Back to Journals » International Journal of Nanomedicine » Volume 15
Effects of TiO2-Coated Stainless Steel Orthodontic Wires on Streptococcus mutans Bacteria: A Clinical Study
Authors Mollabashi V, Farmany A, Alikhani MY , Sattari M, Soltanian AR, Kahvand P, Banisafar Z
Received 16 April 2020
Accepted for publication 14 October 2020
Published 10 November 2020 Volume 2020:15 Pages 8759—8766
DOI https://doi.org/10.2147/IJN.S258440
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster
Vahid Mollabashi,1 Abbas Farmany,2 Mohammad Yousef Alikhani,3 Mohammad Sattari,4 Ali Reza Soltanian,5 Parnian Kahvand,6 Ziba Banisafar7
1Orthodontic Department, Dental Faculty and Dental Research Center, Hamadan University of Medical Sciences, Hamadan, Iran; 2Dental Research Center, Hamadan University of Medical Sciences, Hamadan, Iran; 3Department of Microbiology, Hamadan University of Medical Sciences, Hamadan, Iran; 4Division of Microstructure Physics, Department of Physics, Chalmers University of Technology, Gothenburg SE-412 96, Sweden; 5Modeling of Noncommunicable Diseases Research Center, Department of Biostatistics and Epidemiology, School of Public Health, Hamadan University of Medical Sciences, Hamadan, Iran; 6Orthodontic Department, Dental Faculty, Hamadan University of Medical Sciences, Hamadan, Iran; 7Orthodontic Department, Dental Faculty, Hamadan University of Medical Sciences, Hamadan, Iran
Correspondence: Ziba Banisafar Orthodontic
Department, Dental Faculty, Hamadan University of Medical Sciences, Hamadan, Iran
Email [email protected]
Introduction: The aim of this study was to clinically evaluate Streptococcus mutans adhesion on titanium dioxide–coated stainless steel orthodontic wires to decrease white-spot formation.
Methods: In this study, four groups of 17 patients each (n=68) aged 12– 25 years participated. A titanium dioxide coating layer was deposited on 0.4572 mm stainless steel orthodontic wires using physical vapor deposition. The coated wires were randomly assigned to one jaw, and the opposite jaw received an uncoated wire as control. Patients were divided into groups according to the duration that wires were in their mouths: A) 1 week, B) 2 weeks, C) 3 weeks, and D) 4 weeks. Block randomization was used to assign patients to each group. At the end of the experiment, 20 mm of each wire (canine-to-canine area) was cut and cultured in S. mutans–specific medium. The culture plates were placed in an incubator containing 5% CO2 for 72 hours at 37°C, and then colonies were counted. MTT was used to test the biocompatibility of the coated and uncoated wires. To evaluate the stability of the coated titanium dioxide layer on the wires, titanium concentration on the saliva was determined using inductively coupled plasma mass spectroscopy.
Results: The Kruskal–Wallis test showed that there was no significant difference in colony counts among the coated wires during 1– 4 weeks (p< 0.48). In the uncoated-wire groups, colonys count at week 1 were higher than weeks 24 –(p< 0.022). Wilcoxon’s test showed that the number of colonies was significantly different in groups A and C, but there was no significant difference in groups B or D. MTT-assay results showed that there was not a significant difference between cell viability in the coated-wire group and the control. The Kruskal–Wallis test showed that there was no significant difference in titanium concentration in the studied groups (p< 0.834).
Conclusion: Application of titanium dioxide coating is effective in reducing bacterial adhesion at wire insertion.
Keywords: orthodontic wire, titanium dioxide, Streptococcus mutans, bacterial adhesion
Introduction
Fixed orthodontic appliances are one of the possible causes of changes in the mouth. These changes can be due to etching of the enamel surfaces, excessive composites, or morphological irregularities of the bracket that lead to plaque accumulation and white tooth-lesion formation.1,2 White lesions around brackets develop within 1 month of orthodontic treatment.3 The area of these lesions can be measured by photography and image-analysis software.4
Orthodontic wires are one of the most important orthodontic components, but provide a suitable environment for oral microorganism growth. As shown by Elides et al, stainless steels have the highest surface tension and energy, which increase plaque-retention capacity.5 During orthodontic treatment, there is a significant increase in the number of some microorganisms, such as Streptococcus mutans and Lactobacillus acidophilus,6,7 which have been identified as major pathogens in periodontal diseases and dental caries and increase the risk of declassification.8 In the past decade, the antibacterial activity of some metal and metal oxide films such as TiO2,9 ZnO,10 and Ag,11 has been considered. Among these materials, TiO2 has gained attention due to its specific properties. For example, in an aquatic environment, when TiO2 is exposed to ultraviolet radiation, it undergoes redox reactions and produces hydroxyl radicals that are able to decompose organic compounds.12–15
Today, the biocompatibility of TiO2 has been proven, and a large number of medical devices are made of titanium alloys. Furthermore, no cell-viability loss has been reported in orthodontic appliances, indicating that TiO2 coating is biocompatible.16,17 So far, in vitro investigations of antiadhesion and antibacterial properties of TiO2 have shown less bacterial attachment to TiO2-coated surfaces.17,18 Therefore, using the photocatalytic and antiadhesion capacity of TiO2 in clinical conditions may be effective in preventing bacteria attachment and bacteria-colony growth around orthodontic instruments. As such, this study aimed to evaluate the antiadhesion and antibacterial properties of TiO2-coated stainless steel orthodontic wires against S. mutans bacteria.
Methods
This study was conducted in accordance with the Declaration of Helsinki. All experimental protocols were carried out in accordance with the Institutional Cell Culture and Animal Care and Use Committee of Hamadan University of Medical Sciences, Iran. The Ethics Research Committee of the Faculty of Dentistry, Hamadan University of Medical Sciences (IR.UMSHA.REC.1397.791 and IR.UMSHA.REC.1398.427) approved the research study.
This in vivo study was performed in the orthodontic department at the Faculty of Dentistry and Microbiology Laboratory of Hamadan University of Medical Sciences, Hamadan, Iran in 2019. Four groups of 17 patients each (n=68) aged 12–25 years — 40 female and 28 male — participated in this clinical study. Each patient was considered his/her own control. Titanium dioxide–coated and uncoated stainless steel wires (0.4572 mm) were placed randomly in each patient’s jaws. In each group, nine coated wires in the maxilla and eight coated wires in the mandible were placed.
Inclusion criteria were age 12–25 years, no antibiotic and antibacterial mouthwash use during the treatment, no systemic disease history, no smoking, and no use of interarch elastic, chain, O-ring, or open coil during treatment.Patients’ systemic status was assessed by taking their history. Patients were excluded if they were suspected of poor immunostatus and microbial flora, as well as pregnant women, those with diabetes or AIDS, and immunosuppressive drug users. Also, patients with missing teeth, syndromes, and craniofacial anomalies were excluded. For all patients, informed consent was received from them or their parents (for patients <18 years old). The Ethics Research Committee of Hamadan University of Medical Sciences approved the informed consents taken from patients.
After receiving informed consent and completing the patient-information form, patients who agreed to receive 0.4572 mm stainless steel wire were included in the study. Antiadhesion properties of the wires were investigated as a function of time. For this purpose, patients were divided into four groups: A) 1 week, B) 2 weeks, C) 3 weeks, and D) 4 weeks. Block randomization was used to assign patients to each group.
Wire Preparation
The wires were coated with TiO2 by physical vapor deposition (PVD). For this purpose, the surface of wires were cleaned carefully:19
- ultrasonic cleaning in ethanol and acetone bath for 15 minutes,
- ultrasonic cleaning in water–NaOH solution for 20 minutes,
- rinsing with distilled water for 15 minutes.
An E-gun thermal method at 5,000 W and 1 Å/second was used for coating the wires with a 100 nm layer of TiO2. Vacuum-chamber pressure was 0.02 bar before the start of the coating process and 0.05 bar during coating. The chamber temperature was set at 300°C.
SEM Analysis
To check coating quality and integrity, one of the wires was randomly selected. The sample was first glued between two silicon wafers with a mixture of superglue and Bakelite (for conductivity). After the adhesive had dried, the sample was ground using P800, P100, and P2400 sandpaper to obtain a uniform cross-section. Then, a smooth cross-section of the sample was prepared using a Leica EM TIC 3X broad ion–beam instrument operated at 6 kV. SEM (JEOL JSM-7800F Prime) with energy dispersive X-ray (EDX) spectroscopy was used to image and characterize the sample.
Cell-Viability Tests
In order to test the biocompatibility of the coated and uncoated wires, MTT testing was applied. L929 cells were cultured in a 96-well plate in RPMI supplemented with 10% FBS, 50 IU/mL penicillin, and 50 μg/mL streptomycin and incubated in 5% CO2 at 37°C. Viability of treated cells was measured by MTT.30 After treatment, cells were incubated for 3 hours with MTT solution. Next, DMSO was added to each well to solubilize formazan particles, and ultimately absorbance was read at 580 nm using an ELISA reader (LaSystems Multiskan).
Titanium Dioxide Release
To evaluate titanium dioxide–coating stability, the titanium concentration of treated patients’ saliva was analyzed. Saliva samples of patients were collected before insertion of coated wires by Navazesh technique20 at 1, 2, 3, and 4 weeks after placing the coated wire in patients’ mouths. Saliva samples were kept at −20°C. The concentration of titanium was determined by inductively coupled plasma mass spectrometry (ICP-MS)(ELAN DRC II; PerkinElmer).
Bacteria Counting
At selected times, wires were removed from patients’ mouths with a sterile device. The canine-to-canine area (distance between brackets) was cut and separated (20 mm) and transferred to a sterile tube containing physiological serum. Trypsin (25%) was added to the samples and incubated for 45 minutes to separate bacteria from the wires’ surfaces.21 Different volumes of bacteria suspension (2, 5, and 10 µL) were cultured in S. mutans–specific mitis–salivarius agar base (HiMedia). Plates were incubated for 72 hours in 5% CO2 at 37°C and colony counting performed. An MBT22 system was used in all patients, and brackets were engaged with ligature wires to eliminate wire play. All procedures were performed by one person. This study was blind for the patients and the evaluator of bacterial colonies in the samples.
Statistical Analysis
The Kruskal–Wallis test was used to compare colony counts and titanium release between the studied groups. Wilcoxon’s test was used to compare colony counts within groups. Data were analyzed using SPSS 22.0, and the significance level was set at p≤0.05.
Results
PVD was used to coat a layer of TiO2 film on the stainless steel orthodontic wires. SEM images confirmed the formation of a TiO2 film with thickness of ≈100 nm on the wire’s surface (Figure 1). EDX spectroscopy qualitative point analysis confirmed the presence of titanium and oxygen in the coated layer. The presence of other elements, such as Fe and Cr, in the EDX spectrum is due to the inherent spatial resolution of SEM–EDX spectroscopy, which is in order of 1 µm. Since the thickness of the coated layer is about 100 nm, which is far below the spatial resolution of SEM–EDX spectroscopy, a signal from the wire is inevitably detected, together with a signal from the coated layer. The EDX reference spectrum taken from the uncoated wire showed no Ti, which confirmed that the Ti peak in the coated-layer spectrum indeed originated from the coating process (Figure 2).
 |
Figure 1 SEM of TiO2-coated orthodontic stainless steel wires at two magnifications: (A) ×3,500, (B) ×9,000. |
 |
Figure 2 Images and graphs: (A) SEM of coated area, (B) SEM–EDX spectroscopy of coated area, (C) SEM of uncoated area, (D) SEM–EDX spectroscopy of uncoated area. |
Biocompatibility results for coated- and uncoated-wire cells revealed that viability of L929 cells in the coated-wire group was 90.3%±9.5% and for the uncoated-wire group 86.6%±8.1%. There was not a significant difference between the coated-wire group and control group, with 100% viable cells (Figure 3). The Kolmogorov–Smirnov test showed that titanium concentration in the studied groups did not follow a normal distribution. Therefore, a nonparametric test was used to compare the titanium concentration among the groups. Kruskal–Wallis nonparametric testing showed that there was no significant difference (p<0.834). This test also showed (Table 1) that there was no difference between the mean concentration of titanium at the beginning and end of the study (p<0.386). In addition to these comparisons, the mean difference between titanium concentrations of coated wires before and after placement was investigated by Kruskal–Wallis test, and it was observed that there was no statistically significant difference among the four groups (Table 1 and Figure 4).
 |
Table 1 Descriptive Indices and Comparison of Titanium Concentration in the Groups (Kruskal–Wallis Test) |
 |
Figure 3 Cell viability of coated and uncoated wires. |
 |
Figure 4 Titanium concentration across groups. |
Kruskal–Wallis analysis showed that by increasing treatment time from 1 to 4 weeks, the colony counts on the coated orthodontic wires did not differ significantly (p<0.48, Table 2), but the difference in bacterial colonies in the uncoated wires was significant (p<0.022), especially at the beginning of wire insertion (Table 3). The Wilcoxon signed-rank test was used for intragroup comparison of S. mutans colonies.The number of colonies was significantly different between groups A and C, but there was no significant difference between groups B and D (Table 4). Mean colony numbers in the mandible at the times studied in the coated-wire group were significantly lower than the uncoated-wire group; however, this trend was not seen in the maxilla (Figure 5). Also, no significant difference in colony numbers was found on comparison between the sexes (p<0.24).
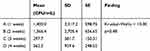 |
Table 2 Comparison of Streptococcus mutans Colonies Amonggroups (Coated Wires) |
 |
Table 3 Comparison of Streptococcus mutans Colonies Among Groups (Uncoated Wires) |
 |
Table 4 Intragroup Comparison of Streptococcus mutans Colonies Among Groups |
 |
Figure 5 Comparison of Streptococcus mutans colonies by upper and lower arch. |
Discussion
Employing an efficient method to reduce bacterial adhesion on arch wires and around orthodontic appliances can be an appropriate method to prevent enamel decalcification and periodontal diseases. However, most studies of antiadhesion and antibacterial properties of TiO2-coated orthodontic instruments have been carried out as in vitro. As seen in groups A and C, the results of this study showed that orthodontic wires coated with TiO2 can decrease the S. mutans colonies on their surfaces. TiO2 nanoparticles have been widely used as efficient catalysts for organic pollutant degradation.13 It is interesting that titanium dioxide has photocatalytic activity, and when exposed to ultraviolet or visible light, two important changes occur on its surface: formation of oxidizing hydroxyl radicals and hydrophilicity.16 Among the various microorganisms of the oral cavity, S. mutans is one of the most important dental microorganisms. It decomposes sucrose into insoluble glucan through a glycosyltransferase reaction. Insoluble glucan is attached to the tooth surface and provides plaque-formation conditions. The resulting complex of glucan and oral bacteria creates a biofilm (oral), which is the mature stage of dental plaque.
Decreased adhesion of S. mutans on TiO2-coated orthodontic wires resulted from bacterial surface organic molecule degradation. This makes the bacteria more fragile by affecting the cell wall. Following cell-wall degradation, osmotic pressure will change, cellular organelles are destroyed, and finally the bacteria disintegrate.3 Also, surface hydrophilicity of wires will increase with photon absorption. Therefore, the contact angle between TiO2 and water is reduced and forms an aqua-thin layer on the wire surface.22 Therefore, any contamination on the wire surface is easily eliminated by rinsing with water or saliva. The results of our study showed that in the mandible, the average number of bacterial colonies around the coated wires was more reduced than in the maxilla. This seems to have been due to the hydrophilicity of the coated-wire surface, self-distinguishing properties in the mandible (due to tongue movement), and saliva flow in that area, which led to more effective wire coating in the lower jaw.
Previous studies have investigated the effect of titanium dioxide coating in vitro, but in this study we studied this effect on human samples. Chhattani et al23 studied the photocatalytic activity of titanium dioxide–coated steel and nickel titanium wires. They used the sol–gel method for coating, which is done at a lower temperature than the PVD coating we used in this study. As such, this method is applicable for nickel titanium wire coating, which loses its structural memory properties at high temperatures. Other aspects of the sol–gel method include higher cost, more time needed, the possibility of cracking the coated thin film, and surface-porosity formation that affects its clinical application.24 In the present study, PVD was used. Surfaces coated using PVD method have excellent abrasion resistance, friction-coefficient reduction, excellent adhesion, and stability in different environments,25 which shows the superiority of our study over other reports that have used the sol–gel method. In a study by Chhattani et al,23 after exposure to UVA radiation, coated wires were put in culture media containing S. mutans, and less adhesion and bacterial survival than than in the control group was seen. In a clinical study of bacterial antiadhesive effects of titanium nitride–coated wires, Amini et al26 showed that colony numbers around the coated wires decreased significantly compared to the control group. Only 20 participants were involved in their study, which does not seem to be large enough to account for the wide range of interpersonal differences, especially those factors related to oral microbial topography, diet, personal hygiene, and genetic variation. In this study, 19×25 steel wires were used. Özyildiz et al27 examined the effect of wire type on the titanium dioxide photocatalytic activity. They used three types of orthodontic wires — 16×22 steel, 16×22 D-Rect, and 14×18 multistrand — which are used as holders, and showed that multistranded wire had the greatest effect on Candida albicans and Enterococcus faecalis, while the steel wire had high S. mutans resistance. This means that the type of wire can affect antiadhesion activity. However, its mechanism is not clear and needs to be investigated. In a study by Amini et al26 bacterial culture was performed onlyafter 4 weeks in all patients in blood agar–culture medium, while in the present study antibacterial study was performed over four time periods. Furthermore, to increase the accuracy of the study, the specific S. mutans culture medium was used.
An important issue regarding the application of nanomaterials in human samples is their biocompatibility. The toxicity of nanoparticles is directly related to their size.28 In this study, a thin film of titanium dioxide with thickness of 100 nm was used. Here, the nanostructure coating was achieved by PVD. In addition, the oral exposure limit of titanium oxide in the age range of 7–69 years is 1.6 µg/kg, which is not clinically toxic.29 Fatani et al17 assessed the biocompatibility of titanium dioxide–coated brackets on the human gingival fibroblast (HGF) cell line with MTT assays in vitro. All coated brackets had higher OD values, which indicated that the survival rate of HGF cells around the coated brackets was higher than that of uncoated brackets, due to the passivating effect of titanium dioxide coating.
In this study, in order to evaluate the bactericidal effect of coated wires over time, we divided the patients into four groups. Bacterial colony counting was done over 4 weeks. The results showed that there was no statistically significant difference in colonization rate at different weeks, but average bacterial colon numbers in the uncoated wires were significantly different, especially in group A. This shows that the maximum antibacterial effect of TiO2-coated wire is seen when wire insertion is done and is beneficial for patients who have recently have started orthodontic treatment and not yet acquired sufficient oral health skills. Simultaneous use of coated wire and brackets is recommended if a time-dependent effect is desired.
The present study did not perform heavy mechanotherapy when the wires were inserted into the patient’s mouth, leading to mechanical deterioration of the wire coating. However, further studies are needed to investigate the mechanical stability of the titanium dioxide coating under mechanical loading during the treatment. Since the dark conditions of the mouth are a limiting factor, the antibacterial activity of titanium dioxide in the oral cavity may not be fully achieved. Other materials, such as silver and platinum, are recommended to be added to the coating layer for increasing light absorption and antibacterial activity.
Conclusion
A thin titanium dioxide–layer coating on stainless steel orthodontic wires decreased S. mutans colonies in the first and third week. It is interesting that surface modification of orthodontic wires with TiO2 is effective in reducing bacterial adhesion at the beginning of wire insertion.
Acknowledgment
The authors would like to acknowledge the Research Center of Dentistry, Hamadan University of Medical Sciences, Hamadan, Iran.
Funding
This study was part of a postgraduate student thesis in orthodontics (thesis number: 9805153697 and 9807095104), which was supported by the Vice-Chancellor of Research and Technology, Hamadan University of Medical Sciences, Hamadan, Iran.
Disclosure
No potential conflict of interest is reported by the authors.
References
1. Gorelick L, Geiger AM, Gwinnett AJ. Incidence of white spot formation after bonding and banding. Am J Orthod. 1982;81(2):93–98. doi:10.1016/0002-9416(82)90032-X
2. Rosenbloom RG, Tinanoff N. Salivary Streptococcus mutans levels in patients before, during, and after orthodontic treatment. Am J Orthod Dentofacial Orthop. 1991;100(1):35–37. doi:10.1016/0889-5406(91)70046-Y
3. Shah AG, Shetty PC, Ramachandr C, Bhat NS, Laxmikanth SM. In vitro assessment of photocatalytic titanium oxide surface modified stainless steel orthodontic brackets for antiadherent and antibacterial properties against Lactobacillus acidophilus. Angle Orthod. 2011;81(6):1028–1035. doi:10.2319/021111-101.1
4. Mahmoudzadeh M, Alijani S, Soufi LR, Farhadian M, Namdar F, Karami S. Effect of CO2 laser on the prevention of white spot lesions during fixed orthodontic treatment: a randomized clinical trial. Turk J Orthod. 2019;32(3):165. doi:10.5152/TurkJOrthod.2019.18052
5. Eliades T, Eliades G, Brantley WA. Microbial attachment on orthodontic appliances: I. Wettability and early pellicle formation on bracket materials. Am J Orthod Dentofacial Orthop. 1995;108(4):351–360.
6. Anhoury P, Nathanson D, Hughes CV, Socransky S, Feres M, Chou LL.Microbial profile on metallic and ceramic bracket materials. Angle Orthod. 2002;72(4):338–343.
7. Liu Y, Xu Y, Song Q, et al. Anti-biofilm activities from Bergenia crassifolia leaves against Streptococcus mutans. Front Microbiol. 2017;8(1738):1–10. doi:10.3389/fmicb.2017.01738
8. Schwaninger B, Vickers-Schwaninger N. Developing an effective oral hygiene program for the orthodontic patient: review, rationale, and recommendations. Am J Orthod Dentofacial Orthop. 1979;75(4):447–452. doi:10.1016/0002-9416(79)90167-2
9. Liqiang J, Xiaojun S, Weimin C, Zili X, Yaoguo D, Honggang F. The preparation and characterization of nanoparticle TiO2/Ti films and their photocatalytic activity. J Phys Chem Solids. 2003;64(4):615–623. doi:10.1016/S0022-3697(02)00362-1
10. Kachoei M, Nourian A, Divband B, Kachoei Z, Shirazi S. Zinc-oxide nanocoating for improvement of the antibacterial and frictional behavior of nickel-titanium alloy. Nanomedicine. 2016;11(19):2511–2527. doi:10.2217/nnm-2016-0171
11. Shah PK, Sharma P, Goje SK. Comparative evaluation of frictional resistance of silver-coated stainless steel wires with uncoated stainless steel wires: an in vitro study. Contemp Clin Dent. 2020;9(2):331–336.
12. Hashimoto K, Irie H, Fujishima A. TiO2 photocatalysis: a historical overview and future prospects. Jpn J Appl Phys. 2005;44(12):8269–8285. doi:10.1143/JJAP.44.8269
13. Chun MJ, Shim E, Kho EH, et al. Surface modification of orthodontic wires with photocatalytic titanium oxide for its antiadherent and antibacterial properties. Angle Orthod. 2007;77(3):483–488. doi:10.2319/0003-3219(2007)077[0483:SMOOWW]2.0.CO;2
14. Xiao G, Zhang X, Zhao Y, Su H, Tan T. The behavior of active bactericidal and antifungal coating under visible light irradiation. App Surf Sci. 2014;292:756–763. doi:10.1016/j.apsusc.2013.12.044
15. Fujishima A, Nakata K, Ochiai T, Manivannan A, Tryk DA. Recent aspects of photocatalytic technologies for solar fuels, self-cleaning, and environmental cleanup. Electrochem Soc Interface. 2013;22(2):51–56. doi:10.1149/2.F06132if
16. Visai L, De Nardo L, Punta C, et al. Titanium oxide antibacterial surfaces in biomedical devices. Int J Artif Organs. 2011;34(9):929–946. doi:10.5301/ijao.5000050
17. Fatani EJ, Almutairi HH, Alharbi AO, et al. In vitro assessment of stainless steel orthodontic brackets coated with titanium oxide mixed Ag for anti-adherent and antibacterial properties against Streptococcus mutans and Porphyromonas gingivalis. Microb Pathogenesis. 2017;112:190–194. doi:10.1016/j.micpath.2017.09.052
18. Prince V, Agrawal J, Agrawal M, et al. Evaluation of photocatalytic titanium oxide surface modified stainless steel and nickel-titanium orthodontic wires for its antiadherent and antibacterial properties-an in vitro study. Int J Sci Study. 2014;4(12):132–136.
19. Ghasemi T, Arash V, Rabiee SM, Rajabnia R, Pourzare A, Rakhshan V. Antimicrobial effect, frictional resistance, and surface roughness of stainless steel orthodontic brackets coated with nanofilms of silver and titanium oxide: a preliminary study. Microsc Res Techniq. 2017;80(6):599–607. doi:10.1002/jemt.22835
20. Bakhtiari S, Toosi P, Samadi S, Bakhshi M. Assessment of uric acid level in the saliva of patients with oral lichen planus. Med Princ Pract. 2017;26(1):57–60. doi:10.1159/000452133
21. Kantorski KZ, Scotti R, Valandro LF, Bottino MA, Koga-Ito CY, Jorge AO. Surface roughness and bacterial adherence to resin composites and ceramics. Oral Hlth Prev Dent. 2009;7(1):29–32.
22. Jinhui Z, Si L, Long C, Yi P, Shuangchun Y. The progress of TiO2 photocatalyst coating. IOSR-JEN. 2012;2(8):50–53. doi:10.9790/3021-02855053
23. Chhattani S, Shetty PC, Laxmikant SM, Ramachandra CS. In vitro assessment of photocatalytic titanium oxide surface-modified stainless steel and nickel titanium orthodontic wires for its antiadherent and antibacterial properties against Streptococcus mutans.J. Ind Orthod Soc. 2014;48:82–87. doi:10.1177/0974909820140202
24. Hench L. The Sol-Gel process. Chem Rev. 1990;90:33–37. doi:10.1021/cr00099a003
25. Richter K, Lorbeer C. A novel approach to prepare optically active ion doped luminescent materials via electron beam evaporation into ionic liquids. Chem Comm. 2015;51:114–117. doi:10.1039/C4CC05817H
26. Amini F, Bahador A, Kiaee B, Kiaee G. The effect of the titanium nitride coating on bacterial adhesion on orthodontic stainless steel wires: in vivo study. Biosci Biotech Res Comm. 2017;10(1):28–33. doi:10.21786/bbrc/10.1/5
27. Özyildiz F, Uzel A, Hazar AS, Güden M, Ölmez S, Aras I. Photocatalytic antimicrobial effect of TiO_2 anatase thin-film-coated orthodontic arch wires on 3 oral pathogens. Turk J Biol. 2014;38(2):289–295.
28. Lin X, Li J, Ma S, et al. Toxicity of TiO2 nanoparticles to Escherichia coli: effects of particle size, crystal phase and water chemistry. PLoS One. 2014;9(10):e110247. doi:10.1371/journal.pone.0110247
29. Heringa MB, Geraets L, van Eijkeren JC, Vandebriel RJ, de Jong WH, Oomen AG. Risk assessment of titanium dioxide nanoparticles via oral exposure, including toxicokinetic considerations. Nanotoxicology. 2016;10(10):1515–1525. doi:10.1080/17435390.2016.1238113
30. Hashemi E, Mahdavi H, Khezri J, Razi F, Shamsara M, Farmany A. Enhanced gene delivery in bacterial and mammalian cells using pegylated calcium doped magnetic nanograin. Int J Nanomedicine. 2019;14:9879–9891. doi:10.2147/IJN.S228396
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
