Back to Journals » International Journal of General Medicine » Volume 10
Extreme leukocytosis and leukemoid reaction associated with the lung sarcomatoid carcinoma: an unusual case report
Authors Wang D, Zhang H, Yu F, Fang B
Received 15 December 2015
Accepted for publication 16 March 2016
Published 23 December 2016 Volume 2017:10 Pages 7—9
DOI https://doi.org/10.2147/IJGM.S102524
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Danyang Wang, Haiyan Zhang, Fengkuan Yu, Baijun Fang
Department of Hematopathy, The Affiliated Cancer Hospital of Zhengzhou University, Zhengzhou, People’s Republic of China
Purpose: To report a rare case of extreme leukocytosis and leukemoid reaction associated with lung sarcomatoid carcinoma (LSC) and increase people’s awareness of the disease.
Patients and methods: A 58-year-old male patient was diagnosed with LSC; however, after the end of the second course of chemotherapy, his white blood cells increased gradually without fever or use of medications such as granulocyte colony-stimulating factor and steroids. A bone marrow biopsy then confirmed it to be a leukemoid reaction.
Results: The patient died of multiple organ failure 2 months after being diagnosed with leukocytosis.
Conclusion: LSC associated with leukemoid reaction is very rare and the prognosis is poor. When a patient’s white blood cells are extremely elevated, we should think of the possible causes of the tumor itself and identify it with other diseases. However, more data and evidence are still needed to find an effective adjuvant therapy for these patients.
Keywords: lung sarcomatoid carcinoma, leukemoid reaction
Introduction
Lung sarcomatoid carcinomas (LSCs) are very rare, mostly reported in the form of case reports, and leukemoid reactions has rarely been described in patients with LSC, to the best of our knowledge. Meanwhile, the mechanism of leukocytosis is not very clear. Here, we discuss an unusual case of extreme leukocytosis and a leukemoid reaction associated with the LSC. We summarize the relevant reports, hoping to provide some help for other clinicians when they encounter similar disease or symptoms.
Case presentation
A 58-year-old male patient had symptoms of intermittent cough and hemoptysis with no incentives in June 2014, and accompanying chest tightness and shortness of breath. He was first admitted to a local hospital, where computed tomography of the chest showed a left lung mass, suggesting a probability of tuberculosis. Therefore, he was treated for tuberculosis, but had no significant relief of symptoms. He then visited our hospital, where he underwent computed tomography-guided biopsy; the pathology results showed poorly differentiated adenocarcinoma (left lung). He then underwent surgery for resection of the mass; pathological examination showed poorly differentiated peripheral pulmonary carcinoma with the tumor being composed of mononuclear and multinucleated tumor giant cells with significant atypia. Immunohistochemistry showed: creatine kinase (CK) (−), P63 (−), thyroid transcription factor (TTF)-1 (−), CK18 (+), Napsin A (−), CK (+), vimentin (+), smooth muscle actin (SMA) (−), desmin (−), epidermal growth factor receptor (EGFR) (++), Ki-67 (+60%), epithelial membrane antigen (EMA) (+); the consideration is sarcomatoid carcinoma (giant cell carcinoma). The patient received two courses of chemotherapy in our hospital after surgery, gemcitabine 1.4 D1and 8 + cisplatin 30 mg D1–4 and tolerated it well.
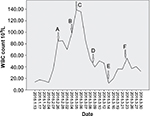
However, the patient’s white blood cells (WBCs) increased gradually without fever or use of medications, such as granulocyte colony-stimulating factor (G-CSF) and steroids, after the end of the second course (Figure 1), and neutrophils accounted for ~93%. While his hemoglobin and platelets were within the normal range. We initially used antibiotics empirically, which was ineffective. Next, a bone marrow biopsy was performed; the results showed myeloid and erythroid focal hyperplasia and significant partial immature myeloid cell proliferation (Figure 2). Immunohistochemistry showed individual lymphocytes CD3 (+), individual lymphocytes CD20 (+), erythroid CD235a (+), megakaryocytes CD61 (+), CD34 (−), individual monocytes CD14 (+), individual plasma cells CD138 (+), a large number of myeloid myeloperoxidase (MPO) (+) (Figure 3). Finally, the patient was diagnosed with leukocytosis, then he and his family refused further chemotherapy, so we put the patient on “hydroxyurea” to control the leukocytosis. The patient died of multiple organ failure 2 months after being diagnosed with leukocytosis. This study was approved by the Ethics Committee of The Affiliated Cancer Hospital of Zhengzhou University.
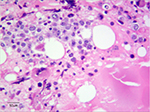  | Figure 2 Bone marrow biopsy (hematoxylin and eosin 10×40). |
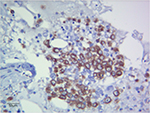  | Figure 3 Immunohistochemistry (MPO 10×40). Abbreviation: MPO, myeloperoxidase. |
Discussion
Sarcomatoid carcinoma is a poorly differentiated malignant cancer, which is composed of both carcinomatous and sarcomatous elements. It can occur in many parts of the body, but most commonly affects the respiratory tract, lungs, breasts, and kidneys. Clinically, sarcomatoid carcinoma originating in the lung (LSC) is very rare, accounting for 0.3%–4.7% of all lung malignancies.1 LSC is a non-small-cell lung carcinoma, which has a high degree of malignancy, and is more aggressive than the average lung cancers. The effects of radiotherapy and chemotherapy are not very good; therefore, the prognosis of LSC is poor.2–4
Leukemoid reaction is defined as a WBC count >50×109/L without evidence of leukemia or infection.5 Leukemoid reaction is a potential cause for leukocytosis, especially in the presence of predominately mature granulocytic leukocytosis. A leukemoid reaction is caused by an exaggerated myeloid; it has been associated with infections, allergies, burns, intoxications, acute hemorrhage, malignant neoplasms, and several other stimuli.6–9
LSC associated with leukemoid reaction is very rare. The mechanism of leukemoid reaction is not very clear. Cancer-related symptoms, may be caused by G-CSF secreted by the tumor cells.10–12 Chen et al13 indicated that, in cases of leukemoid reaction, leukocytosis is frequently mediated by tumor-related cytokines and CSF including G-CSF, granulocyte-macrophage CSF, interleukin 6 or 1α.
Jardin et al14 have reported a case with lung sarcoma and leukemoid reaction. The patient’s G-CSF concentration increase was observed after the first cycle of chemotherapy (>20,000 pg/mL). Dramatically, the highest G-CSF concentration level was observed when the WBC count was lowest, suggesting that a massive cytokine release appeared after tumor lysis related to chemotherapy.14 We also observed a similar phenomenon in our report; after chemotherapy, the patient’s WBC experienced dramatic increases. For various reasons, we have not measured G-CSF concentration level, but we can speculate that perhaps the reason was the same.
Leukemoid reaction always indicates a high degree of malignancy, high probability of metastasis, and recurrence and wretched prognosis. In our report, the patient died of multiple organ failure 2 months after being diagnosed with leukocytosis. This is consistent with other reports correlating leukemoid reactions with advanced and aggressive tumor cell growth and poor outcome.6,15
In conclusion, LSC associated with leukemoid reaction is very rare and the prognosis is poor. When a patient’s WBC is extremely elevated, we should think of possible causes of the tumor itself and identify it with other diseases. However, more data and evidence are still needed to find an effective adjuvant therapy for these patients.
Acknowledgment
Written informed consent was obtained from this patient for publication of this case report and any accompanying images.
Disclosure
The authors report no conflicts of interest in this work.
References
Pelosi G, Sonzogni A, De Pas T, et al. Pulmonary sarcomatoid carcinomas: a practical overview. Int J Surg Patho1. 2010;18(2):103–120. | ||
Martin LW, Correa AM, Ordonez NG, et al. Sarcomatoid carcinoma of the lung: a predictor of poor prognosis. Ann Thorac Surg. 2007;84(3):973–980. | ||
Venissac N, Pop D, Lassalle S, Berthier F, Hofman P, Mouroux J. Sarcomatoid lung cancel(spindle/giant cells): an aggressive disease? J Thorac Cardiovasc Surg. 2007;134(3):619–623. | ||
Yuki T, Sakuma T, Ohbayashi C, et al. Pleomorphic carcinoma of the lung: a surgical outcome. J Thorac Cardiovasc Surg. 2007;134(2):399–404. | ||
McKee L. Excess leukocytosis (leukemoid reactions) associated with malignant diseases. South Med J. 1985;78(12):1475–1482. | ||
Stav K, Leibovici D, Siegel YI, Lindner A. Leukemoid reaction associated with transitional cell carcinoma. Isr Med Assoc J. 2002;4(3):223–224. | ||
Marinella MA. Extreme leukemoid reaction associated with retroperitoneal hemorrhage. Arch Intern Med. 1998;158(3):300–301. | ||
Pina-Ceballos VM, Montemayor-Garza R, Gonzalez-Oritz J. Leukemoid reaction secondary to prenatal use of betamethasone in a premature neonate. Rev Mex Pediatr. 1997;64:197–200. | ||
Abramson N, Melton B. Leukocytosis: basics of clinical assessment. Am Fam Physician. 2000;62(9):2053–2060. | ||
Nimieri HS, Makoni SN, Madziwa FH, Nemiary DS. Leukemoid reaction response to chemotherapy and radiotherapy in a patient with cervical carcinoma. Ann Hematol. 2003;82(5):316–317. | ||
Turalic H, Deamant FD, Reese JH. Paraneoplastic production of granulocyte colony-stimulating factor in a bladder carcinoma. Scand J Urol Nephrol. 2006;40(5):429–432. | ||
Lee MY, Kaushansky K, Judkins SA, Lottsfeldt JL, Waheed A, Shadduck RK. Mechanism of tumor-induced neutroneutrophilia: constitutive production of colony-stimulating factors and their synergistic actions. Blood. 1989;74:115–122. | ||
Chen YM, Whang-Peng J, Liu JM, et al. Leukemoid reaction resulting from multiple cytokine production in metastatic mucoepidermoid carcinoma with central necrosis. Jpn J Clin Oncol. 1995;25(4):168–172. | ||
Jardin F, Vasse M, Debled M, et al. Paraneoplastic neutrophilic leukemoid reaction related to a G-CSF-secreting lung sarcoma. Am J Hematol. 2005;80(3):243–245. | ||
Kasuga I, Makino S, Kiyokawa H, Katoh H, Ebihara Y, Ohyashiki K. Tumor-related leukocytosis is linked with poor prognosis in patients with lung carcinoma. Cancer. 2001;92(9):2399–2405. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.