Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 11
Efficacy of a repair cream containing Rhealba oat plantlets extracts, L-ALA-L-GLU dipeptide, and hyaluronic acid in wound-healing following dermatological acts: a meta-analysis of >2,000 patients in eight countries corroborated by a dermatopediatric clinical case
Authors Saint Aroman M , Guillot P, Dahan S, Coustou D , Mortazawi K, Zourabichvili O , Aardewijn T
Received 19 June 2018
Accepted for publication 27 September 2018
Published 13 November 2018 Volume 2018:11 Pages 579—589
DOI https://doi.org/10.2147/CCID.S177614
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jeffrey Weinberg
M Saint Aroman,1 P Guillot,2 S Dahan,3 D Coustou,3 K Mortazawi,4 O Zourabichvili,5 T Aardewijn1
1Pierre Fabre Dermo-Cosmétique, A-DERMA, Lavaur, France; 2Wallerstein Medico-Surgical Center, Dermatology Department, Arès, France; 3Saint-Jean du Languedoc Clinic, Dermatology Department, Toulouse, France; 4Clinic for Pediatric Surgery, Hospital Karlsruhe GmbH, Karlsruhe, Germany; 5Quanta Medical, Rueil-Malmaison, France
Background: The frequency of dermatological acts is increasing. These procedures often cause injuries and traumatic alterations in specific skin layers, slowing down wound healing.
Patients and methods: An open observational study lasting 1 month was conducted on 2,363 patients who had undergone various dermatological procedures. This study was conducted in eight European countries and an Asian country during which the tolerance and efficacy of a cosmetic cream based on Rhealba oat plantlets’ extract, l-ALA–l-GLU dipeptide, and hyaluronic acid were assessed on patients’ wounds.
Results: Efficacy was observed 5′ after the first application, which leads to an immediate relief, confirmed by the overall efficacy judged by the doctors as good or very good in 96.8% of the cases. In Germany, the efficacy of the same cream was assessed on children suffering from first- or second-degree burns. In this dermatopediatric case, the aim was to support the regeneration process and prevent scarring by using a topical cream rather than a silicon bandage or corticosteroids. A positive effect on skin regeneration and prevention of scaring could already be observed after 4 weeks of application without any undesired complication.
Conclusion: This clinical focus complements the previous meta-analysis by demonstrating that the tested cream containing Rhealba oat plantlets’ extracts, l-ALA–l-GLU dipeptide, and hyaluronic acid could also be used with a great efficacy in children after thermal burns to prevent scaring.
Keywords: observational study, dermatology, meta-analysis, Rhealba oat plantlets’ extract, l-ALA–l-GLU dipeptide
Introduction
The skin has numerous lesions accessible to dermatological procedures, but they have in common to temporarily induce signs and symptoms of skin aggression.
The efficacy and tolerance of the products used to correct these signs and symptoms can be studied by country; however, using aggregated data can provide an explanation to this efficacy.
A wide range of dermatological procedures
Nowadays, a wide range of laser equipment is available to treat a large number of dermatological conditions.
Among dermatological lasers, the surgical ones are the most commonly used especially the CO2 laser in the case of skin or mucosal disease. In contrast, the Er:YAG laser is particularly useful in treating superficial cutaneous lesions and skin refreshing.1
One of the greatest breakthroughs with lasers may have been to treat wrinkles, textures, and scars using fractional lasers. These lasers can be used to treat moderate-to-severe rhytides, moderate dyschromia, eyelid tightening, and so on.
The demand for skin resurfacing and rejuvenating procedures to improve the appearance of scarred, sun damaged, or aging skin has progressively increased during the last decade. Several technological and procedural skin-resurfacing developments are being integrated into clinical practices today.2
Pulsed and scanned CO2 laser systems, along with Er:YAG lasers, were developed to reduce thermal damages during skin procedures. Currently, the YSGG laser has intensified this ablative laser evolution with technologies that allow a balance of depth and thermal impact not achievable with the other ablative wavelengths.
Induced traumatic alterations: be aware of the consequences!
The frequency of dermatological acts is steadily increasing, accompanying a growing demand from patients. These acts have in common to induce troublesome clinical manifestations of the skin.
Cutaneous wound healing is a dynamic process involving complex series of cellular, molecular, and biochemical events. Growth factors synthesized by cells present in the wound form a dense communication network, which coordinates cell proliferation, protein synthesis, and cell migration. Successful healing and reorganization of the scar depend on molecular events that restore the initial functional structures of the tissue.
Rhealba oat plantlets’ extract
The tested product contained Rhealba oat plantlets’ extract and oil associated with L-ALA–L-GLU dipeptide and hyaluronic acid. The oat aerial part essentially contains flavonoids and saponins, two molecules of interest in preventing and treating several diseases.
Certain flavonoids have beneficial repairing and healing properties for fragile skins presenting an imbalanced or damaged skin barrier. In vitro studies showed that they inhibit enzymes, such as phospholipase A2, which is involved in the cellular inflammatory response but also the release of proinflammatory cytokines such as IL-1β and TNF-α.3–5
Saponins are known to reduce transepidermal water loss and skin pH, increase stratum corneum hydration, and inhibit IL-2 and IL-13 productions.6,7
In some fragile skin inflammatory pathologies, we observe increased eicosanoid and arachidonic acid levels, simultaneously with an increased phospholipase A2 (PLA2) activity. PLA2 triggers the release of arachidonic acid in the cell membrane, and cyclooxygenase 2 (COX-2) catalyzes the formation of eicosanoids from arachidonic acid.8–11
The enzymatic action of PLA2 and COX-2 was measured in tubo in the presence or absence of plantlets’ extract. Rhealba oat plantlets’ extract inhibited the enzymatic action of COX-2 in a significant dose-dependent manner. Therefore, Rhealba oat plantlets’ extract has a regulatory effect on the cyclooxygenase pathway, and its anti-inflammatory action manifests itself through direct inhibition of COX-2 enzymatic properties.12,13
Concerning hyaluronic acid, it is a naturally occurring endogenous glycosaminoglycan, which plays an important role in tissue development and wound healing by promoting early inflammation. Recently, its key role was also demonstrated in each phase of corneal epithelial wound healing, including cell proliferation, anti-inflammation, and wound repair.14,15 The hyaluronic acid used in this study was of plant origin, obtained by fermentation, and had molecular weight between 1 and 1.4 MDa.
Clinical studies had been conducted on altered skin to determine the tolerance and efficacy of this topical cream based on Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid.
The tolerance of the tested cream, applied during 15 days, was also evaluated during an open-label study, in which a good cosmetic acceptability was demonstrated in 88% of patients.16,17
Time to re-epidermization in Er:YAG laser-ablated skin was evaluated in an intra-individual randomized controlled study carried out in 22 healthy volunteers. Wound surface area measurement showed that complete wound closure occurred significantly sooner with the test product (12.5±3.1 days) compared to the active comparator (17.6±3.5 days; P<0.0001).18 Similarly, complete healing for at least 50% of subjects occurred earlier with the test product (12 days) than with the active comparator (15 days).
Clinical studies
Patients and methods
The prospective, noninterventional studies considered in this data meta-analysis were conducted from October 2015 to September 2017 in eight countries by dermatologists on 2,363 patients. Questionnaires and data collection methods were strictly similar between different countries.
Patients were seen in consultation following the therapeutic procedure and evaluated on objective signs (erythema, edema, oozing, and presence of crusts) and symptoms’ intensity (pain, itching, burning, tingling, tightness, discomfort, and pruritus). These symptoms were re-evaluated 5 minutes after the repair cream application.
Patients completed a self-questionnaire at the end of the treatment evaluating the changes in the symptomatology and their satisfaction with the treatment. Concomitantly, they were checked by a dermatologist to assess the treatment’s efficacy and safety.
This study was exempt from review by an ethics committee, as it is an observational consumer satisfaction survey of an existing cosmetic product as described in the Decree 2017/884, which means that our study is out of the scope of the 2016/800 of Jardé Act Ordinance. This was conducted in accordance with the regulation 536/2014 of the European parliament, and all participants provided written informed consent.
Studied population
Patients were recruited by dermatologists in the following eight countries: France, DOM-TOM, Portugal, Romania, Korea, Italy, Taiwan, and Spain.
They were included following dermatological acts such as peels, fractional ablative lasers and/or continuous (CO2), pigment lasers, laser tattoo removal, intense pulsed light, cryotherapy, injections, and minor surgery. They were treated with a topical cream based on Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid.
The analyzed population consisted of patients who completed the inclusion sheet, provided information on their use of the tested topical cream, and were evaluated at the final medical consultation.
The exclusion criteria of patients were as follows: pregnant or breastfeeding woman, patient with a known sensitization to at least one of the product components, presence of another condition that can interfere in the assessment of clinical signs, and participation during the previous month to an interventional clinical study.
Statistical analysis
The statistical analysis was performed using the SAS® 9.4 software (SAS Institute Inc., Cary, NC, USA). The efficacy analysis was performed on the Full Analysis Set (FAS) population, that is, to say all patients enrolled. As all patients followed the protocol, the safety population was identical to the recruited population.
Quantitative criteria, baseline, and final values were described using standard error on the mean, quartiles, minimum and maximum values, and 95% CI of the mean. Concerning qualitative criteria, the frequency and percentage of each modality were presented.
Gaussian quantitative variables were analyzed using the parametric ANCOVA.
When the normality assumption was not verified, mean comparisons were performed using a Student’s t-test or a nonparametric test (Wilcoxon rank).
Percentage comparisons were made using the χ2 test or Fisher’s exact test when the theoretical numbers were less than 5.
Mean comparisons subpopulations (more than two) were performed by ANOVA or by Kruskal–Wallis test.
All tests were two sided and used a significance level of 0.05.
A receiver operating characteristic curve was used to check the performance of the combination of the variable and was followed by a multivariate analysis stepwise logistic regression.
Results
At inclusion
Results concern the 2,363 patients included. The mean age of the study populations was 47.3±18.5 years (Table 1).
  | Table 1 Description of the population |
Gender distribution was not homogeneous across countries (P<0.001) and ranged from 2.1 times more women in Italy to 7.2 times more in Taiwan (Figure 1). The aggregated data showed a female population three times larger.
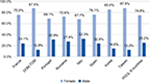  | Figure 1 Gender ratio in each country. Note: POOL 8 countries indicates data compilation of all other countries. Abbreviations: DOM-TOM, French Overseas Department and Territories. |
The most performed dermatological procedures per patient prior to the study were (Table 2) 26.1% for cryotherapy, 15.6% for laser (fractional, continuous, vascular, or laser tattoo removal), and 12.9% for peeling/microabrasion.
  | Table 2 Main dermatological procedures per patient |
Cryotherapy frequency was significantly different regarding countries (Figure 2): more common in France (41.7%) and less common in Korea (1.9%).
  | Figure 2 Cryotherapy prevalence. Abbreviation: DOM-TOM, French Overseas Department and Territories. |
After the procedure, patients had erythema in 93.6% of cases (n=2,165), moderate to severe in 67.5% of cases, and edema in 72.4% of cases (n=1,622). They also had reduced oozing in 22.0% of cases (n=486) or crusts in 25.4% of cases (n=559) (Figure 3).
  | Figure 3 Clinical signs at Day 0. |
Among experienced symptoms after the dermatological procedure, burns, tingling, and pain were the most frequent manifestations, present in 79.3% (n=1,790), 73.3% (n=1,625), and 71.3% (n=1,597). Tightness/discomfort was present in 68.1% of cases (n=1,513) and pruritus was present more rarely in 30.7% of cases (n=669).
Treatment
The mean prescription time of the repair cream was 18.9±14.1 days and differed significantly according to the treated area (P<0.0001): longer for the body (23.0 days) and shorter for the face (17.7 days). The cream was prescribed as a monotherapy in most cases (60.3%) and associated with other products in 39.7% of cases.
Early efficacy results
Symptoms were evaluated on a scale from 0 (none) to 3 (very intense) and were significantly improved (P<0.0001) 5 minutes after having applied the repair cream.
Patients experienced fewer burns, tingling, and pain, 56.5% (n=1,153), 45.7% (n=908), and 49.2% (n=991) of cases, respectively. This was also the case for tightness and pruritus since they were still present only in 42.5% (n=845) and 15.1% (n=289) of patients 5 minutes after having applied the repair cream (early relief – Figure 4).
  | Figure 4 Early relief (mean score). |
Late efficacy results
Late efficacy was observed, on average, 6.6±6.2 days after the first application of the cream. The average duration of follow-up was 28.6±17.6 days. Global efficacy was then judged by the dermatologist and evaluated as very effective or effective in 96.8% of the cases (Figure 5).
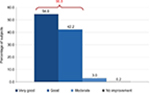  | Figure 5 Efficacy evaluation (medical). |
The medical evaluation significantly differs (P<0.0001) from one country to another. The evaluations effective and very effective range from 82.3% (Korea) to 98.9% (Spain). Medical evaluation was similar to subjects’ appreciation who considered the efficacy as very effective or effective in 98.6% of the cases (Figure 8). Very few of them considered the result as unsatisfying.
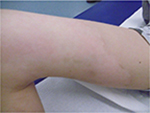  | Figure 8 Results after 2 months of tested topical cream. |
Wound healing duration following a dermatological procedure was appreciated by dermatologists as significantly shortened for 1,743 patients (91.7%), slightly shortened for 148 patients (7.8%), and not shortened for 10 patients (0.5%).
Healing quality was assessed as significantly improved for 1,754 patients (93.0%), slightly improved for 128 patients (6.8%), and not improved for four patients (0.2%).
Safety results
Tolerance appreciated by the physician was rated good or very good for 99.0% of patients. This assessment does not differ according to the treatment regimen but differs significantly between countries (P<0.0001) with the best tolerance in Romania (100%) and the poorest in Korea (91.6%). The average tolerance is more frequently encountered (P<0.0001) after multiple acts (2.2%) or laser (1.9%). Bad tolerance has never been reported.
Factors related to clinical success
Clinical success was defined as the association of “very effective” and “effective” in the doctor’s evaluation and the patient’s self-evaluation, also associated with the absence of adverse events and reduction in at least one symptom.
The very strict definition of clinical success concerns 72.4% of patients. Results of the multi-varied analysis showed that clinical success was significantly associated with:
- the country: the product is twice as effective in France (OR 2.3, P<0.01) and Italy (OR 2.3, P<0.05) and eight times more in Romania (OR 8.3, P<0.001) compared to Taiwan,
- the scars: the fewer scars there are, the more clinical success there is (OR 7.0, P<0.01),
- the cream duration of use (at 2 weeks, OR 1.2, P<0.05),
- having a mild pruritus (OR 1.5, P<0.05), and
- absence of cryotherapy (OR 1.7, P<0.05).
Clinical case
Treatment of thermal injuries in children with a cream containing Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid
Thermal injuries, especially thermal burns, are one of the most common injuries in childhood. Unlike adults, even unboiling fluid such as hot coffee and tea can sometimes cause severe damages to the skin.
Children’s skin has a tremendous potential for regeneration, and factors that have a negative influence on healing process such as impaired circulation, type 2 diabetes, nicotine, and alcohol can usually be taken out of consideration.
We have widely standardized the treatment of thermal injuries in our department and provide an outpatient follow-up service. Child-friendly treatments, avoiding unnecessary and usually painful dressing changes, are important aspects to consider.
Different treatment strategies and products exist, but they are often misapplied. For example, silicon bandages are often not applied long enough in children due to subjective discomforts, which lead to limited therapeutic effects.
Besides, parents usually have a negative opinion on products containing silicon or prednisone.
Since about a year, we have applied a cream containing Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid in the treatment of thermal burns, especially those of the first and the second degrees, in order to support the regeneration process and prevent scaring. The product should be applied during 6–9 months. As there are not enough data due to the short period of observation, the following results are only based on primary clinical experience. The cohort is made of 25 patients aged between 2 and 14 years. The treatments were concluded in three of them, and 14 of them stopped the treatment before its end.
Thermal burns of the first and second degrees are initially treated with hydrocolloid dressing or greased gauze. In case of a second degree thermal burn, a wound debridement followed by a wound covering with an alloplastic skin substitute is usually established under general anesthesia.
After epithelization completion, the cream containing Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid was applied thinly with a gentle massage twice a day. The application is suitable for all body regions including facial area. Clinical follow-ups took place monthly.
A positive effect through a catalyzed epithelization, especially a redness reduction, could already be observed after 4 weeks.
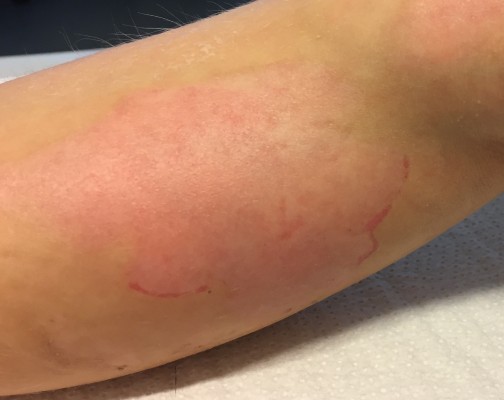
No undesired complications such as allergic reactions, burning, and increased redness occurred. The application was considered comfortable by all patients. Figures 6 and 7 present the initial state of a thermal burn of the second degree, and Figures 8 and 9 show the results after having applied the cream containing Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid for 2 months.
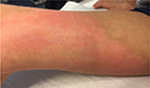  | Figure 6 Initial state of a second degree thermal burn. |
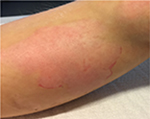  | Figure 7 Initial state of a second degree thermal burn. |
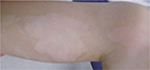  | Figure 9 Results after 2 months of tested topical cream. |
The redness had been largely reduced, and the skin was mostly soft and nonirritated except the area of hypopigmentation.
Another example is presented in Figures 10 and 11. This case was caused by hot steam. The results after having applied the cream containing Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid for 4 weeks are shown in Figures 12 and 13. A reverse from skin deepening and a recovery of the skin structure could be observed. The redness was also considerably reduced.
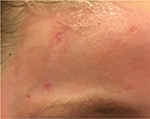  | Figure 10 Initial state of a second degree thermal burn caused by hot steam. |
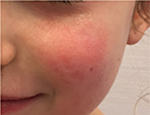  | Figure 11 Initial state of a second degree thermal burn caused by hot steam. |
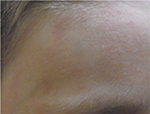  | Figure 12 Recovery after 4 weeks of tested topical cream. |
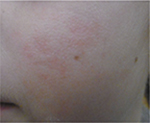  | Figure 13 Recovery after 4 weeks of tested topical cream. |
The last case was caused by boiling water and was also treated initially by an alloplastic skin substitute. Figure 14 shows the result after removing the alloplastic skin substitute. Figures 15 and 16 show the results after the application of the cream containing Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid during 4 weeks and 2 months, respectively. A skin flattening and a significant reduction in the wound area could be observed especially at the edges.
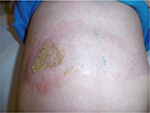  | Figure 14 Burn aspect at removal of the alloplastic skin substitute. |
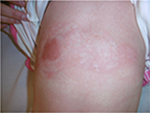  | Figure 15 Recovery after 4 weeks of tested topical cream. |
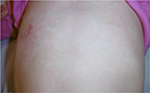  | Figure 16 Recovery after 2 months of tested topical cream. |
In conclusion, the application of a cream containing Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid in children is recommended after thermal burns. It showed no side effects, and a considerable effect could already be observed after 4–8 weeks of application. A positive effect on skin regeneration and a successful prevention of scarring could be observed. A recovery of the skin structure took place, and skin redness was significantly reduced. No exuberant scarring occurred in those patients.
Discussion
The meta-analysis examined the effects of a cosmetic cream based on Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid through data from eight different countries and 2,363 patients who had previously undergone a dermatological procedure.
This meta-analysis was made possible thanks to a standardized questionnaire of data collection adapted to each participating countries. Thanks to the important number of patients analyzed, we can reduce the variability of the parameters that can be encountered in more limited populations without erasing the intrinsic specificities of the participating countries.19 It highlights, robustly and homogeneously, a significant and early efficacy of the tested product on the clinical symptoms induced by the dermatological acts. It also confirms overall efficacy assessed after a mean follow-up of 27 days.
Meta-analyses give us the opportunity to use particular models of statistical analysis because of the large size of available data; in our case, we can use stepwise regression model. Stepwise regression is a method of fitting regression models in which the choice of predictive variables is carried out by an automatic procedure.20 In each step, a variable is considered for addition to or subtraction from the set of explanatory variables based on some prespecified criterion.
Skincare products are often recommended following esthetic resurfacing procedures such as laser, pulsed light, peeling, injection, dermatological surgery, cryotherapy, electro-coagulation, and curettage. Indeed, these techniques temporarily disrupt the skin’s barrier function, leaving it fragile, permeable, often irritated, and sensitive to topical products. The results of this study suggest that the tested topical cream based on Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid can be safely used to speed-up skin healing following resurfacing procedures.
Conclusion
These recommendations can be extended to thermal burns and children. Indeed, the clinical case clearly demonstrates that the cream based on Rhealba oat plantlets’ extract, L-ALA–L-GLU dipeptide, and hyaluronic acid can be used once the epithelization is complete to avoid scaring and support the regeneration process.
Abbreviations
Alex, alexandrite; ANCOVA, analysis of covariance; DOM-TOM, French Overseas Department and Territories; Er:YAG, erbium-doped yttrium aluminum garnet; KTP, potassium-titanyl-phosphate; Nd:YAG neodymium-doped yttrium aluminum garnet; YSGG, yttrium scandium gallium garnet.
Acknowledgments
The authors would like to thank the following investigators involved in the trial: Pierre Fabre Dermo-Cosmétique R&D department and all the investigators involved in the numerous clinical studies. Pierre Fabre Dermo-Cosmétique set up the observational studies and paid for the statistical studies as well as the meta-analysis. The trial was designed and carried out by A-DERMA, Pierre Fabre Dermo-Cosmétique, Lavaur, France. The data management and statistical services were carried out by Quanta Medical SA, Rueil-Malmaison, France.
Disclosure
MSA is the medical director of A-DERMA Dermatological Laboratories and an employee of Pierre Fabre Dermo-Cosmétique, which sponsored the clinical trial. TA is intern at Pierre Fabre Dermo-Cosmétique but is affiliated to Université Toulouse III. OZ is the CEO of Quanta Medical, recruited by Pierre Fabre Dermo-Cosmétique for the statistical services and the meta-analysis. All other authors report no conflicts of interest in this work.
References
Gianfaldoni S, Tchernev G, Wollina U, et al. An Overview of Laser in Dermatology: The Past, the Present and … the Future (?). Open Access Maced J Med Sci. 2017;5(4):526–530. | ||
Loesch MM, Somani AK, Kingsley MM, Travers JB, Spandau DF. Skin resurfacing procedures: new and emerging options. Clin Cosmet Investig Dermatol. 2014;7:231–241. | ||
Lodhi S, Singhai AK. Wound healing effect of flavonoid rich fraction and luteolin isolated from Martynia annua Linn. on streptozotocin induced diabetic rats. Asian Pac J Trop Med. 2013;6(4):253–259. | ||
Middleton E Jr. Effect of plant flavonoids on immune and inflammatory cell function. Adv Exp Med Biol. 1998;439:175–182. | ||
Jeon IH, Kim HS, Kang HJ, et al. Anti-inflammatory and antipruritic effects of luteolin from Perilla (P. frutescens L.) leaves. Molecules. 2014;19(6):6941–6951. | ||
Man MQ, Hupe M, Sun R, Man G, Mauro TM, Elias PM. Topical apigenin alleviates cutaneous inflammation in murine models. Evid Based Complement Alternat Med. 2012;2012:912028. | ||
Stalder JF, Tennstedt D, Deleuran M, et al. Fragility of epidermis and its consequence in dermatology. J Eur Acad Dermatol Venereol. 2014;28(Suppl 4):1–18. | ||
Fogh K, Herlin T, Kragballe K. Eicosanoids in skin of patients with atopic dermatitis: prostaglandin E2 and leukotriene B4 are present in biologically active concentrations. J Allergy Clin Immunol. 1989;83(2 Pt 1):450–455. | ||
Ikai K. Psoriasis and the arachidonic acid cascade. J Dermatol Sci. 1999;21(3):135–146. | ||
Greaves MW, Camp RD. Prostaglandins, leukotrienes, phospholipase, platelet activating factor, and cytokines: an integrated approach to inflammation of human skin. Arch Dermatol Res. 1988;280(Suppl 1): S33–S41. | ||
Lin LL, Lin AY, Knopf JL. Cytosolic phospholipase A2 is coupled to hormonally regulated release of arachidonic acid. Proc Natl Acad Sci U S A. 1992;89(13):6147–6151. | ||
Katsumata M, Gupta C, Goldman AS. A rapid assay for activity of phospholipase A2 using radioactive substrate. Anal Biochem. 1986;154(2):676–681. | ||
Glaser K, Sung ML, O’Neill K, et al. Etodolac selectively inhibits human prostaglandin G/H synthase 2 (PGHS-2) versus human PGHS-1. Eur J Pharmacol. 1995;281(1):107–111. | ||
Petrey AC, de la Motte CA. Hyaluronan, a crucial regulator of inflammation. Front Immunol. 2014;5:101. | ||
Chen WY, Abatangelo G. Functions of hyaluronan in wound repair. Wound Repair Regen. 1999;7(2):79–89. | ||
Rossi AB, Thouvenin MD, Ribet V. Tolerance of a dermo-cosmetic cream after chemical peels, LASERs and light’s procedures. Poster. AAD Meeting. Orlando. 2017;3–7. | ||
Thouvenin MD, Ribet V, Saint Aroman M, Rossi B. Tolerance of a dermo-cosmetic cream containing cicahyalumide® after chemical peels, LASERs and light’s procedures. EADV Meeting Vienne. 2016;2. | ||
Fabbrocini G, Rossi AB, Thouvenin MD, et al. Fragility of epidermis: acne and post-procedure lesional skin. J Eur Acad Dermatol Venereol. 2017;31(Suppl 6):3–18. | ||
Rogozińska E, Marlin N, Thangaratinam S, Khan KS, Zamora J. Meta-analysis using individual participant data from randomised trials: opportunities and limitations created by access to raw data. Evid Based Med. 2017;22(5):157–162. | ||
Draper N, Smith H. Applied Regression Analysis. 2nd ed, New York: John Wiley & Sons, Inc; 1981. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
