Back to Journals » Clinical Ophthalmology » Volume 15
Factors Influencing Response to Aflibercept in Diabetic Macular Oedema Patients in a Diverse North West London Population: A Real-World Study
Authors Sim SY, Ghulakhszian A, Minocha A, Ramcharan D, Nokhostin S , Cheong-Leen R, George S, Posner E, Dinah C
Received 15 April 2021
Accepted for publication 7 May 2021
Published 20 May 2021 Volume 2021:15 Pages 2089—2097
DOI https://doi.org/10.2147/OPTH.S314614
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Sing Yue Sim,1 Arevik Ghulakhszian,2 Amal Minocha,1 Dhannie Ramcharan,2 Soroush Nokhostin,3 Richard Cheong-Leen,1 Sheena George,1 Esther Posner,3 Christiana Dinah2
1Department of Ophthalmology, The Hillingdon Hospitals NHS Foundation Trust, Uxbridge, UK; 2Department of Ophthalmology, London North West University Healthcare NHS Trust, London, UK; 3Department of Ophthalmology, Imperial College Healthcare NHS Trust, London, UK
Correspondence: Christiana Dinah Email [email protected]
Background: Diabetic macular oedema (DMO) is the leading cause of sight impairment in working age populations in developed countries. Current first line treatment for centre-involving DMO involves intravitreal anti-VEGF but treatment response can be variable. In this retrospective, real world, multi-centre cohort study, we aim to identify ocular and systemic characteristics that correlate with anatomical and functional outcomes for treatment-naive DMO patients treated with intravitreal aflibercept.
Methods: Retrospective multicentre cohort study of treatment-naive DMO patients initiated on aflibercept at three North West London hospitals between 2016 and 2018. Baseline systemic and ocular factors, best corrected visual acuity (BCVA) and central macular thickness (CMT) at 12 months were determined and statistically analysed.
Results: A total of 270 eyes of 221 DMO patients met inclusion criteria. Mean age was 62.8 ± 12.1, mean baseline HbA1c was 67 ± 20 mmol/mol, and mean eGFR was 72 mL/min/1.7m2. Mean number of aflibercept injections at 12 months was 6.2. Better baseline BCVA, lower baseline CMT, and absence of epiretinal membrane (ERM) were associated with better BCVA at 12 months whilst lower baseline CMT and proliferative retinopathy status were associated with lower CMT at 12 months.
Conclusion: Our study is the largest real-world dataset examining factors influencing functional and anatomical response to aflibercept in DMO in the UK. Older age, lower baseline BCVA, higher baseline CMT and more severe diabetic retinopathy were associated with poorer visual acuity at 12 months and prioritisation of these patients within a pressured healthcare setting is recommended.
Keywords: diabetic macular oedema, functional and anatomical response to Aflibercept
Background
Diabetic macular oedema (DMO) is a leading cause of sight impairment in the working age population with approximately 21 million individuals affected worldwide.1 Currently, the first line treatment for centre-involving DMO involves intravitreal anti-vascular Endothelial Growth Factor (VEGF). Despite multiple landmark studies demonstrating efficacy of anti-VEGF in the treatment of DMO, it is recognised that a subset of patients do not respond favourably.2 Identifying factors which influence treatment response is important as it would enable us to potentially modify these factors thereby enhancing response, individualise treatment regimens and prognosticate outcomes.
Post-hoc analyses of several RCTs such as DRSS Protocol I,3 RESTORE,4 RESOLVE,5 READ26,7 and BOLT,8 RISE/RIDE9 have previously identified younger age, male sex, better baseline best corrected visual acuity (BCVA) and presence of subretinal fluid as favourable predictors of visual acuity.3,6–9 Meanwhile, duration of diabetes mellitus, worse baseline diabetic retinopathy, presence of epiretinal membrane (ERM) or surface wrinkling have been shown to adversely affect functional and anatomical outcomes;3,4,9 whilst intraretinal cysts (IRC), HbA1c, renal failure and hypercholesterolaemia have not been demonstrated to have an effect on treatment response.3,8,9
In many populations, including the UK, the prevalence of diabetes and diabetic retinopathy is higher in ethnic minorities.10–13 North West London (NWL) is a region of London known to be one of the most ethnically diverse in the country.14
In all eye departments in North West London, intravitreal aflibercept is the first-line treatment for centre-involving macular oedema as described in our previous study.15 The licence for aflibercept for the DMO indication was approved by the European Medicines Agency in June 2014 and it received the UK’s National Institute for Health and Care Excellence (NICE) approval in July 2015 based on encouraging results from the pivotal VIVID and VISTA studies.16 Our recent report confirmed that the real-world efficacy of aflibercept in treatment-naive patients with DMO in North West London was lower than reported in the landmark clinical trials, with a mean gain of +4 letters at 12 months compared to +10.7 in the trials. We note that the mean number of injections in our population was lower (6.2 in the NWL cohort versus 8.7 and 8.4 in VIVID and VISTA) as is often the case in the real world with clinic capacity pressures and lower patient adherence than in clinical trials.15 We also demonstrated that the mean visual acuity gain in our population was higher in the subgroup of patients with baseline BCVA less than 70 letters (mean gain of +7.4 ETDRS letters), with many of our patients having higher visual acuity at baseline than was allowed in the registration trials.15 However, we wanted to go further to investigate baseline ocular and systemic factors that may predict poorer visual and anatomical outcomes. Our goal was to identify patients that may be particularly at risk of poor visual and anatomical outcomes to enable better counselling on initiation of treatment, guide better individualisation of treatment and provide signals to direct further prospective studies. This study is important as previous real-world studies have not included a population as diverse as routinely treated in North West London.
Methods
The methodology of this study has been previously described.15 In summary, we conducted this retrospective, multicentre cohort study at three North West London Hospitals between January 2016 and July 2018 with data analysed between June 2020 and July 2020. The study was approved prospectively by the Research and Development departments of all three hospitals (London North West University Healthcare NHS Trust Research and Development reference no. SE19/016, Imperial College NHS Trust service evaluation reference no. 381 and Hillingdon Hospital service evaluation reference no. 1018 and the study followed the tenets of the Declaration of Helsinki.
Treatment-naive diabetic macular oedema patients initiated on intravitreal aflibercept and followed up for at least 12 months were included in the study. Both Type 1 and Type 2 diabetes mellitus patients of all ages, gender and ethnicity on oral hypoglycaemics and/or insulin were included in the study. For patients with bilateral treatment, both eyes were included. Patients were initiated on aflibercept based on the presence of centre-involving DMO defined as central subfield thickness (CSFT) at initiation over 400 µm, although the CSFT could be less than <400 µm if there was an area greater than 400 µm in the central 3000 µm of the ETDRS grid.
The exclusion criteria were cataract surgery within 3 months of commencing intravitreal aflibercept, other macular diseases such as retinal vein occlusion or age-related macular degeneration, epiretinal membranes greater than stage 3 and the presence of macular oedema of other aetiology.
The treatment protocol in all three hospitals was in alignment with 2014 EU summary of product characteristics label for aflibercept which is loading phase of 5 monthly intravitreal aflibercept (unless treatment success after three injections defined as 20/20 vision or CSFT<250 μm), followed by ongoing injections every 8 weeks until stability was achieved. Stability was defined as in Protocol T as no improvement or worsening in visual acuity or central macular thickness after two consecutive injections.17
Data Collection
Electronic medical records were reviewed for patients’ demographic data, HbA1c values (within 6 months of first injection), creatinine and eGFR (within 6 months of first injection), lens status and retinopathy status using the UK national screening committee (NSC) grades (R1: mild and moderate non-proliferative diabetic retinopathy, R2: severe non-proliferative diabetic retinopathy, R3A: active proliferative diabetic retinopathy, R3S: stable treated proliferative diabetic retinopathy) as documented in the medical records at baseline, total number of injections at 12 months and best corrected visual acuity (BCVA) at baseline and 12 months. Visual acuity was assessed using ETDRS charts at 4 metres at baseline and at all injection visits. SD-OCT was obtained in the standard mode (the enhanced depth imaging mode was not used for the purpose of evaluating DMO): Heidelberg Spectralis, Heidelberg, Germany at Central Middlesex Hospital and Western Eye Hospital and HD OCT Cirrus 5000 (Carl Zeiss AG, Germany) at Hillingdon Hospital. Central subfield thickness CSFT and macular volume were calculated automatically by the instrument and recorded at baseline, 3 months, 6 months and 12 months after the first intravitreal aflibercept. OCT features were classified into the following patterns: presence of intraretinal fluid, subretinal fluid, epiretinal membrane (ERM), vitreomacular adhesion (VMA) and mild vitreomacular traction (VMT). Virtual Caliper function of Spectral Domain-OCT was used for measurement of width and height of the largest intraretinal cyst. All data were collected at scheduled time points ± 1 month to allow for scheduling and capacity issues within each hospital.
Statistical Analysis
Data collected from our cohort were analysed using Microsoft Excel 2010 (Microsoft Corporation, Washington, USA) and SPSS 22.0 (SPSS Inc, Chicago, USA).
Sample measurements were summarised with mean value and standard deviation while categorical variables were expressed as frequencies. To assess associations between outcomes and variables, univariate and multivariate logistic regressions were used with results presented as regression coefficient with a 95% confidence interval. When choosing candidate variables for multivariable logistic regression, entry selection criterion was set at P ≤ 0.10 and stay criterion set at P ≤ 0.05. A backwards selection procedure was used to retain only the statistically significant variables in the final model. A p value of <0.05 was interpreted as statistically significant.
Results
A total of 270 eyes of 221 patients met the inclusion criteria for the study and the overall efficacy is reported elsewhere and summarised in Table 1. For the present report, due to missing data for some baseline variables, only 202 eyes were included in the regression model. The following analysis includes 202 eyes of the total cohort who had complete data of all investigated baseline variables.
 |
Table 1 Baseline Patient Characteristics in Our North West London (NWL) Study Compared with VIVIDa and VISTAb |
Predictors of Functional Outcomes
On univariate analysis, better baseline BCVA was associated with better BCVA at 12 months. In addition, younger age, lower baseline CMT and higher HbA1c were also associated with better BCVA at 12 months, having corrected for baseline BCVA (Table 2). In terms of retinopathy status, eyes that had received previous PRP (R3S) trended towards lower BCVA at 12 months, although this did not reach statistical significance (P = 0.07). On multivariate analysis, better baseline BCVA, lower baseline CMT, higher HbA1c and absence of epiretinal membrane (ERM) were associated with better BCVA at 12 months (Table 3). There was no statistically significant association between gender, ethnicity, baseline renal function (eGFR), baseline macular volume, lens status, presence of subretinal fluid, vitreomacular adhesion (VMA) or vitreomacular traction (VMT) with BCVA at 12 months on univariable and multivariable analysis (Tables 2 and 3).
 |
Table 2 Univariable Analysis of Associations Between Ocular and Systemic Factors with Best Corrected Visual Acuity (BCVA) at 12 Months |
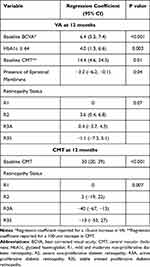 |
Table 3 Multivariable Analysis of Associations Between Ocular and Systemic Factors with BCVA and CMT at 12 Months |
Predictors of Structural Outcomes
On univariate and multivariate analysis, baseline CMT (P < 0.001) and retinopathy status (P = 0.007) were found to be significantly associated with CMT at 12 months (Tables 4 and 3). Patients with R3A status had lowest CMT values at 12 months (P = 0.007), followed by R3S, R2 and R1. Ethnicity was found to be significantly associated with the CMT at 12 months, with Caucasian eyes having higher CMT than other ethnicities, but this did not reach significance once baseline CMT was corrected for. There was no statistically significant association between gender, HbA1c, renal function (eGFR), baseline macular volume, lens status, size of intraretinal cyst, presence of subretinal fluid, vitreomacular adhesion (VMA) or vitreomacular traction (VMT) with CMT values at 12 months(Tables 4 and 3).
 |
Table 4 Univariable Analysis of Associations Between Ocular and Systemic Factors with Central Macular Thickness (CMT) at 12 Months |
Discussion
We previously reported the efficacy of aflibercept in treatment-naïve patients with diabetic macular oedema in North West London. Our intention in the present report was to identify any ocular or systemic characteristics that may be predictive of better functional or anatomical response in our cohort. Such knowledge is useful to help design prospective studies and guide service delivery. The broad diversity of our treatment population cohort within North West London uniquely enables our data to be widely applicable compared with the necessarily homogeneous demographic in randomised controlled trials. Identified factors associated with adverse outcomes may form the basis of a risk stratification tool for DMO patients initiated on intravitreal aflibercept, particularly as many of our findings have been previously confirmed in prospective studies.
Within our cohort, we found that better baseline visual acuity was significantly associated with better visual acuity at 12 months. This finding is consistent with post-hoc analysis of RIDE/RISE which showed a correlation between better baseline BCVA and final BCVA of 6/12 or better and READ2 which showed better baseline BCVA increased the chance of good visual outcome defined as better than 6/30.7,9 It has been hypothesised that good baseline BCVA despite macular oedema suggests that the fluid has not been present for sufficiently long to confer permanent damage thereby resulting in better treatment response to intravitreal aflibercept.9
In terms of patient’s age, we found that 10 years increase in age was associated with 1.5 less ETDRS letters at 12 months. This association was also noted in Diabetic Retinopathy Clinical Research Network Protocol I, albeit at a greater effect of 1.9 less ETDRS letters at 12 months with every 10 years increase in age and the post-hoc analysis of RIDE/RISE which showed that for every 5 years older at baseline, a patient was less likely to gain 15 or more letters at 24 months3,9 This may be because the eyes of younger patients may be more tolerable to fluid accumulation making them more amenable to reversible damage.9 In terms of HbA1c, there was no association with visual outcomes when analysed as a continuous variable but when categorised, we found that patients with HbA1c ≥64 had higher BCVA at 12 months. All published studies to date have either found no association or a deleterious effect of HbA1c on treatment response to aflibercept and it is likely our finding to the contrary is confounded by the inclusion of HbA1c level within 6 months of baseline as a baseline value which does not account for fluctuations in HbA1c levels throughout the study period.8,9
Interestingly, we demonstrated better anatomical response but a trend towards lower BCVA in patients with more advanced diabetic retinopathy. The adverse effects of proliferative disease on visual outcomes is consistent with previous studies including RIDE/RISE and Protocol T.9,17 Postulated mechanisms for this association include diabetic macular ischaemia being more frequently associated with more advanced diabetic retinopathy.18 A recent study by Tsai et al. also confirmed that increased severity of diabetic retinopathy was associated with decreased deep perifoveal vessel density, larger superficial and deep foveal avascular zone (FAZ) and reduced retinal sensitivity while Samara et al. found a correlation between larger FAZ areas and poorer visual acuities.19,20 On the other hand, these eyes likely have higher VEGF drive resulting in better anatomical responses.
We excluded eyes that had vitreoretinal interface abnormalities requiring surgery but evaluated the impact of mild vitreoretinal interface disturbance on functional outcomes. Our finding that the presence of ERM was associated with poorer BCVA mirrors that found in the post hoc analysis of Protocol I.3 ERM is known to reduce visual function independently by distorting the retina and can progress to preretinal macular fibrosis and it is likely that it may restrict functional improvement in DMO via similar mechanisms.21
Anatomical Outcomes
In terms of anatomical outcomes of treatment, baseline CMT and retinopathy status were found to be associated with final CMT at 12 months. We found that higher baseline CMT resulted in higher final CMT despite greater absolute fluid reduction over a 12-month period. The greater CMT reduction within this subset of patients is due to the ceiling effect to treatment of patients with low baseline CMT. Notwithstanding the greater CMT reduction, our study suggests that patients with higher baseline CMT may require more intensive treatment for optimal anatomical outcome at 12 months. Caucasian eyes tended to have higher CMT at 12 months compared with other ethnicities before adjusting for baseline CMT. This may be due to findings that eyes of African descent have lower CMT and broader foveal pits compared with Caucasian eyes but further prospective studies on anatomical response to treatment of macular oedema across ethnicities are warranted.22
Our study is limited by its retrospective nature. In addition, our data on HbA1c and renal function was limited to within 6 months of baseline and only documented at this single time point and thus does not take into account the fluctuations before the study treatment was started or during the study. Our study also lacked the strict visual acuity and OCT acquisition protocols as is the norm in clinical trials but is expected within a real-life pressured health-care setting. The strength of our study, however, lies in the diversity of our study population which is a real-world inner London population with diverse ethnicities and grades of diabetic retinopathy represented. Our results would therefore be more reflective of real-world clinical practice and highlight that older patients, patients with lower BCVA, more advanced retinopathy, epiretinal membrane and higher CMT are likely to have worse outcomes when treated with aflibercept for diabetic macular oedema and could benefit from prompt and timely commencement of treatment to maximise outcomes.
Conclusions
In summary, our study identified several baseline characteristics associated with treatment response to aflibercept in treatment-naive DMO at 12 months. To our knowledge, this is the largest published dataset examining factors influencing treatment response in patients treated with aflibercept for diabetic macular oedema within the UK in a real-world setting. Further work including large prospective studies in diverse populations are required to confirm our findings with the goal of individualising treatment, and prognosticating outlook.
Data Sharing Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Consent to Participate
The study was approved prospectively by the Research and Development departments of all three hospitals (London North West University Healthcare NHS Trust Research and Development reference no. SE19/016, Imperial College NHS Trust service evaluation reference no. 381 and Hillingdon Hospital service evaluation reference no. 1018 and the study followed the tenets of the Declaration of Helsinki.
Acknowledgments
Paul Bassett – Statsconsultancy Ltd for statistical analysis.
Author Contributions
All authors made significant contributions to the work reported including conception, study design, execution, acquisition of data, analysis and interpretation, drafting, revising and critically reviewing the article as shown below. All authors gave final approval of the version to be published and have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Conceptualisation: CD.
Data Curation: CD, AG, SYS, AM, SN.
Formal Analysis: CD, AM.
Investigation: SYS, AM, SN.
Methodology: CD, RCL, SG, EP.
Project Administration: AG, SYS.
Supervision: CD, EP, RCL, SG.
Validation: SYS, AG, CD.
Writing- Original Draft: SYS, AG.
Writing – Review and Editing: CD, AM, EP, RCL, SG.
Funding
The authors received no specific funding for this work.
Disclosure
CD has received travel and meeting grants from Bayer, Novartis and Allergan. She has also served on advisory boards for Novartis and Allergan and received speaker fees from Novartis. EP has received travel and meeting grants from Bayer, Allergan and Alimera Sciences, speaker fees from Allergan and Novartis and has served on advisory board for Alimera Sciences. SG has received travel and meeting grants and served on advisory boards for Novartis and Bayer. The authors report no other conflicts of interest in this work.
References
1. Yau JWY, Rogers SL, Kawasaki R, et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012;35:556–564.
2. Bahrami B, Zhu M, Hong T, Chang A. Diabetic macular oedema: pathophysiology, management challenges and treatment resistance. Diabetologia. 2016;59(8):1594–1608. doi:10.1007/s00125-016-3974-8
3. Elman MJ, Aiello LP, Beck RW, et al. Randomized trial evaluating Ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular oedema. Ophthalmology. 2010;117:1064–77.e35. doi:10.1016/j.ophtha.2010.02.031
4. Mitchell P, Chong V. Baseline predictors of 3-year responses to Ranibizumab and laser photocoagulation therapy in patients with visual impairment due to diabetic macular edema (DME): the RESTORE study. Invest Ophthalmol Vis Sci. 2013;54:2373. doi:10.1167/iovs.12-10757
5. Massin P, Bandello F, Garweg JG, et al. Safety and efficacy of Ranibizumab in diabetic macular edema (RESOLVE Study): a 12-month, randomized, controlled, double-masked, multicenter Phase II study. Diabetes Care. 2010;33:2399–2405. doi:10.2337/dc10-0493
6. Nguyen QD, Shah SM, Khwaja AA, et al. Two-year outcomes of the Ranibizumab for edema of the macula in diabetes (READ-2) study. Ophthalmology. 2010;117:2146–2151. doi:10.1016/j.ophtha.2010.08.016
7. Channa R, Sophie R, Khwaja AA, et al. Factors affecting visual outcomes in patients with diabetic macular edema treated with Ranibizumab. Eye. 2014;28:269–278. doi:10.1038/eye.2013.245
8. Sivaprasad S, Crosby-Nwaobi R, Heng LZ, Peto T, Michaelides M, Hykin P. Injection frequency and response to bevacizumab monotherapy for diabetic macular oedema (BOLT report 5). Br J Ophthalmol. 2013;97(9):1177–1180. doi:10.1136/bjophthalmol-2013-303168
9. Sophie R, Lu N, Campochiaro PA. Predictors of functional and anatomic outcomes in patients with diabetic macular edema treated with ranibizumab. Ophthalmology. 2015;122:1395–1401. doi:10.1016/j.ophtha.2015.02.036
10. Raymond NT, Lakshminarayanan V, Reynold D, et al. Higher prevalence of retinopathy in diabetic patients of South Asian ethnicity compared with white Europeans in the community. Diabetes Care. 2009;32(3):410–415. doi:10.2337/dc08-1422
11. Cheng YJ, Kanaya AM, Araneta MRG, et al. Prevalence of diabetes by race and ethnicity in the United States, 2011–2016. JAMA. 2019;322(24):2389–2398. doi:10.1001/jama.2019.19365
12. Pham T, Carpenter JR, Morris T, Sharma M, Petersen I. Ethnic differences in the prevalence of type 2 diabetes diagnosis in the UK: cross-sectional analysis of the health improvement network primary care database. Clin Epidemiol. 2019;11:1081–1088. doi:10.2147/CLEP.S227621
13. Zhang X, Saaddine JB, Chou C, Cotch MF, Cheng YJ, Geiss LS. Prevalence of diabetic retinopathy in the United States, 2005–2008. JAMA. 2010;304(6):649–656. doi:10.1001/jama.2010.1111
14. Office of National Statistics. 2011 Census Aggregate Data. UK Data Service. June, 2016. doi:10.5257/census/aggregate-2011-1
15. Dinah C, Ghulakhszian A, Sim S, et al. Aflibercept for treatment-naïve diabetic macula oedema in a multi-ethnic population: real-world outcomes from North West London. PLoS One. 2021;16(2):e0246626. doi:10.1371/journal.pone.0246626
16. Korobelnik JF, Do DV, Schmidt-Erfurth U, et al. Intravitreal aflibercept for diabetic macular oedema. Ophthalmology. 2014;121:2247–2254. doi:10.1016/j.ophtha.2014.05.006
17. Wells JA, Glassman AR, Ayala AR, et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema. N Engl J Med. 2015;372:1193–1203.
18. Usman M. An overview of our current understanding of diabetic macular ischemia (DMI). Cureus. 2018;10(7):e3064.
19. Tsai ASH, Gan ATL, Ting DSW, et al. Diabetic macular ischemia: correlation of retinal vasculature changes by optical coherence tomography angiography and functional deficit. Retina. 2019. doi:10.1097/IAE.0000000000002721
20. Samara WA, Shahlaee A, Adam MK, et al. Quantification of diabetic macular ischemia using optical coherence tomography angiography and its relationship with visual acuity. Ophthalmology. 2017;124:235–244. doi:10.1016/j.ophtha.2016.10.008
21. Ghazi-Nouri SM, Tranos PG, Rubin GS, Adams ZC, Charteris DG. Visual function and quality of life following vitrectomy and epiretinal membrane peel surgery. Br J Ophthalmol. 2006;90:559–562. doi:10.1136/bjo.2005.085142
22. Wagner-Schuman M, Dubis AM, Nordgren RN, et al. Race- and sex-related differences in retinal thickness and foveal pit morphology. Invest Ophthalmol Vis Sci. 2011;52(1):625–634. doi:10.1167/iovs.10-5886
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
