Back to Journals » Drug Design, Development and Therapy » Volume 13
Evaluation of pharmacokinetics and pharmacodynamics of sinomenine-hyaluronic acid conjugate after intra-articular administration for osteoarthritis treatment
Authors Liu J, Shao H, Fang S, Cheng Y, Ling P, Chen J
Received 5 September 2018
Accepted for publication 28 December 2018
Published 15 February 2019 Volume 2019:13 Pages 657—665
DOI https://doi.org/10.2147/DDDT.S186558
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sukesh Voruganti
Jie Liu,1,2,* Huarong Shao,1,* Shaoying Fang,1 Yanling Cheng,1 Peixue Ling,1,3 Jianying Chen1,3
1Key Laboratory of Biopharmaceuticals of Shandong Province, Shandong Academy of Pharmaceutical Sciences, Jinan, Shandong Province, China; 2Shandong Provincial Engineering Research Center for Medicines of Orthopedic Pain, Shandong Mingren Freda Pharmaceutical Co. Ltd, Jinan, Shandong Province, China; 3Key Laboratory of Mucosal and Transdermal Drug Delivery Technology in Shandong Province, Shandong Freda Pharmaceutical Group Co. Ltd, Jinan, Shandong Province, China
*These authors contributed equally to this work
Objectives: Intra-articular injection of sinomenine (SN) is an effective treatment method for knee osteoarthritis (OA), however, SN could be eliminated quickly in vivo. To extend the residence time of SN in the joint cavity, the SN-hyaluronic acid (HA) conjugate was prepared previously. This study was performed to evaluate the pharmacokinetics and pharmacodynamics of SN-HA conjugate after intra-articular administration for the treatment of OA.
Methods: A high performance liquid chromatography-mass spectrometry (HPLC-MS/MS) method was established to determine the SN content in rat synovial fluid. One hundred and twenty rats were randomly divided into two groups, the SN-HA group and SN group. The concentration of SN in articular cavity washings was determined by HPLC-MS/MS. The protective effect on the cartilage was evaluated by histological evaluation in a model of papain induced rabbit knee osteoarthritis.
Results: The method was validated with respect to sensitivity, specificity, linearity, precision, accuracy and especially the stability of analytes under various conditions, and was successfully applied in evaluating the pharmacokinetic profiles of SN in the joint cavity. Compared to the SN injection, the drug exposure in joint cavity was significantly increased following SN-HA injection administration, and AUC(0–12h) was 2.9 times of SN injection, mean residence time (MRT) was 1.88 times of SN injection. In the pharmacodynamic study, there was no significant difference between the SN-HA twice-treated group and SH/HA five-times mixture-treated group.
Conclusion: The local bioavailability of SN in joint cavity was improved significantly after conjugated with HA. The SN-HA conjugate showed good synergism effect of OA inhibition. The results indicated that the SN-HA conjugate seemed to be an effective therapeutic means for the treatment of OA.
Keywords: sinomenine-hyaluronic acid conjugate, HPLC-MS/MS, pharmacokinetics, pharmacodynamics, osteoarthritis
Introduction
Osteoarthritis (OA) is a degenerative joint disease that causes progressive damage to articular cartilage. As the population ages, OA has attracted the public’s attention around the world in recent years.1,2 More research and new products are needed in order to control the disease and its side effects.
Sinomenine (SN) is a pure alkaloid derived from the Chinese medicinal plant, Sinomenium acutum, which has been utilized to treat inflammation in China for centuries.3 The pharmacological effect of SN including anti-inflammation,4 immunosuppression,5 arthritis amelioration,6 and protection against hepatitis7 has been proved by previous pharmacological studies. Intra-articular injection of SN is also an effective treatment method for acute pain of knee OA.8,9 However, the short half-life in vivo restricts the application of SN,10 and excessive dosage could cause liver damage and mild myocardial hyperemia.11 So it is a challenge to treat OA with SN.
In recent years, in order to extend the half-life and minimize the side effects of SN, many studies have focused on the attachment of small molecule drugs with carriers that were chemically synthesized or natural polymer.12,13 Hyaluronic acid (HA) is a high molecular weight molecule composed of alternating N-acetyl-d-glucosamine and d-glucuronic acid residues attached by β(1–4) and β(1–3) bonds.2 Due to its good properties, HA has been considered as the first selection for the preparation of conjugate.14–16 Now, HA and its derivative have been widely used as a carrier for many kinds of anticancer drug,17,18 peptide and protein drug.19 HA not only possesses good biocompatibility, but also obviously prolongs residence time in the administration area, improves drug bioavailability, diminishes adverse drug reaction.20 Moreover, HA is the main ingredient of the articular cartilage and synovial fluid, and plays an important role for the normal joint physiological function serving as a lubricant, scavenger for free radicals, and for the regulation of cellular activity such as binding of proteins. In a previous study, our group has prepared SN-HA conjugate with glycine as a spacer for the therapy of OA, and the conjugate showed significant SN delayed-release effect in vitro.20 The conjugate may exert synergistic functions of SN and HA, and improve the therapeutic effect for OA.
In this study, we investigate the local pharmacokinetics (PK) of SN in the rat joint and its therapeutic effect in a rabbit model of OA following SN-HA conjugate injected intra-articularly.
Materials and methods
Reagents
SN was provided by Tiancheng Plant Engineering Co., Ltd. (Shanxi, China). HA was provided by Freda Biopharm Co., Ltd. (Shandong, China). The SN-HA conjugate was prepared according to the method described by Liu et al,20 wherein the degree of substitution (DS) of SN was 26.5%. Theophyllinum was supplied by China Food and Drug Testing Institute (Bejing, China). Methanol, acetonitrile and formic acid were purchased from Thermo Fisher Scientific Inc. (Waltham, USA).
Pharmacokinetics study
Animal experiments
All animal studies were conducted in accordance with Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) guidelines with the approval of the Institutional Animal Care and Use Committee of Drug Safety Evaluation Center of Shandong Institute of Pharmaceutical Industry (Jinan, China). Male Sprague Dawley rats (200–250 g) were acclimatized to laboratory conditions at 20°C–26°C and 35%–75% relative humidity for more than a week before dosing. The animals were given a pelleted diet and were given free access to food and water. Animals were acclimatized for 7 days before the experiments were initiated.
One hundred and twenty rats were randomly divided into two groups, the SN-HA conjugate group and the SN/HA mixture group. The rats were deeply anaesthetized with an isoflurane/O2 gas mixture until the flexor withdrawal reflex was abolished. Left knees were intra-articularly injected with the SN-HA conjugate and SN dissolved in the HA, respectively. The injection volumes of SN and SN-HA were 60 μL (the SN content were both 0.68 mM). Six rats from each group were executed at 5 minutes, 15 minutes, 30 minutes, 1 hour, 1.5 hour, 2 hours, 4 hours, 6 hours, 8 hours, 12 hours post injection, and the joint cavities were washed with 5 mL saline, and diluted to 10 mL with saline. The joint cavity washing fluid was stored at –20°C until analysis.
Quantification of SN in the synovial fluid
Instrumentation
Samples were analyzed on an Agilent 1,260 Infinity LC system (Agilent Technologies, Santa Clara, CA, USA) comprising a 6,460 triple quadrupole equipped with an electrospray ionization source, vacuum degasser, binary pump and thermostatic column compartment. The LC system, mass spectrometer and data analysis are controlled using Agilent Mass Hunter software version B.05.01 (Agilent Technologies).
HPLC-MS/MS conditions
The analytical column was a Zorbax SB-C18 column (Agilent Technologies; 150 mm*4.6 mm, 5 μm). The mobile phase consists of methanol (solvent A) and 0.1% formic acid solution (solvent B) at a flow rate of 1.0 mL/min (A:B, 20:80).
The mass spectrometer was operated in an ESI positive ion mode. Multiple reaction monitoring transitions were performed at m/z 329.9→180.9 for SN, and m/z 181.0→124.0 for theophylline (internal standard, IS, purity >99%, National Institute for Food and Drug Control). The optimized values for collision energy and fragmentor were 35 V and 155 V for RA, 20 V and 110 V for IS. Other ion source conditions were as follows: ion spray voltage, 4500; ion-source temperature, 550°C. Nitrogen was used as both curtain and collision gas.
Stock solutions and sample preparation
The stock solutions of SN (100 μg/mL) and IS (5 μg/mL theophylline solution) were prepared in methanol. 100 μL sample synovial fluid was dispensed into 1.5 mL polyethylene tubes followed by 10 μL of IS (5 μg/mL theophylline solution) and vortexed for 1 minute. Then 390 μL acetonitrile containing 0.1% trifluoroacetic acid was added and vortexed for another 2 minutes. The mixture was centrifuged at 10,000 rpm for 10 minutes at 4°C. Supernatant was injected (5 μL) into the high performance liquid chromatography-mass spectrometry (HPLC-MS/MS) system.
Bioanalytical method validation
The method was validated by determining the following parameters: matrix effects, selectivity, linearity range, recovery, accuracy, precision, lower limit of quantification (LLOQ) and stability studies.
Selectivity and matrix effect
The selectivity was assessed by using six blank rat joint cavity washing fluid samples that were subjected to the precipitation procedure and analyzed to determine the extent to which endogenous synovial fluid components could interfere in the analysis of the SN and IS. Blank (without SN or IS), drug sample (with SN) and IS sample (with IS) were prepared, processed and analyzed for the exclusion of any endogenous co eluting interference at the peak region of both SN and IS.
The matrix effects of SN and IS were evaluated by using the ratios of the mean peak areas of the analytes spiked in posttreatment joint cavity washing fluid to those of the mean peak areas of the pure standard solution at corresponding concentrations (low, medium, and high concentrations).
Calibration curves
Seven-point calibration curves over the concentration range of 10–1,000 ng/mL were constructed to demonstrate the linearity of the method. The ratio of the SN peak area to that of the IS vs the respective standard concentrations was used for plotting the graph, and the linearity was evaluated by a weighted (1/x2) least-squares regression analysis. The acceptance criteria for each calculated standard concentration was not more than 15% deviation from the nominal value, except for the LLOQ which was set at 20%.
Accuracy and precision
To evaluate the interday precision and accuracy, the quality control (QC) samples with SN in different concentrations were analyzed together with one independent calibration standard curve for three assay batches. The intraday precision and accuracy were evaluated through analysis of QC samples at three different concentrations in six replicates in the same batches. Inter- and intraday precision was expressed as relative standard deviation (RSD). The accuracy was expressed as the percent ratio between the experimental concentration and the nominal concentration for each sample. The evaluation of precision was based on the criteria that the deviation of each concentration level should be within 15%.
Recovery
Protein precipitation extraction procedure of the joint cavity washing fluid followed as described before. The peak areas of extracted joint cavity washing fluid (pre-spiked) standard QC samples (N=6) were compared to those of the post-spiked standards at equivalent concentrations to determine the extraction recovery of SN. Recoveries of SN and IS were determined by analyzing six replicates at three concentration levels (low, medium, and high concentrations).
To determine whether SN could be released completely from the conjugate through the current sample preparation method, the joint cavity washing fluid containing SN-HA samples (the SN content at low, medium, and high concentrations) were prepared and processed for SN determination.
Limits
The lowest standard concentration on the calibration curve should be accepted as the limit of quantification if the following conditions are met: the analyte response at the LLOQ should be at least five times the response compared to blank response and analyte peak (response) should be identifiable, discrete, and reproducible with a precision of 20% and accuracy of 80%–120%.21
Stability
Two concentrations (high and low) were selected to carry out all the stability studies using six replicates at each of the two concentration levels. Replicate injections of processed samples were analyzed up to 12 hours to establish autosampler (AS) stability of SN and IS at 4°C. The stability of SN in rat joint cavity washing fluid during 6 hours exposure at room temperature was determined. Freeze/thaw (FT) stability was evaluated up to three cycles. In each cycle, samples were frozen for at least 12 hours at −20°C. Freezer stability was assessed by analyzing the QC samples stored at −20°C for at least 15 days. The stability was determined using initial concentrations of SN determined for the freshly prepared samples. Samples were considered to be stable if assay values were within the acceptable limits of accuracy (ie, ±15% deviation.) and precision (ie, ±15% RSD).
Dilution integrity
The dilution integrity experiment was performed for validation of the routine procedure to dilute samples in which the observed concentration is higher than the upper limit of quantification of the method. Dilution integrity experiments were carried out by 10 times dilution of joint cavity washing fluid samples containing 5,000 ng/mL of SN with blank matrix to obtain samples containing 500 ng/mL of SN.
Pharmacokinetic parameters
The data were subjected to non-compartmental pharmacokinetic analysis using Phoenix WinNonlin (version 5.1, Pharsight Corporation, St Louis, MO, USA). The observed maximum concentration (Cmax) and the time to reach the maximum concentration (Tmax) were obtained by visual inspection of the experimental data. Area under the synovial fluid concentration-time curve (AUC) from time zero to the last quantifiable concentration was calculated using linear trapezoidal rule. The MRT is calculated as area under the first moment curve (AUMC)/AUC.
Pharmacodynamics study
Experimental OA model and treatment regimens
Forty adult male New Zealand White rabbits (2.5±0.2 kg) were housed individually in steel cages of 45×30×60 cm dimensions and maintained under the same environmental conditions (temperature 19°C–21°C, relative humidity of 50%–60%) for 2 weeks. The animals were fed with a standard rabbit diet (14% protein, 7% fat, 15% cellulose, 1%–1.2% calcium and phosphorus) and filtered water.
Papain and L-cysteine were dissolved in normal saline at a concentration of 2% (w/v) and 0.03 mol/L before use and the solution was filtrated with a 0.22 μm membrane. IA injections of the solution into the left knees of 30 rabbits were performed three times on days 1, 3 and 5, respectively. Then these animals were randomly divided into three groups of 10 each. The SN suspended in HA were injected intra-articularly into the left knees once a week for 5 weeks (SN/HA mixture group). The SN-HA conjugate were given intra-articularly into the left knees in weeks 1 and 3 (SN-HA conjugate group), while normal saline were injected once a week for 5 weeks (NS control group). The injection volumes of SN and SN-HA were 300 μL (the SN content were both 3.4 mM). Animals in the sham group (N=10) were just given injection practices and not induced OA or received drug treatment.
Histological evaluation of the cartilage
Femoral condyle and tibial plateau were collected and stained for histological evaluation using standard procedures. Briefly samples were fixed with 10% (w/v) buffered formalin for 48 hours, and subsequently decalcified in a solution containing 5% formic acid, 8.5% hydrochloric acid and 7% (w/v) AlCl3 for 7 days, dehydrated, embedded in paraffin, and cut into 5 μm-thick serial sections. Serial sections were collected from the same anatomical site and stained with H&E for the analysis of cartilage structure. The histology was evaluated through double-blind observations following the method described previously.
Statistical analysis
Data were presented as mean ± SD. SPSS 16.0 statistical software was used (SPSS Inc., Chicago, IL, USA). Data were initially evaluated for normal distribution. Statistical significance among groups was then tested using ANOVA analysis and differences between groups were further confirmed by the Student’s t-test. P<0.05 was considered to be significant.
Results
Selectivity
In the present study, the selectivity was studied by using rat joint cavity washing fluid samples from six different rats. Figure 1 shows a typical chromatogram for the drug-free rat joint cavity washing fluid and drug-free joint cavity washing fluid spiked with SN and IS. As shown in Figure 1, there is no significant interference from synovial fluid found at retention time of either the SN or the IS.
Sensitivity, linearity, accuracy, and precision
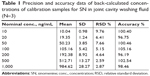
LLOQ was the lowest quantifiable concentration whose signal-to-noise ratio was greater than five and it was found to be 10 ng/mL for the SN. Seven calibration standards (10–1,000 ng/mL) were used to construct the synovial fluid calibration curve. The average regression (N=3) was found to be ≥0.998. The accuracy observed for the mean of back-calculated concentrations for three calibration curves was within 96.19% to 105.16%, while the precision values ranged from 2.59% to 9.76% (Table 1). Accuracy and precision data for intra- and interday synovial samples are presented in Tables 2 and 3. The assay values on both the occasions (intra- and interday) were found to be within the accepted variable limits.
  | Table 2 Intraday assay precision and accuracy for SN in rat joint cavity washing fluid |
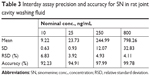  | Table 3 Interday assay precision and accuracy for SN in rat joint cavity washing fluid |
Stability
The predicted concentrations for SN at 25 and 800 ng/mL samples deviated within the nominal concentrations in all the stability tests, viz., AS stability (12 hours), bench top (BT) stability (6 hours), repeated three freeze/thaw cycles (FT-3) and at −20°C for 15 days (Table 4). The results were found to be within the assay variability limits during the entire process.
  | Table 4 Stability of SN in rat joint cavity washing fluid |
Recovery and matrix effect
The extraction recoveries of SN ranged from 92.82% to 97.54%, and the extraction recovery of the IS was 94.44% to 103.26%. The matrix effect of SN ranged from 94.53% to 98.22%, and the matrix effect of IS ranged from 93.29% to 101.02%. The recovery of SN released from the SN-HA conjugate was 95.96%–102.84% (Table 5).
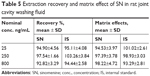  | Table 5 Extraction recovery and matrix effect of SN in rat joint cavity washing fluid |
Clearance kinetics of SN from joint cavity
The validated HPLC-MS/MS method was successfully used to measure the synovial concentration of SN after intra-articular administration of SN-HA and SN. The synovial fluid concentration-time curve was shown as Figure 2. Almost at all the time-points, the SN concentration of SN-HA conjugate group was higher than that of the SN group. The main pharmacokinetics parameters of SN are listed in Table 6. When equally available SN was administrated, the AUC0–12h values were 22,449.97 μg/L*h and 7,726.91 μg/L*h, the MRT were 1.07 hour and 0.57 hour. Compared with the SN injection, the AUC and the MRT has been increased about 2.9 times and 1.88 times by the SN-HA injection. The MRT of the SN-HA was 1.07 hours, which was also prolonged.
Histological evaluation
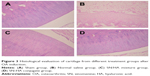
Figure 3 presents a comparison of different treatment groups. In the normal group, the superficial layer of articular cartilage was smooth and integrity, overlaying the subchondral bone plate and the chondrocytes were flattened and arranged in neat rows. However, in the saline group, the joint cartilage showed noticeable degradation, including irregular superficial layer, complete degenerative and disarranged chondrocytes. The SN-HA conjugate and SN/HA mixture treatment groups showed significant improvement of fissures, disorganization and loss of chondrocytes (P<0.05, Table 7). There was no significant difference between the SN-HA conjugate twice-treated group and SH-treated five times group.
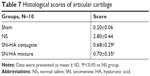  | Table 7 Histological scores of articular cartilage |
Discussions
In this study, we investigate the sustained-release effect of SN from the SN-HA conjugate in the rat joint and its therapeutic effect in a rabbit model of OA following intra-articular injection. The kinetic study results indicated that the MRT and AUC have been increased by the SN-HA conjugate, and the pharmacodynamic (PD) study indicated that the SN-HA conjugate twice-treated group and SN/HA mixture-treated five times group have no significant differences.
Intra-articular injection of papain could induce a reliable and reproducible rabbit OA model and was widely used for the efficacy study.22 However, synovial fluid collection was difficult to operate using living animals and would affect the SN content at the subsequent time-point. In this study, 6 animals each group were killed at each time-point for sample collection, rats were chosen rather than rabbits for the pharmacokinetics study considering the animal welfare and suitability for operation.23,24
Release of SN depended on the hydrolysis of SN-HA conjugate and hyaluronidase degradation of HA in the joint cavity. In our study, the content of total SN was detected, including the free SN and SN in the conjugate which could be completely released in the acidic treatment condition. A simple and fast HPLC-MS/MS method for the determination of SN in synovial fluid was developed and validated by allowing its determination in the 10–1,000.00 ng/mL range. The assay is rapid, the analysis time is only 6.5 minutes, and this method involves a single-step protein precipitation procedure using theophyllinum as IS. The results of the validation studies show that the method possesses selectivity, sensitivity, linearity, precision, and accuracy. The proposed method was successfully applied to the pharmacokinetic study of SN and SN-HA after intra-articular injection into the rat knee joint. Compared with the SN injection, the AUC and the MRT have been increased about 2.9 times and 1.88 times by the SN-HA injection.
Intra-articular injection of SN has therapeutic effect on knee OA by decreasing inflammatory cytokines, such as the IL–1β, IL-6 and tumor necrosis factor-α (TNF-α) in the synovial fliud.25,26 The primary mechanism of HA injection is to restore the viscoelasticity of synovial fluid and increase the ability of synovial fluid to lubricate and protect articular tissues, and to absorb joint loads, inhibiting the further progression of OA.27,28 In the pharmacodynamics study, intra-articular administration of SN-HA conjugate and SN/HA mixture solutions both showed significant improvement of fissures, disorganization and loss of chondrocytes. An important finding was that SN-HA conjugate could be injected fewer times than SN/HA mixture or HA to get similar OA treatment effect, because no significant difference was seen between the SN-HA conjugate twice-treated group and SN/HA mixture-treated five times group. However, in our further work, it is necessary to compare the effect of treatment twice with SN-HA conjugate with that of the treatment twice with SN/HA mixture, SN and HA, separately.
Conclusion
Intra-articular injection of SN-HA conjugate might be a novel effective therapeutic approach for the treatment of OA. In the future work, the PK/PD study in the same animal model was needed to illuminate the synergism effect and clear the relationship between PK and PD of SN-HA conjugate after intra-articular administration for OA treatment.
Acknowledgment
This work was supported by the Key Research And Development Plan of Shandong Province (2018GSF118045) and the Natural Science Foundation of Shandong Province (ZR2017BH051).
Disclosure
The authors report no conflicts of interest in this work.
References
Berenbaum F. Osteoarthritis as an inflammatory disease (osteoarthritis is not osteoarthrosis!). Osteoarthritis Cartilage. 2013;21(1):16–21. | ||
Altman RD, Manjoo A, Fierlinger A, Niazi F, Nicholls M. The mechanism of action for hyaluronic acid treatment in the osteoarthritic knee: a systematic review. BMC Musculoskelet Disord. 2015;16(1):321. | ||
Yamasaki H. Pharmacology of sinomenine, an anti-rheumatic alkaloid from Sinomenium acutum. Acta Med Okayama. 1976;30(1):1–20. | ||
Vieregge B, Resch K, Kaever V. Synergistic effects of the alkaloid sinomenine in combination with the immunosuppressive drugs tacrolimus and mycophenolic acid. Planta Med. 1999;65(1):80–82. | ||
Liu L, Buchner E, Beitze D, et al. Amelioration of rat experimental arthritides by treatment with the alkaloid sinomenine. Int J Immunopharmacol. 1996;18(10):529–543. | ||
Liu L, Riese J, Resch K, Kaever V. Impairment of macrophage eicosanoid and nitric oxide production by an alkaloid from Sinomenium acutum. Arzneimittelforschung. 1994;44(11):1223–1226. | ||
Kondo Y, Takano F, Yoshida K, Hojo H. Protection by sinomenine against endotoxin-induced fulminant hepatitis in galactosamine-sensitized mice. Biochem Pharmacol. 1994;48(5):1050–1052. | ||
Liang LJ, Xq J, Zhu H. Effect of intra-articular cavity injection of sinomenine hydrochloride for acute pain of knee osteoarthritis. China Mod Med. 2015;25:111–113. | ||
Feng S, Zhu L, Huang Z, et al. Controlled release of optimized electroporation enhances the transdermal efficiency of sinomenine hydrochloride for treating arthritis in vitro and in clinic. Drug Des Devel Ther. 2017;11:1737–1752. | ||
Yao Y-M, Tan Z-R, Hu Z-Y, et al. Determination of sinomenine in human plasma by HPLC/ESI/ion trap mass spectrum. Clinica Chimica Acta. 2005;356(1–2):212–217. | ||
Ye MR, Liu L, Zeng YE, et al. Studies on the relationship between sinomenine distribution and its organic toxicology. Chinese Pharmacol Bull. 2001;(1):65–69. | ||
Oh EJ, Park K, Kim KS, et al. Target specific and long-acting delivery of protein, peptide, and nucleotide therapeutics using hyaluronic acid derivatives. J Control Release. 2010;141(1):2–12. | ||
Mero A, Pasqualin M, Campisi M, Renier D, Pasut G. Conjugation of hyaluronan to proteins. Carbohydr Polym. 2013;92(2):2163–2170. | ||
Kim KS, Park SJ, Yang JA, et al. Injectable hyaluronic acid-tyramine hydrogels for the treatment of rheumatoid arthritis. Acta Biomater. 2011;7(2):666–674. | ||
Kong JH, Oh EJ, Chae SY, Lee KC, Hahn SK. Long acting hyaluronate – exendin 4 conjugate for the treatment of type 2 diabetes. Biomaterials. 2010;31(14):4121–4128. | ||
Galer CE, Sano D, Ghosh SC, et al. Hyaluronic acid-paclitaxel conjugate inhibits growth of human squamous cell carcinomas of the head and neck via a hyaluronic acid-mediated mechanism. Oral Oncol. 2011;47(11):1039–1047. | ||
Homma A, Sato H, Tamura T, et al. Synthesis and optimization of hyaluronic acid-methotrexate conjugates to maximize benefit in the treatment of osteoarthritis. Bioorg Med Chem. 2010;18(3):1062–1075. | ||
Saravanakumar G, Choi KY, Yoon HY, et al. Hydrotropic hyaluronic acid conjugates: synthesis, characterization, and implications as a carrier of paclitaxel. Int J Pharm. 2010;394(1–2):154–161. | ||
Ferguson EL, Alshame AM, Thomas DW. Evaluation of hyaluronic acid-protein conjugates for polymer masked-unmasked protein therapy. Int J Pharm. 2010;402(1–2):95–102. | ||
Liu J, Dw L, Cao XK. Preparation and characterization of hyaluronic acid-sinomenine conjugate. Chin Pharm J. 2015;50(20):1816–1821. | ||
Abbott RW, Townshend A, Gill R. Determination of morphine in body fluids by high-performance liquid chromatography with chemiluminescence detection. Analyst. 1987;112(4):397–406. | ||
Han G, Wang G, Zhu X, et al. Preparation of xanthan gum injection and its protective effect on articular cartilage in the development of osteoarthritis. Carbohydrate Polymers. 2012;87(2):1837–1842. | ||
Nemirovskiy O, Zheng YJ, Tung D, et al. Pharmacokinetic/pharmacodynamic (PK/PD) differentiation of native and PEGylated recombinant human growth hormone (rhGH and PEG-rhGH) in the rat model of osteoarthritis. Xenobiotica. 2010;40(8):586–592. | ||
Vugmeyster Y, Wang Q, Xu X, et al. Disposition of human recombinant lubricin in naive rats and in a rat model of post-traumatic arthritis after intra-articular or intravenous administration. Aaps J. 2012;14(1):97–104. | ||
Huang BD, He AS, Fu M, Sheng P, Liao WM. Sinomenine suppresses expression of interleukin-1beta-induced matrix metalloproteinases in human osteoarthritic chondrocytes. J Med Plants Res. 2010;4(18):1830–1836. | ||
Yang HQ, Chen LR. Effects of sinomenine on synovial fluid and serum content of interleukin-1β in rabbits with osteoarthritis. J Chin Integr Med. 2008;6(12):1275–1279. | ||
Moreland LW. Intra-articular hyaluronan (hyaluronic acid) and hylans for the treatment of osteoarthritis: mechanisms of action. Arthritis Res Ther. 2003;5(2):54–67. | ||
Bannuru RR, Natov NS, Dasi UR, Schmid CH, Mcalindon TE. Therapeutic trajectory following intra-articular hyaluronic acid injection in knee osteoarthritis – meta-analysis. Osteoarthritis Cartilage. 2011;19(6):611–619. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.