Back to Journals » Cancer Management and Research » Volume 11
FOXM1 promotes the growth and metastasis of colorectal cancer via activation of β-catenin signaling pathway
Authors Yang K , Jiang B, Lu Y, Shu Q, Zhai P, Zhi Q , Li Q
Received 28 August 2018
Accepted for publication 1 February 2019
Published 1 May 2019 Volume 2019:11 Pages 3779—3790
DOI https://doi.org/10.2147/CMAR.S185438
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Beicheng Sun
Kankan Yang,1,* Bing Jiang,1,* Yecai Lu,1,* Qingbing Shu,1 Pan Zhai,1 Qiaoming Zhi,2 Qixin Li1
1Department of Gastrointestinal Surgery, Chaohu Hospital of Anhui Medical University, Hefei 238000, Anhui, China; 2Department of General Surgery, The First Affiliated Hospital of Soochow University, Suzhou 215006, Jiangsu, China
*These authors contributed equally to this work
Purpose: Our previous study proved that FOXM1 regulates colorectal cancer (CRC) cell metastasis through epithelial–mesenchymal transition program. The aim of this study is to further explore the underlying mechanism of FOXM1 in CRC.
Materials and methods: In this study, we detected the mRNA and protein expressions of FOXM1 and β-catenin in CRC tissues and their corresponding normal-appearing tissues (NATs) by quantitative reverse transcription-PCR and western blot analysis, respectively. Then the potential link between FOXM1 and β-catenin in CRC tissues was analyzed. Furthermore, we systematically analyzed the biological functions of FOXM1 in CRC cells after reconstitution of FOXM1 expression in vitro. Moreover, the mechanism of FOXM1-promoted CRC progression by improving β-catenin nuclear translocation was also discussed.
Results: Our data demonstrated that FOXM1 and β-catenin were upregulated in CRC tissues compared with the corresponding NATs (P<0.05). Clinicopathologic analysis revealed that increased FOXM1 (or β-catenin) expression positively correlated with some clinicopathologic features, such as tumor size, TNM stage, lymphatic metastasis, and distant metastasis (P<0.05). Meanwhile, the possible relationships between FOXM1 and β-catenin in CRC samples were evaluated using SPSS software, and a significant positive correlation was found (P<0.05). In vitro data demonstrate that elevated FOXM1 expression exerted oncogenic effects on CRC via activation of β-catenin signaling pathway. The inhibition of β-catenin by siRNAs significantly attenuates FOXM1-induced malignant activities.
Conclusion: The data suggested that FOXM1/β-catenin is critical for malignancy of CRC, which may constitute a potential therapeutic strategy for CRC.
Keywords: FOXM1, β-catenin, colorectal cancer, signaling pathway
Introduction
Colorectal cancer (CRC) is the third most common cancer and the fourth leading cause of cancer-related deaths in broad areas of the world, accounting for nearly 1.2 million new patients and 600,000 deaths/year.1 Despite many improvements in early detection, neoadjuvant therapy, and surgical treatment, metastatic CRC still has a poor prognosis, and 5-year survival rate is not exceeding 12%.2 Therefore, identification of new prognostic factors to improve therapeutic approaches of CRC is imperative.
FOXM1, which belongs to a winged-helix transcription factor family, is a typical proliferation-associated transcription factor. FOXM1 has been proved to control both G1/S and G2/M phase progression by transcriptional regulation of a set of genes that are essential for cell cycle progression, such as p21Cip1, p27Kip1 proteins, Cdc25A, and Cdc25B.3,4 Emerging evidence demonstrated that FOXM1 was aberrantly overexpressed in multiple types of malignancies, which suggested that FOXM1 might play a critical role in tumor initiation.5–8 Additionally, FOXM1 promoted tumor metastasis in various types of tumors, such as pancreatic cancer, CRC, lung cancer, ovarian cancer, and glioma.7–11 For example, in 2007, Dai et al reported that FOXM1 contributed to glioma progression by enhancing MMP-2 gene transcription.12 Li et al and Zhang et al found that FOXM1 directly and significantly correlated with transactivation of vascular endothelial growth factor (VEGF) expression and promoted the angiogenic ability of gastric cancer and glioma cells.13,14 A study by Yang et al in 2015 showed that knockdown of FOXM1 regulated CRC metastasis through reversing the acquisition of epithelial–mesenchymal transition phenotype.15 Furthermore, FOXM1 overexpression was considered as a molecular marker that predicted increased invasive/metastatic potential of CRC and a poorer prognosis.16
Wnt signaling pathway is commonly considered to play an important role in the carcinogenic process, especially in CRC. Approximately 90% of CRCs have an activating mutation of the canonical Wnt signaling pathway, ultimately leading to the nuclear accumulation of β-catenin through inactivating APC mutations or activating β-catenin mutations, which subsequently combines with T-cell factor (TCF) and lymphoid enhancing factor transcription factors to activate downstream gene transcription, such as c-Myc and cyclin D1.17,18 Previously, many studies revealed that the expression of β-catenin was regulated by FOXM1.33,34 FOXM1 was identified as a new downstream target of Wnt signaling, which was essential for β-catenin/TCF4 transactivation. In glioma, FOXM1 interacted with β-catenin and promoted the nuclear accumulation of β-catenin to induce glioma stem cell self-renewal and tumorigenesis through controlling Wnt target-genes expression. Yoshida et al used FOXM1B transgenic mice and conditional FOXM1 knockout mice to examine the role of FOXM1B in colon cancer development and proliferation, and found that specific deletion of FOXM1 inhibited colorectal tumorigenesis and potential regulation of β-catenin/TCF4 signaling.19 Though there have been many studies to investigate the role of FOXM1 and β-catenin independently, the interaction of FOXM1 and β-catenin on CRC progression in vitro has still not been studied.
In this study, we first detected the mRNA and protein expressions of FOXM1 and β-catenin in 124 CRC tissues and their corresponding normal-appearing tissues (NATs) by quantitative reverse transcription-PCR (qRT-PCR) and western blot analysis, respectively. Then the potential link between FOXM1 and β-catenin in CRC tissues was analyzed. Furthermore, we systematically analyzed the biological functions of FOXM1 in CRC cells after reconstitution of FOXM1 expression in vitro. Moreover, the mechanism of FOXM1-promoted CRC progression by improving β-catenin nuclear translocation was also discussed. Our new findings advanced our understanding of FOXM1-mediated oncogenic mechanisms of CRC.
Materials and methods
Cell lines and tumor samples
Human CRC cell lines (HCT116 and DLD-1) were purchased from the Cell Resource Center, Chinese Academy of Sciences Committee (Shanghai, China) and maintained in PRIM-1640 supplemented with 10% fetal bovine serum (Sijiqing Biological Engineering Materials Co. Ltd., Hangzhou, China) at 37˚C in a humidified atmosphere containing 5% CO2. Human CRC tissues and their corresponding NATs (at least 2 cm distant from the tumor site) were obtained from 124 patients at the Department of General Surgery, the First Affiliated Hospital of Soochow University from October 2008 to October 2016. All patients underwent surgery and had a clear histological diagnosis. None of the patients had received radiotherapy or chemotherapy before surgery. The study was approved by Institute Research Ethics Committee of the First Affiliated Hospital of Soochow University and written informed consent was obtained from all patients prior to the study.
Cell transfection
To obtain FOXM1 overexpressing cells, we transfected pcDNA3.1-FOXM1 or control vector pcDNA3.1 plasmids into DLD-1 cells with the Lipofectamine® 2000 (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s instructions.11 To silence FOXM1 expression, FOXM1 siRNA oligonucleotides (sense: UGGUUAAUAAUCUUGAUCCCA and antisense: GGAUCAAGAUUAUUAACCACC) were designed, synthesized, and transfected into HCT116 cells. Additionally, RNA interference targeting β-catenin (sense: UUACAACUGCAUGUUUCAGCA and antisense: CUGAAACAUGCAGUUGUAAAC) were also synthesized by Shanghai Gene Pharma Co., Ltd. (Shanghai, China) and transfected into FOXM1 overexpressing DLD-1 cells. After 48 hours transfection, cells were collected and the transfection efficiencies of overexpressing plasmids or siRNAs were detected by western blot and qRT-PCR.
qRT-PCR
Total RNA from cultured cells or CRC tissues was extracted using TRIzol™ Reagent (Thermo Fisher Scientific). cDNA was synthesized from 2 µg of RNA using the the RevertAid™ First Strand cDNA Synthesis Kit (Thermo Fisher Scientific). The PCR analysis was performed using PowerUp™ SYBR® Green PCR Master Mix (Thermo Fisher Scientific) on the 7,500 real-time PCR system (Thermo Fisher Scientific). The sequences of the PCR primers were listed as follows: FOXM1 5′-GGAGGAAATGCCACACTTAGCG-3′ (sense) and 5′-TAGGACTTCTTGGGTCTTGGGGTG-3′ (antisense); β-actin 5′-CCACACTGTGCCCATCTACG-3′ (sense) and 5′-AGGATCTTCATGAGGTAGTCAGTCAG-3′ (antisense); β-catenin 5′-AAA GCGGCTGTTAGTCACTGG-3′ (sense) and 5′-GACTTG GGAGGTATCCACATCC-3′ (antisense); cyclin D1 5′-TGAAGCCAGCTCACAGTGCT-3′ (sense) and 5′-AGCCAGGATGGTTGAGGTAA-3′ (antisense); c-Myc 5′-CTTGAACAGCTACGGAACTC-3′ (sense) and 5′-GAGGCAGTTTACATTATGGC-3′ (antisense); TCF-4 5′-GCTGAGCTGCCCAGGAATAT-3′ (sense) and 5′-GCAGAGGCCTGAGTAATTATCAGAA-3′ (antisense). β-actin was used as a reference to obtain the relative fold change for the targets using the 2−ΔΔCt method. Each sample was tested in triplicate and each experiment was performed at least three times.
Western blot analysis
Tumor tissues or cells were washed with ice-cold PBS and lysed in ice-cold radioimmunoprecipitation assay lysis buffer (Beyotime Institute of Biotechnology, Nantong, China) to obtain whole protein extracts. The nuclear protein lysates were prepared with a NE-PER™ Nuclear and Cytoplasmic Extraction Reagents (Thermo Fisher Scientific) according to the manufacturer’s instructions. Proteins in the lysates were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred to polyvinylidene difluoride membranes. Membranes were incubated with primary antibodies: polyclonal rabbit antibody against human FOXM1 (1:1000; Abcam, Cambridge, UK), rabbit anti-Cyclin D1 (1:1000; Sigma-Aldrich Co., St Louis, MO, USA), rabbit anti-c-Myc (1:1000; Sigma-Aldrich Co.), rabbit anti-TCF-4 (1:500; Sigma-Aldrich Co.), rabbit anti-Lamin B1 (1:1000; Abcam), and rabbit anti-β-actin (1:1000; Beyotime Institute of Biotechnology). The signals from the primary antibody were amplified by incubating with horse radish peroxidase conjugated anti-rabbit IgG (1:1000; Beyotime Institute of Biotechnology) and detected with Amersham™ ECL™ Western Blotting Detection Reagents (GE Healthcare, Boston, MA, USA).
Cell proliferation and colony formation assay
Cell proliferation was determined by MTT assay. After transfection with overexpression plasmids or siRNAs for 24 hours, cells were digested, resuspended, and seeded at a density of 4×103 cells/well in 96-well culture plates at 37˚C in 5% CO2. Then cells were incubated with 20 µL MTT solution (5 mg/mL) at 37˚C for 4 hours. Formazan crystals were dissolved with dimethylsulfoxide and the absorbance was measured at 450 nm using a microplate reader at 24, 48, and 72 hours. Each sample was four replicate wells and the experiment was repeated at least three times. For colony formation assay, cells were digested to a single-cell suspension by trypsin. Then cells were plated into 6-well plates at a density of 2×103 cells/well. After 2 weeks of incubation in complete culture medium at 37°C in 5% CO2, the colonies were fixed and stained with 0.1% crystal violet. Finally, the colonies containing at least 50 cells were counted.
Migration and invasion assays
Transwell assays were used to detect the cell migration and invasion ability. This experiment was performed by using the transwell chambers that equipped with a pore size of 8μm (Corning Systems, Inc., San Jose,CA, USA) which were pre-coated with or without Matrigel (BD Biosciences, San Jose, CA, USA). Briefly, cells were digested, resuspended, and seeded into the upper wells at a density of 2×104 cells/well, while medium containing 10% FBS was placed in the lower wells as a chemoattractant. After 24 hours incubation at 37°C, cells on the upper side of the insert filter were completely removed by wiping with a cotton swab and cells on the bottom surface of filters were fixed in methanol, then stained with 0.1% crystal violet. The migrated or invaded cells were counted microscopically in five randomly selected fields at 200× magnification.
Statistical analyses
All statistical analyses were performed with SPSS 17.0 software (SPSS Inc., Chicago, IL, USA). The association between FOXM1 (or β-catenin) expression and clinicopathologic characteristics were analyzed using χ2-test or Fisher’s exact test. Spearman test was used for analyzing the correlation. Significant differences between the groups were determined using the student’s t-test. P<0.05 was considered statistically significant. Values for all measurements were expressed as the mean ± SD with at least three independent experiments.
Results
FOXM1 and β-catenin expressions levels were upregulated in CRC tissues and associated with clinicopathologic parameters of CRC
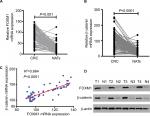
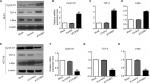
First, the mRNA expressions levels of FOXM1 and β-catenin in 124 CRC tissues and their corresponding NATs were determined by qRT-PCR. As shown in Table 1, our results showed that FOXM1 or β-catenin mRNA levels were overexpressed in 62.9% (78/124) or 67.7% (84/124) of CRC tissues, respectively, compared to those in adjacent nontumorous tissues (FOXM1 37.9% [47/124] or β-catenin 34.7% [43/124]) (P<0.05). Meanwhile, the results of Figure 1A and 1B also demonstrated that the mRNA expressions levels of FOXM1 or β-catenin in CRC tissues were significantly higher than those in corresponding NATs (P<0.05). Furthermore, we chose four CRC tissues and detected the protein expressions levels of FOXM1 and β-catenin. Our western blot analysis revealed that protein levels of FOXM1 and β-catenin were markedly upregulated in tumor samples compared to normal tissues (Figure 1D). These results indicate that FOXM1 and β-catenin might be involved in human colorectal carcinogenesis.
  | Table 1 The expression of FOXM1 and β-catenin in 124 CRCs and NATs Abbreviations: CRCs, colorectal cancers; NATs, normal-appearing tissues. |
To further explore the clinical roles of FOXM1 and β-catenin in CRC, we analyzed the potential relationships between FOXM1 (or β-catenin) expression and clinicopathologic parameters in CRC patients. As shown in Table 2, we observed that increased FOXM1 expression positively correlated with the tumor size (P=0.015), lymphatic metastasis (P=0.004), and distant metastasis (P=0.012). However, FOXM1 expression did not correlate to the age, gender, tumor location, tumor differentiation, and TNM stage of CRC patients (all P>0.05). Similar phenomena were also observed in β-catenin expression and our data implied that the high mRNA level of β-catenin was significantly associated with these clinicopathologic parameters, including the tumor size (P=0.011), TNM stage (P=0.003), lymphatic metastasis (P=0.006), and distant metastasis (P<0.0001).
  | Table 2 Correlation between the expression of FOXM1 and β-catenin with clinicopathological parameters in colorectal carcinomas Note: *P<0.05. |
FOXM1 positively correlated with β-catenin in CRC tissues
The possible relationships between FOXM1 and β-catenin in 124 CRC samples were evaluated using SPSS software and a significant positive correlation between FOXM1 and β-catenin mRNA expressions was found (R2=0.664, P<0.0001) in CRC patients (Figure 1C, Table 3).
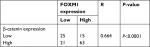  | Table 3 The relationship of FOXM1 and β-catenin in colorectal carcinomas |
FOXM1 promoted the tumorigenesis of CRC in vitro
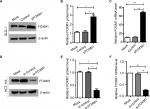
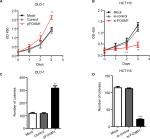
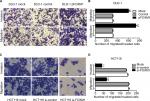
To explore the effects of FOXM1 on CRC progression in vitro, we obtained FOXM1 upregulating and downregulating cells for our subsequent experiments in vitro. When pcDNA3.1-FOXM1 plasmid or FOXM1-siRNA oligonucleotides were successfully transfected into DLD-1 or HCT116 cells, respectively, our results from western blot and PCR analysis confirmed the transfection efficiencies of FOXM1 plasmids or siRNAs (P<0.05, Figure 2A–F). The results of MTT and colony formation assay showed that FOXM1 enhanced the abilities of cell proliferation and colony formation in DLD-1 cells, whereas silencing FOXM1 expression in HCT116 cells significantly inhibited the cell proliferation and colony formation compared to the control group (P<0.05, Figure 3A–D). Meanwhile, the abilities of metastasis were evaluated by cell migration and invasion assay. Our data demonstrated that upregulation of FOXM1 significantly increased the abilities of cell migration and invasion in DLD-1 cells, whereas FOXM1 knocking down attenuated the migration and invasion capacities of HCT116 cells (P<0.05, Figure 4A–D). Taken together, these results suggested that FOXM1 could promote CRC growth and progression in vitro.
FOXM1 provoked the β-catenin signaling pathway to promote CRC progression
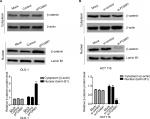
Our previous clinical data implied that FOXM1 positively correlated with β-catenin in CRC. To uncover the underlying mechanism of FOXM1-mediated oncogenic activity, we examined the effect of FOXM1 on the activation of β-catenin signaling. First, we examined the protein and mRNA levels of cyclin D1, c-Myc, and TCF-4, which are critical downstream targets of β-catenin signaling pathway when FOXM1 was restored or silenced in CRC cells. Our results showed that FOXM1 overexpression gave rise to an increase of cyclin D1, TCF-4, and c-Myc in DLD-1 cells in both protein and mRNA levels (Figure 5A–D) but were inhibited by FOXM1 siRNAs in HCT116 cells (Figure 5E–H). We detected the expression of FOXM1 and β-catenin in both the cytoplasm and the nucleus by western blots. Interestingly, the results showed that the nuclear protein level of β-catenin was increased by FOXM1 overexpression in DLD-1 cells but decreased in HCT116 cells with FOXM1 knockdown, whereas the cytoplasmic level was not changed (Figure 6A, B). These data indicate that FOXM1 triggers β-catenin signaling pathway potential by improving nuclear translocation of β-catenin in colon cancer cells.
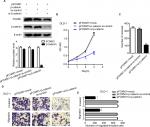
Silencing β-catenin rescued the FOXM1-induced tumorigenicity in CRC cells
The previous study of FOXM1 on activation of β-catenin signaling prompted us to investigate the effect of β-catenin inhibition on FOXM1-mediated malignant activities. Cells with FOXM1 overexpression were transfected with β-catenin siRNA. Multiple biological experiments in vitro were performed, such as MTT, colony formation, and transwell assays. The data demonstrated that silencing β-catenin expression did not change the levels of FOXM1 in FOXM1-overexpression cells, compared to the control cells which were transfected with mock or β-catenin-si-control plasmid (Figure 7A, P>0.05). Additionally, FOXM1-promoted colon cancer growth and formation were eliminated by β-catenin siRNA (Figure 7B, 7C). Similarly, increased cell migration and invasion by FOXM1 overexpression was also eliminated (Figure 7D, E). Taken together, our data suggest that FOXM1 and β-catenin are important for CRC cell progression and inhibition of their expression is a promising strategy for CRC therapy.
Discussion
There is growing evidence that FOXM1 serves as a key oncogene and novel prognostic biomarker in the initiation and progression of many cancers.9,20–23 However, the underlying mechanism of FOXM1 in CRC is still not fully elucidated. To explore the significance of altered FOXM1 in CRC, we detected the mRNA expressions of FOXM1 in 124 CRC tissues by qRT-PCR and found that FOXM1 was significantly upregulated in CRC tissues. As described in previous study, β-catenin is a key molecule in the canonical Wnt signaling pathway in many human cancers.24 Our results from PCR analysis also showed that β-catenin mRNA expression was markedly enhanced in CRC tissues, whereas lower expression of β-catenin was found in adjacent normal tissues. We consistently chose four CRC tissues and detected the protein expressions of FOXM1 and β-catenin. Our western blot analysis revealed that protein levels of FOXM1 and β-catenin were also markedly upregulated in tumor samples compared to normal tissues. Besides, our clinicopathologic analysis revealed that increased FOXM1 (or β-catenin) expression positively correlated with some clinical features, such as the tumor size, TNM stage, lymphatic metastasis, or distant metastasis. Interestingly, the possible relationships between FOXM1 and β-catenin in CRC samples were evaluated using SPSS software and a significant positive correlation was found. Hence, these results indicated that FOXM1 and β-catenin might work together in human colorectal carcinogenesis.
The long-term survival of CRC patients is extremely unsatisfactory because of recurrences and distant metastases. It has been reported that liver metastasis after curative colectomy occurs in nearly 50% of colon cancer patients.25 Cell growth, invasion, and metastasis are now regarded as important characteristics of cancers. To further explore the effects of FOXM1 on CRC progression, we obtained FOXM1 upregulating and downregulating cells in vitro. Our experiments demonstrated that upregulation of FOXM1 significantly increased the abilities of cell proliferation, colony formation, migration, and invasion in DLD-1 cells, whereas silencing FOXM1 expression exerted an obvious inhibition of tumor cell growth and metastasis in HCT116 cells. Thus, there was strong evidence that FOXM1 plays an important role in the development of CRCs.
The diverse behavior of FOXM1 depends on its various target genes in different cancer types. It has been reported that a variety of FOXM1 downstream target molecules are involved in regulating tumor progression and invasive processes, such as MMP-2, MMP-9, uPAR, VEGF, PLAUR, Snail,10,26–28 or some microRNAs.29–31 Furthermore, alterations in FOXM1 signaling pathway have been reported to be associated with tumor initiation and progression. For instance, the aberrant activation of Hedgehog (Hh) signaling promoted CRC cell proliferation by directly binding to the promoter of FOXM1 and transactivating the activity of FOXM1, and the dysregulation of the Hh-Gli1-FOXM1 axis was essential for human CRC tumorigenesis.32 In 2007, Yoshida et al constructed FOXM1 transgenic and knockout mice models, and initially found that FOXM1 was critical for the proliferation and growth of CRC with decreased expression of cyclin A2, cyclin B1, survivin, and TCF4 genes. β-catenin has proved to be a subunit of the cadherin protein complex, which mediates the Wnt transcriptional response.24 Our current data demonstrated that overexpression of FOXM1 resulted in the accumulation of nuclear β-catenin and facilitated the expression of β-catenin downstream target genes, including TCF-4, c-Myc, and cyclin D1, which indicates that FOXM1 might manifest its protumor activity by enhancing the transactivation of β-catenin signaling pathway. These results were partly consistent with previous studies in other cancers. For example, FOXM1 was proved to be a downstream component of Wnt signaling and was critical for β-catenin transcriptional function in glioma.33 A similar study was also reported by Chen in 2016. They found that FOXM1 accumulation in the nucleus promoted recruitment of β-catenin to Wnt target-gene promoter and activated the Wnt signaling pathway by protecting the β-catenin/TCF4 complex in glioma.34
Conclusion
In summary, our study demonstrated that FOXM1 and β-catenin were both upregulated in CRC tissues and enhanced FOXM1 expression positively correlated with β-catenin in CRC. In vitro, restoring FOXM1 expression significantly promoted the CRC growth and metastasis by activating the β-catenin downstream effectors, whereas silencing β-catenin could diminish the FOXM1-induced tumorigenicity. Our results provided convincing evidence that elevated FOXM1 expression exerted oncogenic activities towards CRC via activation of β-catenin signaling, which suggests that FOXM1 and β-catenin are important for CRC cell progression and that inhibition of their expression is a promising strategy for CRC therapy.
Acknowledgment
This study was supported by grants from the Special Subject of Diagnosis Treatment of Key Clinical Diseases of Suzhou City Science and Technology Bureau (LCZX201401) and Jiangsu Province’s Youth Provincial Talents Program (QNRC2016723).
Disclosure
The authors report no conflicts of interest in this work.
References
Brenner H, Kloor M, Pox CP. Colorectal cancer. Lancet. 2014; 383(9927):1490–1502. | ||
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63(1):11–30. | ||
Kalin TV, Ustiyan V, Kalinichenko VV. Multiple faces of FoxM1 transcription factor: lessons from transgenic mouse models. Cell Cycle. 2011;10(3):396–405. | ||
Teh MT. FoxM1 coming of age: time for translation into clinical benefits? Front Oncol. 2012;2:146. | ||
Ahmad A, Wang Z, Kong D, et al. FoxM1 down-regulation leads to inhibition of proliferation, migration and invasion of breast cancer cells through the modulation of extra-cellular matrix degrading factors. Breast Cancer Res Treat. 2010;122(2):337–346. | ||
Kim IM, Ackerson T, Ramakrishna S, et al. The forkhead box M1 transcription factor stimulates the proliferation of tumor cells during development of lung cancer. Cancer Res. 2006;66(4):2153–2161. | ||
Kong X, Li L, Li Z, et al. Dysregulated expression of FoxM1 isoforms drives progression of pancreatic cancer. Cancer Res. 2013;73(13):3987–3996. | ||
Zhang HG, Xu XW, Shi XP, et al. Overexpression of forkhead box protein M1 (FoxM1) plays a critical role in colorectal cancer. Clin Transl Oncol. 2016;18(5):527–532. | ||
Xu N, Jia D, Chen W, et al. FoxM1 is associated with poor prognosis of non-small cell lung cancer patients through promoting tumor metastasis. PLoS One. 2013;8(3):e59412. | ||
Wen N, Wang Y, Wen L, et al. Overexpression of FoxM1 predicts poor prognosis and promotes cancer cell proliferation, migration and invasion in epithelial ovarian cancer. J Transl Med. 2014;12(1):134. | ||
Liu M, Dai B, Kang SH, et al. FoxM1B is overexpressed in human glioblastomas and critically regulates the tumorigenicity of glioma cells. Cancer Res. 2006;66(7):3593–3602. | ||
Dai B, Kang SH, Gong W, et al. Aberrant FoxM1B expression increases matrix metalloproteinase-2 transcription and enhances the invasion of glioma cells. Oncogene. 2007;26(42):6212–6219. | ||
Li Q, Zhang N, Jia Z, et al. Critical role and regulation of transcription factor FoxM1 in human gastric cancer angiogenesis and progression. Cancer Res. 2009;69(8):3501–3509. | ||
Zhang Y, Zhang N, Dai B, et al. FoxM1B transcriptionally regulates vascular endothelial growth factor expression and promotes the angiogenesis and growth of glioma cells. Cancer Res. 2008;68(21):8733–8742. | ||
Yang K, Jiang L, Hu Y, et al. Short hairpin RNA-mediated gene knockdown of FOXM1 inhibits the proliferation and metastasis of human colon cancer cells through reversal of epithelial-to-mesenchymal transformation. J Exp Clin Cancer Res. 2015;34(1):40. | ||
Chu XY, Zhu ZM, Chen LB, et al. FOXM1 expression correlates with tumor invasion and a poor prognosis of colorectal cancer. Acta Histochem. 2012;114(8):755–762. | ||
Giles RH, Van Es JH, Clevers H. Caught up in a Wnt storm: Wnt signaling in cancer. Biochim Biophys Acta. 2003;1653(1):1–24. | ||
Behrens J, Lustig B. The Wnt connection to tumorigenesis. Int J Dev Biol. 2004;48(5-6):477–487. | ||
Yoshida Y, Wang IC, Yoder HM, Davidson NO, Costa RH. The forkhead box M1 transcription factor contributes to the development and growth of mouse colorectal cancer. Gastroenterology. 2007;132(4):1420–1431. | ||
Pilarsky C, Wenzig M, Specht T, Saeger HD, Grützmann R. Identification and validation of commonly overexpressed genes in solid tumors by comparison of microarray data. Neoplasia. 2004;6(6):744–750. | ||
Uddin S, Ahmed M, Hussain A, et al. Genome-wide expression analysis of middle Eastern colorectal cancer reveals FoxM1 as a novel target for cancer therapy. Am J Pathol. 2011;178(2):537–547. | ||
Xia L, Huang W, Tian D, et al. Upregulated FoxM1 expression induced by hepatitis B virus X protein promotes tumor metastasis and indicates poor prognosis in hepatitis B virus-related hepatocellular carcinoma. J Hepatol. 2012;57(3):600–612. | ||
Xia JT, Wang H, Liang LJ, et al. Overexpression of FoxM1 is associated with poor prognosis and clinicopathologic stage of pancreatic ductal adenocarcinoma. Pancreas. 2012;41(4):629–635. | ||
Logan CY, Nusse R. The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol. 2004;20(1):781–810. | ||
van Cutsem E, Nordlinger B, Adam R, et al. Towards a pan-European consensus on the treatment of patients with colorectal liver metastases. Eur J Cancer. 2006;42(14):2212–2221. | ||
Wang Z, Banerjee S, Kong D, Li Y, Sarkar FH. Down-regulation of forkhead box M1 transcription factor leads to the inhibition of invasion and angiogenesis of pancreatic cancer cells. Cancer Res. 2007;67(17):8293–8300. | ||
Li D, Wei P, Peng Z, et al. The critical role of dysregulated FOXM1-PLAUR signaling in human colon cancer progression and metastasis. Clin Cancer Res. 2013;19(1):62–72. | ||
Wei P, Zhang N, Wang Y, et al. FoxM1 promotes lung adenocarcinoma invasion and metastasis by upregulating SNAIL. Int J Biol Sci. 2015;11(2):186–198. | ||
Ma N, Zhang W, Qiao C, et al. The tumor suppressive role of MiRNA-509-5p by targeting FOXM1 in non-small cell lung cancer. Cell Physiol Biochem. 2016;38(4):1435–1446. | ||
Sun Y, Yu X, Bai Q. miR-204 inhibits invasion and epithelial-mesenchymal transition by targeting FOXM1 in esophageal cancer. Int J Clin Exp Pathol. 2015;8(10):12775–12783. | ||
Duan N, Hu X, Yang X, Cheng H, Zhang W. MicroRNA-370 directly targets FoxM1 to inhibit cell growth and metastasis in osteosarcoma cells. Int J Clin Exp Pathol. 2015;8(9):10250–10260. | ||
Wang D, Hu G, Du Y, et al. Aberrant activation of hedgehog signaling promotes cell proliferation via the transcriptional activation of forkhead Box M1 in colorectal cancer cells. J Exp Clin Cancer Res. 2017;36(1):23. | ||
Zhang N, Wei P, Gong A, et al. FoxM1 promotes β-catenin nuclear localization and controls Wnt target-gene expression and glioma tumorigenesis. Cancer Cell. 2011;20(4):427–442. | ||
Chen Y, Li Y, Xue J, et al. Wnt-induced deubiquitination FoxM1 ensures nucleus β-catenin transactivation. EMBO J. 2016;35(6):668–684. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.