Back to Journals » Journal of Blood Medicine » Volume 11
Evaluation of Peripheral Blood Parameters of Pulmonary Tuberculosis Patients at St. Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia: Comparative Study
Authors Kahase D , Solomon A, Alemayehu M
Received 4 November 2019
Accepted for publication 9 March 2020
Published 1 April 2020 Volume 2020:11 Pages 115—121
DOI https://doi.org/10.2147/JBM.S237317
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Martin H Bluth
Daniel Kahase, Absra Solomon, Mihret Alemayehu
Department of Medical Laboratory Sciences, College of Medicine and Health Sciences, Wolkite University, Gubrie, Wolkite, Ethiopia
Correspondence: Daniel Kahase
Department of Medical Laboratory Sciences, College of Medicine and Health Sciences, Wolkite University, P.O. Box: 07, Gubrie, Wolkite, Ethiopia
Tel +251 9 1241 4564
Fax +251 11 322 0041
Email [email protected]
Background and Aim: Pulmonary tuberculosis is still among the leading cause of morbidity and mortality in Ethiopia. Different hematological abnormalities are commonly associated with pulmonary tuberculosis even though inconsistent results have been described. Hence, this study aimed to evaluate the hematological parameters of pulmonary tuberculosis patients visited St. Paul’s hospital millennium medical college, Addis Ababa, Ethiopia.
Methods: From April to September 2018, a comparative cross-sectional study was conducted among pulmonary tuberculosis patients (n=40) and control patients (n=40). About 5 mL venous blood and 2– 5 mL sputum samples were collected and examined by Cell Dyn 1800 hematology analyzer and cultured using Mycobacteria Growth Indicator Tube (BACTEC MGIT 960), respectively. Independent t-test was performed with the help of SPSS version 20 software, and p-value < 0.05 was considered as statistically significant difference.
Results: The proportion of male to female in the pulmonary tuberculosis patients (PTB) and the control patients was 1.7 (25/15). Two-sample independent t-test revealed that the mean values of hemoglobin level (P=0.002), hematocrit (P=0.018), mean cell hemoglobin concentration (P=0.001) and relative lymphocyte percentage (P=0.036) of PTB were significantly lower than the control group. Moreover, significantly higher mean values were also observed in total white blood cell count (P=0.004), platelet count (P< 0.001) and erythrocyte sedimentation rate (P< 0.001). Among the hematologic abnormalities detected, thrombocytosis and anemia presented in 65% and 25% of PTB patients, respectively.
Conclusion: Statistically significant mean differences were observed in hemoglobin, hematocrit (HCT), MCHC, relative lymphocyte percentage, WBC, platelet count, relative neutrophil percentage and ESR values. So, the utilization of such data is important in providing preliminary information for diagnosis and management of pulmonary tuberculosis. In fact, a further large scale study is needed to substantiate this finding.
Keywords: hematologic parameter, Mycobacterium tuberculosis, thrombocytosis, anemia
Introduction
Mycobacterium tuberculosis is an intracellular pathogen that is able to cause tuberculosis and humans are the only natural reservoir.1 During M. tuberculosis infection, host immunity plays an important role in the host–pathogen interaction. Neutrophils migrate early on the site of infection, followed by monocytes, which can be observed to differentiate into macrophages. Then, macrophages present processed Mycobacterium tuberculosis antigen to T-lymphocyte which is a type of white blood cell. These all host responses are more likely responsible for clinical manifestation of tuberculosis.2,3
Tuberculosis (TB) is one of the top ten causes of death, and the leading cause from a single infectious agent worldwide. Currently, world health organization reported the fastest regional declines of tuberculosis from 2013 to 2017 were in the WHO European Region followed by WHO African Region. Despite the decline in the WHO African Region, 2.5 million people fell ill with all forms of tuberculosis in the African region, accounting for a quarter of new TB cases worldwide in 2017.4
The severity of tuberculosis epidemics varies widely among countries. Ethiopia ranked 10th among the thirty high burden tuberculosis countries worldwide, with an estimated 164 incidents of all forms of TB cases per 100 000 population.4 Ethiopia ministry of health also reveals that TB is the eighth leading cause of hospital admission and the third leading cause of hospital deaths in Ethiopia in 2011.5
Studies present hematological and biochemical abnormalities are common in pulmonary tuberculosis patients.6,7 Leukocytosis, monocytosis, lymphocytosis, thrombocytosis, lymphopenia, and anemia are among the reported hematologic abnormalities.8–11 Modulating a normal hematopoiesis process is mentioned as a mechanism for peripheral blood abnormalities to occur.12 These hematological changes can act as important markers for diagnosis and persistent excretion of acid-fast bacilli, which is associated with failure of these indices to return to normal.6 In addition, these changes have correlation with the severity of clinical findings of pulmonary tuberculosis.13
If clinical laboratory assays such as hematologic parameters are interpreted carefully, it can be useful in assisting diagnosis and prognosis at a low cost. To the best of our knowledge, few studies are available in Ethiopia. Studies done in Jima and Gonder, Ethiopia stated the occurrence of hematological changes in pulmonary tuberculosis patients,14,15 even though their results are inconsistent in some of the hematological indices. As well, to the best of our knowledge, no study has been conducted in the study area. Therefore, this study planned to evaluate peripheral blood findings among PTB patients at St. Paul’s hospital millennium medical college, Addis Ababa, Ethiopia.
Materials and Methods
Study Design and Study Area
From April to September 2018, hospital-based comparative cross-sectional study was implemented at St. Paul’s hospital millennium medical college, which is the second largest referral hospital in Ethiopia. It is located in Addis Ababa, the capital city of Ethiopia. The hospital has different departments and annually offers diagnosis and treatment for approximately 100,000 up to 150,000 patients. All patients triaged in pulmonology department during the study period, with signs and symptoms suggestive of pulmonary tuberculosis were considered as a source population for PTB group.
Study Population
From the presumptive PTB patients who provided blood and sputum samples, culture confirmed consecutive pulmonary tuberculosis (PTB) patients were taken as PTB group. The control patients were those matched for sex and age of PTB patients and who visit the hospital for other medical cases which majorly have no effect on hematological parameters such as HIV and other chronic diseases. In addition, the control groups were also negative for pulmonary tuberculosis by culture.
Inclusion Criteria
Patients who did no take anti-tuberculosis drug, no record of any other chronic disease, non-pregnant women and HIV negative patients were included in the study.
Exclusion Criteria
Patients with signs of concomitant chronic or acute infection other than pulmonary tuberculosis, bleeding manifestations, endocrine disorders, other organ dysfunction or systemic disorders and chronic inflammatory disease on clinical examination were not included as PTB group.
Data Collection
After written informed consent was obtained from each study subjects, all subjects were asked to provide a detailed history and were subjected to a physical examination. Then, morning sputum and venous blood samples were collected.
Sputum Sample Collection, Processing and Culturing
Each collected morning sputum sample (2–5 mL) was mixed with an equal volume of N-acetyl-L-cysteine-sodium hydroxide (NALC-NaOH) solution, vortexing for 20 s was done and it was kept for 15 mins at 20–25°c for decontamination process. Then, Phosphate Buffered saline was filled to 50 mL mark on falcon tube and vortexing was done. After it was centrifuged at 3000 g for 15 mins, the supernatant was discarded and a portion of the deposits was used for culture.16,17
Portion of each specimen sediment (0.5mL) was inoculated into BACTEC MGIT 960 (Becton Dickinson, Franklin Lakes, NJ07417, USA) media and incubated in an automated BACTEC MGIT 960TM machine (Becton Dickinson Diagnostic Instrument Systems) for a maximum of 42 days. Cultures exhibiting growth were subjected to light microscopy for the presence of acid-fast bacteria using Ziehl-Neelsen stain and tested by Capilia TB-Neo rapid test. Finally, both Acid-fast bacteria and Capilia TB-Neo rapid test positive isolates were considered as Mycobacterium tuberculosis complex.16
Blood Collection and Analysis
Five milliliter blood samples were collected aseptically from each of the study participants and correctly labeled with the patient’s identification number. Then, complete blood count (hemoglobin, MCH, hematocrit, MCV, MCHC, platelet count, RBC count, total leukocyte count, and differential leukocyte count (Lymphocyte, Neutrophil and MID cell) were measured using Cell Dyn 1800 hematology analyzer. In addition, Erythrocyte sedimentation rate measurement was performed using Westergreen method).
Quality Assurance
The validity of the study findings was assured by properly using established standard operating procedures (SOPs) and manufactures instruction for both hematological tests and TB culture. Control organisms Mycobacterium tuberculosis H37Rv (ATCC 27294) reference strain was also used as a quality control for sputum culture. The quality performance of the hematology analyzer was checked before running the patients' sample by performing normal, low and high blood controls.
Operational Definitions
Hematological reference values were utilized from study done in Addis Ababa, Ethiopia. Accordingly, Leukocytosis, lymphopenia and thrombocytosis were defined as WBC > 10.2 × 103 cells/µL for both sex, lymphocyte percentage < 27.4 for male and < 25.3 for female and platelet count > 337 × 103 cells/µL for both sex, respectively.18
Anemia was defined per WHO guidelines as Hgb <13 g/dl for males and Hgb <12 g/dl for females. Hemoglobin values of 9.0–11.0 g/dl for women and 9.0–12.0 g/dl for men were considered as mild while Hemoglobin values 8.0–9.9 g/dl were considered as moderate anemia.19
Data Analysis
Data were entered into Microsoft Excel, exported to SPSS version 20 for further analysis. Descriptive statistics were used to describe the distribution of age, sex among the pulmonary tuberculosis cases and the control patients. Two-sample independent t-tests were utilized for comparison of the hematological parameters mean values between the pulmonary tuberculosis cases and the control group. A P-value of < 0.05 was considered as statistically significant difference.
Results
Characteristics of Study Subjects
This study enrolled forty culture-positive pulmonary tuberculosis patients and an equal number of control patients. Of the pulmonary tuberculosis patients (cases), males accounted for 25 (62.5%) and females accounted for 15 (37.5%). The proportion of male to female in the control patients was similar to the cases as shown in Table 1. The mean age of the pulmonary tuberculosis patients was 40 years (Sd. ±15). There was no statistically significant difference between the mean age (p=0.850) of the pulmonary tuberculosis patient (PTB) and the control group.
 |
Table 1 Sex and Age Group Distribution Among Eighty (n=80) Study Participants |
Comparison of Hematological Parameters
In the current study, the mean values for blood hemoglobin level (P=0.002), hematocrit (HCT) (P=0.018), mean cell hemoglobin concentration (MCHC) (P=0.001) and relative lymphocyte percentage (P=0.036) in PTB patients were found to be less than that of control patients. These differences were statistically significant as seen in Table 2. Other measured hematologic parameters like absolute total white blood cell count (WBC) (P=0.004), platelet count (P<0.001) and erythrocyte sedimentation rate (ESR) (P<0.001) values found higher than the control patients, which were also statistically significant. However, there were no statistically significant differences observed in red cell (RBC) count, mean cell volume (MCV), and mean cell hemoglobin (MCH) and MID cells (%) (Table 2).
 |
Table 2 Two Sample Independent t-Test for Hematological Parameters Among the Pulmonary Tuberculosis Patients and Control Patients at St. Paul’s Hospital Millennium Medical College, 2018 |
Hematologic Abnormalities in Pulmonary Tuberculosis Patients
Of the total forty pulmonary tuberculosis patients (PTB), leukocytosis, thrombocytosis, and lymphopenia were observed in 27.5%, 65%, and 60% of the patients, respectively (Table 3). About 25% of PTB study subjects showed anemia. Of these, the majority (70%) were with mild and the remaining 30% were with moderate anemia.
 |
Table 3 The Proportion of PTB Patients (n=40) with Low, Normal and High Values of Some Hematological Profile at St. Paul’s Hospital Millennium Medical College, 2018 |
Discussion
A range of diseases can have an effect on hematological indices including tuberculosis. Limited studies are available on blood parameters of pulmonary tuberculosis patients in Ethiopia and even the results of the studies vary in some of the hematological parameters. Thus, the current study evaluated hematological values of forty newly diagnosed pulmonary tuberculosis patients who were nonpregnant and HIV negative.
This study depicted a significant decrement of mean hemoglobin (p꞊0.002) and hematocrit values (p꞊0.018) of the pulmonary tuberculosis patients as compared to the control patients. Similar results with our findings also explained in studies done by Atomsa et al, Rohini et al, and Akpan et al.14,20,21 Scientifically different studies demonstrate that Mycobacterium tuberculosis can utilize heme as an iron source for survival22,23 which can be the reason for hemoglobin decrement.
It is known that hemoglobin measurement is important to define and classify anemia, which is an associated problem in tuberculosis patients.9,19 Anemia was found in 25% of our cases, more frequently mild anemia. This percentage is with little difference from study done by Bozóky et al, which showed as 32% of pulmonary tuberculosis patients had anemia.12 Other studies report high proportion of anemia in tuberculosis patients as 45.8%,14 46%15 and 60%.6 Those differences could reflect the differences of the study participants in their nutritional status, malabsorption syndrome and stage of the disease during diagnosis. Indeed, the small sample size of the current study may have also an effect on the differences observed.
Despite high percentage (72.5%) of PTB patients had normal total WBC count in this study, the mean total WBC count showed significantly higher (p=0.004) along with neutrophilia in those with PTB than the control patients. This finding is supported by previous studies done by Morris et al,6 Olaniyi et al,8 and Amilo et al.10 In fact, increased polymorphonuclear leukocytes and macrophages occur as a part of cell-mediated immunity to combat bacterial pathogen.2 However, the mean lymphocyte percentage which is a subpopulation of WBC showed significantly lower (p=0.036) than that of control subjects in our finding. This significant decrement of lymphocyte is in agreement with previous studies done by Akintunde et al,24 and Johnson et al.25 But, studies done by Shafee et al,11 and Amilo et al,10 contradict our result. These differences could reflect the variation in the study population.
Erythrocyte sedimentation rate raised with changes in plasma proteins, particularly increases in fibrinogen, immunoglobulins, and C-reactive protein in a wide range of infectious, inflammatory, degenerative, and malignant conditions.26 In this study, as expected, significantly elevated (p<0.001) ESR values were observed in the cases as compared to the control patients, with 85% of PTB patients had abnormal ESR values. Mandal et al also suggest that active TB is associated mostly with high ESR values.27
Based on our findings, thrombocytosis was detected in majority (65%) of the PTB patients, and the remaining patients had normal platelet count. One study conducted by Rathod et al reported as 75% of tuberculosis patients were with thrombocytosis.28 Lower thrombocytosis percentage (22%) also reported by Shafee et al.11 The percentage difference could be due to different definition was applied in this study for thrombocytosis (>337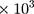 /µL). This is due to platelet count of Ethiopian is lower than the commonly utilized standard values.18
/µL). This is due to platelet count of Ethiopian is lower than the commonly utilized standard values.18
The increment of platelet count in the PTB than the control group in this study was statistically significant (p<0.001) which coincide with studies done by Amilo et al,10 and Al-Omar et al.29 A study done by Rohini et al on forty pulmonary tuberculosis patients showed statistically significant decrement of platelet count20 which contradicts our result. The variation could be the early diagnosis of patients in the current study which could have an effect on platelet count. Moreover, a study hypothesizes that increased platelet count in many cases is causally related to elevated interleukin‐6 (IL‐6) which is known to promote megakaryocytopoiesis during the acute phase of infection.30
This study was limited to a small number of participants due to the study laboratory techniques are time-consuming and costly. Therefore, this might have an effect in our conclusion.
Conclusion
This study revealed mean values of hemoglobin level, hematocrit (HCT), MCHC, relative lymphocyte percentage of PTB showed statistical decrements whereas total WBC, platelet count, relative neutrophil percentage and ESR values of PTB patients showed statistically significant increments over the control patients. Moreover, PTB patients found to have lymphopenia (60%) rather than lymphocytosis (10%) and relative to different studies low burden of anemia (25%) was observed in our PTB patients. So, these data are important to provide preliminary information in the diagnosis and management of pulmonary tuberculosis. Indeed, this study needs to be substantiated by further large-scale study.
Abbreviations
CBC, complete blood count; ESR, erythrocyte sedimentation rate; MCV, mean cell volume; MCH, mean cell hemoglobin; MCHC, mean cell hemoglobin concentration; PTB, pulmonary tuberculosis; SPSS, Statistical Package for Social Sciences; RBC, red blood cells; TB, tuberculosis; WBC, white blood cell; WHO, World Health Organization.
Data Sharing Statement
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics and Consent Statement
Ethical clearance obtained from Ethical Review Committee of Medical Laboratory Sciences, Allied Health Sciences, Faculty of Medicine; Addis Ababa University, Ethiopian Public Health Institute and St. Paulo’s Hospital Millennium Medical College. Written informed consent was also obtained from all eligible study participants, for patients aged below 18 years old from their guardian. The study was carried out in accordance with the Declaration of Helsinki. Finally, Laboratory confirmed cases were reported to and managed by the clinicians.
Acknowledgments
We would like to acknowledge the support of St. Paul’s Millennium Medical College Hospital staff and Ethiopian Public Health Institute particularly, national tuberculosis laboratory staff for their technical support. The authors are also thankful to all the study participants for their collaboration.
Author Contributions
Daniel Kahase conceived, designed the study and drafted the manuscript. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
The study was supported by Addis Ababa University. The funder had no role in data collection, study design, data analysis, and interpretation.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Murray PR, Rosenthal KS, Kobayashi GS, Pfaller MA. Medical Microbiology.
2. Schluger NW, Rom WN. The host immune response to tuberculosis. Am J Respir Crit Care Med. 1998;157(3):679–691. doi:10.1164/ajrccm.157.3.9708002
3. Cooper AM. Cell-mediated immune responses in tuberculosis. Annu Rev Immunol. 2009;27:393–422. doi:10.1146/annurev.immunol.021908.132703.
4. World Health Organization. WHO Report 2018 for 30 High-Burden Countries Profile. Geneva: World Health Organization; 2018.
5. Federal Democratic Republic of Ethiopia, Ministry of Health. Health and Health-Related Indicators. Addis Ababa, Ethiopia: Federal Ministry of Health; 2011.
6. Morris CD, Bird AR, Nell H. The haematological and biochemical changes in severe pulmonary tuberculosis. Q J Med. 1989;73(3):1151–1159.
7. Kurup R, Flemming K, Daniram S, Marks-james S, Roberts Martin R. Hematological and biochemistry profile and risk factors associated with pulmonary tuberculosis patients in Guyana. Tuberc Res Treat. 2016;2016:6983747. doi:10.1155/2016/6983747
8. Olaniyi JA, Aken’ova YA. Haematological profile of patients with pulmonary tuberculosis in Ibadan, Nigeria. Afr J Med Med Sci. 2003;32(3):239–242.
9. Lee SW, Kang Y, Yoon YS, et al. The prevalence and evolution of anemia associated with tuberculosis. J Korean Med Sci. 2006;21(6):1028–1032. doi:10.3346/jkms.2006.21.6.1028
10. Amilo GI, Meludu SC, Ele PU, Ezechukwu C, Onyenekwe C, Chukwu MI. Haematologic indices in pulmonary tuberculosis with or without HV Co-infection in South Eastern Nigeria. Adv Sci Technol Res J. 2013;11:1–7.
11. Shafee M, Abbas F, Ashraf M, et al. Hematological profile and risk factors associated with pulmonary tuberculosis patients in Quetta, Pakistan. Pak J Med Sci. 2014;30(1):36–40. doi:10.12669/pjms.301.4129
12. Schlossberg D. Tuberculosis and Non-Tuberculous Mycobacterial Infection.
13. Bozoky G, Ruby E, Goher I, Tóth J, Mohos A. Hematologic abnormalities in pulmonary tuberculosis. Orv Hetil. 1997;138(17):1053–1056.
14. Atomsa D, Abebe G, Sewunet T. Immunological markers and hematological parameters among newly diagnosed tuberculosis patients at Jimma University Specialized Hospital. Ethiop J Health Sci. 2014;24(4):311–318. doi:10.4314/ejhs.v24i4.6
15. Abay F, Yalew A, Shibabaw A, Enawgaw B. Hematological abnormalities of pulmonary tuberculosis patients with and without HIV at the University of Gondar Hospital, Northwest Ethiopia: a comparative cross-sectional study. Tuberc Res Treat. 2018;2018:5740951. doi:10.1155/2018/5740951
16. Mycobacteriology laboratory manual. A publication of the Global Laboratory Initiative a Working Group of the Stop TB Partnership. First Edition, April 2014. Available from: www.who.int/tb/laboratory/mycobacteriology-laboratory-manual.pdf.
17. Sankar MM, Kumar P, Munawwar A, Singh J, Parashar D, Singh S. Recovery of Mycobacterium tuberculosis from sputum treated with cetyl pyridinium chloride. J Clin Microbiol. 2009;47(12):4189–4190. doi:10.1128/JCM.01295-09
18. Tsegaye A, Messele T, Tilahun T, et al. Immunohematological reference ranges for adult Ethiopians. Clin Diagn Lab Immunol. 1999;6(3):410–414. doi:10.1128/CDLI.6.3.410-414.1999
19. World Health Organization. Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity. WHO/NMH/NHD/MNM/11.1; 2011.
20. Rohini K, Bhat MS, Srikumar PS, Kumar AM. Assessment of hematological parameters in pulmonary tuberculosis patients. Indian J Clin Biochem. 2016;31(3):332–335. doi:10.1007/s12291-015-0535-8
21. Akpan PA, Akpotuzor JO, Akwiwu EC. Some haematological parameters of tuberculosis (TB) infected Africans: the Nigerian perspective. J Natural Sci Res. 2012;2:50–57.
22. Tullius MV, Harmston CA, Owens CP, et al. Discovery and characterization of a unique mycobacterial heme acquisition system. Proc Natl Acad Sci USA. 2011;108(12):5051–5056. doi:10.1073/pnas.1009516108
23. Jones CM, Niederweis M. Mycobacterium tuberculosis can utilize heme as an iron source. J Bacteriol. 2011;193(7):1767–1770. doi:10.1128/JB.01312-10
24. Akintunde EO, Shokunbi WA, Adekunle CO. Leucocyte count, platelet count and erythrocyte sedimentation rate in pulmonary tuberculosis. Afr J Med Med Sci. 1995;24(2):131–134.
25. Jemikalajah JD, Okogun GA. Hematological indices in human immunodeficiency virus and pulmonary tuberculosis infections in parts of Delta State, Nigeria. Saudi Med J. 2009;30(2):253–256.
26. Cheesbrough M. District Laboratory Practice in Tropical Countries Part Two.
27. Mandal SK, Chavan L. Erythrocyte sedimentation rate values in cases of active tuberculosis without HIV co-infection. JMSCR. 2016;4(10):13156–13159. doi:10.18535/jmscr/v4i10.58
28. Rathod S, Samel DR, Kshirsagar P, Pokar M. Thrombocytosis: can it be used as a marker for tuberculosis? Int J Res Med Sci. 2017;5(7):3082–3086. doi:10.18203/2320-6012.ijrms20172991
29. Al-omar IA, Al-ashban R, Shah A. Hematological abnormalities in Saudis suffering from pulmonary tuberculosis and their response to the treatment. Res J Pharma. 2009;3(4):78–85.
30. Hollen CW, Henthorn J, Koziol JA, Burstein SA. Elevated serum interleukin‐6 levels in patients with reactive thrombocytosis. Br J Haematol. 1991;79(2):286–290. doi:10.1111/j.1365-2141.1991.tb04534.x
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
