Back to Journals » Infection and Drug Resistance » Volume 15
Effects of General Anesthesia on Changes of Serum Hepatitis B Virus-DNA Levels in Infected Patients Underwent Non-Hepatobiliary Minimally Invasive Surgery: A Pilot Observational Study
Authors Quan J, Chen X , Tang X , Liu X, Li J, Yi B, Li P, Lu K, Gu J
Received 21 June 2022
Accepted for publication 21 October 2022
Published 11 November 2022 Volume 2022:15 Pages 6631—6640
DOI https://doi.org/10.2147/IDR.S379350
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Héctor Mora-Montes
Junxian Quan1 *, Xingtong Chen1 *, Xi Tang,1 Xianzhe Liu,1 Jieyu Li,1 Bin Yi,1 Peng Li,2 Kaizhi Lu,1 Jianteng Gu1
1Department of Anesthesiology, The First Affiliated Hospital, Third Military Medical University (Army Medical University), Chongqing, People’s Republic of China; 2Department of Pharmacognosy and Traditional Chinese Medicine, Third Military Medical University (Army Medical University), Chongqing, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Jianteng Gu; Kaizhi Lu, Department of Anesthesiology, The First Affiliated Hospital, Third Military Medical University (Army Medical University), 30 Gao Tanyan Street, Chongqing, 400038, People’s Republic of China, Tel +86 23 68766122, Fax +86 23 65463270, Email [email protected]; [email protected]
Objective: To investigate the effects of different general anesthesia methods on the changes of serum hepatitis B virus deoxyribonucleic acid (HBV-DNA) levels in the hepatitis B virus (HBV) infected patients.
Methods: This pilot prospective observational study was carried out from March 2021 to January 2022. Forty patients infected by HBV, who underwent non-hepatobiliary minimally invasive surgery, were allocated into an intravenous anesthesia group maintained with propofol (Group P, n = 20) and an inhalation anesthesia group maintained with sevoflurane (Group S, n = 20) by a random envelope method. Patient’s blood was drawn before operation (Tbaseline), at 24 hours (Tpost 24hrs) and 48 hours (Tpost 48hrs) after operation to detect the serum HBV-DNA levels and analyze the subset levels of T, B lymphocytes and NK cells (TBNK) in two groups. The serum HBV-DNA level was used as the major outcome, and it was analyzed by repeated-measures analysis of variance after natural logarithm transformation.
Results: In Group P and Group S, compared with the baseline, the serum HBV-DNA levels decreased significantly at Tpost 24hrs and Tpost 48hrs, P< 0.05. The total ratio of CD4+T cells and the ratio of CD4+T to CD8+T cells (CD4+/CD8+) were lower at Tpost 24hrs, P< 0.05, and the total ratio of B cells was significantly increased at Tpost 24hrs, P< 0.05. Compared with the values at Tpost 24hrs, the total ratio of CD4+ T cells, CD4+/CD8+ T cells, and the total ratio of B cells were significantly increased at Tpost 48hrs in both groups, P< 0.05. However, there were no statistical differences between Group P and Group S.
Conclusion: The levels of serum HBV-DNA decreased within 48 hours after general anesthesia. There were no significant differences between the effects of intravenous and inhalation anesthesia on the serum HBV-DNA levels.
Trial Registration: This study has been prospectively registered in the Clinical Trials Registry (NCT02038088, 1/28/2015).
Keywords: general anesthesia, hepatitis B virus deoxyribonucleic acid, lymphocyte subsets, immune function
Background
The modulation of general anesthesia on the immune function is a double-edged sword: it can relieve the inflammatory response by reducing pro-inflammatory cytokines secretion, as well as increasing the susceptibility of immunosuppression which may be associated with the recurrence and metastasis of tumors.1 The previous studies demonstrated that the immunosuppressive effects of general anesthesia were stronger than those of regional anesthesia.2–4 As an antigen, anesthetics can directly and indirectly affect the immune function. Propofol, a commonly used intravenous drug for induction and maintenance of general anesthesia, has been proved to promote the tissue infiltration abilities of natural killer (NK) cells and T helper cells (Th cell) without affecting T cell counts or leukocyte apoptosis.5 As one of fluoride inhalation anesthetics, sevoflurane can change the ratio between the Th cell subtype Th1 and Th2, and reduce the counts of circulatory lymphocytes and NK cells, thereby inhibiting the immune function.6 Other intravenous anesthetics, such as midazolam and opioid analgesics, may also orchestrate immune response by regulating the chemotaxis and phagocytosis of lymphocytes and macrophages, which are associated with the decrease of mitochondrial membrane potential, the inhibition of nuclear transcription factor activity, the attenuation of phosphorylation of p38 mitogen kinase and the increase of superoxide production.7,8
Hepatitis B virus (HBV) infection is one of the major public health problems worldwide, with approximately 30% population in the world having serological evidence of previous or current infection.9 A recent study indicated that the global prevalence of HBV infection had risen to 3.9%, and about 292 million people worldwide were affected by HBV.10 The epidemiological investigation of HBV in China showed that the rate of Hepatitis B Surface Antigen (HBsAg) carrier reached 7.18%, giving an estimation of over 93 million HBV carriers, of which 30 million were the chronic hepatitis B patients.11 At present, the detection of serum HBV-DNA levels has become a basic practice to determine the degree of virus replication and guide the formulation of treatment plans.12 The replication and clearance of HBV are closely related to the immune function.13 NK cells, monocytes, CD4+ and CD8+ T cells all play important roles during the HBV infection, among which virus-specific CD8+ T cells are more vital.14,15
It is assumed that the effects of general anesthesia on immune system may affect the HBV replication cycle, and thus influencing the strategy of perioperative HBV antiviral therapy. Therefore, this study aimed to investigate the effects of general anesthesia on serum HBV-DNA levels and the immune function in patients with chronic hepatitis B virus infection perioperatively.
Methods and Materials
General Information
Patients who underwent non-hepatobiliary minimally invasive surgery under general anesthesia in our hospital during March 2021 to January 2022 were selected as the research subjects for our pilot prospective observational study. According to the inclusion and exclusion criteria, after obtaining informed consent and anesthesia consent, the patients included in the study were divided into two groups by the random envelope method: an intravenous general anesthesia group (Group P, n = 20) and an inhalation general anesthesia group (Group S, n = 20).
This research protocol complies with the ethical guidelines of international clinical research trials and the Helsinki Standard, and it has been approved by the Ethics Committee of the First Affiliated Hospital of Army Medical University (KY2021035) and registered on the clinical trial website (NCT02038088).
Inclusion and Exclusion Criteria
Inclusion Criteria
(1) age is 18–65 years old; (2) ASA grade I–II; (3) voluntarily participate in the study and with the ability to comply with the study protocol; (4) HBsAg–positive and the HBV-DNA load is higher than the lower limit of detection (LLOD); (5) NYHA classification I–II, satisfied pulmonary function examined by pulmonary function tests and blood gas analysis [Vital capacity (VC) or Maximal voluntary ventilation (MVV) %>71, Forced expiratory volume in the first second (FEV1.0%)>61, SaO2%>94, PaO2>75mmHg, and PaCO2<45 mmHg], Child-Pugh modified grade A-B, and CKD stage 1–3; (6) without the history of hepatitis B vaccination; (7) the duration of general anesthesia is more than 1 hour.
Exclusion Criteria
(1) participating in other clinical trials; (2) received regular anti-HBV therapy or received antiviral therapy in the past; (4) combined with cancer; (5) taking immunomodulatory drugs in the past 2 weeks (6) for other reasons deemed inappropriate for this clinical trial by the investigators.
Drop-Out Cases
All patients who have filled out the informed consent form and are screened as eligible to enter the randomized trial, no matter when and why they drop out are defined as drop-out cases, for example ①with adverse events occurred; ②patients withdrawn voluntarily; ③violation of the trial protocol (including poor compliance); ④lost to follow-up after the surgery; ⑤surgery is cancelled.
Anesthesia Protocol
The same protocol was used for intubation and induction in the two groups: 0.5 μg/kg IV bolus sufentanil (Renfu Pharmaceutical Co., Ltd., Yichang, China); 0.03 mg/kg IV bolus midazolam (Renfu Pharmaceutical Co., Ltd., Yichang), 0.15 mg/kg IV bolus cis-atracurium (Dongying Pharmaceutical Co., Ltd., Shanghai PharmaP, China), 0.3 mg/kg IV bolus etomidate (Enhua Pharmaceutical Co., Ltd. Company, Jiangsu, China). The anesthesia maintenance intraoperatively in Group P: target-controlled infusion (TCI) propofol 2–3 ug/mL (Corden Pharma SPA, German), TCI remifentanil 3–4 ng/mL (Renfu Pharmaceutical Co., Ltd., Yichang, China), cis-atracurium 0.1 mg/kg IV per 30–40 minutes; In Group S, anesthesia was maintained by inhalation of sevoflurane (Maruishi Pharmaceutical Co., Ltd., Japan) at 1.0–1.7 minimum alveolar concentration (MAC), while the dose and methods of remifentanil and cis-atracurium administration were the same as those in Group P. During the operation, the Bispectral Index (BIS) was maintained between 40 and 60, and the hemodynamic stability was maintained at 10% fluctuations of the basic value. The patient was sent back to the general ward for further specialist treatment after the surgery.
Observation Indicators
Serum HBV-DNA level was used as the major outcome, and the distributions of lymphocyte subsets of T, B lymphocytes and NK cells (TBNK) were used as the secondary outcomes. Perioperative clinical data were used as other outcomes.
5mL venous blood was collected from patients before surgery (Tbaseline), 24 hours after surgery (Tpost 24hrs), and 48 hours after surgery (Tpost 48hrs), of which 3mL was used to detect the level of HBV-DNA, and 2mL was used to detect the level of TBNK. Preoperative results of virus serological markers (HBsAg, HBsAb, HBeAg, HBeAb, and HBcAb) were collected from the medical record system. Serum HBV-DNA levels were measured by real-time quantitative polymerase chain reaction (RT-qPCR), and the independent technicians involved in the measurement were blinded to the groupings. After the harvested blood samples were centrifuged, 200 μL of serum samples were mixed with 450 μL of DNA extract. Then the mixture was preheated in a 100°C incubator for 10 min and then centrifuged in a refrigerated centrifuge. Finally, 20 μL of supernatant was aspirated and transferred to a PCR tube, and the detection was completed by a RT-qPCR amplifier [Bio-Rad CFX96 Deep Well Dx System; lower limit of detection (LLOD) = 50 IU/mL]. The distribution of lymphocyte subsets of T, B lymphocytes, and NK cells (TBNK) was examined by flow cytometry using the flow cytometer (FACSCalibur, Becton, Dickinson and Company, United States) and its corresponding detection kit. Viral serological markers (HBsAg, HBsAb, HBeAg, HBeAb, and HBcAb) were quantitatively measured by the standard commercial enzyme-linked immunosorbent assay (ELISA) method (Jianglai Biotechnology Co., Ltd., Shanghai, China). All operating procedures were carried out in strict accordance with the instructions of the detection instrument and detection kit.
Sample Size Estimation and Statistical Methods
Taking HBV-DNA level as the major outcome, the statistical analysis was carried out after the natural logarithmic transformation of the HBV-DNA level. In the preliminary trial of 3 patients who met the inclusion and exclusion criteria, the baseline level of ln (HBV-DNA, UI/mL) was 8.81±0.89, and the level of ln (HBV-DNA, UI/mL) at postoperative 24 hours was 6.64±1.23. PASS 15.0 software (PASS, NCSS, LLC) was used to calculate the sample size. The α value was set as 0.05 and the β value was set as 0.2. The test power was determined to be 1-β=0.8. Considering the dropout rate of 10%, the calculated sample size was 14 people with 7 patients in each group.
All the data collected in this study were analyzed using SPSS 23.0 software. Normally distributed measurement data were expressed as mean±standard deviation (SD), while non-normally distributed measurement data were expressed as median (interquartile range). The comparisons were examined by Student's t-test, Mann–Whitney test (non-parametric distribution), and repeated measures analysis of variance (ANOVA). The categorical data were expressed as n (%), and the differences between the two groups were examined by chi-square analysis or Fisher’s exact test. P<0.05 was considered statistically significant.
Results
During March 2021 and January 2022, a total of 43 patients met the criteria. However, 2 patients were excluded for refusal to participate and 3 patients dropped out halfway (1 for cancellation of the surgery and 2 for refusal to follow up after the surgery). This results in a total of 38 patients that were included and analyzed (Figure 1).
 |
Figure 1 The flow diagram of the patients’ enrollment. |
Basic Information and Perioperative Clinical Outcomes of the Two Groups
There were no significant differences in gender, age, body weight, American Society of Anesthesiologists (ASA) classification, anesthesia time, blood loss, type of surgery, post-operative infection, and qualitative analysis of hepatitis B antigen and antibody between the two groups, P>0.05. (Table 1)
 |
Table 1 Basic Information and Perioperative Clinical Outcomes of the Two Groups |
Changes of HBV-DNA Levels in the Two Groups
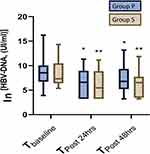
In Group P, the HBV-DNA level decreased from the baseline (8.74±2.95) to 6.66±3.00, P<0.05, at Tpost 24hrs, and to 7.21±2.86, P<0.05, at Tpost 48hrs. Accordingly, it decreased from 8.32±2.61 to 6.02±2.84, P<0.05, and to 6.57±2.87, P<0.05, in Group S. There was no significant difference between the two groups, P>0.05 (Figure 2).
Comparison of TBNK Levels in the Two Groups
At Tpost 24hrs, the levels of CD4+ T cells in the two groups decreased from the baseline value (Group P: 39.11% vs Group S: 41.82%) to (Group P: 34.26% vs Group S: 36.11%), P<0.05; then at Tpost 48 hrs, the levels of CD4+ T cells in both groups evaluated to the level around the baseline (Group P: 39.11% vs Group S: 41.82%) (Figure 3A).
The basal levels of CD4+/CD8+ were significantly decreased at Tpost 24hrs in Group P from 1.69±0.58 to 1.45±0.64, P<0.05; accordingly, in the Group S, CD4+/CD8+ decreased from 1.82±0.45 to 1.48±0.50, P<0.05. Then the ratio came back to the approximate level of the baseline at Tpost 48hrs (Figure 3B).
Compared with the preoperative level, the B cells in Group P increased to 15.89% and 17.42% at Tpost 24hrs and Tpost 48hrs, respectively, P<0.05; in Group S, it increased to 14.82% and 16.29% accordingly, P<0.05 (Figure 3C).
Compared with the level at Tpost 24hrs (Group P: 18.21% vs Group S: 17.71%), NK cell level decreased from 18.21% to 15.47% in Group P and reduced from 17.71% to 13.18% in Group S at Tpost 48hrs, P<0.05 (Figure 3D).
There were no significant differences in CD8+ T cells and total T cells percent between and within groups at each time point, P>0.05 (Figure 3E and F).
Discussion
HBV is a very stealth virus. If initial exposure to HBV fails to induce a significant innate immune response and immunosuppressive microenvironment in hepatocytes, it can lead to incomplete clearance of HBV and chronic infection.16 Stresses caused by surgery and pain are associated with immunosuppression,17 and each class of anesthetics has the potential to affect both innate and adaptive immunity.18 Previous studies have suggested that the volatile agents, isoflurane and sevoflurane, can alter innate immunity by reducing NK cell cytotoxicity; whereas propofol may have a mix impact on cells of the innate immune system, especially macrophage cells and NK cells. Exogenous opioids, such as morphine and fentanyl, can impair the function of macrophages, NK cells, and T cells, and can weaken the gut barrier, thus inhibiting immune response.18,19 Therefore, HBV carriers who underwent general anesthesia may experience suppressed or unaffected body immune function, and, in theory, may increase or maintain the serum level of HBV-DNA. To verify the theoretical derivation, this pilot observational study was conducted to study the effects of different general anesthesia methods on the changes in serum HBV-DNA levels. In the study design, to control confounding factors as much as possible, this study avoided the inclusion of hepatobiliary patients and tumor patients, and selected the patients who did not receive the hepatitis B vaccine, anti-HBV drugs, and immunomodulators as the research objects. Taking surgical-related factors into account,1 the minimal invasive surgeries were selected into this study to minimize the effect of intraoperative infusion on serum HBV levels. However, the final results of this study deviated from the theoretical deduction. This study is the first to report that within 48 hours after general anesthesia, the serum HBV-DNA level was significantly lower than that before surgery, and it was observed that both intravenous and inhalation of general anesthesia led to the decrease in serum HBV-DNA level after the operation. In addition, there was no significant difference between two general anesthesia methods on serum HBV-DNA level.
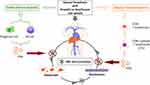
The results of this study suggest that the immune clearance of HBV is transiently enhanced after general anesthesia. The putative mechanism may be as follows: First, as an antigen, anesthetic drugs stimulate some immune cells in a short period, which in turn leads to a transient enhancement of immune clearance of hepatitis B virus. The immune clearance follows the body’s immune response that heavily relies on CD4+ T and CD8+ T cells, where CD4+T cells induce and maintain the activity of CD8+T cells, and CD8+T cells can kill the infected cells and inhibit the proliferation of the virus. Moreover, CD8+T, CD4+T, NK cells and other immune cells secrete antiviral cytokine gamma interferon (INF-γ) that inhibit virus replication.20 In addition, studies have shown that propofol can up-regulate the expression of INF-γ by inhibiting the production of prostaglandin E2 (PGE2)21 and can also significantly inhibit the expression of the viral protein and further inhibit the release of viral particles.22 Inhalation anesthetic sevoflurane can also increase the expression of INF-γ in mouse serum.23 Therefore, the increased expression of INF-γ may also be an important reason for the reduction of serum HBV-DNA level after general anesthesia. Second, anesthetics are mostly metabolized in the liver, which may cause changes in liver cell function and may disrupt the balance of HBV-DNA level. HBV-DNA in serum is at a dynamic equilibrium, and its main source is virions containing relaxed circular DNA (rcDNA) released by infected hepatocytes. The virions can exist either in the blood circulation or return to hepatocytes to replenish the covalently closed circular (cccDNA) pool.24 Metabolism of anesthetics in hepatocytes results in the activation of hepatocyte function. This may inhibit the release of rcDNA-containing virus particles from infected hepatocytes and hence subsequently slow down the replication of HBV within hepatocytes. As a result, the balanced HBV level in and out of hepatocytes is disrupted, leading to a rapid transfer of serum HBV-DNA into the hepatocytes and thus temporarily reducing the serum HBV-DNA level (Figure 4). The above two hypotheses are rough and require more detailed research for further verification.
To control confoundings, we strictly control the use of opioids in the two groups to ensure that the methods and doses of use were comparable. This could be the reason why the differences in the HBV-DNA level were not statistically significant between two groups. However, in this study, the temporary enhancement of immune clearance of HBV after general anesthesia is inconsistent with the conclusion that opioids suppress immune function in vitro and in animal studies, indicating that the impact of opioids on the immune system still requires further clinical research to clarify.
This study still has the following limitations: First, our research on the relevant immune components is not in detail, especially the investigation of CD8+ T cells is insufficient, and we had not detected whether they were activated to form cytotoxic T cells and play a role in the clearance of HBV.25 Second, there was no monitoring of perioperative changes in INF-γ levels in patients, and there was a lack of relevant evidence for the underlying mechanism of the decline in serum HBV-DNA levels. Third, we only studied the HBV viral load in the blood but not the HBV viral load in hepatocytes. Fourth, the follow-up time was short, and the changes in serum levels of HBV-DNA were not studied for a longer time.
Conclusion
Within 48h after the general anesthesia, serum HBV-DNA levels decreased while CD4+ T cells and CD4+/CD8+ levels decreased significantly at first, and then returned to normal levels. There were no significant differences between the two general anesthesia, intravenous anesthesia and inhalation anesthesia, on postoperative HBV-DNA levels and TBNK levels.
Abbreviations
ANOVA, analysis of variance; ASA, American Society of Anesthesiologists; BIS, Bispectral Index; cccDNA, covalently closed circular deoxyribonucleic acid; rcDNA, relaxed circular deoxyribonucleic acid; FEV1.0%, Forced expiratory volume in the first second; HBV, hepatitis B virus; HBV-DNA, hepatitis B virus deoxyribonucleic acid; LLOD, lower limit of detection; MAC, minimum alveolar concentration; MVV, Maximal voluntary ventilation; NK cell, natural killer cell; SD, standard deviation; TBNK, T, B lymphocytes and NK cells; Th cell, T helper cell; VC, Vital capacity.
PI Statement
The authors confirm that the PI for this paper is Jianteng Gu, who had direct clinical responsibility for patients.
Data Sharing Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Consent to Participate
The study was approved by the Ethics Committee of the First Affiliated Hospital of Army Medical University (KY2021035), and the written informed consents were obtained from all patients participated in this study. This study has been prospectively registered in the Clinical Trials Registry (NCT02038088, 1/28/2015). All methods performed in this study were in accordance with the relevant guidelines and regulations. All procedures performed in this study involving human participants were in accordance with the Ethical Standards of the Institutional Ethics Committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent for Publication
All authors have read and approved the manuscript in its current state and are all approved for publication.
Acknowledgments
The authors would like to thank Qian Chen and Ryan Wong, Division of Anaesthetics, Pain Medicine & Intensive Care, Imperial College London for their dedicated efforts and valuable contributions to the manuscript revision. We appreciate the support from the Excellent Talents Foundation of Army Medical University (XZ-2019-505-028) and the National Key R&D Program of China (No. 2018YFC0116700 & No. 2018YFC0116704).
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This study was supported by the Excellent Talents Foundation of Army Medical University (XZ-2019-505-028) and the National Key R&D Program of China (No. 2018YFC0116700 & No. 2018YFC0116704). The funding agencies had no direct role in the conduct of the study; the collection, management, analyses, or interpretation of the data; or preparation or approval of the manuscript.
Disclosure
The authors declare that they have no competing interests.
References
1. Rossaint J, Zarbock A. Anesthesia-induced immune modulation. Curr Opin Anaesthesiol. 2019;32(6):799–805. doi:10.1097/ACO.0000000000000790
2. Chen YH, Rau RH, Keller JJ, et al. Possible effects of anaesthetic management on the 1 yr followed-up risk of herpes zoster after caesarean deliveries. Br J Anaesth. 2012;108(2):278–282. doi:10.1093/bja/aer386
3. Tsai PS, Hsu CS, Fan YC, et al. General anaesthesia is associated with increased risk of surgical site infection after caesarean delivery compared with neuraxial anaesthesia: a population-based study. Br J Anaesth. 2011;107(5):757–761. doi:10.1093/bja/aer262
4. Kopp SL, Berbari EF, Osmon DR, et al. The impact of anesthetic management on surgical site infections in patients undergoing total knee or total hip arthroplasty. Anesth Analg. 2015;121(5):1215–1221. doi:10.1213/ANE.0000000000000956
5. Lim JA, Oh CS, Yoon TG, et al. The effect of propofol and sevoflurane on cancer cell, natural killer cell, and cytotoxic T lymphocyte function in patients undergoing breast cancer surgery: an in vitro analysis. BMC Cancer. 2018;18(1):1–8. doi:10.1186/s12885-018-4064-8
6. Pirbudak Cocelli L, Ugur MG, Karadasli H. Comparison of effects of low-flow sevoflurane and desflurane anesthesia on neutrophil and T-cell populations. Curr Ther Res Clin Exp. 2012;73(1–2):41–51. doi:10.1016/j.curtheres.2012.02.005
7. Ninković J, Roy S. Role of the mu-opioid receptor in opioid modulation of immune function. Amino Acids. 2013;45(1):9–24. doi:10.1007/s00726-011-1163-0
8. Horiguchi Y, Ohta N, Yamamoto S, et al. Midazolam suppresses the lipopolysaccharide-stimulated immune responses of human macrophages via translocator protein signaling. Int Immunopharmacol. 2019;66(December 2018):373–382. doi:10.1016/j.intimp.2018.11.050
9. Trépo C, Chan HLY, Lok A. Hepatitis B virus infection. Lancet. 2014;384(9959):2053–2063. doi:10.1016/S0140-6736(14)60220-8
10. Nguyen MH, Wong G, Gane E, et al. Hepatitis B virus: advances in prevention, diagnosis, and therapy. Clin Microbiol Rev. 2020;33(2):1–38. doi:10.1128/CMR.00046-19
11. Wang H, Men P, Xiao Y, et al. Hepatitis B infection in the general population of China: a systematic review and meta-analysis. BMC Infect Dis. 2019;19(1):811. doi:10.1186/s12879-019-4428-y
12. Loomba R, Liang TJ. Hepatitis B reactivation associated with immune suppressive and biological modifier therapies: current concepts, management strategies, and future directions. Gastroenterology. 2017;152(6):1297–1309. doi:10.1053/j.gastro.2017.02.009
13. Meng Z, Chen Y, Lu M. Advances in targeting the innate and adaptive immune systems to cure chronic hepatitis B virus infection. Front Immunol. 2020;10(February):1–16. doi:10.3389/fimmu.2019.03127
14. Rehermann B. Pathogenesis of chronic viral hepatitis: differential roles of T cells and NK cells. Nat Med. 2013;19(7):859–868. doi:10.1038/nm.3251
15. Peeridogaheh H, Meshkat Z, Habibzadeh S, et al. Current concepts on immunopathogenesis of hepatitis B virus infection. Virus Res. 2018;245(October 2017):29–43. doi:10.1016/j.virusres.2017.12.007
16. Koffas A, Kumar M, Gill US, et al. Chronic hepatitis B: the demise of the ‘inactive carrier’ phase. Hepatol Int. 2021;15(2):290–300. doi:10.1007/s12072-021-10137-2
17. Kurosawa S. Anesthesia in patients with cancer disorders. Curr Opin Anaesthesiol. 2012;25(3):376–384. doi:10.1097/ACO.0b013e328352b4a8
18. Ackerman RS, Luddy KA, Icard BE, et al. The effects of anesthetics and perioperative medications on immune function: a narrative review. Anesth Analg. 2021;133(3):676–689. doi:10.1213/ANE.0000000000005607
19. Plein LM, Rittner HL. Opioids and the immune system - friend or foe. Br J Pharmacol. 2018;175(14):2717–2725. doi:10.1111/bph.13750
20. Guidotti LG, Isogawa M, Chisari FV. Host-virus interactions in hepatitis B virus infection. Curr Opin Immunol. 2015;36:61–66. doi:10.1016/j.coi.2015.06.016
21. Inada T, Kubo K, Shingu K. Promotion of interferon-gamma production by natural killer cells via suppression of murine peritoneal macrophage prostaglandin E2 production using intravenous anesthetic propofol. Int Immunopharmacol. 2010;10(10):1200–1208. doi:10.1016/j.intimp.2010.06.027
22. Shen TJ, Chen CL, Jhan MK, et al. Antiviral efficacy of the anesthetic propofol against dengue virus infection and cellular inflammation. J Immunol Res. 2021;2021:1–8. doi:10.1155/2021/6680913
23. Wang L, Zha B, Shen Q, et al. Sevoflurane inhibits the Th2 response and NLRP3 expression in murine allergic airway inflammation. J Immunol Res. 2018;2018:1–8. doi:10.1155/2018/9021037
24. Tu T, Budzinska MA, Shackel NA, et al. HBV DNA integration: molecular mechanisms and clinical implications. Viruses. 2017;9(4):75. doi:10.3390/v9040075
25. Reina-Campos M, Scharping NE, Goldrath AW. CD8+ T cell metabolism in infection and cancer. Nat Rev Immunol. 2021;21(11):718–738. doi:10.1038/s41577-021-00537-8
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.