Back to Journals » Patient Related Outcome Measures » Volume 11
Feasibility and Utility of Incorporating Patient-Reported Outcomes into Surveillance Strategies for Advanced Lung Cancer
Authors Cavanna L , Citterio C, Orlandi E
Received 7 December 2019
Accepted for publication 6 February 2020
Published 13 February 2020 Volume 2020:11 Pages 49—66
DOI https://doi.org/10.2147/PROM.S179185
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Robert Howland
Luigi Cavanna, Chiara Citterio, Elena Orlandi
Oncology and Hematology Department, Oncology Unit, Piacenza General Hospital, Piacenza 29121, Italy
Correspondence: Luigi Cavanna
Oncology and Hematology Department, Piacenza General Hospital, Via Taverna 49, Piacenza 29121, Italy
Tel +39 0523302697
Fax +39 0523302141
Email [email protected]
Purpose: To identify and to describe patient-reported outcomes (PROs) in lung cancer patients and to evaluate the feasibility and utility of PROs into surveillance strategies, a review was carried out.
Patients and Methods: A systematic search in bibliographic databases evaluating the instruments used in PROs of non-small-Cell lung cancer (NSCLC) patients was done.
Results: From August 2014 to August 2019, 33 studies were included in this review and 16,491 patients were evaluated. PROs were divided into 6 different categories: 1) PROs as a guide in therapeutic choice, 2) PROs as indicator of disease progression, 3) agreement between PROs and the evaluated parameters, 4) PROs to evaluate the effects of immunotherapy, 5) need to deepen the knowledge of PROs, and 6) use of new electronic PROs.
Conclusion: The most frequently used instruments are EORTC QLQ-30 (16, 50%) and EORTC LC-13 (14, 43.75%) and in some studies (37.5%) they are used together. For different reasons (disease progression, adverse event, death, incomplete participation, etc.), the completion of these instruments decreased over time from baseline to subsequent measurements. This review demonstrates that PROs can play an important role as part of health care, and that routine use implementation could improve patient management in addition to the traditionally collected outcome.
Keywords: quality of life, lung cancer, patient-reported outcomes, PROs
Introduction
Lung cancer still has a particularly poor prognosis: 60-month survival ranges are between 68% in patients with stage I disease, to 0–10% in stage IV patients. It is the leading cause of cancer deaths in Western countries.1,2 Due to its symptom-free course, lung cancer is often diagnosed in an advanced stage and the most frequent and clinically relevant disease-related symptoms experienced by patients in an advanced stage are pain, fatigue, dyspnea, and cough, with a significant impact on the health-related quality of life (HR QoL).3,4 It must be emphasized that patients with advanced/metastatic lung cancer during the course of the disease develop devastating physical and psychosocial symptoms, though new target therapies and immunotherapies are changing the situation of advanced/metastatic lung-cancer patients, improving disease-free survival (DFS) and overall survival (OS).5 However, old and new therapies for metastatic lung cancer show adverse effects that can worsen the quality of life and worsen the prognosis if not promptly diagnosed and treated. Some studies reported that incorporating patient-reported outcomes (PROs) into surveillance strategies for advanced lung cancers appears to improve both quality of life and outcome in patients with advanced/metastatic lung cancer.6 Therefore, to analyze the role of PROs in the management of patients with advanced/metastatic lung cancer, we performed a review regarding feasibility and utility of incorporating patient-reported outcomes into surveillance strategies for advanced lung cancer.
Materials and Methods
A systematic search in the bibliographic databases, PubMed, Cochrane Library and EMBASE to identify all relevant publications was performed. In accordance with the guideline Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA),7 the latest database search was conducted on 19 Aug 2019 using the following search terms: “reported outcomes” AND “advanced lung cancer” with the restricted publication date “last 5 years”. For each study, data about authors, year of publication, type of study, number of patients included, treatment, instruments were used to asses QoL and timing for data collection were extracted. Only prospective observational studies, randomized-controlled trials and a post-hoc analysis are reported in this review. Two authors (LC and CC) independently evaluated the paper for the potential eligibility in this systematic review based on the inclusion criteria, with disagreements resolved by consensus with the third author. We cannot evaluate utility and feasibility of PROs directly, using a parameter common to all PROs, so we reported them evaluating clinical applicability but also the limits.
Instruments Utilized in the Reviewed Studies
In the articles analyzed, a total of 21 different PROs were described focused on different dimensions of the QoL (ie functional, physical, emotional and social function). The quality-of-life instruments evaluated can be categorized in general (ESAS), cancer-specific (EORTC QLQ-C30, RALS, PMC, FACT - G, SF-36, PRO-CTCAE, MDASI), lung-cancer-specific (EORTC LC-13, LCSS, FACT - L, LCS, NSCLC-SAQ, MDASI-LC), specific symptoms (FACT- F and SCFS for fatigue, FAACT and PG-SGA for Anorexia/Cachexia/nutrition and EAT-10 and SWAL-QoL for dysphagia), specific for Work Productivity and Activity Impairment (WPAIGH) and instruments to evaluate the caregivers’ burdens (ZBI).
The European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30)8 is a standardized cancer-specific instrument for measuring HRQL and symptom and functional domains consisting of 30 items, with subscales related to physical, role, emotional, cognitive and social functioning, global health status/quality of life, symptoms and financial issues. Scores are reported on a scale of 0–100.
The EORTC Lung Cancer-13 (EORTC LC-13)9 is a standardized instrument that measures lung-cancer-specific symptoms (dyspnea, cough, hemoptysis and site-specific pain), chemotherapy/radiotherapy-related side effects and medication use for pain. Scores are reported on a scale of 0–100.
The Lung Cancer Symptom Scale (LCSS)10,11 is a standardized instrument for measuring lung cancer symptoms and HRQL; it evaluates different symptoms (pain, loss of appetite, fatigue, cough, dyspnea and hemoptysis) and general effects related to QoL. Each item is measured using a visual analogue scale, with scores reported on a scale of 0–100.
The MD Anderson Symptom Inventory (MDASI) and lung cancer module of the MDASI (MDASI-LC)12,13 is a multi-symptom patient-reported outcome measurement for clinical and research use. It includes symptoms found to have the highest frequency and/or severity in patients with various cancers and treatment types. The MDASI-LC is specific for lung cancer. It includes coughing.
The Rotterdam Activity Level Scale (RALS) of the Rotterdam Symptom Checklist (RSCL)14 was developed to measure the symptoms reported by cancer patients participating in clinical trials but it is also applicable to monitoring the levels of the patient’s anxiety and depression, and reflects the presence of psychological illness.
The Patients’ symptoms via the Patient Care Monitor (PCM) v2.015–17 is a review of system surveys with different lengths depending on gender. Each question is scored on a scale ranging from 0 to 10. This instrument has been validated and demonstrates a significant correlation with other validated instruments.
The FACT measurement system includes FACT - G Functional Assessment of Cancer Therapy – General, FACT - L Functional Assessment of Cancer Therapy – Lung Cancer, FACT - F Functional Assessment of Cancer Therapy – Fatigue, FAACT Functional Assessment of Anorexia/Cachexia Treatment.18–20 Since 1987 the FACT measurement system has been developed; FACT - G was developed to measure QoL in cancer patients receiving therapy. Now it is also a well-established instrument in cancer-related treatment evaluations and clinical interventions. To this questionnaire, problems that are specific to a particular type of cancer can be added. The FACT - L questionnaire asks about symptoms reported by lung cancer patients which may affect their quality of life. The FACT - F questionnaire assessing fatigue and anemia-related concerns in people with cancer; the FAACT uses an anorexia-cachexia subscale as a marker of QoL in patients with advanced lung cancer and cancer anorexia-cachexia syndrome.
The 36-item Short-Form Health Survey (SF-36)21,22 is a generic questionnaire that generates eight subscale scores with values from 0 to 100. It can be used to evaluate the physical and mental condition of the patient.
The Lung Cancer Subscale (LCS) of the Functional Assessment of Cancer Therapy – Lung (FACT - L)23,24 is an independently validated tool to assess symptoms of lung cancer. The maximum score is 28; lower scores represent more severe symptoms.
The Schwartz Cancer Fatigue Scale (SCFS)25 is a reliable and valid patient-reported measure of fatigue severity with a 5-point scale to generate a score ranging from 6 to 30.
The Patient-Generated Subjective Global Assessment (PG-SGA)26 is a highly sensitive and specific screening tool to identify the need for nutritional intervention in patients with cancer; data produce 3 categories: well-nourished, moderately malnourished and severely malnourished.
The Patient-Reported Outcomes version of Common Terminology Criteria for Adverse Events (PRO-CTCAE)27 is a validated PRO measurement system developed by the US NCI to assess symptoms possibly related to cancer treatments. PRO-CTCAE items are listed in publicly available libraries and cover 78 symptoms, each item reflects specific symptom attributes included in the corresponding CTCAE.
The Eating Assessment Tool (EAT-10)28 is used to obtain a self-reported prevalence estimate for both oropharyngeal and esophageal dysphagia in the samples.
The Swallowing Quality of Life instrument SWAL-QoL is used to measure the impact of dysphagia on QoL.29
The Non-Small Cell Lung Cancer Symptom Assessment Questionnaire (NSCLC-SAQ)30 is a 7-item PRO measurement for use in advanced NSCLC. It uses a 7-day recall period and verbal rating scale.
The Work Productivity and Activity Impairment: General Health (WPAI: GH)31 is a validated, non-disease-specific tool and consists of items about work (employment status, hours missed from work, etc.) used to give four data: work time missed; impairment while working; overall work impairment; and activity impairment.
The Zarit Burden Interview (ZBI)32 is a validated scale that measures feelings of the burdens of caregivers for patients with a range of medical and psychological conditions.
The Edmonton Symptom Assessment Scale (ESAS)33 consists of 9 common symptoms that patients rate on an 11-point scale, from 0 to 10: pain, fatigue, nausea, depression, anxiety, drowsiness, inappetence, malaise and dyspnea.
Results
The search yielded a total of 179 articles (Figure 1). After removing duplicates and case reports, 21 were excluded. We screened 158 articles and 126 were excluded since they did not meet the inclusion criteria: 84 used instruments not patient-reported, 39 included patients suffering from different types of cancer, not lung cancer or not only lung cancer, 2 reports were available only in abstract form. Finally, 33 studies were included (Table 1). Four articles were published in 2015, 5 in 2016, 10 in 2017, 8 in 2018 and 6 in 2019. Seventeen studies are randomized clinical trials, 15 observational studies and 1 is a post-hoc analysis. The number of patients included in the studies ranges from a minimum of 32 to a maximum of 1466 for a total of 16,491 included patients. In most studies, the PROs reported data on standard chemotherapy (first or second line), or target therapy, such as tyrosine kinase inhibitors (TKIs), or immunotherapy. Other studies also use chemoradiotherapy, second-line and maintenance therapies. Fifteen (45.45%) compare the QoL of patients treated with two different therapeutic regimens; 1 (3.03%) was a validation study, 2 (6.06%) evaluated PROs ability to detect symptom, 4 (12.12%) evaluated symptom severity, 2 (6.06%) evaluated QoL of patients treated with a specific regimen (nivolumab and brigatinib), 1 (3.03%%) evaluated the QoL of patients treated with concurrent chemoradiation, 8 (24.24%) analyzed the correlation between QoL and different factors: age, EGFR mutation, brain metastasis, Cancer Anorexia-Cachexia Syndrome, dysphagia, disease progression, physical activity and patients’ and caregivers’ burdens.
 |  |  |  |  |  |
Table 1 Identified Literature Overview |
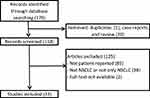 |
Figure 1 Flowchart of study selection. |
We can classify the results of these articles into six categories (Table 2); the first includes all those studies where results indicate the use of PROs as a guide in therapeutic choice: the PRO-CTCAE has the potential to bring the patient’s voice to clinical trials, and to provide insights into the patient’s experiences of treatment,34 the use of PROs measures in addition to PFS in the development of new treatments and as a consideration when choosing available treatment options for patients in the out-patient clinics were provided;35 the integration of PROs generated in clinical trials as well as at an individual patient level is required to enable shared decision-making and personalized health care based on a mutual understanding of treatment objectives and expectations.36 The ESAS questionnaire has a key role for the data it provides not only as a guide to symptom management for improved quality of life, but also in the development of optimal treatment plans and estimation of OS.37 In addition, it was reported that converting QLQ-C30 scores into utilities in trials using established mapping algorithms can improve the evaluation of medicines from a patient’s perspective.38
 |
Table 2 Results Classification in Categories About the Topic of the Article |
The second part of the article concerns the use of PROs as an indicator of disease progression, patient’s QoL and survival. The differences of HR QoL evaluated with different PROs for patients treated with the two treatments under evaluation and their concordance about the outcome evaluated in the study were reported.39 Disease progression had a significant adverse impact on many PROs’ endpoints;40 PROs showed that they were useful in identifying patients with advanced NSCLC who are likely to have significantly lower survival.41 The prognostic power of overall QoL on the survival of lung cancer patients and the advantages for lung cancer survivors reporting to be physically active was demonstrated versus those who were not physically active;42 the early detection of symptomatic relapse and management of symptoms through a web-mediated individualized follow-up strategy provided an improvement in quality of life and overall survival.43
The third group included the studies that evaluated the agreement between the PROs and the evaluated parameters: concordance between LCSS and ECOG PS measurements and tumor-related symptoms is reported.44,45 The use of PROs for the accurate assessment of health-related QoL in patients with lung cancer and to identify instruments to improve the value of care delivered was reported.46,47 The utility of PROs to evaluate improvements in HR QoL and symptoms burden of the subgroups of patients treated with nivolumab and to analyze the AEs between the two treatment groups was reflected in PRO outcomes.48 The correlation between PRO scores and deterioration of HR QoL in patient with brain metastases at the diagnosis of lung cancer was evaluated.49 Using the EAT-10 and SWAL-QoL questionnaires, dysphagia was demonstrated a potential symptom in advanced lung cancer which may impact QoL, and some studies reported change in the mental component of HR QoL related to PROs outcomes50 or showed the symptoms that need to be addressed in routine care of advanced NSCLC;51 different studies reported that PROs are arguably more representative of the patient perspective than physician-reported outcomes.52–54
The fourth group concerned the evaluation of the role of PROs in assessing the effects of immunotherapy: the EORTC QLQ-LC13 instrument may not adequately reflect the experiences of patients who receive new therapies.55 They concluded that commonly occurring symptoms attributed to immunotherapy can be found by using the EORTC QLQ-C30 and EORTC QLQ-LC13 questionnaires. PROs disease questionnaire find some, but not all relevant symptoms in a disease area and are unlikely to discover several common toxicities related to immunotherapy drugs.56 PROs assessments should be considered standard tools in the future of cancer immunotherapy research because their use will lead to a better understanding of the effect of immunotherapy on patients’ outcomes. Beyond the traditional parameters of OS and radiographic endpoints,57 some authors reported the need to develop instruments capable of evaluating the treatment-related symptoms hand-in-hand with the introduction of new therapies with specific related symptoms,58 the prospective evaluation of PROs with the appropriate hypothesis and instruments, is vital particularly in clinical trials that evaluate new therapeutics in incurable cancers.59
Another group of studies expresses the need to deepen the knowledge of PROs: the MDASI scale was used to evaluate patient-reported symptoms. This scale does not evaluate coughing, a common symptom in lung cancer; as a standard of care and in clinical trials, the MDASI-LC test, which includes coughing, must be used to a complete evaluation of the patient’s symptoms.60 Widely used PRO-based symptom assessment tools are needed to facilitate a comparison of results with those of other published studies.61 Only one study62 reported the use of a new electronic PRO: the NSCLC-SAQ, after Food and Drug Administration (FDA) qualification, will be publicly available to capture patient-reported NSCLC-related symptoms via electronic data entry platforms. The use of PROs in the studies evaluated, it is not always a primary outcome. In some cases, especially for studies that evaluate PROs as a guide in therapeutic choice, PROs as indicators of disease progression and agreement between PROs and the evaluated parameters, the data obtained from the use of PROs is a secondary outcome of the study.
Discussion
The purpose of this systematic review was to identify and describe peer‐reviewed PROs used to evaluate QoL in lung cancer patients and to evaluate the feasibility and utility of PROs in surveillance strategies. Worldwide, lung cancer remains a disease with severe morbidity and mortality. Therefore, in addition to survival, the QoL of the patient is of great importance. There is a growing interest in measuring the QoL with the aid of PROs.63 The subjective data about how therapies and diseases can modify patients’ lives can be useful for the physicians and can compensate for the lack of time the physicians have for outpatients to understand the patients’ point of view. In the studies reviewed, PRO-based endpoints are indicative of clinical benefit in terms of patient symptoms and overall quality of life. The addition of PROs to traditionally collected outcome measures (OS, PFS, DFS) can offer a comprehensive overview of patient status. In an optimal setting, the PROs should allow for an overall assessment of QoL, along with specific questionnaires to assess specific effects associated with the disease and treatment. The most frequently used instruments are EORTC QLQ-30 (16, 50%) and EORTC LC-13 (14, 43.75%) and in some studies (37.5%) EORTC QLQ-C30 was supplemented with the EORTC QLQ-LC13, result in agreement with the European literature.64
There was great dispersion in data collection timing: the baseline is often collected, and subsequent checks ranging from a few weeks to some years or until PD or the start of a new treatment.
It must be emphasized that there is much evidence in the literature about the benefits of collecting and using PROs in lung cancer populations, for treatment monitoring,35,38-40,44–48,51,55,57-59,65–67 detection of symptoms,36,37,41,50,53,54,60,65 the role of patient or pathology characteristics on QoL,40,42,56,68 to improve patient-clinician communication and patient satisfaction.43,61,62 Only one study about the effect of disease progression on QoL was identified49 assessing the use of PROs related to efficacy outcomes (PFS, OS). Data on PRO completion rates are available for 20 studies (60.61%) (Table 3), only in 3 studies (15%) was it lower than 80%;44,47,59 for the others 17 (85%) it was higher than 80%.35,36,39,40,45,48,49,51,52,55-58,61,66-68 Only in one study (5%) did the authors report a completion rate of 100%.52 In most cases, frequencies decreased over time from baseline to subsequent measurements. The reasons given in the different studies were the increasing number of patients who discontinued the study due to disease progression, physician decision, adverse events, or death and the incomplete patient participation at each time point.
 |
Table 3 Literature Data |
PROs are primarily collected in the context of scientific research. Moreover, the majority of the clinical trials do not use generic instruments but give preference to disease-specific instruments as they are more sensitive to subtle changes. Assessment of HRQL can help to better understand the physical, mental, and emotional implications of cancer and the effects of treatment on patients. PRO use was developed in scientific research to understand the efficacy of a treatment and to evaluate symptom type and impact on QoL and to act in a timely manner to control the symptom and its consequences on QoL. They become important also to evaluate new therapies taking into consideration, in addition to clinical results, also the quality of life of the patient. Since the association of QoL with survival PROs use appears to be a strong and independent factor in the prediction of survival in lung cancer.64,69
The literature demonstrates that the use of PROs continues to increase, the collected data support the hypothesis that PROs can play an important role as part of the health care and that routine use implementation could improve patient management: early identification of symptoms and adverse events due to treatment, monitor the patient’s response to therapy and improve communication between patient, health care professional and caregivers70 with a consequent reduction of costs for exams, therapies and hospitalizations avoidable with a timely intervention. Steps for routine implementation of PROs were previously reported71 but cost-effectiveness of the use of PROs is still under evaluation. Despite the good psychometric properties and all the possible advantages of instruments already mentioned, the feasibility of the routine implementation of PROs finds some practical difficulties: the availability of personnel, programming of training for the correct administration and interpretation of PROs, costs, time required and the need for people able to analyze the data collected.72 Without proper preparation and organization, their use is disruptive to normal work routines. One of the things that made the PROs easier to administer, but mostly simplified the data collection and analysis phase, was the transition from paper versions to electronic platforms, with a significant reduction of time.73
Considering the fact that there are almost 200 tumor types, PROs are not cancer-specific because they do not consider that different cancers involved different symptoms, as well as they do not specify selected treatment benefits or toxicity. There is no objective consideration and no comparison between the questionnaires that can be formulated. Further studies would be useful to assess the symptoms associated with different therapies such as immunotherapy.
However, there are many considerations that need attention to enable long-term, quality collection and use of PRO data within routine clinical settings.71 These include identifying the goals of PROs collection; patient selection; setting and timing of assessments; choice of questionnaire; scale of interpretation and the way to facilitate it, developing strategies for responding to issues detected by the questionnaires and evaluating the impact of the PROs in the practice. To our knowledge, there are no comparison parameters between the various PROs and therefore it is difficult to find a method of evaluation of utility and feasibility applicable in the comparison between the different used tools. Utility and feasibility are described by the assessment of costs, times, staff required, limits of applicability and interpretation issues.
The limitations of this revision are the reduced time period of publication, the choice of PROs also not specific for lung cancer and the exclusion of studies in which PROs were not the primary objective. PROs results, obtained in a routine collection could provide the basis on which adapt therapy and interventions to the needs of the patient, to improve both QoL and the probability of survival. A study carried out in 201174 shows the improvement of the communication between the patient and the health care professional, and the monitoring of the response to treatment and the satisfaction of the patient when routinely collecting PROs. More recently, a randomized clinical trial investigated the influence of routinely collecting PROs in cancer patients.75 It evaluated the use of e-mail alerts to severe symptoms or to usual care; the e-mail system allowed an immediate action such as medications or access to the emergency room. The results were a better control of symptoms in the e-mail group, resulted in a better HR QoL, fewer visits to the emergency room, fewer hospitalizations, longer duration of palliative chemotherapy and both improved one-year survival and quality-adjusted survival.76 PROs are also applied in the field of palliative care, where different studies have tried to evaluate the impact on the OS and patients QoL but the factors to be evaluated, including those related to the families and the patient’s caregivers are complex. The number of studies is still reduced and the results are difficult to compare.77,78
The inclusion of PROs as endpoints in clinical trials is encouraged by the FDA, the European Medicines Agency (EMA)79 and scientific societies as the European Society of Medical Oncology.80 US Food and Drug Administration has recently published guidelines on the review and evaluation of PROs to encourage their appropriate use in regulatory studies and decisions.
Conclusion
A great variety of PROs are used with lung cancer patients in order to improve quality of care. A general questionnaire to assess overall QoL, which can be supplemented with disease-specific questionnaires allowing for the assessment of QoL of different treatment methods seems to be most effective. PROs can be used for different purposes and can be focused on the specific disease or symptoms or related to the progression of the disease. The standard routine use of PROs is still not widely recognized, despite the positive aspects reported. This can be related to organization, timing and personnel difficulties. A step towards solving these problems is the introduction of electronic PROs. We emphasized the unmet need for focused research to justify and to guide the analytic method of PROs to facilitate the interpretation of patient experience. Future research should assess the applicability of PROs in routine clinical practice.
Abbreviations
SF-36, 36-item Short-Form Health Survey; MDASI, MD Anderson Symptom Inventory; DFS, disease-free survival; EAT-10, Eating Assessment Tool; ESAS, Edmonton Symptom Assessment Scale; EMA, European Medicines Agency; EORTC QLQ-C30, European Organization for Research and Treatment of Cancer Quality of Life Questionnaire; FDA, Food and Drug Administration; FAACT, Functional Assessment of Anorexia/Cachexia Treatment; FACT - F, Functional Assessment of Cancer Therapy - Fatigue; FACT - G, Functional Assessment of Cancer Therapy - General; FACT - L, Functional Assessment of Cancer Therapy - Lung Cancer; HR QoL, health-related quality of life; LCSS, Lung Cancer Symptom Scale; NSCLC-SAQ, Non-Small Cell Lung Cancer Symptom Assessment Questionnaire; OS, overall survival; PROs, Patient Reported Outcomes; PG-SGA, Patient-Generated Subjective Global Assessment; PRO-CTCAE, Patient-Reported Outcomes version of Common Terminology Criteria for Adverse Events; PCM, Patients’ symptoms via the Patient Care Monitor; RALS, Rotterdam Activity Level Scale; SWAL-QoL, Swallowing Quality of Life instrument; LCS, The Lung Cancer Subscale; SCFS, The Schwartz Cancer Fatigue Scale; TKIs, tyrosine kinase inhibitors; WPAIGH, Work Productivity and Activity Impairment: General Health; ZBI, Zarit Burden Interview.
Author Contributions
All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
Luigi Cavanna reports consulting and advisory roles for AstraZeneca and Merck; travel and accommodation expenses from Celgene, Pfizer, and Ipsen. The other authors have no financial support or relationships that may pose a conflict of interest with this work.
References
1. Duma N, Santana-Davila R, Molina JR. Non-small cell lung cancer: epidemiology, screening, diagnosis, and treatment. Mayo Clin Proc. 2019;94(8):1623–1640. doi:10.1016/j.mayocp.2019.01.013
2. Torre LA, Siegel RL, Jemal A. Lung cancer statistics. Adv Exp Med Biol. 2016;893:1–19.
3. Tishelmann C, Degner LF, Rudman A, et al. Symptoms in patients with lung carcinoma: distinguishing distress from intensity. Cancer. 2005;104:2013–2021. doi:10.1002/(ISSN)1097-0142
4. Cooley ME. Symptoms in adults with lung cancer: a systematic research review. J Pain Symptom Manage. 2000;19:137–153. doi:10.1016/S0885-3924(99)00150-5
5. Giroux Leprieur E, Dumenil C, Julie C, et al. Immunotherapy revolutionises non-small-cell lung cancer therapy: results, perspectives and new challenges. Eur J Cancer. 2017;78:16–23. doi:10.1016/j.ejca.2016.12.041
6. Bouazza YB, Chiairi I, El Kharbouchi O, et al. Patient-reported outcome measures (PROMs) in the management of lung cancer: a systematic review. Lung Cancer. 2017;113:140–151. doi:10.1016/j.lungcan.2017.09.011
7. Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535. doi:10.1136/bmj.b2535
8. Aaronson NK, Ahmedzai S, Bergman B, et al. The European organization for research and treatment of cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85(5):365–376. doi:10.1093/jnci/85.5.365
9. Bergman B, Aaronson NK, Ahmedzai S, Kaasa S, Sullivan M. The EORTC QLQ-LC13: a modular supplement to the EORTC Core Quality of Life Question- naire (QLQ-C30) for use in lung cancer clinical trials. EORTC study group on quality of life. Eur J Cancer. 1994;30A(5):635–642. doi:10.1016/0959-8049(94)90535-5
10. Hollen PJ, Gralla RJ, Kris MG, Potanovich LM. Quality of life assessment in indi- viduals with lung cancer: testing the Lung Cancer Symptom Scale (LCSS). Eur J Cancer. 1993;29A(Suppl 1):S51–8. doi:10.1016/S0959-8049(05)80262-X
11. Hollen PJ, Gralla RJ, Kris MG, et al. Measurement of quality of life in patients with lung cancer in multicenter trials of new therapies. Psychometric assess- ment of the lung cancer symptom scale. Cancer. 1994;73(8):2087–2098. doi:10.1002/(ISSN)1097-0142
12. Cleeland CS, Mendoza TR, Wang XS, et al. Assessing symptom distress in cancer patients: the M.D. Anderson symptom inventory. Cancer. 2000;89:1634–1646. doi:10.1002/(ISSN)1097-0142
13. Mendoza TR, Wang XS, Lu C, et al. Measuring the symptom burden of lung cancer: the validity and utility of the lung cancer module of the M. D. Anderson symptom inventory. Oncologist. 2011;16:217–227. doi:10.1634/theoncologist.2010-0193
14. de Haes JCJM, Olschewski M, Fayers PM, et al. Measuring the Quality of Life of Cancer Patients with the Rotterdam Symptom Checklist: a Manual. Groningen, The Netherlands: Northern Centre for Healthcare Research (NCH); 1996.
15. Abernethy AP, Herndon JE
16. Abernethy AP, Herndon JE
17. Abernethy AP, Zafar SY, Uronis H, et al. Validation of the patient care monitor (Version 2.0): a review of system assessment instrument for cancer patients. J Pain Symptom Manag. 2010;40(4):545–558. doi:10.1016/j.jpainsymman.2010.01.017
18. Brucker PS, Yost K, Cashy J, Webster K, Cella D. General population and cancer patient norms for the Functional Assessment of Cancer Therapy-General (FACT-G). Eval Health Prof. 2005;28:192e211. doi:10.1177/0163278705275341
19. Cella D, Eton DT, Fairclough DL, et al. What is a clinically meaningful change on the Functional Assessment of Cancer Therapy-Lung (FACT-L) Questionnaire? Results from Eastern Cooperative Oncology Group (ECOG) Study 5592. J Clin Epidemiol. 2002;55:285e295. doi:10.1016/S0895-4356(01)00477-2
20. Cella D, Eton DT, Lai JS, Peterman AH, Merkel DE. Combining anchor and distribution-based methods to derive minimal clinically important differences on the Functional Assessment of Cancer Therapy (FACT) anemia and fatigue scales. J Pain Symptom Manage. 2002;24:547e561. doi:10.1016/S0885-3924(02)00529-8
21. Hopman WM, Towheed T, Anastassiadeset T, et al. Canadian normative data for the SF-36 health survey. Can Med Assoc J. 2000;163(3):265–271.
22. Ware JE
23. Cella DF, Bonomi E, Lloyd SR, Tulsky DS, Kaplan E, Bonomi P. Reliability and validity of the Functional Assessment of Cancer Therapy-Lung (FACT-L) quality of life instrument. Lung Cancer. 1995;12(3):199–220. doi:10.1016/0169-5002(95)00450-F
24. Cella D, Hahn EA, Dineen K. Meaningful change in cancer-specific quality of life scores: differences between improvement and worsening. Qual Life Res. 2002;11(3):207–221. doi:10.1023/A:1015276414526
25. Schwartz A, Meek P. Additional construct validity of the schwartz cancer fatigue scale. J Nurs Meas. 1999;7(1):35–45. doi:10.1891/1061-3749.7.1.35
26. Gabrielson DK, Scaffidi D, Leung E, et al. Use of an abridged scored patient-generated subjective global assessment (abPG-SGA) as a nutritional screening tool for cancer patients in an outpatient setting. Nutr Cancer. 2013;65(2):234–239. doi:10.1080/01635581.2013.755554
27. US National Cancer Institute. Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE™). Available from: https://healthcaredelivery.cancer.gov/pro-ctcae/.
28. Belafsky PC, Mouadeb DA, Rees CJ, et al. Validity and reliability of the eating assessment tool (EAT-10). Ann Otol Rhinol Laryngol. 2008;117(12):919–924. doi:10.1177/000348940811701210
29. McHorney CA, Robbins J, Lomax K, et al. The SWAL- QOL and SWAL- CARE outcomes tool for oropharyngeal dysphagia in adults: III. Documentation of reliability and validity. Dysphagia. 2002;17(2):97–114. doi:10.1007/s00455-001-0109-1
30. McCarrier KP, Atkinson TM, DeBusk KP, et al. Qualitative development and content validity of the Non-small Cell Lung Cancer Symptom Assessment Questionnaire (NSCLC-SAQ), a patient-reported outcome instrument. Clin Ther. 2016;38(4):794–810. doi:10.1016/j.clinthera.2016.03.012
31. Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. Pharmacoeconomics. 1993;4(5):353–365. doi:10.2165/00019053-199304050-00006
32. Zarit SH, Reever KE, Bach-Peterson J. Relatives of the impaired elderly: correlated of feelings of burden. Gerontologist. 1980;20:649–655. doi:10.1093/geront/20.6.649
33. Bruera E, Kuehn N, Miller MJ, et al. The Edmonton Symptom Assessment System (ESAS): a simple method for the assessment of palliative care patients. J Palliat Care. 1991;7:6–9. doi:10.1177/082585979100700202
34. Sebastian M, Rydén A, Walding A, Papadimitrakopoulou V. Patient-reported symptoms possibly related to treatment with osimertinib or chemotherapy for advanced non-small cell lung cancer. Lung Cancer. 2018;122:100–106. doi:10.1016/j.lungcan.2018.05.003
35. Geater SL, Xu CR, Zhou C, et al. Symptom and quality of life improvement in LUX-Lung 6: an open-label phase III study of afatinib versus cisplatin/gemcitabine in Asian patients with EGFR mutation-positive advanced non-small-cell lung cancer. J Thorac Oncol. 2015;10(6):883–889. doi:10.1097/JTO.0000000000000517
36. Sztankay M, Giesinger JM, Zabernigg A, et al. Clinical decision-making and health-related quality of life during first-line and maintenance therapy in patients with advanced non-small cell lung cancer (NSCLC): findings from a real-world setting. BMC Cancer. 2017;17:565. doi:10.1186/s12885-017-3543-7
37. McGee SF, Zhang T, Jonker H, et al. The impact of baseline edmonton symptom assessment scale scores on treatment and survival in patients with advanced non-small-cell lung cancer. Clin Lung Cancer. 2018;19(1):e91–e99. doi:10.1016/j.cllc.2017.05.018
38. Kawata AK, Lenderking WR, Eseyin OR, et al. Converting EORTC QLQ-C30 scores to utility scores in the brigatinib ALTA study. J Med Econ. 2019;22(9):924–935. doi:10.1080/13696998.2019.1624080
39. Reck M, Schenker M, Lee KH, et al. Nivolumab plus ipilimumab versus chemotherapy as first-line treatment in advanced non-small-cell lung cancer with high tumour mutational burden: patient-reported outcomes results from the randomised, open-label, phase III CheckMate 227 trial. Eur J Cancer. 2019;116:137–147. doi:10.1016/j.ejca.2019.05.008
40. Walker MS, Wong W, Ravelo A, Miller PJE, Schwartzberg LS. Effectiveness outcomes and health related quality of life impact of disease progression in patients with advanced nonsquamous NSCLC treated in real-world community oncology settings: results from a prospective medical record registry study. Health Qual Life Outcomes. 2017;15(1):160. doi:10.1186/s12955-017-0735-4
41. LeBlanc TW, Nipp RD, Rushing CN, et al. Correlation between the international consensus definition of the Cancer Anorexia-Cachexia Syndrome (CACS) and patient-centered outcomes in advanced non-small cell lung cancer. J Pain Symptom Manage. 2015;49:4. doi:10.1016/j.jpainsymman.2014.09.008
42. Sloan JA, Cheville AL, Liu H, et al. Impact of self-reported physical activity and health promotion behaviors on lung cancer survivorship. Health Qual Life Outcomes. 2016;14:66. doi:10.1186/s12955-016-0461-3
43. Denis F, Lethrosne C, Pourel N, et al. Randomized trial comparing a web-mediated follow-up with routine surveillance in lung cancer patients. JNCI J Natl Cancer Inst. 2017;109(9):djx029. doi:10.1093/jnci/djx029
44. Pérol M, Ciuleanu TE, Arrieta O, et al. Quality of life results from the Phase 3 REVEL randomized clinical trial of ramucirumab-plus-docetaxel versus placebo-plus-docetaxel in advanced/metastatic non-small cell lung cancer patients with progression after platinum-based chemotherapy. Lung Cancer. 2016;93:95–103. doi:10.1016/j.lungcan.2016.01.007
45. Boye M, Wang X, Srimuninnimit V, et al. First-line pemetrexed plus cisplatin followed by gefitinib maintenance therapy versus gefitinib monotherapy in East Asian never-smoker patients with locally advanced or metastatic nonsquamous non-small-cell lung cancer: quality of life results from a randomized phase III trial. Clin Lung Cancer. 2016;17(2):150–160. doi:10.1016/j.cllc.2015.12.004
46. Nguyen PAH, Vercauter P, Verbeke L, Beelen R, Dooms C, Tournoy KG. Health outcomes for definite concurrent chemoradiation in locally advanced non-small cell lung cancer: a prospective study. Respiration. 2019;97(4):310–318. doi:10.1159/000493984
47. Novello S, Kaiser R, Mellemgaard A, et al. LUME-LUNG 1 study group. Analysis of patient-reported outcomes from the LUME-Lung 1 trial: a randomised, double-blind, placebo-controlled, Phase III study of second-line nintedanib in patients with advanced non-small cell lung cancer. Eur J Cancer. 2015;51(3):317–326. doi:10.1016/j.ejca.2014.11.015
48. Felip E, Hirsh V, Popat S, et al. Symptom and quality of life improvement in LUX-lung 8, an open-label phase III study of second-line afatinib versus erlotinib in patients with advanced squamous cell carcinoma of the lung after first-line platinum-based chemotherapy. Clin Lung Cancer. 2017;19–1:74–83.
49. Walker MS, Wong W, Ravelo A, Miller PJE, Schwartzberg LS. Effect of brain metastasis on patient-reported outcomes in advanced NSCLC treated in real-world community oncology settings. Clin Lung Cancer. 2017;19–2:139–147.
50. Shallwani SM, Simmondsc MJ, Kasymjanova G, Spahija J. Quality of life, symptom status and physical performance in patients with advanced non-small cell lung cancer undergoing chemotherapy: an exploratory analysis of secondary data. Lung Cancer. 2016;99:69–75. doi:10.1016/j.lungcan.2016.06.018
51. von Verschuera U, Schnellb R, Tessenc HW, et al. Treatment, outcome and quality of life of 1239 patients with advanced non-small cell lung cancer-final results from the prospective German TLK cohort study. Lung Cancer. 2017;112:216–224. doi:10.1016/j.lungcan.2017.07.031
52. Blackhall F, Kim DW, Besse B, et al. Patient-reported outcomes and quality of life in PROFILE 1007: a randomized trial of crizotinib compared with chemotherapy in previously treated patients with nALK-Positive advanced non–small-cell lung cancer. Thorac Oncol. 2014;9:1625–1633. doi:10.1097/JTO.0000000000000318
53. LeBlanc TW, Nickolich M, Rushing CN, Samsa GP, Locke SC, Abernethy AP. What bothers lung cancer patients the most? A prospective, longitudinal electronic patient-reported outcomes study in advanced non-small cell lung cancer. Support Care Cancer. 2015;23:3455–3463. doi:10.1007/s00520-015-2699-4
54. Wood R, Taylor-Stokes G, Lees M. The humanistic burden associated with caring for patients with advanced non-small cell lung cancer (NSCLC) in three European countries-a real-world survey of caregivers. Support Care Cancer. 2019;27(5):1709–1719. doi:10.1007/s00520-018-4419-3
55. Barlesi F, Garon EB, Kim DW, et al. Health-related quality of life in KEYNOTE-010: a phase II/III study of pembrolizumab versus docetaxel in patients with previously treated advanced, programmed death ligand 1-expressing NSCLC. J Thorac Oncol. 2019;14(5):793–801. doi:10.1016/j.jtho.2019.01.016
56. King-Kallimanis BL, Kanapuru B, Blumenthal GM, Theoret MR, Kluetz PG. Age-related differences in patient-reported outcomes in patients with advanced lung cancer receiving anti-PD-1/PD-L1 therapy. Semin Oncol. 2018;45:201–209. doi:10.1053/j.seminoncol.2018.06.003
57. Bordoni R, Ciardiello F, vonPawel J, et al. Patient-reported outcomes in OAK: a phase III study of atezolizumab versus docetaxel in advanced non–small-cell lung cancer. Clin Lung Cancer. 2018;19:441–449.e4. doi:10.1016/j.cllc.2018.05.011
58. Brahmer JR, Rodríguez-Abreu D, Robinson AG, et al. Health-related quality-of-life results for pembrolizumab versus chemotherapy in advanced, PD-L1-positive NSCLC (KEYNOTE-024): a multicentre, international, randomised, open-label phase 3 trial. Lancet Oncol. 2017;18:1600–1609. doi:10.1016/S1470-2045(17)30690-3
59. Lee CK, Novello S, Rydén A, Mann H, Mok T. Patient-reported symptoms and impact of treatment with osimertinib versus chemotherapy in advanced non-small-cell lung cancer: the AURA3 trial. J Clin Oncol. 2018;36(18):1853–1860. doi:10.1200/JCO.2017.77.2293
60. Mendoza TR, Kehl KL, Bamidele O, et al. Assessment of baseline symptom burden in treatment-naïve patients with lung cancer: an observational study. Support Care Cancer. 2019;27(9):3439–3447. doi:10.1007/s00520-018-4632-0
61. Wang XS, Shi Q, Williams LA, et al. Prospective study of patient-reported symptom burden in patients with non-small-cell lung cancer undergoing proton or photon chemoradiation therapy. J Pain Symptom Manage. 2016;51(5):832–838. doi:10.1016/j.jpainsymman.2015.12.316
62. McCarrier KP, Atkinson TM, Debusk KPA, Liepa AM, Scanlon M, Coons SJ. Qualitative development and content validity of the Non‐small Cell Lung Cancer Symptom Assessment Questionnaire (NSCLC‐SAQ), A patient‐reported outcome instrument. Clin Ther. 2016;38(4):
63. Damm K, Roeske N, Jacob C. Health-related quality of life questionnaires in lung cancer trials: a systematic literature review. Health Econ Rev. 2013;3(1):15. doi:10.1186/2191-1991-3-15
64. Sloan JA, Zhao X, Novotny PJ, et al. Relationship between deficits in overall quality of life and non-small-cell lung cancer survival. J Clin Oncol. 2012;30:
65. Brady GC, Roe JWG, O’ Brien M, Boaz A, Shaw C. An investigation of the prevalence of swallowing difficulties and impact on quality of life in patients with advanced lung cancer. Support Care Cancer. 2018;26:515–519. doi:10.1007/s00520-017-3858-6
66. Reck M, Brahmer J, Bennett B, et al. Evaluation of health-related quality of life and symptoms in patients with advanced non-squamous non-small cell lung cancer treated with nivolumab or docetaxel in CheckMate 057. Eur J Cancer. 2018;102:23–30.
67. Spigel DR, McCleod M, Jotte RM, et al. Safety, efficacy, and patient-reported health-related quality of life and symptom burden with nivolumab in patients with advanced non-small cell lung cancer, including patients aged 70 years or older or with poor performance status (CheckMate 153). J Thorac Oncol. 2019;14(9):1628–1639. doi:10.1016/j.jtho.2019.05.010
68. Wu Y-L, Hirsh V, Lecia V, et al. Does EGFR mutation type influence patient-reported outcomes in patients with advanced EGFR mutation-positive non-small- cell lung cancer? Analysis of two large, phase iii studies comparing afatinib with chemotherapy (LUX-Lung 3 and LUXLung 6). Patient. 2018;11:131–141. doi:10.1007/s40271-017-0287-z
69. Ediebah DE, Coens C, Zikos E, et al. Does change in health-related quality of life score predict survival? Analysis of EORTC 08975 lung cancer trial. Br J Cancer. 2014;110:
70. Boyce MB, Browne JP. Does providing feedback on patient-reported outcomes to healthcare professionals result in better outcomes for patients? A systematic review. Qual Life Res. 2013;22:
71. Snyder CF, Aaronson NK, Choucair AK, et al. Implementing patient-reported outcomes assessment in clinical practice: a review of the options and considerations. Qual Life Res. 2012;21:
72. Gralla RJ. Quality-of-life evaluation in cancer: the past and the future. Cancer. 2015;121:
73. Hollen PJ, Gralla RJ, Stewart JA, Meharchand JM, Wierzbicki R, Leighl N. Can a computerized format replace a paper form in PROSand HRQL evaluation? Psychometric testing of the computer-assisted LCSS instrument (eLCSS-QL). Support Care Cancer. 2013;21:
74. Chen J, Ou L, Hollis SJ. A systematic review of the impact of routine collection of patient reported outcome measures on patients, providers and health organisations in an oncologic setting. BMC Health Serv Res. 2013;13:
75. Basch E, Deal AM, Kris MG, et al. Symptom monitoring with patient-reported outcomes during routine cancer treatment: a randomized controlled trial. J Clin Oncol. 2016;34:
76. Basch E, Deal AM, Dueck AC, et al. Overall survival results of a trial assessing patient-reported outcomes for symptom monitoring during routine cancer treatment. JAMA. 2017;318(2):197–198. doi:10.1001/jama.2017.7156
77. Dudgeon D. The Impact of Measuring Patient-Reported Outcome Measures on Quality of and Access to Palliative Care. J Palliat Med. 2018;21(S1):S76–S80. doi:10.1089/jpm.2017.0447
78. Ambroggi M, Biasini C, Toscani I, et al. Can early palliative care with anticancer treatment improve overall survival and patient-related outcomes in advanced lung cancer patients? A review of the literature. Support Care Cancer. 2018;26(9):2945–2953. doi:10.1007/s00520-018-4184-3
79. Marquis P, Caron M, Emery MP, Scott JA, Arnould B, Acquadro C. The role of health-Related quality of life data in the drug approval processes in the US and Europe. Pharm Med. 2012;25:
80. Cherny NI, Sullivan R, Dafni U, et al. A standardised, generic, validated approach to stratify the magnitude of clinical benefit that can be anticipated from anti-cancer therapies: the European Society for Medical Oncology Magnitude of Clinical Benefit Scale (ESMO-MCBS). Ann Oncol. 2015;26:
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
