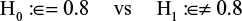Back to Journals » Journal of Pain Research » Volume 10
Efficacy and safety of 0.75% ropivacaine instillation into subinguinal wound in patients after bilateral microsurgical varicocelectomy: a bi-center, randomized, double-blind, placebo-controlled trial
Authors Cui WS, Shin YS , You JH, Doo AR , Soni KK, Park JK
Received 5 January 2017
Accepted for publication 11 March 2017
Published 3 July 2017 Volume 2017:10 Pages 1515—1519
DOI https://doi.org/10.2147/JPR.S131692
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Katherine Hanlon
Wan Shou Cui,1,* Yu Seob Shin,2,3,* Jae Hyung You,3 A Ram Doo,4 Kiran Kumar Soni,3 Jong Kwan Park3
1Andrology Center, Peking University First Hospital, Beijing, People’s Republic of China; 2Department of Urology, Armed Forces Capital Hospital, Seongnam, 3Department of Urology, Chonbuk National University and Research Institute of Clinical Medicine of Chonbuk National University-Biomedical Research Institute and Medical Device Clinical Trial Center of Chonbuk National University Hospital, Jeonju, 4Department of Anesthesiology and Pain Medicine, Chonbuk National University Medical School, Jeonju, Republic of Korea
*These authors contributed equally to this work
Objective: To evaluate the efficacy and safety of 0.75% ropivacaine instillation into inguinal wound in patients who have undergone bilateral microsurgical varicocelectomy.
Patients and methods: Eighty-five men who were screened for bilateral varicoceles from March 2015 to July 2016 were randomized for the treatment. All patients underwent inguinal varicocelectomy by general anesthesia. After ligation of the internal spermatic veins from the spermatic cord, additional delivery of testis through inguinal incision site was done to ligate external spermatic veins and gubernacular veins. Before repairing external oblique aponeurosis, 6 mL of 0.75% ropivacaine and 6 mL of normal saline were instilled under the fascia and around the funiculus (spermatic cord) by a randomized and double-blind method. Visual analog scale (VAS) pain score and Prince Henry Pain Score (PHPS) were used for evaluating operative sites at 1, 2, 4, and 8 hours and 7 days after surgery. Safety and tolerability were evaluated throughout the course of this study by assessing adverse events.
Results: A total of 55 men completed the study. Of these 55 men, 31 received instillation of ropivacaine on the left operative site, while 24 received instillation of ropivacaine on the right operative site. VAS pain scores and PHPS in the ropivacaine-instilled operative site were significantly lower compared to those obtained with placebo at 2, 4, and 8 hours after surgery. In general, instillation of ropivacaine was safe and well tolerated in patients.
Conclusion: Ropivacaine instillation into inguinal surgical site wound significantly reduced postoperative pain after microsurgical varicocelectomy.
Keywords: pain, ropivacaine, microsurgical varicocelectomy
Introduction
Varicocele is an abnormal enlargement of the pampiniform venous plexus in the scrotum.1 The goal of varicocele treatment is to stop the backward flow of blood from the body to the scrotum.1 Microsurgical varicocelectomy could be considered in the patient who had suffered from severe testicular pain, marked discrepancy in testicular volume, and impaired spermatogenesis in spite of the controversies.2–4
Currently, the main methods employed for surgical repair of varicocele include microsurgical varicocelectomy, laparoscopic varicocelectomy, and radiological percutaneous embolization.5 Among various surgical methods, microsurgical varicocelectomy appears to be the optimal treatment in most cases.6 However, acute postoperative pain is common after microsurgical varicocelectomy.
Various methods including systemic analgesics and nerve block have been utilized to manage postoperative pain. Reducing pain after microsurgical varicocelectomy can be done using systemic analgesics. However, systemic analgesics such as opioids may have side effects such as nausea, vomiting, itching, respiratory problems, and sedation. They might lengthen the duration of postoperative ileus.7 There is a controversy regarding the application of a local anesthetic on the operative site after inguinal surgery to reduce postoperative pain.8,9 However, results from a meta-analysis of Wu et al7 have shown that instilling a local anesthetic infusion pump following an inguinal hernia repair can reduce postoperative pain in comparison to the placebo group.7 To the best of our knowledge, no study has reported the effect of instilling a local anesthetic agent following microsurgical varicocelectomy. Therefore, the objective of this study was to compare postoperative pain after bilateral microsurgical varicocelectomy by applying a local anesthetic agent on a unilateral operative site through a randomized and double-blind method.
Patients and methods
Study design and sample
This study was conducted with a bi-center, prospective, randomized, double-blind, and placebo-controlled setting at the Department of Urology in Chonbuk National University Hospital and Armed Forces Capital Hospital in Korea. This study was reviewed and approved by the institutional review board of the Chonbuk National University Hospital (approval no: CUH 2014-05-020-003) and the Armed Forces Medical Command (approval no: AFMC-16027-IRB-16-020). The study was conducted in accordance with the Good Clinical Practice and the International Conference on Harmonization guidelines and in conformity with the ethical principles of the Declaration of Helsinki. Patients were recruited prospectively and consecutively. Written informed consent was obtained from each patient before entering the study. A group of men with clinical bilateral varicoceles were identified. All men who were presented to our clinic with one or more years of infertility, a clinically palpable varicocele, and abnormal semen parameters (reduced sperm concentration, motility, or morphology on 2 or more semen samples) were suitable candidates for varicocele repair. All varicoceles were evaluated and graded in accordance with the Dubin grading system (grades I–III). They were based solely on clinical examination. All varicoceles were confirmed and measured by Doppler ultrasonography to detect the reflux of blood and their actual sizes. Patients who had other causes of scrotal pain such as testis trauma, testicular torsion, epididymitis, prostatitis, sexually transmitted disease, urinary tract disease, inguinal hernia repair, stone disease, and any other testicular pathology were excluded.
Power analysis for sample size
The following hypothesis was formulated:
|
where ∈ = was the mean difference in VAS pain score between the ropivacaine-instilled site and the placebo site at 8 hours after the surgery. Aiming at a power of 80% with a 2-sided alpha level at 0.05, considering a VAS pain score difference of 0.8 as clinically significant with a standard deviation (SD) of 2.25, and assuming a dropout rate of 20%, 78 patients were required based on Bonferroni test with correction for multiple comparisons.
Procedure and data collection
Bilateral microsurgical varicocelectomy was performed by 2 surgeons (Jong Kwan Park and Yu Seob Shin). All patients underwent inguinal varicocelectomy by general anesthesia induced with propofol (target-controlled infusion 3–5 μg/mL), fentanyl (1.5–2.5 μg/kg), and cisatracurium (0.15 mg/kg). Anesthesia was maintained with propofol (target-controlled infusion 3–5 μg/mL) and remifentanil (continuous infusion 0.05–0.3 μg/kg/min). Patients were intubated and ventilated with a mix of oxygen and air. Anesthesia management was left to the discretion of the attending anesthesiologist. No other analgesics were administered intraoperatively. For surgery, incision over the inguinal canal, opening the external oblique aponeurosis, encirclement, and delivery of the spermatic cord were performed. After ligation of the internal spermatic veins from the spermatic cord, additional delivery of testis through inguinal incision site was done for ligation of external spermatic veins and gubernacular veins. Before repairing external oblique aponeurosis, 6 mL of 0.75% ropivacaine and 6 mL of normal saline were instilled under the fascia and around the funiculus (spermatic cord) of both sites using randomized and double-blind method. Visual analog scale (VAS) pain score and Prince Henry Pain Score (PHPS) were used to evaluate both operative sites at 1, 2, 4, and 8 hours and 7 days after the surgery. Safety and tolerability were evaluated throughout the course of the study by assessing adverse events.
Statistical analysis
VAS pain score and PHPS were compared using independent t-test. All statistical analyses were performed using statistical software package SPSS 18.0 (IBM Corp., Armonk, NY, USA). Statistical significance was considered when P-value was <0.05.
Results
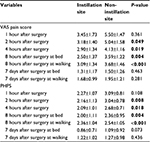
From March 2015 to July 2016, a total of 96 patients who were screened for bilateral varicoceles were recruited into the study (Figure 1). Of these patients, 11 were excluded due to declining to participate (n=9) or consuming analgesics (n=2). Therefore, a total of 85 patients were randomized for the treatment. However, 30 of the 85 patients were excluded due to missing pain score assessment (n=22) or receiving other analgesics (n=8). The remaining 55 men fully completed the study. Of these 55 men, 31 received instillation of ropivacaine on the left operative site, while 24 received instillation of ropivacaine on the right. Characteristics of these 55 patients are listed in Table 1. The mean age of these patients was 26.25±5.35 years (range: 16.0–43.0 years). The VAS pain scores in the ropivacaine-instilled operative site were significantly lower compared to those in the placebo site at 2, 4, and 8 hours after the surgery (Table 2). However, there was no statistically significant difference in VAS pain score between the ropivacaine-instilled site and placebo site at 1 hour and 7 days after the surgery (Table 2). The values of PHPS at 2, 4, and 8 hours after surgery in ropivacaine-instilled operative site were significantly lower compared to those at placebo site (Table 2). However, there was no statistically significant difference in PHPS between the ropivacaine-instilled site and the placebo site at 1 hour and 7 days after the surgery (Table 2). In general, instillation of ropivacaine was safe and well tolerated in patients. No clinically significant changes were observed in patients. There was no systemic or surgical site infection in patients.
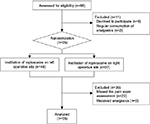  | Figure 1 Cohort study flow diagram. |
  | Table 1 Patient characteristics Abbreviation: SD, standard deviation. |
Discussion
Acute postoperative pain is a common complication after open inguinal surgery. Application of a local anesthetic agent to the operative site might reduce postoperative pain. Clinical experience and other studies have suggested that application of a local anesthetic agent to the operative site might reduce postoperative pain after open inguinal surgery.7,10,11 In the field of urology, microsurgical varicocelectomy is the most common open inguinal surgery. In our experience, patients frequently report acute postoperative pain after microsurgical varicocelectomy. The majority of patients with varicocele are young males. The reason that young male patients suffer from more pain compared to older patients is possibly due to their hypersensitivity. For this reason, postoperative pain management is important in patients after microsurgical varicocelectomy.
Ropivacaine is a new amino amide with structure similar to bupivacaine. It is a long-acting local anesthetic. Ropivacaine is prepared as an s-enantiomer rather than a racemic mixture such as bupivacaine.12 Ropivacaine also produces cutaneous vasoconstriction that restricts systemic absorption of the drug and increases its local duration of action. Moreover, ropivacaine produces anti-inflammatory effect that may further reduce the pain when administered locally.13,14 Results from our study revealed that pain scores at 2, 4, and 8 hours after surgery in 0.75% ropivacaine-instilled site were significantly lower compared to those in the placebo site, similar to existing findings.7,10 Moreover, elimination or reduction of postoperative pain following microsurgical varicocelectomy can enhance patients’ quality of life and allow patients to quickly return to normal daily activities.
This prospective, randomized, double-blind trial investigated the clinical effects of applying ropivacaine to operative site for microsurgical varicocelectomy. The design of our study was very unique and creative. We compared the pain score between ropivacaine injection site and placebo site of the same patient. Furthermore, our study was performed at the same time. Such a design is the strength of our study.
We also used both VAS score and PHPS to assess postoperative pain in patients after microsurgical varicocelectomy. Many studies have used either VAS or PHPS to evaluate the patient.7,10,11 We believe that the use of both scores in our study can improve the accuracy of assessing pain in patients, thus more accurately evaluating the effect of applying local anesthetic to the surgical site.
In spite of our findings in favor of analgesic efficacy of ropivacaine, the main limitation of our study was its small sample size that precluded us from reaching a definite conclusion. Further studies with a larger sample size and different application routes are required to investigate the efficacy and the most efficient application route of ropivacaine for postoperative pain control after microsurgical varicocelectomy.
Conclusion
This study showed that instillation of 6 mL of 0.75% ropivacaine into inguinal wound significantly reduced postoperative pain after microsurgical varicocelectomy. The reduction in postoperative pain reduced the use of analgesics in patients. In addition, the elimination or reduction in pain following microsurgical varicocelectomy enhanced patients’ quality of life and allowed them to quickly return to normal daily activities.
Acknowledgments
We thank the members of the Medical Device Clinical Trial Center of Chonbuk National University Hospital for helpful discussion. This study was supported by fund from the Biomedical Research Institute of Chonbuk National University Hospital, Jeonju, Republic of Korea. However, Biomedical Research Institute of Chonbuk National University Hospital had no role in study design, data collection, data management, data analysis, data interpretation, manuscript preparation, manuscript reviewing, or approval of this manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
Kass EJ, Reitelman C. Adolescent varicocele. Urol Clin North Am. 1995;22(1):151–159. | ||
Choi WS, Kim SW. Current issues in varicocele management: a review. World J Mens Health. 2013;31(1):12–20. | ||
Cayan S, Shavakhabov S, Kadioğlu A. Treatment of palpable varicocele in infertile men: a meta-analysis to define the best technique. J Androl. 2009;30(1):33–40. | ||
Cho KS, Seo JT. Effect of varicocelectomy on male infertility. Korean J Urol. 2014;55(11):703–709. | ||
González R. Proposal for describing procedures to correct varicocele. A new terminology. Front Pediatr. 2014;2:47. | ||
Chiba K, Fujisawa M. Clinical outcomes of varicocele repair in infertile men: a review. World J Mens Health. 2016;34(2):101–109. | ||
Wu CC, Bai CH, Huang MT, Wu CH, Tam KW. Local anesthetic infusion pump for pain management following open inguinal hernia repair: a meta-analysis. Int J Surg. 2014;12(3):245–250. | ||
LeBlanc KA, Bellanger D, Rhynes VK, Hausmann M. Evaluation of continuous infusion of 0.5% bupivacaine by elastomeric pump for postoperative pain management after open inguinal hernia repair. J Am Coll Surg. 2005;200(2):198–202. | ||
Bailey ME, Jourdan IC. Randomized placebo-controlled trial of local anaesthetic infusion in day-case inguinal hernia repair. Br J Surg. 1998;85(10):1451. | ||
Stewart A, Fan MM, Fong MJ, Louie A, Lynch JP, O’Shea M. Randomized trial of a pain control infusion pump following inguinal hernia repair. ANZ J Surg. 2004;74(10):873–876. | ||
Lau H, Patil NG, Lee F. Randomized clinical trial of postoperative subfascial infusion with bupivacaine following ambulatory open mesh repair of inguinal hernia. Dig Surg. 2003;20(4):285–289. | ||
Scott DB. Evaluation of the toxicity of local anaesthetic agents in man. Br J Anaesth. 1975;47(1):56–61. | ||
Stienstra R. The place of ropivacaine in anesthesia. Acta Anaesthesiol Belg. 2003;54(2):141–148. | ||
Owen MD, Dean LS. Ropivacaine. Expert Opin Pharmacother. 2000;1(2):325–336. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.