-fold, 1.2-fold, and 1.4-fold increase in autophagy when treated with 0.1 μM, 1 μM, and 5 μM ALS for 24 hours (Figure 5C and D). Taken together, these results demonstrate that ALS induces autophagy in both PANC-1 and BxPC-3 cells.
ALS suppresses PI3K/Akt/mTOR axis in PANC-1 and BxPC-3 cells
Since we have observed the autophagy-inducing effect of ALS in PANC-1 and BxPC-3 cells, we further investigated the possible mechanisms for the autophagy-inducing effect of ALS. First, we examined the phosphorylation of PI3K, an upstream signaling molecule of Akt/mTOR pathway with an important role in the regulation of cell proliferation and autophagy,5,10,28 which catalyzes the formation of phosphatidylinositol-3,4,5-triphosphate, initiating the activation of a number of signaling pathways related to cell proliferation, cell migration, cell survival, and cell death.29 As shown in Figures 6–8, S1A–P, and S2A–P, ALS treatment causes significant alterations in the expression and phosphorylation levels of key functional proteins regulating autophagy signaling pathway. There was a concentration-dependent decrease in the phosphorylation levels of PI3K, Akt, and mTOR in PANC-1 and BxPC-3 cells with ALS treatment at 0.1 μM, 1 μM, and 5 μM. There was a 18.9%, 37.3%, and 57.8% decline in the ratio of p-PI3K over PI3K, 52.6%, 73.4%, and 71.1% decrease in the ratio of p-Akt over Akt, and 18.1%, 29.8%, and 47.7% reduction in the ratio of p-mTOR over mTOR in PANC-1 cells when treated with ALS at 0.1 μM, 1 μM, and 5 μM, respectively (Figures 6 and 7). Similarly, there was a 44.1%, 50.3%, and 58.8% decrease in the ratio of p-PI3K over PI3K, 39.1%, 43.0%, and 53.7% decline in the ratio of p-Akt over Akt, and 29.6%, 44.2%, and 53.2% reduction in the ratio of p-mTOR over mTOR in BxPC-3 cells treated with ALS at 0.1 μM, 1 μM, and 5 μM, respectively (Figures 6 and 8).
Furthermore, there was a concentration-dependent increase in the phosphorylation level of Erk1 and Erk2, after the cells were incubated with ALS. In comparison to the control cells, incubation of PANC-1 cells with ALS at 0.1 μM, 1 μM, and 5 μM led to a 1.5-fold, 1.9-fold, and 2.2-fold increase in the ratio of p-Erk1 over Erk1 and 1.5-fold, 1.7-fold, and 2.0-fold increase in the ratio of p-Erk2 over Erk2, respectively (Figures 6 and 7). There was a similar regulatory effect of ALS on the phosphorylation of Erk1 and Erk2 in BxPC-3 cells. Treatment of cells with ALS at 0.1 μM, 1 μM, and 5 μM increased the ratio of p-Erk1 over Erk1 1.4-fold, 2.0-fold, and 2.5-fold and elevated the ratio of p-Erk2 over Erk2 1.9-fold, 3.4-fold, and 4.6-fold compared to the control cells, respectively (Figures 6 and 8).
In addition, the effect of ALS on the expression levels of PTEN, beclin 1, LC3-I, and LC3-II was examined in PANC-1 and BxPC-3 cells. ALS treatment concentration-dependently increased the expression level of PTEN and beclin 1 in PANC-1 cells (Figures 6–8). In comparison to the control cells, treating PANC-1 cells with ALS at 0.1 μM, 1 μM, and 5 μM led to a 1.1-fold, 1.4-fold, and 1.7-fold increase in the expression of PTEN and 1.2-fold, 1.4-fold, and 1.6-fold rise in the expression of beclin 1, respectively. The ratio of LC3-II over LC3-I was elevated 1.2-fold, 1.7-fold, and 3.1-fold when PANC-1 cells were treated with ALS at 0.1 μM, 1 μM, and 5 μM, respectively (Figures 6–8). There was a similar regulatory effect of ALS on the expression levels of PTEN, beclin 1, LC3-I, and LC3-II in BxPC-3 cells. In comparison to the control cells, incubation of BxPC-3 cells with ALS at 0.1 μM, 1 μM, and 5 μM increased 1.1-fold, 1.2-fold, and 1.4-fold the expression level of PTEN and elevated 1.1-fold, 1.2-fold, and 1.6-fold the expression level of beclin 1, respectively (Figures 6–8). The ratio of LC3-II over LC3-I was increased 2.0-fold when BxPC-3 cells were treated with 5 μM ALS. Taken together, the regulatory effect of ALS on PI3K/Akt/mTOR signaling pathway contributes to its pancreatic cancer cells-killing effect.
ALS activates AMPK but inhibits p38 MAPK signaling pathways in PANC-1 and BxPC-3 cells
AMPK and p38 MAPK have important roles in the regulation of cell survival and cell death and interact with Akt/mTOR signaling pathway.30,31 We speculated that ALS can regulate the AMPK and p38 MAPK signaling pathways in PANC-1 and BxPC-3 cells, which may contribute to the cancer cell-killing effect of ALS. As shown in Figures 6–8, treatment of both cell lines with ALS resulted in differential effects on the phosphorylation and expression levels of AMPK and p38 MAPK. In comparison to the control cells, there was a 1.3-fold, 1.4-fold, and 2.6-fold increase in the ratio of p-AMPK over AMPK but 25.8%, 62.3%, and 77.8% decline in the ratio of p-p38 MAPK over p38 MAPK, when PANC-1 cells were treated with ALS at 0.1 μM, 1 μM, and 5 μM, respectively (Figures 6–8). In BxPC-3 cells, there was a 1.7-fold, 2.1-fold, and 2.9-fold rise in the ratio of p-AMPK over AMPK but 28.4%, 49.5%, and 65.1% reduction in the ratio of p-p38 MAPK over p38 MAPK, when cells were incubated with ALS at 0.1 μM, 1 μM, and 5 μM, respectively (Figures 6–8). The results show that ALS can inhibit p38 MAPK signaling but activate AMPK signaling pathways in PANC-1 and BxPC-3 cells.
ALS inhibits EMT phenotype in PANC-1 and BxPC-3 cells
EMT is a critical process playing an important role in cancer invasion and metastasis, with a reduction in the expression of cell adhesion molecules.11,12,32 Therefore, the effect of ALS on the expression of key EMT markers and regulators was examined in PANC-1 and BxPC-3 cells. ALS treatment results in differential effects on the expression level of EMT-related proteins (Figures 9–11). Incubation of PANC-1 cells with ALS at 0.1 μM, 1 μM, and 5 μM increased by 1.2-fold, 1.4-fold, and 1.5-fold the expression level of E-cadherin but decreased by 11.0%, 18.3%, and 24.2% the expression level of N-cadherin, respectively. Besides, treating PANC-1 cells with 5 μM ALS significantly decreased the expression level of slug, snail, and TCF-8 by 34.5%, 46.8%, and 31.1%, respectively, whereas, there was a 1.6-fold, 1.5-fold, and 1.5-fold increase in the expression level of Zona occludin-1 (ZO-1), β-catenin, and claudin 1 when PANC-1 cells were treated with 5 μM ALS, respectively (Figures 9–11). There was no significant change in the expression level of vimentin when PANC-1 cells were treated with ALS at 0.1 μM, 1 μM, and 5 μM.
As shown in Figures 9–11, ALS exhibits a similar modulating effect on the expression of the key EMT markers and regulators in BxPC-3 cells. The expression level of E-cadherin was increased 1.1-fold, 2.4-fold, and 2.5-fold, but the expression level of N-cadherin was reduced 5.3%, 32.7%, and 58.4%, when cells were treated with ALS at 0.1 μM, 1 μM, and 5 μM, respectively. There was a 24.4%, 32.5%, and 37.8% reduction in the expression level of slug, 24.1%, 26.3%, and 35.9% decrease in the expression level of snail, and 28.2%, 58.8%, and 73.7% decline in the expression level of TCF-8 in BxPC-3 cells when treated with ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours, respectively (Figures 9–11). In addition, incubation of BxPC-3 cells with ALS at 0.1 μM, 1 μM, and 5 μM increased 1.2-fold, 1.3-fold, 1.4-fold and 1.2-fold, 2.1-fold, and 3.0-fold the expression level of β-catenin and vimentin compared to the control, respectively. Treating BxPC-3 cells with 5 μM ALS increased 1.8-fold the expression level of claudin 1. Taken together, the results show that ALS inhibits the expression of key functional proteins regulating EMT, contributing to the anticancer effect of ALS in PANC-1 and BxPC-3 cells.
ALS suppresses the expression of PBEF and Sirt1 in PANC-1 and BxPC-3 cells
Recently, accumulating evidence shows that Sirt1-mediated signaling pathway plays an important role in the regulation of cellular autophagy and cancer development.33,34 PBEF, a rate limiting enzyme responsible for the NAD+ biosynthesis pathway, plays an essential role in the regulation of Sirt1 activation.35 Recently, studies have shown that Sirt1 deacetylates both histone and nonhistone proteins, such as p53, and deacetylation of p53 and inhibition of p53-regulated cell death indicate that Sirt1 is a negative regulator of p53.36 We speculate that ALS may regulate expression of PBEF and Sirt1 and acetylation of p53 (Ac-p53) in PANC-1 and BxPC-3 cells. Thus, we examined the effect of ALS on the expression level of PBEF, Sirt1, and Ac-p53 in both cell lines. There was a significant decrease in the expression level of PBEF and Sirt1 in PANC-1 and BxPC-3 cells when treated with ALS (Figure 12A and B). There was a 13.1%, 23.1%, and 31.1% reduction in the expression level of PBEF, and 8.4%, 16.3%, and 27.8% decline in the expression of Sirt1 in PANC-1 cells when treated with ALS at 0.1 μM, 1 μM, and 5 μM, respectively (Figure 12A and B). Similarly, there was a 15.2%, 24.9%, and 25.7% decrease in the expression of PBEF, and 12.8%, 24.6%, and 36.0% decline in the expression of Sirt1 in BxPC-3 cells when treated with ALS at 0.1 μM, 1 μM, and 5 μM, respectively (Figure 12A and B). In contrast, there was a significant increase in the level of Ac-p53 in PANC-1 and BxPC-3 cells when treated with ALS (Figure 12A and B). Treatment of PANC-1 cells with ALS at 0.1 μM, 1 μM, and 5 μM resulted in 1.8-fold, 2.0-fold, and 1.7-fold increase in the level of Ac-p53, and 1.4-fold, 1.4-fold, and 1.6-fold rise in the ratio of Ac-p53 over p53 compared to the control cells, respectively. In BxPC-3 cells, incubation of cells with ALS at 0.1 μM, 1 μM, and 5 μM led to a 1.2-fold, 1.2-fold, and 1.3-fold increase in the level of Ac-p53, and 1.2-fold, 1.3-fold, and 1.4-fold elevation in the ratio of Ac-p53 over p53 compared to the control, respectively (Figure 12A and B).
ALS suppresses the expression of Nrf2 in PANC-1 and BxPC-3 cells
Nrf2 is a critical nuclear transcript factor controlling a wide range of antioxidant response element-dependent genes to regulate the physiological and pathophysiological outcomes of oxidant exposure, including cell death and EMT.37–39 Thus, we further determined the effects of ALS on its expression. As shown in Figure 13A and B, there was a concentration-dependent reduction in the expression of Nrf2. Incubation of PANC-1 cells with ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours reduced the expression level of Nrf2 by 25.1%, 32.8%, and 41.2%, respectively. Similarly, treatment with ALS at 0.1 μM, 1 μM, and 5 μM for 24 hours significantly suppressed the expression level of Nrf2 by 9.9%, 25.9%, and 38.2% in BxPC-3 cells, respectively (Figure 13A and B). These results suggested that the anticancer effect of ALS in PANC-1 and BxPC-3 cells may be ascribed, at least in part, to its regulatory effect on Nrf2-mediated signaling pathway.
Discussion
The pancreatic cancer is the most aggressive cancer with a very poor prognosis in patients with this disease, and the treatment of pancreatic cancer remains a major challenge. Therefore, there is an urgent need for new therapeutic strategies for pancreatic cancer treatment. Recently, AURKA has become a promising therapeutic target for cancer treatment,20 which plays an important role in centrosome maturation and separation, bipolar spindle assembly, and mitotic entry. Abnormalities in the expression and activities of AURKA have been implicated in the initiation, development, and progression of various cancers, including the breast, colon, pancreatic, ovarian, and gastric cancers.40
With accumulating evidence showing the role of the Aurora kinases in the pathogenesis of cancer, AURKA becomes a more important therapeutic target in the treatment of cancer. It has been reported that there is a correlation of the expression and gene amplification of AURKA with tumor proliferation rates and prognostic markers.41 Furthermore, increasing studies show that AURKA can induce chemotherapeutic resistance and regulate several key signaling pathways related to cell cycle, autophagy, and EMT in cancer cells,16,41,42 suggesting its pivotal role in cancer cell signaling.43 Currently, there are a number of AURKA inhibitors that have been developed and are being tested in different Phase I and Phase II trials for cancer treatment.18 In the present study, we have investigated the effect of ALS, a potent and selective inhibitor of AURKA, on pancreatic cancer cells. Our findings show that ALS exhibits potent inhibitory effects on pancreatic cancer cell growth and cell cycle progression. ALS also exerts a potent pancreatic cancer cell-killing effect via autophagic cell death with the involvement of PI3K/Akt/mTOR, p38 MAPK, AMPK, and Sirt1-mediated signaling pathways. Moreover, ALS shows a substantial suppressing effect on EMT in pancreatic cancer cells.
AURKA exerts an important role in the regulation of chromosome assembly and segregation during mitosis.44 It regulates the G2-to-M transition and inactivation of AURKA results in G2/M arrest.45 In the present study, our findings clearly showed that ALS treatment remarkably decreased the ratio of p-AURKA over AURKA in PANC-1 and BxPC-3 cells, suggesting that ALS may induce cell cycle arrest in G2/M phase. Indeed, we have observed the inducing effect on cell cycle arrest in G2/M phase in PANC-1 and BxPC-3 cells treated with ALS. Notably, it has been reported that the regulatory effect of Aurora kinases on cell cycle distribution is associated with p53.46,47 Activation of AURKA instigates Mdm2-mediated destabilization and inhibition of p53,46 and AURKB phosphorylates and induces degradation of p53.47 In our study, the phosphorylation level of p53 was significantly decreased, but the total expression level of p53 was remarkably increased in PANC-1 and BxPC-3 cells when treated with ALS, which in turn results in a reduction in the ratio of p-p53 over p53.
Furthermore, our findings show that ALS treatment can significantly increase the expression levels of p21 Waf1/Cip1 that is a tumor suppressor protein and serves as an inhibitor of cell cycle progression via the inhibition of CDK activity.25 Therefore, increased expression of p21 Waf1/Cip1 contributes to cell cycle arrest in PANC-1 and BxPC-3 cells. Moreover, treating PANC-1 and BxPC-3 cells with ALS also results in a significant increase in the expression level of p27 Kip1 that is a member of the Cip/Kip family of CDK inhibitors, inducing cell cycle arrest via its inhibitory binding to CDK2/cyclin E and other CDK/cyclin complexes.48 Taken together, inducing effect of ALS on cell cycle arrest can prevent cell proliferation by modulation of a number of cell cycle regulators in PANC-1 and BxPC-3 cells, which can eventually lead to pancreatic tumor regression.49–51
Recently, the role of autophagy in pancreatic cancer cell death is controversial.26 Increasing evidence shows that targeting autophagy is a promising strategy for pancreatic cancer treatment via regulation of autophagy-related key functional proteins and signaling pathways.9,26,52 Autophagy is a complicated process executed through multiple steps from intracellular membrane/vesicle reorganization to form double-membraned autophagosomes that fuse with lysosome to form autophagolysosomes, which eventually degrade the contents via acidic lysosomal hydrolases.5,10,53 Increasing evidence demonstrates that the PI3K/Akt/mTOR signaling pathway is a central signaling pathway regulating cellular autophagy and that targeting this signaling pathway has been proposed to be a promising strategy for cancer treatment.54–56 In the present study, our findings show that ALS exhibits a significant inhibitory effect on PI3K/Akt/mTOR signaling pathway in both PANC-1 and BxPC-3 cells, contributing to promoting effect of ALS on autophagy in both PANC-1 and BxPC-3 cells. It suggests that ALS may be a promising agent, which can be used to induce autophagic cell death for pancreatic cancer treatment. Notably, AMPK and p38 MAPK are two upstream signaling molecules, which can regulate cellular autophagy via the activation of p53, inhibition of mTOR, and other related signaling pathways.5 In our study, ALS treatment significantly activates AMPK, contributing to the inhibition of mTOR. It can exacerbate the ALS-induced autophagy in PANC-1 and BxPC-3 cells. On the other hand, our findings show that ALS significantly inactivates p38 MAPK signaling, which is responsive to a variety of stress stimuli and regulates autophagy.5,54 Our previous study also showed that activation of AMPK and inactivation of p38 MAPK signaling contribute to the autophagic cell death in PANC-1 and BxPC-3 cells.57 Collectively, AMPK and p38 MAPK contribute to the mTOR-mediated autophagy in PANC-1 and BxPC-3 cells.
Increasing number of studies have shown that modulation of EMT may represent an emerging therapeutic strategy for the treatment of pancreatic cancer,12,32 due to the role in initiation, development, progression, invasion, migration, and metastasis and drug resistance in pancreatic cancer.12,14,58,59 Thus, the key functional molecules that regulate EMT may serve as a therapeutic target. Cadherins are a superfamily of transmembrane glycoproteins regulating calcium-dependent cell–cell adhesion, including N-, P-, R-, B-, and E-cadherins.11,12 E-cadherin acts as a suppressor of invasion and migration of many epithelial cancers,11,12,32 and E-cadherin-mediated calcium-dependent cell–cell adhesion is negatively regulated by snail/slug. Inhibition of snail/slug expression can suppress the EMT progression in pancreatic cancer.60 Our findings show that ALS treatment significantly increases the expression of E-cadherin but suppressed the expression of snail and slug in PANC-1 and BxPC-3 cells. It suggests that ALS can inhibit EMT, reducing drug resistance and suppressing pancreatic cancer progression. Moreover, it has been reported that activation of Erk1/2 signaling pathway is correlated with EMT progress in pancreatic cancer.60 We have observed that ALS inactivates Erk1/2 signaling pathways with a marked inhibition on the phosphorylation of Erk1/2 in PANC-1 and BxPC-3 cells, which also may contribute to the inhibitory effect of ALS on EMT. Therefore, ALS may act as a putative EMT-targeting agent that will be clinically helpful to reduce the morbidity and mortality of pancreatic cancer.
There is emerging evidence showing that Sirt1 promotes pancreatic cancer cell viability and instigates drug resistance in pancreatic cancer,61,62 indicating that inhibition on the activity of Sirt1 and Sirt1-mediated signaling pathway may be a novel strategy for pancreatic cancer treatment via reverse of the chemoresistance and promotion of cancer cell death in pancreatic cancer. Sirt1, an NAD+-dependent DNA repair enzyme originally discovered in yeast (Sir2), plays an important role in the regulation of autophagy and EMT via deacetylation of a number of functional proteins, such as p53, claudin 1, TCF-8, and FoxOs.36,63–65 PBEF/visfatin, a rate-limiting enzyme in the NAD+ biosynthesis salvage pathway, is responsible for the catalysis of nicotinamide with 5-phosphoribosyl-1-pyrophosphate, producing nicotinamide mononucleotide.66 PBEF/visfatin is important in NAD+ biosynthesis which is essential for Sirt1-mediated signaling pathway.66 In the present study, ALS decreased the expression levels of PBEF/visfatin and Sirt1 in PANC-1 and BxPC-3 cells. Notably, there was a significant rise in the level of Ac-p53 and the ratio of Ac-p53 over p-p53 in both cells treated with ALS (Figure 12). Taken together, the results show that ALS-induced autophagy and EMT inhibition in pancreatic cancer cells may be attributed, partially, to the Sirt1-mediated pathway. In addition, it has been reported that enhanced Nrf2 activity facilitates the tumorigenesis in pancreatic cancer67 and that overexpression of Nrf2 promotes pancreatic cancer cell viability and drug resistance.68 Therefore, manipulation of the expression level and/or activity of Nrf2 may represent a potential strategy to reduce pancreatic tumor growth and to increase sensitivity to therapeutics. In our study, we also found that ALS significantly decreased the expression of Nrf2 in PANC-1and BxPC-3cells, suggesting that inhibition of Nrf2 expression contributes, at least in part, to the anticancer effect of ALS in pancreatic cancer treatment.
In summary, we have observed the anticancer effect of ALS and investigated the potential molecular mechanisms of its cancer cell-killing effect in human pancreatic cancer PANC-1 and BxPC-3 cells. The mechanisms of action of ALS were mainly ascribed to cell cycle arrest, autophagy induction, and EMT inhibition, and their related signaling pathways. ALS induced the inhibition of PI3K/Akt/mTOR, p38 MAPK, and Erk1/2 signaling pathways, but activation of AMPK signaling pathway contributes to the autophagy-inducing and EMT-suppressing effects of ALS in PANC-1 and BxPC-3 cells. Collectively, ALS may represent a promising anticancer drug that can be used for pancreatic cancer treatment. More studies are warranted to reveal other potential targets and potential mechanisms of ALS in the treatment of pancreatic cancer.
Acknowledgments
The authors appreciate the financial support from the Startup Funds of the College of Pharmacy, University of South Florida, Tampa, FL, USA. Dr Zhi-Wei Zhou is a holder of a postdoctoral scholarship from College of Pharmacy, University of South Florida, Tampa, FL, USA. The authors appreciate the help from Mr Jeffrey L Edelman from University of South Florida (Tampa, FL) for manuscript proofreading.
Disclosure
The authors report no conflicts of interest in this work.
References
Ferlay J, Soerjomataram I, Ervik M, et al. GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No 11 [Internet]. Lyon, France; 2013. | ||
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9–29. | ||
Guo X, Cui Z. Current diagnosis and treatment of pancreatic cancer in China. Pancreas. 2005;31(1):13–22. | ||
Long J, Luo GP, Xiao ZW, et al. Cancer statistics: current diagnosis and treatment of pancreatic cancer in Shanghai, China. Cancer Lett. 2014;346(2):273–277. | ||
Denton D, Nicolson S, Kumar S. Cell death by autophagy: facts and apparent artefacts. Cell Death Differ. 2012;19(1):87–95. | ||
Westphal S, Kalthoff H. Apoptosis: targets in pancreatic cancer. Mol Cancer. 2003;2:6. | ||
Grasso D, Garcia MN, Iovanna JL. Autophagy in pancreatic cancer. Int J Cell Biol. 2012;2012:760498. | ||
Fulda S. Targeting apoptosis signaling in pancreatic cancer. Cancers (Basel). 2011;3(1):241–251. | ||
Iacobuzio-Donahue CA, Herman JM. Autophagy, p53, and pancreatic cancer. N Engl J Med. 2014;370(14):1352–1353. | ||
Klionsky DJ, Emr SD. Autophagy as a regulated pathway of cellular degradation. Science. 2000;290(5497):1717–1721. | ||
Zheng H, Kang Y. Multilayer control of the EMT master regulators. Oncogene. 2014;33(14):1755–1763. | ||
Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol. 2014;15(3):178–196. | ||
Nauseef JT, Henry MD. Epithelial-to-mesenchymal transition in prostate cancer: paradigm or puzzle? Nat Rev Urol. 2011;8(8):428–439. | ||
Arumugam T, Ramachandran V, Fournier KF, et al. Epithelial to mesenchymal transition contributes to drug resistance in pancreatic cancer. Cancer Res. 2009;69(14):5820–5828. | ||
Vader G, Lens SM. The Aurora kinase family in cell division and cancer. Biochim Biophys Acta. 2008;1786(1):60–72. | ||
Do TV, Xiao F, Bickel LE, et al. Aurora kinase A mediates epithelial ovarian cancer cell migration and adhesion. Oncogene. 2014;33(5):539–549. | ||
Sehdev V, Katsha A, Ecsedy J, Zaika A, Belkhiri A, El-Rifai W. The combination of alisertib, an investigational Aurora kinase A inhibitor, and docetaxel promotes cell death and reduces tumor growth in preclinical cell models of upper gastrointestinal adenocarcinomas. Cancer. 2013;119(4):904–914. | ||
Katayama H, Sen S. Aurora kinase inhibitors as anticancer molecules. Biochim Biophys Acta. 2010;1799(10–12):829–839. | ||
Manfredi MG, Ecsedy JA, Chakravarty A, et al. Characterization of Alisertib (MLN8237), an investigational small-molecule inhibitor of aurora A kinase using novel in vivo pharmacodynamic assays. Clin Cancer Res. 2011;17(24):7614–7624. | ||
Dar AA, Goff LW, Majid S, Berlin J, El-Rifai W. Aurora kinase inhibitors – rising stars in cancer therapeutics? Mol Cancer Ther. 2010;9(2):268–278. | ||
Neel NF, Stratford JK, Shinde V, et al. Response to MLN8237 in pancreatic cancer is not dependent on RalA phosphorylation. Mol Cancer Ther. 2014;13(1):122–133. | ||
Lecoeur H. Nuclear apoptosis detection by flow cytometry: influence of endogenous endonucleases. Exp Cell Res. 2002;277(1):1–14. | ||
Li YC, He SM, He ZX, et al. Plumbagin induces apoptotic and autophagic cell death through inhibition of the PI3K/Akt/mTOR pathway in human non-small cell lung cancer cells. Cancer Lett. 2014;344(2):239–259. | ||
Hu X, Moscinski LC. Cdc2: a monopotent or pluripotent CDK? Cell Prolif. 2011;44(3):205–211. | ||
Warfel NA, El-Deiry WS. p21WAF1 and tumourigenesis: 20 years after. Curr Opin Oncol. 2013;25(1):52–58. | ||
Yang S, Kimmelman AC. A critical role for autophagy in pancreatic cancer. Autophagy. 2011;7(8):912–913. | ||
Mujumdar N, Saluja AK. Autophagy in pancreatic cancer: an emerging mechanism of cell death. Autophagy. 2010;6(7):997–998. | ||
Muilenburg D, Parsons C, Coates J, Virudachalam S, Bold RJ. Role of autophagy in apoptotic regulation by Akt in pancreatic cancer. Anticancer Res. 2014;34(2):631–637. | ||
Cantley LC. The phosphoinositide 3-kinase pathway. Science. 2002;296(5573):1655–1657. | ||
Dunlop EA, Tee AR. The kinase triad, AMPK, mTORC1 and ULK1, maintains energy and nutrient homoeostasis. Biochem Soc Trans. 2013;41(4):939–943. | ||
Arthur JS, Ley SC. Mitogen-activated protein kinases in innate immunity. Nat Rev Immunol. 2013;13(9):679–692. | ||
Cannito S, Novo E, di Bonzo LV, Busletta C, Colombatto S, Parola M. Epithelial-mesenchymal transition: from molecular mechanisms, redox regulation to implications in human health and disease. Antioxid Redox Signal. 2010;12(12):1383–1430. | ||
Lee IH, Cao L, Mostoslavsky R, et al. A role for the NAD-dependent deacetylase Sirt1 in the regulation of autophagy. Proc Natl Acad Sci U S A. 2008;105(9):3374–3379. | ||
Ng F, Tang BL. Sirtuins’ modulation of autophagy. J Cell Physiol. 2013;228(12):2262–2270. | ||
Skokowa J, Lan D, Thakur BK, et al. NAMPT is essential for the G-CSF-induced myeloid differentiation via a NAD(+)-sirtuin-1-dependent pathway. Nat Med. 2009;15(2):151–158. | ||
Preyat N, Leo O. Sirtuin deacylases: a molecular link between metabolism and immunity. J Leukoc Biol. 2013;93(5):669–680. | ||
Suzuki T, Motohashi H, Yamamoto M. Toward clinical application of the Keap1-Nrf2 pathway. Trends Pharmacol Sci. 2013;34(6):340–346. | ||
Ma Q. Role of Nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol. 2013;53:401–426. | ||
Kensler TW, Wakabayashi N, Biswal S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu Rev Pharmacol Toxicol. 2007;47:89–116. | ||
Macarulla T, Ramos FJ, Tabernero J. Aurora kinase family: a new target for anticancer drug. Recent Pat Anticancer Drug Discov. 2008;3(2):114–122. | ||
Zhou N, Singh K, Mir MC, et al. The investigational Aurora kinase A inhibitor MLN8237 induces defects in cell viability and cell-cycle progression in malignant bladder cancer cells in vitro and in vivo. Clin Cancer Res. 2013;19(7):1717–1728. | ||
D’Assoro AB, Liu T, Quatraro C, et al. The mitotic kinase Aurora a promotes distant metastases by inducing epithelial-to-mesenchymal transition in ERα+ breast cancer cells. Oncogene. 2014;33(5):599–610. | ||
Melaiu O, Cristaudo A, Melissari E, et al. A review of transcriptome studies combined with data mining reveals novel potential markers of malignant pleural mesothelioma. Mutat Res. 2012;750(2):132–140. | ||
Bolanos-Garcia VM. Aurora kinases. Int J Biochem Cell Biol. 2005;37(8):1572–1577. | ||
Hilton JF, Shapiro GI. Aurora kinase inhibition as an anticancer strategy. J Clin Oncol. 2014;32(1):57–59. | ||
Katayama H, Sasai K, Kawai H, et al. Phosphorylation by aurora kinase A induces Mdm2-mediated destabilization and inhibition of p53. Nat Genet. 2004;36(1):55–62. | ||
Gully CP, Velazquez-Torres G, Shin JH, et al. Aurora B kinase phosphorylates and instigates degradation of p53. Proc Natl Acad Sci U S A. 2012;109(24):E1513–E1522. | ||
Yoon MK, Mitrea DM, Ou L, Kriwacki RW. Cell cycle regulation by the intrinsically disordered proteins p21 and p27. Biochem Soc Trans. 2012;40(5):981–988. | ||
Williams GH, Stoeber K. The cell cycle and cancer. J Pathol. 2012;226(2):352–364. | ||
Rengarajan T, Rajendran P, Nandakumar N, Balasubramanian MP, Nishigaki I. Cancer preventive efficacy of marine carotenoid fucoxanthin: cell cycle arrest and apoptosis. Nutrients. 2013;5(12):4978–4989. | ||
Diaz-Moralli S, Tarrado-Castellarnau M, Miranda A, Cascante M. Targeting cell cycle regulation in cancer therapy. Pharmacol Ther. 2013;138(2):255–271. | ||
Yang A, Rajeshkumar NV, Wang X, et al. Autophagy is critical for pancreatic tumor growth and progression in tumors with p53 alterations. Cancer Discov. 2014;4(8):905–913. | ||
Chen Y, Yu L. Autophagic lysosome reformation. Exp Cell Res. 2013;319(2):142–146. | ||
Taylor RC, Cullen SP, Martin SJ. Apoptosis: controlled demolition at the cellular level. Nat Rev Mol Cell Biol. 2008;9(3):231–241. | ||
Estaquier J, Vallette F, Vayssiere JL, Mignotte B. The mitochondrial pathways of apoptosis. Adv Exp Med Biol. 2012;942:157–183. | ||
Rodon J, Dienstmann R, Serra V, Tabernero J. Development of PI3K inhibitors: lessons learned from early clinical trials. Nat Rev Clin Oncol. 2013;10(3):143–153. | ||
Wang F, Wang Q, Zhou ZW, et al. Plumbagin induces cell cycle arrest and autophagy and suppresses epithelial to mesenchymal transition involving PI3K/Akt/mTOR-mediated pathway in human pancreatic cancer cells. Drug Des Devel Ther. In press 2014. | ||
Yamada S, Fuchs BC, Fujii T, et al. Epithelial-to-mesenchymal transition predicts prognosis of pancreatic cancer. Surgery. 2013;154(5):946–954. | ||
Krantz SB, Shields MA, Dangi-Garimella S, Bentrem DJ, Munshi HG. Contribution of epithelial-mesenchymal transition to pancreatic cancer progression. Cancers (Basel). 2010;2(4):2084–2097. | ||
Hotz B, Arndt M, Dullat S, Bhargava S, Buhr HJ, Hotz HG. Epithelial to mesenchymal transition: expression of the regulators snail, slug, and twist in pancreatic cancer. Clin Cancer Res. 2007;13(16):4769–4776. | ||
Zhang JG, Hong DF, Zhang CW, et al. Sirtuin 1 facilitates chemoresistance of pancreatic cancer cells by regulating adaptive response to chemotherapy-induced stress. Cancer Sci. 2014;105(4):445–454. | ||
Wauters E, Sanchez-Arévalo Lobo VJ, Pinho AV, et al. Sirtuin-1 regulates acinar-to-ductal metaplasia and supports cancer cell viability in pancreatic cancer. Cancer Res. 2013;73(7):2357–2367. | ||
Byles V, Zhu L, Lovaas JD, et al. SIRT1 induces EMT by cooperating with EMT transcription factors and enhances prostate cancer cell migration and metastasis. Oncogene. 2012;31(43):4619–4629. | ||
Zhang LH, Huang Q, Fan XS, Wu HY, Yang J, Feng AN. Clinicopathological significance of SIRT1 and p300/CBP expression in gastroesophageal junction (GEJ) cancer and the correlation with E-cadherin and MLH1. Pathol Res Pract. 2013;209(10):611–617. | ||
Nihalani D, Susztak K. Sirt1-Claudin-1 crosstalk regulates renal function. Nat Med. 2013;19(11):1371–1372. | ||
Montecucco F, Cea M, Bauer I, et al. Nicotinamide phosphoribosyltransferase (NAMPT) inhibitors as therapeutics: rationales, controversies, clinical experience. Curr Drug Targets. 2013;14(6):637–643. | ||
DeNicola GM, Karreth FA, Humpton TJ, et al. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature. 2011;475(7354):106–109. | ||
Lister A, Nedjadi T, Kitteringham NR, et al. Nrf2 is overexpressed in pancreatic cancer: implications for cell proliferation and therapy. Mol Cancer. 2011;10:37. |
Supplementary materials
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.