Back to Journals » Cancer Management and Research » Volume 11
Establishment and characterization of the GC-030-35 cell line derived from gastric hepatoid adenocarcinoma
Authors Wei J, Xue Y , Huo X, Han R, Su X , Jin Y, Zhao W, Chen Y, Zhang H, Dai J , Chen J
Received 4 September 2018
Accepted for publication 10 January 2019
Published 8 February 2019 Volume 2019:11 Pages 1275—1287
DOI https://doi.org/10.2147/CMAR.S186416
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Beicheng Sun
Jingsun Wei,* Yiqi Xue,* Xinying Huo, Rongbo Han, Xinyu Su, Yan Jin, Wenjing Zhao, Yuetong Chen, Honghong Zhang, Jiali Dai, Jinfei Chen
Department of Oncology, Nanjing First Hospital, Nanjing Medical University, Nanjing, China
*These authors contributed equally to this work
Purpose: Gastric hepatoid adenocarcinoma is a rare subtype of primary gastric cancer and is a high-grade form of malignancy. However, the pathogenesis and molecular biology of gastric hepatoid adenocarcinoma remain poorly understood. The aim of this study was to establish and characterize a new human gastric hepatoid adenocarcinoma cell line, GC-030-35.
Materials and methods: The GC-030-35 cell line was established from tumor cells from a 58-year-old Chinese man with gastric hepatoid adenocarcinoma. The cultured cells underwent immunocytochemistry and flow cytometry to confirm the tumor cell phenotype. RNA sequencing was performed to analyze the differences in gene expression between GC-030-35 cells compared with normal gastric epithelial cells. A zebrafish assay was performed. Gene enrichment analysis and interrogation of the bioinformatics databases, the Gene Ontology (GO) database and the Kyoto Encyclopedia of Genes and Genomes (KEGG) database, were used for pathway analysis.
Results: Flow cytometry analysis of the GC-030-35 cells showed a positive expression rate for CD44+ of 10.7%, high cell clonality, an average plating efficiency of 32%, cell-doubling time of 29.2 hours, and cell proliferation for >15 generations in serial culture. The zebrafish assay showed the ability of the GC-030-35 cells to proliferate, promote angiogenesis, and metastasize. RNA sequencing identified the functional clustering of 6,601 differentially expressed genes of GC-030-35, which were significantly different when compared with nonneoplastic gastric epithelial cells. Pathway enrichment analysis and interrogation of the GO and KEGG bioinformatics databases identified genes for microbial metabolism in diverse environments (63 genes), metabolism of xenobiotics by cytochrome P450 (CYP450; 25 genes), and the drug metabolism cytochrome P450 (28 genes).
Conclusion: A human gastric hepatoid adenocarcinoma cell line, GC-030-35, was developed and characterized by comparison with normal gastric epithelial cells. Bioinformatics and gene analysis data showed that the CYP450 gene was significantly differentially expressed by GC-030-35 cells.
Keywords: gastric hepatoid adenocarcinoma, cell line, differentially expressed genes, bioinformatics, carcinogenesis, cytochrome P450
Introduction
Worldwide, gastric cancer is one of the most common malignancies of the gastrointestinal tract, and the incidence of gastric cancer is ranked as the fourth most commonly diagnosed malignant tumor, and the third most common cause of mortality from gastrointestinal cancer.1 The prevalence of gastric cancer is greater in East Asia and South America than in Europe and North America.2 Gastric hepatoid adenocarcinoma, which is also known as gastric adenocarcinoma with enteroblastic differentiation, is a rare type of primary gastric carcinoma that has a poor prognosis, possibly associated with the difficulty in diagnosing cancer at an early stage. Previous reports on the pathology, pathogenesis, and diagnosis of this rare primary gastric tumor have come from small case series or case reports. Gastric hepatoid adenocarcinoma expresses alpha-fetoprotein (AFP) in the serum and in the tumor cells, but the relationship between gastric hepatoid adenocarcinoma and the expression of AFP is not specific or diagnostic.3–5 Currently, the clinical and pathological features of gastric hepatoid adenocarcinoma have not been fully elucidated.
Hepatoid adenocarcinomas have now been described to originate from the stomach, esophagus, colon, gallbladder, uterus, bladder, pancreas, and ovary, but gastric hepatoid adenocarcinoma is the most commonly found, with a reported incidence of 83.9% of all hepatoid forms of epithelial adenocarcinoma.6 Gastric hepatoid adenocarcinoma has a poor prognosis, with a reported mean survival time from diagnosis of between 10 and 18 months, and a 5-year survival rate of only about 10%.7 Because the pathogenesis and molecular biology of gastric hepatoid adenocarcinoma remain to be determined, further studies are needed to explore the precise mechanisms of gastric hepatoid adenocarcinoma and to find accurate ways to diagnose and treat this disease. It is extremely necessary to establish a stable and effective gastric hepatoid adenocarcinoma cell line.
Herein, we established and characterized the GC-030-35 cell line. The cell line has been studied in the following: the property of growth, cellular morphology and ultrastructure, karyotype, the expression of tumor-related antigens, CD44 and CD326 expression, neoplastic behavior in zebra fish, RNA sequencing, cell line authentication by short tandem repeat (STR) profiling, no mycoplasma contaminated.
RNA sequencing is a recently developed transcriptome analysis method used to detect the gene expression differences in a particular biological process (BP). Research that has utilized RNA sequence analysis has changed the perception of the extent and complexity of eukaryotic transcription. More importantly, RNA sequencing can also result in more accurate measurements of transcripts and their isomers when compared with other methods.8,9
In the present study, RNA sequencing was chosen as a method to determine the gene expression profiles of gastric hepatoid adenocarcinoma cells and to identify differentially expressed genes (DEGs) in this malignancy. Also, the functions of DEGs were analyzed by bioinformatics data from Gene Ontology (GO) annotation and the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment data. The study of DEGs in gastric hepatoid adenocarcinoma may provide new molecular biomarkers that can be used in the diagnosis, prognosis, and treatment of this rare but aggressive gastric cancer.
Therefore, the aim of this study was to establish and characterize a new human gastric hepatoid adenocarcinoma cell line, GC-030-35, and to investigate the mechanisms, biological features, and genetic characteristics of gastric hepatoid adenocarcinoma cells using RNA sequence analysis and bioinformatics data analysis.
Materials and methods
Establishment and cell culture conditions
In 2016, a 58-year-old Chinese man underwent radical surgery to resect a 2 cm diameter tumor mass of the gastric antrum. The pathologic diagnosis was gastric antral hepatoid adenocarcinoma, stage IV with liver metastases. The patient provided informed consent for his tumor to be used to produce a cell line. The medical ethics review report was approved by Nanjing First Hospital Ethics Committee.
The gastric tumor tissue from the patient was washed and placed in a transport medium consisting of Dulbecco’s PBS (Thermo Fisher Scientific, Waltham, MA, USA) with 1,000 U/mL penicillin G, 1,000 µg/mL streptomycin, and 2.5 µg/mL amphotericin B and immediately transported to the laboratory. Nonneoplastic gastric mucosal tissues (≥5 cm from the edge of the tumor) were also obtained from the gastric resection specimen from the patient.
Tumor tissues were washed twice with the transport medium and maintained in the medium for 1–2 hours at 4°C. Small tissue samples were plated in 6-cm sterile culture dishes and cultured in RPMI 1640 (Thermo Fisher Scientific) supplemented with 10% heat-inactivated FBS containing 1,000 U/mL penicillin G, 1,000 µg/mL streptomycin, and 2.5 µg/mL amphotericin B and RPMI 1640 complete medium. Then, the tissue pieces were incubated in a humidified atmosphere containing 5% CO2 at 37°C.
After 2 weeks, tumor cell growth occurred and an initial cell passage was performed. Fibroblasts were removed using trypsin. Tumor cells were obtained and identified as the GC-030-35 cell line (human gastric hepatoid adenocarcinoma). Manual selection of the monoclonal tumor cell populations of the GC-030-35 cell line was performed from the semisolid medium. First, 103 viable GC-030-35 single cells were plated in 3 mL of RPMI 1640 complete medium containing 0.3% agarose at 37°C on a basal layer of RPMI 1640 complete medium containing 0.6% agarose and incubated in a 37°C incubator with 5% CO2. Colony formation was observed after 12–15 days.
Single colonies were selected and expanded in RPMI 1640 complete medium in a 37°C incubator containing 5% CO2, and the cells were fed with additional medium every 2–3 days, depending on the pH. Finally, a highly pure tumor cell line was established that could undergo stable passage. The human gastric hepatoid adenocarcinoma cell line was named GC-030-35, which is one of the four sublines of GC-030.
Ultrastructural studies of the GC-030-35 cells
Cell pellets were fixed in 2.5% glutaraldehyde, postfixed in 1% osmium tetroxide, stained with 1% uranyl acetate, and thin sections were examined using a Siemens 1A electron microscope (Siemens, Munich, Germany).
GC-030-35 cell line authentication by STR profiling
The GenePrint® 10 system (Promega Corporation, Fitchburg, WI, USA) was used to amplify human genomic DNA extracted from the GC-030-35 cell lines. The amplification products were assessed using the ABI 3730xl automated DNA analyzer (Thermo Fisher Scientific). GeneMapper software version 4.0 (Thermo Fisher Scientific) was used to analyze the data, which were compared with the American Type Culture Collection (ATCC, Manassas, VA, USA), DSMZ cell culture collection, and the JCRB Cell Bank database for reference matching. STR testing was performed at GENEWIZ (South Plainfield, NJ, USA).
Chromosome analysis of the GC-030-35 cells
The karyotype analysis of the 15th passage of logarithmic growth phase GC-030-35 cells was undertaken. These cells were treated with colchicine for 2 hours, and then with 0.075 mol/L potassium chloride solution and fixed with glacial acetic acid. The cells were placed on cold glass slides and digested at room temperature with zymogen before staining with Giemsa stain for chromosomal banding analysis.
Immunohistochemical analysis of the GC-030-35 cells
Immunohistochemical staining analysis of the 15th passage of logarithmic growth phase GC-030-35 cells was undertaken using primary antibodies directed against specific proteins. Cell morphology was evaluated by light microscopy. The cells were cultured in 100-mm well plates and developed until they reached 90% confluence. For immunohistochemical staining, tumor cells were collected by centrifuging the cell suspension to form a cell pellet. The cell pellet was fixed overnight in neutral buffered formalin before being processed and embedded in paraffin wax and sectioned onto glass slides. Sections were dewaxed with xylene and rehydrated with graded alcohol, and then briefly immersed in water. Endogenous peroxidase activity was blocked by incubation with 3% hydrogen peroxide for 10 minutes.
Microwave heat treatment was used for antigen retrieval and was carried out by heating the sections, which were immersed in 0.01 mol/L citrate buffer, pH 6.0, in the microwave oven (450 W) for 10 minutes. Then, the slides were washed with PBS before incubation with the respective primary antibody, overnight at 4°C. The slides were incubated for 1 hour with the secondary antibody after being washed with PBS, and then washed a further three times for 5 minutes with PBS.
An immunoperoxidase method was used with the following primary antibodies obtained from Abcam (Cambridge, UK): anti-alpha 1 fetoprotein antibody (AFP-01) (ab3980), anti-glypican-3 (GPC3) antibody (ab66596), anti-cytokeratin 19 antibody (EP1580Y) (ab52625), anti-gastric mucin/MUC6 antibody (MUC6/916) (ab212648), anti-MUC2 antibody (EPR6145) (ab134119), anti-CDX2 antibody (EPR2764Y) (ab76541), and anti-EpCAM antibody (MOC-31) (ab187270). The primary antibody to Hep Par 1 (MAB7927) was obtained from Abnova (Taipei, Taiwan). The negative control replaced the primary antibody with PBS.
The peroxidase reaction was developed in PBS using hydrogen peroxide as a substrate and 3,3′-diaminobenzidine as the chromogen (brown). The sections were stained with hematoxylin, dehydrated, and examined by light microscopy.
Flow cytometry analysis of the GC-030-35 cells
The cultured cells that reached 90% confluence in a 100 mm diameter cell plate were dissociated from the plate by trypsin and EDTA. The cells were centrifuged in 30 mL of PBS and resuspended to 107 nucleated cells per 100 µL of FcR Blocking Reagent (130-059-901). The cells were stained with 10 µL of the following reagents, which were labeled with the fluorophores, fluorescein isothiocyanate (FITC) or phycoerythrin (PE): CD44-FITC (130-098-210), CD326 (EpCAM)-PE (130-098-115), and the isotype control antibodies IgG1-FITC (130-098-847) or IgG1-PE (130-098-845). The reagents used in this experiment were all from Miltenyi Biotec, Bergisch Gladbach, Germany. A BD FACSCanto II system (BD Biosciences, San Jose, CA, USA) was used to perform the flow cytometry analysis.
Cell proliferation and colony formation assay of the GC-030-35 cells
A 1,000-µL cell suspension containing 2,000 cells, which were in the logarithmic phase, was obtained from the cells cultured in the 24-well plates. The GC-030-35 cells were placed in the xCELLigence® RTCA DP instrument (ACEA Biosciences Inc, San Diego, CA, USA) which used electrical impedance to monitor cell morphology, quantify cell proliferation, and cell attachment, every hour for 8 days. From the growth curve, the cell population doubling time (PDT), the cell index, and integration times were calculated according to the formula: PDT = (T − T0) lg2/(lgNt − lgN0), where PDT represents the cell PDT, and T0 and T represent the starting time and end of the cell culture.
The cell index was used to indicate cell impedance, which reflected the number of adherent cells. To explore the plating efficiency of cells, 100, 500, 1,000, and 2,000 single-cell suspensions were placed in 60-mm diameter culture dishes and cultured for 9 days. The ratio of clone number to total inoculated cell number was calculated, and ≥50 cells were considered to represent plating efficiency.
Zebrafish assay for tumor formation by the GC-030-35 cells
Zebrafish zygotes were harvested and incubated at 28.5°C for 24 hours. Water was mixed with 1-phenyl-3-(2-thiazolyl)-2-thiourea (Sigma-Aldrich Co, St Louis, MO, USA) to a final concentration of 0.2 mM, which was added to the incubator and the zygotes were incubated for another 24 hours. The zebrafish embryonic membrane was removed, and the GC-030-35 cells were injected into the zebrafish yolk sac, which had been pretreated with the red fluorescent dye, CellTracker™ CM-Dil (C7000) (Thermo Fisher Scientific). Four days later, fluorescence microscopy was performed, using a D-eclipse C1 fluorescence microscope (Nikon Corporation, Tokyo, Japan), and the fluorescence intensity was analyzed using the Image-Pro Plus version 6.0 software (Media Cybernetics, Rockville, MD, USA).
Nude mouse assay for tumor formation by the GC-030-35 cells
Aliquots of the GC-030-35 cell populations were counted, and cell viability was determined using a conventional trypan blue test. Cells were resuspended in an ice-cold 1:1 mixture of growth medium and Matrigel matrix (BD Biosciences), and 5×106 cells/site were injected subcutaneously into the flanks of five 6- to 8-week-old mice. Two of them were BALB/C nude mice, and three of them were nonobese diabetic/severe combined immunodeficiency mice (supplied by the Comparative Medicine Center of Yangzhou University, Jiangsu, China). The mice were observed weekly for 3 months. The study followed the principles of the Constitution of the laboratory animal ethics committee of Southeast University (school notice [2007] no 51; date: April 24, 2007), and the animal experimental ethical inspection form was approved by the Animal Care and Welfare Committee of Southeast University (no 20170214003; application date: February 14, 2017).
RNA sequence analysis of the GC-030-35 cells
RNA sequence library preparation and sequencing were performed at BioNova (Beijing, China). Total RNA was extracted and digested with DNase, the mRNA in the total RNA was purified by Oligo d(T) beads, and the cDNA was synthesized by fragments of the obtained mRNA. Then, the entire RNA sequence library preparation was complete after purification, modification, and PCR amplification. The constructed RNA sequence library used the Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA, USA) and the ABI StepOnePlus Real-Time PCR System (Thermo Fisher Scientific) for quality and yield testing. Sequencing was performed after quality testing.
Results
Cytomorphology of the GC-030-35 cells
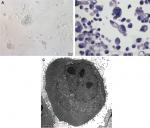
Phase-contrast microscopy showed that the cells of the GC-030-35 cell line showed no contact inhibition. Most cells were about 10 µm in size. A large number of vacuoles could be seen in the cytoplasm, the nucleus was central, with distinct nucleoli. There were 3%–5% of the large cells that were >50 µm in diameter. The nuclear size was about 8–10 µm in diameter, with inequality of size (Figure 1A and B). Transmission electron microscopy showed that the cell nuclei were heterogeneous, many nucleoli were visible, and microvilli-like protrusions were found on the cell surface, with cytoplasmic mitochondria that were swollen with abundant glycogen (Figure 1C).
STR matching analysis showed specificity of the GC-030-35 cell line
GC-030-35 homologous cells and mycoplasma were not found by comparing the STR matching analysis of the ATCC, DSMZ cell culture collection, and the JCRB Cell Bank database for reference matching. These results suggested that the cell line was unique.
Chromosome analysis of the GC-030-35 cells
The aim of the assay was to study the chromosomal changes in gastric hepatoid adenocarcinoma. The results showed that in GC-030-35 cells, chromosome numbers ranged from hypo-pentaploid (about 93–114) to chromosome numbers that were hypo-triploid (about 64–66) (Figure S1A and B). However, analysis of chromosome karyotypes could not be performed because the chromosomes were too large to analyze.
Immunohistochemical analysis of the GC-030-35 cells
Immunohistochemistry is routinely used in diagnostic pathology to type tumors, including gastric hepatoid adenocarcinoma. Immunohistochemistry showed positive immunostaining for AFP (Figure 2A), and GPC3 (+) in tissues (Figure 2B) and in cells (Figure 2C). Most of the gastric hepatoid adenocarcinoma cells were positive for AFP, and the positive staining for GPC3 was almost 100%. Of the epithelial markers, CK l9 was positive (Figure 2D), but the specificity was poor. Hep Par 1 immunostaining was an important marker as immunostaining for Hep Par 1 was positive in GC-030-35 cells (Figure 2E). Immunostaining was also positive for MUC6 and MUC2 but was negative for CDX2 (Figure 2F–H), which confirmed that the GC-030-35 cells were of a gastric epithelial phenotype.10 Also, 90% of the cells of the GC-030-35 cell line were positive for the cell proliferation marker, Ki-67 (Figure 2I).
CD44 and CD326 expression assay
Flow cytometry analysis was performed to study the phenotype of the cell lines and the proportion of cancer stem cells in the GC-030-35 cell line. When compared with the control group of normal gastric epithelial cells, the percentage of CD44-positive cells in the GC-030-35 cell line was 10.7% (Figure 3A and B). Almost 100% (98.2%) of the cells in the GC-030-35 cell line were CD326 (EpCAM) positive compared with minimal positivity in the control group (Figure 3C and D). These findings supported the notion that GC-030-35 cells were derived from the epithelial cells and that the GC-030-35 cell line might have the potential for future study of cancer stem cells.
Cell proliferation and clone formation of the GC-030-35 cells
The assay was to study the growth property of the GC-030-35 cells. The average doubling time of GC-030-35 cells was 29.2 hours, according to the fusion of four proliferation curves (data not shown) (Figure 4A). When 100, 500, 1,000, and 2,000 cells were placed in 60-mm diameter culture dishes, there was a plating efficiency of 39%, 29.4%, 30.55%, and 28%, respectively (Figure 4B), with the average plating efficiency of GC-030-35 cells being 31.7%.
Tumorigenicity of the GC-030-35 cells in vivo
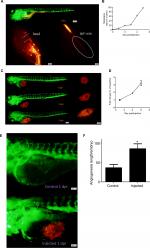
Zebrafish embryos were used to study the tumorigenicity of GC-030-35 cells in vivo. A total of 411 zebrafish were injected, 217 of which could be used for follow-up observation. The success rate of tumor formation following cell injection was 52.80%. After 1 day postinjection (dpi), 24 fish were randomly selected to be placed in 24-well plates, one embryo per well, to observe the tumor growth in the zebrafish. With time, in this in vivo model, tumor cells began to metastasize in the zebrafish (Figure 5A and B).
Cell proliferation and angiogenesis were studied. Cell proliferation was quantified from 1 dpi. The fold-change at 6 dpi was three times more than at 1 dpi (Figure 5C and D). The cells also showed an increased role in inducing the abnormal neovascularization (Figure 5E and F).
The nude mouse tumorigenicity assay was also performed, but the results were negative (Figure S2A and B).
RNA sequence analysis of the GC-030-35 cells
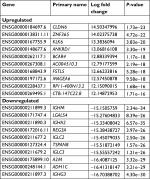
To investigate differences in the gene expression of the GC-030-35 cells, GC-030-35-C (cancer cell) and GC-030-35-N (nonneoplastic gastric mucosal tissue) were compared to screen out DEGs in GC-030-35 cancer cells. All of the 6,000 DEGs were identified, including 2,149 upregulated and 4,451 downregulated DEGs. The top 10 upregulated and downregulated DEGs are listed in Table 1. Enriched items identified from the GO database were divided into cellular component (CC), BP, and molecular function (MF) ontology, and these GO terms are shown in Figure 6.
In the CC ontology, the most related categories were plasma membrane (1,778 genes) and extracellular space (536 genes) (Figure 6A). In the BP ontology, immune response (639 genes) and cell adhesion (667 genes) were the most related gene categories (Figure 6B). In the MF ontology, binding-related items were the major enriched categories and included calcium ion binding (909 genes) and receptor binding (1,452 genes) (Figure 6C). Also, pathway analysis was undertaken using the KEGG bioinformatics database for the assessment of potential dysfunctional pathways, including those for microbial metabolism in diverse environments (63 genes), metabolism of xenobiotics by cytochrome P450 (CYP450; 25 genes), and drug metabolism by CYP450 (28 genes) (Figure 6D).
Discussion
Gastric hepatoid adenocarcinoma is a rare form of primary gastric adenocarcinoma when compared to other types of gastric cancer, with a reported incidence of 0.38%–0.73%.11 Currently, there are no evidence-based clinical guidelines on the diagnosis and management of gastric hepatoid adenocarcinoma, and treatment is based on those for gastric adenocarcinoma and includes surgery and chemotherapy. Gastric hepatoid adenocarcinoma is associated with early lymph node metastasis and hepatic metastasis, resulting in poor prognosis. Also, this tumor is often diagnosed at a late stage, which also contributes to its poor prognosis. However, the tumor lacks special clinical manifestations, so it is already in the late stage of cancer when diagnosed. Gastric hepatoid adenocarcinoma has a high rate of lymphatic and distant metastasis,12 especially the early development of liver metastasis and portal vein tumor thrombosis.13
Currently, there are no specific diagnostic markers for gastric hepatoid adenocarcinoma. Extensive loss of heterozygosity deficiency and high allelic loss could account for the development of invasive clones of gastric hepatoid adenocarcinoma, and this is one of the reasons that this tumor is more aggressive than other adenocarcinomas.14 Because of the combination of these factors that result in the poor prognosis for patients with this type of gastric cancer, it is important to study the biological and molecular mechanisms of this disease. The establishment of a specific cell line may provide the basis for further research into the biological and molecular mechanisms involved in gastric hepatoid adenocarcinoma.
In the present study, we established the human GC-030-35 gastric hepatoid adenocarcinoma cell line, derived from cells from a man with a histologically confirmed tumor of the gastric antrum. Currently, although there are at least 20 available human gastric cancer cell lines, most of them are derived from ascites, lymph node metastases, and pleural effusions.15 The findings of this study showed that the tumorigenicity of the GC-030-35 cell line was negative in nude mice, and positive in a zebrafish assay, which might indicate that the cell line may only show part of the tumor characteristics.16 However, further studies are needed to further establish the in vitro characteristics of this cell model of gastric hepatoid adenocarcinoma.
Immunohistochemistry is an important technique that is used clinically to determine the immunophenotype of gastric hepatoid adenocarcinoma, with a diagnostic panel of primary antibodies to detect the expression of AFP, alpha 1-anti-chymotrypsin, alpha 1-antitrypsin, and GPC3 in tumor cells using immunohistochemistry.7 AFP expression is increased in 59.8%–93% of tumors from patients with gastric hepatoid adenocarcinoma, and AFP is a helpful auxiliary diagnostic marker.17 Increased expression of AFP is characteristic of gastric hepatoid adenocarcinoma but is not specific for this tumor.3 In the present study, GPC3 was positively expressed by gastric hepatoid adenocarcinoma cells. In gastric hepatoid adenocarcinoma, regardless of positive or negative AFP, the positive immunostaining rate of tumor cells for GPC3 has been reported to be as high as 100%, which is an important factor in diagnosis and treatment.18 The findings of the present study also showed that Hep Par 1 was focally positive in tumor cells. Hep Par 1 is an effective immunohistochemical marker for hepatoid differentiation, and positive immunostaining can occur in extrahepatic tumors, especially in adenocarcinomas with hepatoid morphological features.19 Maitra et al showed that Hep Par 1 was focally positive in six out of seven cases of gastric hepatoid adenocarcinoma20 Therefore, the detection of Hep Par 1 expression can be regarded as supportive for the diagnosis of extrahepatic hepatoid adenocarcinoma. Also, because gastric hepatoid adenocarcinoma is a lesion of the gastric mucosa,21 the expression of epithelial markers, including CK19, were evaluated and supported that the GC-030-35 cells were derived from gastric epithelial cells. Also, Ki-67 is commonly used as a cell proliferation marker and increased expression of Ki-67 is associated with high-grade tumors with a poor prognosis.22,23 Ki-67 was positive in the cells of the GC-030-35 cell line, and so this cell line might be useful in future studies of Ki-67 expression in gastric cancer.
The tumorigenicity of GC-030-35 in metastasis, cell proliferation, and angiogenesis was demonstrated using the zebrafish assay. The rate of development of tumor metastases following injection of GC-030-35 cells was nearly 100% by 5 dpi, which indicated the strong metastatic capability of gastric hepatoid adenocarcinoma cells, and might explain the poor prognosis of gastric hepatoid adenocarcinoma.13 GC-030-35 cells induced abnormal neovascularization by 1 dpi in the zebrafish model. Angiogenesis is a highly regulated process, and in malignant tumors, the properties of angiogenesis and invasion are prerequisites for metastasis. Control of tumor angiogenesis is now recognized to be an approach to treating cancer and to improving patient prognosis by preventing metastases.24,25
Park et al showed that some of the cell lines in human colorectal carcinoma, including NCI-H958 and NCI-H742, were not tumorigenic in nude mice.26 In the present study, the tumorigenicity of GC-030-35 was confirmed in the zebrafish assay, but was not confirmed in nude mice. Further studies are required to determine whether GC-030-35 cells in different in vivo models and different microenvironments might show different tumor behavior. In the present study, the GC-030-35 cells showed the ability of metastasis in the zebrafish model, which is a vertebrate tumor model, in terms of genomics and histopathology.27,28 Therefore, further studies with zebrafish might be useful in the study of the biological characteristics of cells of the human GC-030-35 cell line.
The transcriptome of the GC-030-35 cell line was sequenced and the total results of DEGs in GC-030-35 were acquired and compared with nonneoplastic gastric mucosal cells. The biological functions of these enriched items identified from the GO database were divided into CC, BP, and MF ontology. In CC oncology, the majority of these DEGs were enriched in the plasma membrane and extracellular space, indicating that the development of gastric hepatoid adenocarcinoma might be caused by complicated cellular mechanisms associated with membrane structure. In BP ontology, the most relevant items were the immune response and cell adhesion, which are closely associated with the progression and prognosis of cancer, as the immune system inhibits tumor growth. However, tumors can make use of immunomodulatory mechanisms to form an immunosuppressive microenvironment that suppresses immune responses, such as the tumor infiltrating lymphocyte.29
Cell adhesion molecules are associated with tumor invasion and metastasis and include calcium-dependent adhesion molecules and cadherins. In the present study, calcium ion binding was one of the most meaningful items in the MF ontology. A previously reported study showed that plasma membrane Ca2+-ATPase (PMCA) was overexpressed in some malignant tumors, including breast cancer, colorectal cancer, and liver cancer.30–32 Therefore, PMCA may represent a specific biological biomarker for the differentiation of cancer cells, including gastric hepatoid adenocarcinoma. The KEGG pathway analysis showed that CYP450 was the most significant one in expression differences and the degree of enrichment of the pathway. So, CYP450 may be closely linked to gastric hepatoid adenocarcinoma. CYP450 not only participates in the metabolic activation of carcinogens but also inactivated anticancer drugs, resulting in treatment-resistant cancers.33 The CYP450 gene plays a major role in the development of multidrug resistance in tumors, and some exogenous drugs can induce abnormal expression of CYP450 and promote its own metabolism. Therefore, the role of CYP450 and the expression of the CYP450 gene require further studies to determine whether this may be a new therapeutic target for patients with gastric hepatoid adenocarcinoma.
Conclusion
A human gastric hepatoid adenocarcinoma cell line, GC-030-35, was developed and characterized by comparison with normal gastric epithelial cells. Using gene analysis and bioinformatics data, CYP450 was identified as a significant DEG. Although gastric hepatoid adenocarcinoma is very rare, GC-030-35 was shown to be a mature cell line with unique biological characteristics, which may also serve as a future model for the study of the molecular biology of this malignancy, to provide insight into potential targets for therapy. RNA sequencing of GC-030-35 supported by interrogation of bioinformatics data provided a preliminary finding for future study, as was identified. The findings of this preliminary study should be developed further, including further bioinformatics analysis and also by whole-genome sequencing analysis. It is hoped that this new gastric hepatoid adenocarcinoma cell line, GC-030-35, will be of use in future studies.
Acknowledgments
The work was partly supported by grants from the National Natural Science Foundation of China (grant no 81572928 and 81772978) and the Science and Technology Support Program of Jiangsu Province (BE017611).
Disclosure
The authors report no conflicts of interest in this work.
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. | ||
Ferro A, Peleteiro B, Malvezzi M, et al. Worldwide trends in gastric cancer mortality (1980-2011), with predictions to 2015, and incidence by subtype. Eur J Cancer. 2014;50(7):1330–1344. | ||
Matsunou H, Konishi F, Jalal RE, Yamamichi N, Mukawa A. Alpha-fetoprotein-producing gastric carcinoma with enteroblastic differentiation. Cancer. 1994;73(3):534–540. | ||
Govender D, Ramdial PK, Clarke B, Chetty R. Clear cell (glycogen-rich) gastric adenocarcinoma. Ann Diagn Pathol. 2004;8(2):69–73. | ||
Ghotli ZA, Serra S, Chetty R. Clear cell (glycogen rich) gastric adenocarcinoma: a distinct tubulo-papillary variant with a predilection for the cardia/gastro-oesophageal region. Pathology. 2007;39(5):466–469. | ||
Su JS, Chen YT, Wang RC, Wu CY, Lee SW, Lee TY. Clinicopathological characteristics in the differential diagnosis of hepatoid adenocarcinoma: a literature review. World J Gastroenterol. 2013;19(3):321–327. | ||
Nagai E, Ueyama T, Yao T, Tsuneyoshi M. Hepatoid adenocarcinoma of the stomach. A clinicopathologic and immunohistochemical analysis. Cancer. 1993;72(6):1827–1835. | ||
Wang Z, Gerstein M, Snyder M. RNA-Seq: a revolutionary tool for transcriptomics. Nat Rev Genet. 2009;10(1):57–63. | ||
Mortazavi A, Williams BA, Mccue K, Schaeffer L, Wold B. Mapping and quantifying mammalian transcriptomes by RNA-seq. Nat Methods. 2008;5(7):621–628. | ||
Suzuki A, Koide N, Kitazawa M, Mochizuka A, Ota H, Miyagawa S. Gastric composite tumor of alpha fetoprotein-producing carcinoma/hepatoid adenocarcinoma and endocrine carcinoma with reference to cellular phenotypes. Patholog Res Int. 2012;2012(5):1–8. | ||
Gao YB, Zhang DF, Jin XL, Xiao JC. Preliminary study on the clinical and pathological relevance of gastric hepatoid adenocarcinoma. J Dig Dis. 2007;8(1):23–28. | ||
Yang J, Wang R, Zhang W, Zhuang W, Wang M, Tang C. Clinicopathological and prognostic characteristics of hepatoid adenocarcinoma of the stomach. Gastroenterol Res Pract. 2014;2014(6):1–5. | ||
Liu X, Sheng W, Wang Y. An analysis of clinicopathological features and prognosis by comparing hepatoid adenocarcinoma of the stomach with AFP-producing gastric cancer. J Surg Oncol. 2012;106(3):299–303. | ||
Qu BG, Bi WM, Qu BT, et al. PRISMA-Compliant article: clinical characteristics and factors influencing prognosis of patients with hepatoid adenocarcinoma of the stomach in China. Medicine. 2016;95(15):e3399. | ||
Nakanishi H, Yasui K, Ikehara Y, et al. Establishment and characterization of three novel human gastric cancer cell lines with differentiated intestinal phenotype derived from liver metastasis. Clin Exp Metastasis. 2005;22(2):137–147. | ||
Elsässer HP, Lehr U, Agricola B, Kern HF. Structural analysis of a new highly metastatic cell line PaTu 8902 from a primary human pancreatic adenocarcinoma. Virchows Arch B Cell Pathol Incl Mol Pathol. 1993;64(4):201–207. | ||
Metzgeroth G, Ströbel P, Baumbusch T, Reiter A, Hastka J. Hepatoid adenocarcinoma: review of the literature illustrated by a rare case originating in the peritoneal cavity. Onkologie. 2010;33(5):263–269. | ||
Hishinuma M, Ohashi KI, Yamauchi N, et al. Hepatocellular oncofetal protein, glypican 3 is a sensitive marker for alpha-fetoprotein-producing gastric carcinoma. Histopathology. 2006;49(5):479–486. | ||
Liu X, Cheng Y, Sheng W, et al. Analysis of clinicopathologic features and prognostic factors in hepatoid adenocarcinoma of the stomach. Am J Surg Pathol. 2010;34(10):1465–1471. | ||
Maitra A, Murakata LA, Albores-Saavedra J. Immunoreactivity for hepatocyte paraffin 1 antibody in hepatoid adenocarcinomas of the gastrointestinal tract. Am J Clin Pathol. 2001;115(5):689–694. | ||
Akiyama S, Tamura G, Endoh Y, et al. Histogenesis of hepatoid adenocarcinoma of the stomach: molecular evidence of identical origin with coexistent tubular adenocarcinoma. Int J Cancer. 2003;106(4):510–515. | ||
Scholzen T, Gerdes J. The Ki-67 protein: from the known and the unknown. J Cell Physiol. 2000;182(3):311–322. | ||
Ko GH, Go SI, Lee WS, et al. Prognostic impact of Ki-67 in patients with gastric cancer: the importance of depth of invasion and histologic differentiation. Medicine. 2017;96(25):e7181. | ||
Carmeliet P, Jain RK. Angiogenesis in cancer and other diseases. Nature. 2000;407(6801):249–257. | ||
Rao N, Lee YF, Ge R. Novel endogenous angiogenesis inhibitors and their therapeutic potential. Acta Pharmacol Sin. 2015;36(10):1177–1190. | ||
Park JG, Oie HK, Sugarbaker PH, et al. Characteristics of cell lines established from human colorectal carcinoma. Cancer Res. 1987;47(24 Pt 1):6710–6718. | ||
Stern HM, Zon LI. Cancer genetics and drug discovery in the zebrafish. Nat Rev Cancer. 2003;3(7):533–539. | ||
Goessling W, North TE, Zon LI. New waves of discovery: modeling cancer in zebrafish. J Clin Oncol. 2007;25(17):2473–2479. | ||
Chang WJ, du Y, Zhao X, Ma LY, Cao GW. Inflammation-related factors predicting prognosis of gastric cancer. World J Gastroenterol. 2014;20(16):4586–4596. | ||
Peters AA, Milevskiy MJ, Lee WC, et al. The calcium pump plasma membrane Ca(2+)-ATPase 2 (PMCA2) regulates breast cancer cell proliferation and sensitivity to doxorubicin. Sci Rep. 2016;6:25505. | ||
Aung CS, Ye W, Plowman G, Peters AA, Monteith GR, Roberts-Thomson SJ. Plasma membrane calcium ATPase 4 and the remodeling of calcium homeostasis in human colon cancer cells. Carcinogenesis. 2009;30(11):1962–1969. | ||
Delgado-Coello B, Santiago-García J, Zarain-Herzberg A, Mas-Oliva J. Plasma membrane Ca2+-ATPase mRNA expression in murine hepatocarcinoma and regenerating liver cells. Mol Cell Biochem. 2003;247(1–2):177–184. | ||
Yao D, Ding S, Burchell B, Wolf CR, Friedberg T. Detoxication of vinca alkaloids by human P450 CYP3A4-mediated metabolism: implications for the development of drug resistance. J Pharmacol Exp Ther. 2000;294(1):387–395. |
Supplementary materials
  | Figure S1 Chromosomal analysis of the GC-030-35 cell line. Note: The hypo-pentaploid (A) and hypo-triploid (B) phenomenon in the GC-030-35 cell line. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.