Back to Journals » Drug Design, Development and Therapy » Volume 13
Efficacy and safety of sodium cantharidinate and vitamin B6 injection for the treatment of digestive system neoplasms: a meta-analysis of randomized controlled trials
Received 12 October 2018
Accepted for publication 6 December 2018
Published 28 December 2018 Volume 2019:13 Pages 183—203
DOI https://doi.org/10.2147/DDDT.S190674
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Tuo Deng
Meirong Liu,1 Chunhong Xu,2 Yingying Sun3
1Department of Oncology, Liaocheng People’s Hospital, Liaocheng Clinical School of Taishan Medical University, Liaocheng 252000, Shandong Province, China; 2Department of Gastroenterology, Liaocheng People’s Hospital, Liaocheng Clinical School of Taishan Medical University, Liaocheng 252000, Shandong Province, China; 3Department of Radiotherapy, Liaocheng People’s Hospital, Liaocheng Clinical School of Taishan Medical University, Liaocheng 252000, Shandong Province, China
Objective: To systematically evaluate the efficacy and safety of sodium cantharidinate and vitamin B6 (SC/B6) combined with conventional medical treatment (CMT) for the treatment of patients with advanced digestive system neoplasms (DSNs).
Methods: The Cochrane Library, Embase, PubMed, Web of Science, Chinese Scientific Journal Database (VIP), China National Knowledge Infrastructure, and Wanfang databases were searched for clinical trials using SC/B6 for DSNs. Outcome measures, including therapeutic efficacy, quality of life (QoL), and adverse events, were extracted and systematically evaluated.
Results: Data from 24 trials including 1,825 advanced DSN patients were included. Compared with CMT alone, its combination with SC/B6 significantly improved the patients’ overall response rate (OR =2.25, 95% CI =1.83–2.76, P<0.00001), disease control rate (OR =2.41, 95% CI =1.85–3.15, P<0.00001), and QoL improvement rate (OR =2.75, 95% CI =2.13–3.55, P<0.00001). Moreover, adverse events caused by chemotherapy, including leukopenia, nausea and vomiting, gastrointestinal side effects, hepatotoxicity, diarrhea, transaminase disorder, myelosuppression, anorexia, and anemia, were significantly alleviated (P<0.05) when SC/B6 was applied to DSN patients. Nephrotoxicity, thrombocytopenia, hand-foot syndrome, and oral mucositis were not significantly alleviated in patients receiving combination therapy (P>0.05).
Conclusion: The combination of SC/B6 and CMT is more effective in treating DSNs than CMT alone. This combination alleviates the adverse effects associated with chemotherapy and improves the QoL of DSN patients, and its application in the clinic is worth promoting.
Keywords: sodium cantharidinate and vitamin B6, conventional medical treatment, digestive system neoplasms, meta-analysis
Introduction
Digestive system neoplasms (DSNs) are the leading cause of cancer-related death worldwide, and cause 3,056,412 deaths in 2018, which accounts for 32% of all cancer deaths worldwide.1–3 This category comprises colorectal cancer, gastric cancer, liver cancer, esophageal cancer, and pancreatic cancer, which are the fourth, sixth, seventh, ninth, and fourteenth most common cancers, respectively.1 Despite improvements in diagnostic and therapeutic methods in the past decades,4 the prognosis of DSNs is still poor, because they are mostly diagnosed at advanced stages, which may be accompanied by extensive invasion and distant metastasis.4–6 Therefore, effective therapeutic approaches should be developed.
In recent years, traditional Chinese medicine has been more widely used as auxiliary treatment in tumor therapy and has shown promising therapeutic effects in many clinical studies.7–9 Sodium cantharidinate/vitamin B6 (SC/B6) is a combination of sodium cantharidinate (SC) and vitamin B6, and has the pharmacologic characteristics of both.7,8 SC is a derivative of cantharidin, which is extracted from the body of meloidae insects such as Mylabris phalerata pallas and Mylabris cichorii linnaeus.10 SC preserves the unique anticancer activity of cantharidin and has lower toxicity and fewer adverse effects.7,10 Its combination with vitamin B6 can even further lower the side effects.7 In recent years, SC has been used as a safe auxiliary antitumor drug for malignancies such as gastric cancer, liver cancer, and non-small-cell lung cancer.7–9,11 Tao et al12 indicated that SC induces HepG2 cells to undergo apoptosis through the LC3 autophagy pathway. Liang et al13 showed that SC can inhibit tumor growth by downregulating vascular endothelial growth factor expression and blocking tumor angiogenesis. In addition, SC can also have an anticancer effect by blocking progression through the cell cycle, inhibiting invasion/metastasis, and improving the immunity of cancer patients.14–18
Several clinical studies8,19–41 have revealed the prominent therapeutic effects of SC/B6 and conventional medical treatment (CMT, including chemotherapy, symptomatic, and supporting therapy) for advanced DSNs but clinical efficacy and safety have not been systematically evaluated. In this study, we performed a meta-analysis to evaluate the efficacy and safety of SC/B6 for DSN treatment, with a comparison between SC/B6 and CMT combined therapy and CMT alone, in order to provide scientific reference for the design of future clinical trials.
Materials and methods
Search strategy and selection criteria
Publications were searched across the Cochrane Library, Embase, Pubmed, Web of Science, Chinese Scientific Journal Database (VIP), China National Knowledge Infrastructure, and Wanfang databases, using the search terms “sodium cantharidinate” or “disodium cantharidinate” and “vitamin B6” combined with “gastric cancer” or “colorectal cancer” or “gastrointestinal cancer” or “liver cancer” or “esophageal cancer” or “pancreatic cancer” or “digestive system neoplasms” without restriction on the language. The retrieval was initiated in May 2018 and updated in August 2018.
All of the clinical trials brought into this analysis were randomized controlled trials with reference to advanced DSNs, in which patients in the experimental groups were treated by SC/B6 and CMT combined therapy, and patients in the control groups were treated by CMT alone.
Data extraction and quality assessment
Literature screening and data extraction were carried out by two independent investigators (Meirong Liu and Chunhong Xu) and verified by a third reviewer (Yingying Sun). All included studies were summarized as follows: first author name, year of publication, study location, Karnofsky Performance Score (KPS), number of cases, patient ages, study parameter type, treatment regimen and enrollment period, and administration route and dosage of SC/B6. The quality of the included trials was evaluated as described in the Cochrane Handbook.42
Outcome definition
Clinical responses, including therapeutic effects, quality of life (QoL), and adverse events, were analyzed. Therapeutic effects were evaluated by overall survival (OS) rate, complete response (CR) rate, partial response (PR) rate, stable disease (SD) rate, progressive disease (PD) rate, overall response rate (ORR, ORR = CR + PR), and disease control rate (DCR = CR + PR + SD). OS was defined as the length of time from the start of treatment to death from any cause; QoL was assessed using KPS scales and the European Organization for Research and Treatment of Cancer core quality-of-life questionnaire. The QoL improvement rate (QIR) was defined as the improvement in QoL after treatment. Adverse events, including leukopenia, nausea and vomiting, gastrointestinal side effects, hepatotoxicity, nephrotoxicity, diarrhea, thrombocytopenia, transaminase disorder, myelosuppression, hand-foot syndrome, oral mucositis, anorexia, and anemia, were also assessed.
Statistical analysis
Review Manager 5.3 (Nordic Cochran Centre, Copenhagen, Denmark) and Stata 13.0 (Stata Corp., College Station, TX, USA) were the main statistical analysis tools in this study. P<0.05 indicated statistically significant differences. Cochran’s Q test was used to determine heterogeneity among studies,43 and publication bias was analyzed by Begg’s and Egger’s regression asymmetry tests and presented by funnel plots.44 I2<50% or P>0.1 indicated study homogeneity. Therapeutic effects were mainly represented by HRs and ORs presented with 95% CIs. HRs were collected for survival data. If HRs can neither be collected directly nor calculated, survival curve plots were extracted by Engauge Digitizer software and then transformed by specialized form.45–47
Pooled analysis with publication bias determined that the trim-and-fill method would be applied to coordinate the estimates of unpublished studies, and the adjusted results were compared with the original pooled OR.48 Sensitivity analysis (subgroup analyses) was conducted to evaluate the impact of different cancer types, SC/B6 dosages, therapeutic regimens, sample sizes, and study types on clinical efficacy.
Results
Search results
A total of 974 articles were identified with the initial search, and 602 papers were excluded due to duplication. After title and abstract review, 269 articles were further excluded because they did not include clinical trials (n=209), were reviews or meta-analyses (n=6), were unrelated studies (n=43), or were case reports (n=11), leaving 103 studies as potentially relevant. After detailed assessment of full texts, articles without a control group (n=11), studies with inappropriate criteria in the experimental or control group (n=16), studies with insufficient data (n=5), and studies including patients with non-digestive system tumors (n=47) were excluded. Finally, data from 24 trials8,19–41 (gastric cancer, n=7; colorectal cancer, n=5; gastrointestinal cancer, n=3; liver cancer, n=7; esophageal cancer, n=1; and pancreatic cancer, n=1) including 1,825 advanced DSN patients were included in the present analysis (Figure 1).
  | Figure 1 Flow diagram of the selection process. |
Patient characteristics
All studies involved in this analysis were carried out in different hospitals in China. These trials include 1,825 patients with advanced DSNs; of these, 933 were treated by combined SC/B6 and CMT, and 892 were treated by CMT alone. Detailed information on the included trials and patients is presented in Tables 1 and 2.
Quality assessment
The evaluation of bias risk is presented in Figure 2. Twenty-two studies had low risk, and the other two articles did not have a clear description of the randomization process. None of the included trials provided a clear description of the performance and detection risks. Two studies were regarded as high-risk due to the absence of follow-up and seven trials were considered as unclear risk owing to selective reporting.
Therapeutic efficacy assessments
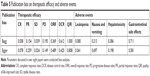
As shown in Figures 3 and 4, Table 3, and Figure S1, patients who underwent combined therapy had a significantly improved CR rate (OR =2.06, 95% CI =1.41–3.00, P=0.0002), PR rate (OR =1.85, 95% CI =1.50–2.29, P<0.00001), ORR (OR =2.25, 95% CI =1.83–2.76, P<0.00001), and DCR (OR =2.41, 95% CI =1.85–3.15, P<0.00001), and significantly decreased SD and PD rates (SD, OR =0.77, 95% CI =0.63–0.93, P=0.009; PD, OR =0.45, 95% CI =0.35–0.59, P<0.00001) compared to patients receiving CMT alone. The OS rates of patients who received combination treatment (HR =0.74, 95% CI =0.47–1.17, P=0.20) did not differ significantly from those in patients who received CMT alone.
QoL assessment
QoL evaluation demonstrated that SC/B6 and CMT combined therapy-treated DSN patients had improved QoL compared to those treated by CMT alone (Figure 5A, OR =2.75, 95% CI =2.13–3.55, P<0.00001).
Adverse events assessment
As shown in Table 4 and Figure S2, patients treated by SC/B6 and CMT combined therapy had lower incidences of leukopenia, nausea and vomiting, gastrointestinal side effects, hepatotoxicity, diarrhea, transaminase disorder, myelosuppression, anorexia, and anemia than those treated with CMT alone (leukopenia: OR =0.29, 95% CI =0.21–0.39, P<0.00001; nausea and vomiting: OR =0.30, 95% CI =0.22–0.40, P<0.00001; gastrointestinal side effects: OR =0.42, 95% CI =0.29–0.62, P<0.00001; hepatotoxicity: OR =0.49, 95% CI =0.30–0.78, P=0.003; diarrhea: OR =0.37, 95% CI =0.23–0.60, P<0.0001; transaminase disorder: OR =0.23, 95% CI =0.09–0.62, P=0.003; myelosuppression: OR =0.33, 95% CI =0.18–0.60, P=0.0003; anorexia: OR =0.37, 95% CI =0.20–0.68, P=0.001; anemia: OR =0.54, 95% CI =0.32–0.91, P=0.02). No significant difference was found in the occurrence of nephrotoxicity, thrombocytopenia, hand-foot syndrome, and oral mucositis (nephrotoxicity: OR =0.70, 95% CI =0.38–1.30, P=0.26; thrombocytopenia: OR =0.77, 95% CI =0.31–1.92, P=0.57; hand-foot syndrome: OR =0.75, 95% CI =0.40–1.40, P=0.36; oral mucositis: OR =0.45, 95% CI =0.13–1.62, P=0.22) between patients receiving combination treatment and those receiving CMT alone.
  | Table 4 Comparison of adverse events between the SC/B6 + CMT and SC/B6 group |
Publication bias
Publication bias of primary outcomes (CR, PR, SD, PD, ORR, DCR, QIR, and adverse events) was evaluated and presented by funnel plots. All plots were approximately symmetrical, indicating generally controlled publication bias (Figures 6 and S3).
We also assessed the publication bias by Begg’s and Egger’s regression asymmetry tests, and SD and leukopenia were found to have bias (SD, Egger: 0.024, Begg: 0.039; leukopenia, Egger: 0.041, Begg: 0.080; Table 5). To determine whether the bias affected the pooled risk, we conducted trim-and-fill analysis. The adjusted OR indicated the same trend as the primary analysis (SD, before: P=0.010, after: P<0.0001; leukopenia, before: P<0.0001, after: P<0.0001), reflecting the reliability of our primary conclusions, except those based on a small number of trials.
Sensitivity analysis
Subgroup analysis was performed for ORR and DCR heterogeneity assessment concerning cancer types, SC/B6 dosages, therapeutic regimens, sample sizes, and study types of involved trials. No significant difference was observed in the sample sizes, study types, or SC/B6 dosages (Table 6). SC/B6 combined with CMT was more effective in treating gastric cancer, colorectal cancer, and liver cancer. Moreover, SC/B6 combined with oxaliplatin and capecitabine (XELOX) or capecitabine regimens was more effective for DSN treatment.
Discussion
The chemotherapeutic regimens commonly used to treat DSNs cause serious side effects, such as myelosuppression, hepatotoxicity, and gastrointestinal side effects, which severely affect the QoL of DSN patients.7,9 Therefore, seeking a therapy that can improve treatment outcomes and decrease the adverse effects of chemotherapy is a major direction in the development of tumor treatment. Traditional Chinese medicine plays a unique role in improving host immunity and lowering the toxic effects of chemotherapy.7,9,49–52 In recent decades, SC/B6 has been clinically applied as an adjuvant therapy for malignancies and has been beneficial for advanced DSN patients in several trials.7–9,11 Despite the published reports on clinical trials using SC/B6, its therapeutic effects have not been systematically demonstrated. In the present study, we performed an extensive literature search followed by rigorous contrasting and combining data analysis for categorization to provide clear and systematic conclusions.
Our meta-analysis revealed that SC/B6 and CMT combined therapy for DSN patients achieved more beneficial effects than CMT alone. Combined therapy-treated patients exhibited markedly improved ORR and DCR (P<0.05 for all) and also significantly improved QoL. These results indicated that intravenous infusion of SC/B6 improved the curative effects of CMT for advanced DSNs.
Our analysis indicates that most of the adverse events caused by chemotherapy, including leukopenia, nausea and vomiting, gastrointestinal side effects, and hepatotoxicity, were alleviated with SC/B6 combination therapy (P<0.05). Therefore, SC/B6 is a safe auxiliary antitumor medicine for DSN and can effectively alleviate the adverse events associated with chemotherapy.
The analysis of therapeutic effects may be influenced by several factors. In our study, no difference was found between sample sizes, study types, and SC/B6 dosages. SC/B6 combined with CMT was more effective in treating gastric cancer, colorectal cancer, and liver cancer than it was in treating esophageal cancer and pancreatic cancer. Moreover, our subgroup analysis showed that SC/B6 combined with XELOX/capecitabine was more effective for DSN treatment. However, recent studies on the impact of these factors on the curative effect of SC/B6 adjuvant therapy remain insufficient, and further investigations should be performed.
There are some limitations in our analysis. First, the follow-up durations of the included studies were not long enough. Second, as a traditional medicine, SC/B6 was mainly applied in China, which may bring an unavoidable regional bias and subsequently influence the clinical application of SC/B6 worldwide. Furthermore, treatment/medical history is very important for evaluating the efficacy of SC/B6-mediated therapy. However, our data were extracted from published papers rather than from the original patient records; therefore, analytical bias may possibly exist. More original data would be valuable to achieve a higher reliability of statistical analysis on SC/B6 for DSN treatment.
In summary, this meta-analysis indicated that SC/B6 and CMT combined therapy was effective in treating advanced DSNs. Intravenous infusion of SC/B6 not only greatly improved the therapeutic effects of CMT but also effectively alleviated the toxicity and most of the side effects associated with chemotherapy. Therefore, SC/B6 has potential for development as a new adjuvant therapy for the treatment of DSN.
Author contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. | ||
Fidler MM, Bray F, Soerjomataram I. The global cancer burden and human development: a review. Scand J Public Health. 2018;46(1):27–36. | ||
Gong J, Lv L, Huo J. Roles of F-box proteins in human digestive system tumors (review). Int J Oncol. 2014;45(6):2199–2207. | ||
Li MX, Bi XY, Huang Z, et al. Prognostic role of phospho-STAT3 in patients with cancers of the digestive system: a systematic review and meta-analysis. PLoS One. 2015;10(5):e0127356. | ||
Dai C, Wang M, Lu J, et al. Prognostic and predictive values of PD-L1 expression in patients with digestive system cancer: a meta-analysis. Onco Targets Ther. 2017;10:3625–3634. | ||
Fu M, Zou C, Pan L, et al. Long noncoding RNAs in digestive system cancers: Functional roles, molecular mechanisms, and clinical implications (review). Oncol Rep. 2016;36(3):1207–1218. | ||
Wang B, Cui J. Treatment of mid-late stage NSCLC using sodium cantharidinate/vitamin B6/GP regimen in clinic. J Cancer Res Ther. 2014;10(Suppl 1):C79–C81. | ||
Shao H, Hong G, Luo X. Evaluation of sodium cantharidinate/vitamin B6 in the treatment of primary liver cancer. J Cancer Res Ther. 2014;10(Suppl 1):75–78. | ||
Shi Z, Song T, Wan Y, et al. A systematic review and meta-analysis of traditional insect Chinese medicines combined chemotherapy for non-surgical hepatocellular carcinoma therapy. Sci Rep. 2017;7(1):4355. | ||
Wen SQ, Chen Q, Hu M. Experimental study on the inhibitory effect of sodium cantharidinate on human hepatoma HepG2 cells. AfrJ Tradit Complement Altern Med. 2014;11(1):131–134. | ||
Zhang D, Wu J, Wang K, Duan X, Liu S, Zhang B. Which are the best Chinese herbal injections combined with XELOX regimen for gastric cancer?: A PRISMA-compliant network meta-analysis. Medicine. 2018;97(12):e0127. | ||
Tao R, Sun WY, Yu DH, et al. Sodium cantharidinate induces HepG2 cell apoptosis through LC3 autophagy pathway. Oncol Rep. 2017;38(2):1233–1239. | ||
Liang F, Wang MY, Huang WB, Li AJ. Effect of sodium cantharidinate on the angiogenesis of nude mice with human gastric cancer. Zhong Yao Cai. 2011;34(3):343–346. | ||
Zhao RL, Chen MJ, Zhao FM, Xu DQ, Zhang X, Zhou KF. Study on synergistic effect of sodium cantharidinate combined with chemotherapeutic drugs on hepatic carcinoma and its effective mechanism. Zhong Yao Cai. 2014;37(11):1938–1946. | ||
Tao R, Wang ZF, Qiu W, et al. Role of S100A3 in human hepatocellular carcinoma and the anticancer effect of sodium cantharidinate. Exp Ther Med. 2017;13(6):2812–2818. | ||
Zang GH, Li R, Zhou RS, et al. Effects of disodium cantharidinate on dendritic cells of patients with bladder carcinoma. Oncol Lett. 2018;15(2):2273–2277. | ||
Verma AK, Prasad SB. Changes in glutathione, oxidative stress and mitochondrial membrane potential in apoptosis involving the anticancer activity of cantharidin isolated from redheaded blister beetles, epicauta hirticornis. Anticancer Agents Med Chem. 2013;13(7):1096–1114. | ||
Li W, Xie L, Chen Z, et al. Cantharidin, a potent and selective PP2A inhibitor, induces an oxidative stress-independent growth inhibition of pancreatic cancer cells through G2/M cell-cycle arrest and apoptosis. Cancer Sci. 2010;101(5):1226–1233. | ||
Chen Y, Zhu WM, Zhou DX, Shan YF. Clinical study on disodium cantharidinate and vitamin B6 combined with raltitrexed and oxaliplatin in treatment of advanced colorectal cancer. Drugs Clinic. 2016;31(11):1784–1787. | ||
Fan LJ. Efficacy of sodium cantharidinate and vitamin B6 combined with 5-fluorouracil and calcium folinate in treatment of advanced colorectal cancer. Mod Med J China. 2009;11(10):64–65. | ||
Fan QL, Wang Y, Zhao KX, Kao J. Clinical observation on effects of sodium cantharidate vitamin B6 injection combined with S-1 treatment of advanced gastrointestinal tract tumor patients. Guangming J Chin Med. 2013;28(4):786–788. | ||
Fang XH, Xu J. Cantharidin sodium vitamin B6 injection as assistant therapy for treating liver cancer in 37 cases. China Pharm. 2016;25(1):25–27. | ||
Guan LY, Ma J, Yuan SF, Qu J. Disodium cantharidinate and vitamin B6 combined with tegafur, gimeracil and oteracil porassium for treating stage IV pancreatic cancer in 27 cases. Tradit Med. 2015;24(20):124–125. | ||
Jia JM, Li ZH. Efficacy of sodium cantharidinate and vitamin B6 combined with intravenous chemotherapy for advanced esophageal cancer. Med Inf. 2013;26(9):188–189. | ||
Li GP, Li XR, Wang JH. Clinical observation on sodium cantharidate vitamin B6 injection combined with postoperative chemotherapy in treatment of gastric cancer. Pract Prev Med. 2010;17(1):105–106. | ||
Liu GW, Ren WD. Cantharis acid sodium vitamin B6 injection combined with maintain capecitabine synchronization for the treatment of advanced colorectal cancer. World Latest Med Inf. 2017;17(26):12–14. | ||
Liu SH, Feng W, Zhu RX, Feng ZC. Comparative study of disodium cantharidinate and vitamin B6 injection combined with oxaliplatin and tegafur in the treatment of advanced gastric cancer. Pract Clin J Integr Tradit Chinese West Med. 2008;8(4):3–4. | ||
Mao WD. Clinical study of sodium cantharidinate and vitamin B6 combined with capecitabine in the treatment of advanced gastric cancer. J Clin Med. 2016;3(45):9034–9036. | ||
Shi XY, Zhang J, Meng W, Ma F, Li ZL, Zhao JF. Clinical observation of sodium cantharidinate and vitamin B6 injection combined with XELOX protocol in treatment of patients with advanced colorectal carcinoma. Med & Pharm J Chin PLA. 2017;29(6):36–39. | ||
Tian XL, Yang P. Clinical efficacy of treatment of liver cancer with combined sodium cantharidate vitamin B6 injection and TACE therapy. Chin J Clin Oncol Rehabil. 2006;13(4):351–353. | ||
Wang JH, Li GP, Wu JH. Application of sodium cantharidinate and vitamin B6 injection in patients with advanced colorectal cancer. J Mod Med Health. 2010;26(6):844–845. | ||
Wang YW, Yang RL. The effect observation of sodium cantharidate vitamin B6 combined with capecitabine in the treatment of advanced stomach and colorectal carcinoma. Med Innov China. 2017;14(36):23–26. | ||
Wei YF, Wu JS, Huo ZG, Jia ZY, Qi SS, Wang XX. Efficacy evaluation of sodium cantharidinate and vitamin B6 injection combined with TACE in the treatment of 48 patients with intermediate and advanced primary liver cancer. China Pharm. 2015;24(5):72–74. | ||
Wu ZM, Liu Q, Shi SM, Yu SY. Clinical study of ai yishu combined with FOLFIRI regimen in the treatment of recurrent gastric cancer. Med Inf. 2013;26(6):256. | ||
Xie ZX, Wang JS, Liu YB. Short-term efficacy and adverse effects of sodium cantharidinate and vitamin B6 injection combined with chemotherapy in the treatment of advanced gastric cancer. Hebei Med J. 2016;38(10):1554–1559. | ||
You ZY, Liu Y, Shu XH, Liu BN, Liu XF. Malignant ascites of gastrointestinal tumor treated with sodium cantharidinate and vitamin B6 injection and hyperthermic intraperitoneal chemotherapy. World J Integr Tradit West Med. 2015;10(9):1249–1255. | ||
Zeng L, Liu YL, Liu XT, Yang ZF. Clinical observation of sodium cantharidinate and vitamin B6 injection in the treatment of liver cancer. J Hebei Med Univ. 2009;30(6):596–597. | ||
Zhang MJ, Zuo CF. Clinical efficacy of sodium cantharidate vitamin B6 injection combined with chemotherapy in treatment of liver cancer. J Pract Oncol. 2011;26(1):50–52. | ||
Zhang W, Yang J, Zhong CS. Efficacy of sodium cantharidinate vitamin B6 combined with xeloda in the treatment of advanced gastric cancer. J Huaihai Med. 2012;30(5):446–447. | ||
Zhang W, Lao WW. Efficacy of sodium cantharidinate vitamin B6 combined with CapeOX in the treatment of advanced gastric cancer. Med Front. 2015;5(16):108–110. | ||
Zhu WQ. Sodium cantharindinate and vitamin B6 injection in combination with TACE for primary liver cancer. Int Med Health Guid News. 2014;20(14):2130–2132. | ||
Zeng X, Zhang Y, Kwong JSW, et al. The methodological quality assessment tools for preclinical and clinical studies, systematic review and meta-analysis, and clinical practice guideline: a systematic review. J Evid Based Med. 2015;8(1):2–10. | ||
Jackson D, White IR, Riley RD. Quantifying the impact of between-study heterogeneity in multivariate meta-analyses. Stat Med. 2012;31(29):3805–3820. | ||
Zhang L, Mu Y, Zhang A, et al. Cytokine-induced killer cells/dendritic cells-cytokine induced killer cells immunotherapy combined with chemotherapy for treatment of colorectal cancer in China: a meta-analysis of 29 trials involving 2,610 patients. Oncotarget. 2017;8(28):45164–45177. | ||
Williamson PR, Smith CT, Hutton JL, Marson AG. Aggregate data meta-analysis with time-to-event outcomes. Stat Med. 2002;21(22):3337–3351. | ||
Zhou L, Wang XL, Deng QL, Du YQ, Zhao NQ. The efficacy and safety of immunotherapy in patients with advanced NSCLC: a systematic review and meta-analysis. Sci Rep. 2016;6(1):32020. | ||
Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 2007;8(1):16. | ||
Liang M, Chen Q, Zhang Y, et al. Impact of diabetes on the risk of bedsore in patients undergoing surgery: an updated quantitative analysis of cohort studies. Oncotarget. 2017;8(9):14516–14524. | ||
Gu Y, Jiang L, Miao JH, Liang TS, Kan QC, Yang DK. Clinical effects of thermotherapy in combination with intracavitary infusion of traditional Chinese medicine in the treatment of malignant pleural effusion. J Biol Regul Homeost Agents. 2016;30(4):1023–1028. | ||
Yan Z, Lai Z, Lin J. Anticancer properties of traditional Chinese medicine. Comb Chem High Throughput Screen. 2017;20(5):423–429. | ||
Xu J, Song Z, Guo Q, Li J. Synergistic effect and molecular mechanisms of traditional Chinese medicine on regulating tumor microenvironment and cancer cells. Biomed Res Int. 2016;2016(5):1–14. | ||
Liao YH, Li CI, Lin CC, Lin JG, Chiang JH, Li TC. Traditional Chinese medicine as adjunctive therapy improves the long-term survival of lung cancer patients. J Cancer Res Clin Oncol. 2017;143(12):2425–2435. |
Supplementary materials
  | Figure S3 Funnel plot of percentage of complete response rates (A), partial response rates (B), stable disease rates (C), and progressive disease rates (D). |
References
Chen Y, Zhu WM, Zhou DX, Shan YF. Clinical study on disodium cantharidinate and vitamin B6 combined with raltitrexed and oxaliplatin in treatment of advanced colorectal cancer. Drugs Clinic. 2016; 31(11):1784–1787. | ||
Fan LJ. Efficacy of sodium cantharidinate and vitamin B6 combined with 5-fluorouracil and calcium folinate in treatment of advanced colorectal cancer. Mod Med J China. 2009;11(10):64–65. | ||
Fan QL, Wang Y, Zhao KX, Kao J. Clinical observation on effects of sodium cantharidate vitamin B6 injection combined with S-1 treatment of advanced gastrointestinal tract tumor patients. Guangming J Chin Med. 2013;28(4):786–788. | ||
Fang XH, Xu J. Cantharidin sodium vitamin B6 injection as assistant therapy for treating liver cancer in 37 cases. China Pharm. 2016;25(1):25–27. | ||
Guan LY, Ma J, Yuan SF, Qu J. Disodium cantharidinate and vitamin B6 combined with tegafur, gimeracil and oteracil porassium for treating stage IV pancreatic cancer in 27 cases. Tradit Med. 2015;24(20):124–125. | ||
Jia JM, Zh L. Efficacy of sodium cantharidinate and vitamin B6 comwith intravenous chemotherapy for advanced esophageal cancer. Med Inf. 2013;26(9):188–189. | ||
Liu GW, Ren WD. Cantharis acid sodium vitamin B6 injection comwith maintain capecitabine synchronization for the treatment of advanced colorectal cancer. World Latest Med Inf. 2017;17(26):12–14. | ||
Liu SH, Feng W, Zhu RX, Feng ZC. Comparative study of disodium cantharidinate and vitamin B6 injection combined with oxaliplatin and tegafur in the treatment of advanced gastric cancer. Pract Clin J Integr Tradit Chinese West Med. 2008;8(4):3–4. | ||
Mao WD. Clinical study of sodium cantharidinate and vitamin B6 combined with capecitabine in the treatment of advanced gastric cancer. J Clin Med. 2016;3(45):9034–9036. | ||
Shao H, Hong G, Luo X. Evaluation of sodium cantharidinate/vitamin B6 in the treatment of primary liver cancer. J Cancer Res Ther. 2014; 10(Suppl 1):75–78. | ||
Shi XY, Zhang J, Meng W, Ma F, Li ZL, Zhao JF. Clinical observation of sodium cantharidinate and vitamin B6 injection combined with XELOX protocol in treatment of patients with advanced colorectal carcinoma. Med & Pharm J Chin PLA. 2017;29(6):36–39. | ||
Tian XL, Yang P. Clinical efficacy of treatment of liver cancer with combined sodium cantharidate vitamin B6 injection and TACE therapy. Chin J Clin Oncol Rehabil. 2006;13(4):351–353. | ||
Wang JH, Li GP, Wu JH. Application of sodium cantharidinate and vitamin B6 injection in patients with advanced colorectal cancer. J Mod Med Health. 2010;26(6):844–845. | ||
Wang YW, Yang RL. The effect observation of sodium cantharivitamin B6 combined with capecitabine in the treatment of advanced stomach and colorectal carcinoma. Med Innov China. 2017;14(36):23–26. | ||
Wei YF, Wu JS, Huo ZG, Jia ZY, Qi SS, Wang XX. Efficacy evaluation of sodium cantharidinate and vitamin B6 injection combined with TACE in the treatment of 48 patients with intermediate and advanced primary liver cancer. China Pharm. 2015;24(5):72–74. | ||
Wu ZM, Liu Q, Shi SM, Yu SY. Clinical study of ai yishu combined with FOLFIRI regimen in the treatment of recurrent gastric cancer. Med Inf. 2013;26(6):256. | ||
Xie ZX, Wang JS, Liu YB. Short-term efficacy and adverse effects of sodium cantharidinate and vitamin B6 injection combined with chein the treatment of advanced gastric cancer. Hebei Med J. 2016;38(10):1554–1559. | ||
You ZY, Liu Y, Shu XH, Liu BN, Liu XF. Malignant ascites of gastumor treated with sodium cantharidinate and vitamin B6 injection and hyperthermic intraperitoneal chemotherapy. World J Integr Tradit West Med. 2015;10(9):1249–1255. | ||
Zeng L, Liu YL, Liu XT, Yang ZF. Clinical observation of sodium cantharidinate and vitamin B6 injection in the treatment of liver cancer. J Hebei Med Univ. 2009;30(6):596–597. | ||
Zhang MJ, Zuo CF. Clinical efficacy of sodium cantharidate vitamin B6 injection combined with chemotherapy in treatment of liver cancer. J Pract Oncol. 2011;26(1):50–52. | ||
Zhang W, Yang J, Zhong CS. Efficacy of sodium cantharidinate vitamin B6 combined with xeloda in the treatment of advanced gastric cancer. J Huaihai Med. 2012;30(5):446–447. | ||
Zhang W, Lao WW. Efficacy of sodium cantharidinate vitamin B6 combined with CapeOX in the treatment of advanced gastric cancer. Med Front. 2015;5(16):108–110. | ||
Zhu WQ. Sodium cantharindinate and vitamin B6 injection in combinawith TACE for primary liver cancer. Int Med Health Guid News. 2014;20(14):2130–2132. | ||
Li GP, Li XR, Wang JH. Clinical observation on sodium cantharidate vitamin B6 injection combined with postoperative chemotherapy in treatment of gastric cancer. Pract Prev Med. 2010;17(1):105–106. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.