Back to Journals » International Journal of Nanomedicine » Volume 9 » Issue 1
Effective inhibition of colon cancer cell growth with MgAl-layered double hydroxide (LDH) loaded 5-FU and PI3K/mTOR dual inhibitor BEZ-235 through apoptotic pathways
Authors Chen J, Shao R, Li L, Xu ZP , Gu W
Received 30 January 2014
Accepted for publication 29 March 2014
Published 16 July 2014 Volume 2014:9(1) Pages 3403—3411
DOI https://doi.org/10.2147/IJN.S61633
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Jiezhong Chen,1,2 Renfu Shao,3 Li Li,4 Zhi Ping Xu,4 Wenyi Gu4
1School of Biomedical Sciences, University of Queensland, St Lucia, Queensland, 2Faculty of Science, Medicine and Health, University of Wollongong, Wollongong, New South Wales, 3GeneCology Research Centre, Faculty of Science, Health, Education and Engineering, University of the Sunshine Coast, Maroochydore, Queensland, 4Australian Institute of Bioengineering and Nanotechnology, University of Queensland, St Lucia, Queensland, Australia
Abstract: Colon cancer is the third most common cancer and the third largest cause of cancer-related death. Fluorouracil (5-FU) is the front-line chemotherapeutic agent for colon cancer. However, its response rate is less than 60%, even in combination with other chemotherapeutic agents. The side effects of 5-FU also limit its application. Nanoparticles have been used to deliver 5-FU, to increase its effectiveness and reduce side effects. Another common approach for colon cancer treatment is targeted therapy against the phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt) pathway. A recently-invented inhibitor of this pathway, BEZ-235, has been tested in several clinical trials and has shown effectiveness and low side effects. Thus, it is a very promising drug for colon cancer treatment. The combination of these two drugs, especially nanoparticle-packed 5-FU and BEZ-235, has not been studied. In the present study, we demonstrated that nanoparticles of layered double hydroxide (LDH) loaded with 5-FU were more effective than a free drug at inhibiting colon cancer cell growth, and that a combination treatment with BEZ-235 further increased the sensitivity of colon cancer cells to the treatment of LDH-packed 5-FU (LDH-5-FU). BEZ-235 alone can decrease colon cancer HCT-116 cell viability to 46% of the control, and the addition of LDH-5-FU produced a greater effect, reducing cell survival to 8% of the control. Our data indicate that the combination therapy of nanodelivered 5-FU with a PI3K/Akt inhibitor, BEZ-235, may promise a more effective approach for colon cancer treatment.
Keywords: layered double hydroxide (LDH), BEZ-235, 5-FU, colon cancer HCT-116 cells, cell death, apoptosis, PI3K/Akt/mTOR
Introduction
Colon cancer is the third most common cancer in humans and has a high mortality rate. The five-year survival rate for metastatic colon cancer is less than 10%. Many therapeutic approaches have been used for the treatment of colon cancer, including chemotherapy, radiotherapy, targeted therapy, and immune therapy. Nanoparticles (NPs) have been used to control drug release. For example, polymer microsphere delivery allows slower and delayed drug release of valdecoxib, an inhibitor of cyclooxyygenase-2 enzyme.1 Nanodelivery can also avoid the release of drug at pH 1.2–6.8, so that the drug is not released in the stomach, ensuring colonic release.2 Fluorouracil is the drug of first choice in chemotherapy for colon cancer. However, its response rate is less than 60%, even in combination with other chemotherapeutic agents.3 Drug resistance is a major issue in the clinical application of 5-FU.4 In addition, the severe side effects of 5-FU limit its clinical application at high dosages. The bioavailability of 5-FU is poor and its metabolism is rapid. Thus, an effective delivery mechanism for 5-FU is needed for clinical application.5 In order to increase the effectiveness of 5-FU and reduce its side effects, several approaches have been employed, including combinatorial application with other anticancer agents and the application of nanotechnology for its delivery.
Recently, several types of NPs have been used to deliver 5-FU to increase treatment efficacy and reduce side effects, including layered double hydroxide (LDH), liposomes and silica-based NPs.6–9 LDH is an inorganic NP with low toxicity, which has been used to deliver chemotherapeutic agents, including 5-FU.10–13 Delivery by NPs can increase treatment efficacy and reduce side effects, as NP-5-FU can accumulate in tumor sites (due to vascular structural differences between the tumor and normal tissues). Nanoparticle-loaded drugs can also be taken up by cells through endocytosis. Therefore, their concentrations in cells are increased.14 LDH-packed 5-FU has been used together with curcumin and AllStars Hs Cell Death small interfering RNAs (siRNAs) to increase treatment efficacy,5,15 but has not been used with other anticancer agents, such as BEZ-235.
5-FU has been used most often together with oxaliplatin, to increase patient response to 5-FU.16,17 Furthermore, folic acid has been included in a treatment regime containing 5-FU and oxaliplatin. The chemotherapeutic agents irinotecan and leucovorin have been added to the combinational therapeutic regime with 5-FU.18,19 In addition, 5-FU has been used in combination with immune therapy and targeted therapies against B-cell lymphoma 2 (Bcl-2), such as interferon-α, cetuximab, rapamycin, and siRNA.20–22 A phase III clinical trial showed that cetuximab significantly increased the effect of 5-FU in combination with the FOLFIRI regimen.23 Fluorouracil has also been used with a dual inhibitor of the phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt)/mammalian target of rapamycin (mTOR) pathway, PI-103.24
PI3K/Akt is a major survival pathway and plays a key role in many cancers, including colon cancer.25–28 Targeted therapy against the PI3K/Akt/mTOR pathway has been extensively studied in colon cancer. The pathway is associated also with resistance to chemotherapeutic drugs.29,30 BEZ-235 is a new and very promising inhibitor of the PI3K/Akt pathway (due to its effectiveness and low side effects). It can inhibit the activity of both PI3K and mTOR and thus eliminate the feedback activation of PI3K activity due to inhibition of mTOR. A recent study showed that BEZ-235 is effective on both PIK3CA-mutated and unmutated colon cancer cell lines.31 Therefore, BEZ-235 may soon be incorporated into the present treatment regime for colon cancer. However, combinational treatment with BEZ-235 and nanoparticle-delivered 5-FU, especially the additive or even synergistic effect of their treatment, has not been well studied. In the present study, we investigated the treatment effect of LDH-loaded 5-FU and BEZ-235 in the colon cancer cell line HCT-116. We found that the combination treatment could dramatically increase cancer cell death, both through cell cycle changes and apoptotic pathways.
Materials and methods
MgCl2 ·6H2O, AlCl3·6H2O, NaOH, 5-FU, Tween® 20, protein lysis buffer, and Triton® 100 were purchased from Sigma-Aldrich Corp (St Louis, MO, USA). Dulbecco’s Modified Eagle’s Medium (DMEM), PBS buffer, and Tris buffer were obtained from Life Technologies Corporation (Mulgrave, Victoria, Australia). BEZ-235 was obtained from Cayman Chemical Company (Ann Arbor, MI, USA). The Annexin V-FITC Apoptosis detection kit was obtained from BD Biosciences (Franklin Lakes, NJ, USA). Mouse monoclonal antihuman Bcl-2 antibody and rabbit antihuman α-tubulin antibody were obtained from Cell Signaling Technology Inc (Danvers, MA, USA).
Preparation of LDH and LDH-5-FU
LDH was made by a standard method.5,12,13 The 5-FU/Mg2Al-LDH hybrid (LDH-5-FU) was synthesized via ion exchange methods with a hydrothermal treatment. The synthetic procedure involved mixing 0.3 M MgCl2 and 0.1 M AlCl3 solution before quickly adding it to 0.15 M NaOH solution and stirring for 10 minutes. Then, LDH slurry was collected by centrifugation. The precipitation was resuspended into 40 mL of the basic solution containing 0.2 mm of 5-FU (neutralized with dilute NaOH solution, with pH 8–9) for 1 hour. After washing two times, the LDH-5-FU was resuspended in 40 mL of distilled water and transferred to an autoclave (stainless steel with a teflon lining) and incubated at 100°C for 16 hours. After hydrothermal treatment, a transparent, homogeneous suspension of LDH-5FU was obtained.
Physicochemical characterization
The hydrodynamic particle size and the zeta potential of the LDH and LDH-5-FU nanohybrids were determined by dynamic light scattering (DLS). All measurements were carried out at 25°C with photon correlation spectroscopy using a Zetasizer Nano ZS (Malvern Instruments Ltd, Malvern, UK). Each parameter was measured in triplicate and the average value was calculated. Transmission electron microscopy (TEM) images were obtained using a JEM-1010A (JEOL, Tokyo, Japan) at an acceleration voltage of 100 kV. The loading amount of 5-FU into LDH was determined by ultraviolet-visible absorbance (UV-Vis) at 265 nm.
Cell lines and cell viability test: MTT assay
The colon cancer HCT-116 cell line was purchased from American Type Culture Collection (ATCC®) (Manassas, VA, USA) and the cells were maintained in complete DMEM, supplemented with 10% fetal calf serum, 1% penicillin, and 1% streptomycin, in 75 mL flasks at 37°C and 5% CO2. For the cytotoxicity assay, cells were trypsinized for 5 minutes and resuspended in a medium to dilute the concentration to 1×105/mL. Then 100 μL of cells was added to each well in a 96-well plate for the cytotoxicity assay, under treatments of various concentrations of BEZ-235 and LDH-5-FU. An MTT assay was used to measure cell viability, according to the manufacturer’s (Sigma-Aldrich, St louis; MO) instructions.
Cell cycle analysis
Propidium iodide (PI) was used to stain the DNA content. HCT-116 cells were harvested by trypinization and were washed with phosphate buffered saline (PBS). The cells were resuspended in PBS at 2×106/mL. With 1 mL of the resuspended cells, 1 mL of 100% cold ethanol was added dropwise and vortexed gently. The mixture was stored overnight at −20°C. After washing the cells with cold PBS, they were resuspended in a 300 μL PI/Triton buffer containing 0.01% Triton 100 (by volume) and 6 μg PI. After incubation at room temperature for 40 minutes, the stained cells were analyzed with flow cytometry (Accuri™, BD Biosciences, Franklin Lakes, NJ, USA) and (FlowJo software, Tree Star Inc, Ashland, OR, USA).
Cell apoptosis analysis
Cell apoptosis and cell death were detected using the Annexin V apoptosis kit, following the procedure recommended by the manufacturer (BD; Bioscienes, Franklin Lakes, NJ, USA). HCT-116 cells were cultured in a 6-well plate with 2×105 cells in each well. Cells were treated for 24 hours with various concentrations of BEZ-235 ranging from 25–400 nm, and LDH-5-FU from 3–12 μg/mL. As well, a combinational treatment of BEZ-235 and LDH-5-FU was used. Treated cells were collected by centrifugation and suspended in 100 μL of the provided buffer and stained with Annexin V-FITC conjugates and PI for 15 minutes. Cell death and apoptosis were then detected by flow cytometry.
Data analysis
Data collected from each group (experimental and control) were expressed as mean ± standard deviation. The one-way ANOVA and unpaired Student’s t-test (Prism version 6, GraphPad Software Inc, La Jolla, CA, USA) were used to analyze the differences between groups and discriminate the significant differences between experimental and control groups (one-way ANOVA: P<0.05).
Results
Characterization of LDH and LDH-5-FU
As shown in Figure 1A, the synthesized LDH suspension had platelike particles with a hexagonal shape. The DLS data in Figures 1C and 1D indicated that the average size of LDH nanoparticles was ~109 nm with a zeta potential of +42.9 mV, consistent with our previous reports.15 After loading 5-FU into LDH nanoparticles, LDH-5-FU nanohybrids presented a narrow particle size distribution in the range of 50–150 nm and a zeta potential of +40.6 mV (Figures 1C and 1D). Compared with LDH, the average size of LDH-5-FU decreased from 109 nm to 85 nm, as reported previously,15 due to inhibition of the hydroxide layer growth by the anionic 5-FU. The TEM image in Figure 1B shows that the LDH-5-FU nanohybrid had a similar platelike morphology to the LDH nanoparticle. The amount of 5-FU loaded in LDH nanoparticles was 9.6% by weight, determined by UV-Vis.
Cytotoxicity induced by 5-FU, LDH-5-FU, and BEZ-235
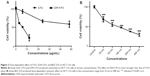
We compared the cytotoxicity of LDH-5-FU with free 5-FU on the colon cancer cell line HCT-116. In Figure 2A, it can be seen that both 5-FU and LDH-5-FU produced a dose-dependent cytotoxic effect on HCT-116 cells after 24 hour treatment. LDH-5-FU has much higher cytotoxicity than free 5-FU. It started to show the effect at 3 μg/mL, with an increased effect at 6 μg/mL, and the number of surviving cells decreased to about 20% at 12 μg/mL. The half maximal inhibitory concentration (IC50) for the inhibitory effect of LDH-5-FU on HCT-116 is 5.585 μg/mL. The free 5-FU had much lower cytotoxicity and it only reduced cell viability to 56%, even at 48 μg/mL. We chose 6 μg/mL for the study because of its combinatorial effect with BEZ-235.
In order to select a dose of BEZ-235 to test the effect of combination treatment of LDH-5-FU and BEZ-235, the effect of BEZ-235 on HCT-116 cells was tested at different doses. After 24 hour incubation, BEZ-235 produced dose-dependent cytotoxicity on HCT-116 cells in the dose range from 50–400 nm (Figure 2B). IC50 for the inhibitory effect of BEZ-235 on HCT-116 cells is 78.2 nm. We chose the concentration of 100 nm for the combination treatment.
More effective inhibition by the combination of LDH-5-FU and BEZ-235
Figure 3 showed that LDH-5-FU and BEZ-235 together produced a higher cytotoxic effect. It is shown that both LDH-5-FU and BEZ-235 alone produced a middle range of cytotoxicity to HCT-116 cells. Treatment with BEZ-235 alone at 100 nm decreased cell survival to 46% of the control. Treatment with LDH-5-FU at 6 μg/mL alone decreased the survival rate to 56%. However, the combination use of the two drugs resulted in massive cell death, reducing survival rate to 8% of the control. The cell survival rate was significantly lower than both LDH-5-FU and BEZ-235 treatments alone. Therefore, BEZ-235 increases the sensitivity of HCT-116 to LDH-5-FU treatment. These data clearly show that a combination treatment with BEZ-235 and LDH-5-FU remarkably suppresses HCT-116 cell growth.
Combination treatment of LDH-5-FU and BEZ-235 caused more G1 arrest
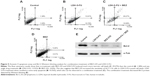
It was reported that BEZ-235 could induce cell cycle G1 arrest in the prostate cancer cell line PC3M.32 To examine whether combination treatment with LDH-5-FU and BEZ-235 has the same effect, we carried out a cell cycle analysis. The result showed that the combination treatment remarkably increased the Sub-G1 (apoptotic) population (51.85%) (Figure 4), compared with single drug treatments (16.75% and 23.05%, for LDH-5-FU and BEZ-235, respectively) and the untreated control (Figure 4). Instead of G1 arrest, we observed a remarkable decrease in the G1 phase and an increase of population at the G2/Mitotic (G2/M) phase after the combination treatment (Figure 4).
LDH-5-FU and BEZ-235 caused cell death and apoptosis
We further assayed the combination treatment effect of LDH-5-FU and BEZ-235 on cell death using the Annexin V apoptosis kit. As shown in Figure 5, both LDH-5-FU and BEZ-235 produced cell death and apoptosis after 24 hour treatment. LDH-5-FU alone, at 6 μg/mL, resulted in 28% cell apoptosis and death. Treatment with BEZ-235 alone, at 100 nm, led to 9% cell apoptosis and cell death. However, the combinatorial application of both agents produced a greater effect, resulting in 46% cell apoptosis and death. Compared with the control, the LDH-5-FU treatment caused some early apoptosis (10.76% versus 1.09%) (Figures 5A and 5B) and this effect was carried on into the combination treatment (10.68%) (Figure 5C). BEZ-235 treatment alone did not induce much early apoptosis (Figure 5D). In addition, LDH-5-FU (Figure 5B) and combination treatments (Figure 5C) caused much more necrotic cell death (right upper quarters) than either BEZ-235 alone (Figure 5D) or the control (Figure 5A). We further analyzed Bcl-2 protein levels (an antiapoptotic protein associated with mitochondria apoptosis pathways) and found that treatment with a single drug led to the reduction of this protein. The most significant reduction was seen in the combination treatment (Figure 5E), which is consistent with the above results.
Discussion
Fluorouracil is a common chemotherapeutic agent used in the treatment of colon cancer. It is a suicide inhibitor of thymidylate synthase, an enzyme necessary for DNA replication.33,34 Fluorouracil is very toxic to normal cells and causes severe side effects at the therapeutic dosage, including damage to bone marrow, the gastrointestinal tract, and skin.35,36 To reduce side effects, its dose must be decreased through combinational application with other drugs or drug delivery systems. In this study, we used LDH to lower the dose of 5-FU, at which higher cytotoxicity to cancer cells still remained. We also tested the combination treatment of LDH-loaded 5-FU with BEZ-235.
Nanoparticles have been extensively used for drug delivery, to increase drug efficacy and reduce side effects. In our study, we showed that nanodelivered 5-FU increased cytotoxicity to HCT-116 markedly. This may be due to the endocytosis effect. In this process, LDH-5-FU is engulfed by HCT-116 cells as a particle, which contains more 5-FU molecules than free 5-FU.37 This process facilitates entry of 5-FU into cells and therefore results in higher cytotoxicity. So far, LDH-5-FU has only been tested with curcumin as a combinational treatment on cancer cells, where LDH-5-FU has been shown to have a synergistic effect with solid lipid nanoparticle-curcumin.5 In our study, we tested the effect of LDH-5-FU on HCT-116 cells in combination with BEZ-235, a newly-invented PI3K/Akt inhibitor for colon cancer treatment.
The rationale behind applying 5-FU and a PI3K/Akt inhibitor is based on the effectiveness of inhibition of the pathway in colon cancer treatment.38–40 We have shown that activation of the PI3K/Akt pathway can reduce the effect of chemotherapeutic agents, including 5-FU, on colon cancer cell lines.41,42 Inhibition of the pathway increased the sensitivity of HT-29 cells to 5-FU. Leptin has also been shown to increase colon cancer resistance to 5-FU treatment, via activation of the PI3K/Akt pathway.43 Chen et al showed that siRNA knockdown of focal adhesion kinase (FAK) reduced Akt activity and increased colon cancer sensitivity to 5-FU.44 Recently, Liao et al showed that regular use of aspirin increased the survival time of colorectal cancer patients with the mutated PIK3CA gene but not of those with the wild-type PIK3CA.45 The effectiveness of BEZ-235 has been extensively tested in PIK3CA-mutated colon cancer cell lines.31 Therefore, it is useful for the combination study with LDH-5-FU.
Our study shows for the first time that the dual inhibitor of the PI3K/Akt/mTOR pathway, BEZ-235, can sensitize HCT-116 cells to LDH loaded with 5-FU. This is a novel approach for the treatment of colon cancer. HCT-116 cells have a mutation in PIK3CA. Therefore, their PI3K pathway is highly-activated and thus is sensitive to inhibition of the pathway. BEZ-235 has been used for several clinical trials in colon cancer treatment. The approach we tested could be applicable for clinical trials, as both BEZ-235 and 5-FU are approved for clinical use as anticancer agents. BEZ-235 is known to have minor side effects. Several studies showed that it is well-tolerated in clinical trials. 5-FU is a traditional therapy drug for colon cancer. LDH is also a low toxicity nanoparticle. In our study, the concentration of 5-FU is 300 μg/mL and the concentration of LDH is 4 mg/mL. At the concentration of 6 μg/mL of 5-FU, the concentration of LDH is 80 μg/mL. At this concentration of LDH, it has no toxicity to normal cells.
Muller et al studied the effect of BEZ-235 and irinotecan on HT-29 cells and showed that BEZ-235 increased the effect of irinotecan.46 This study indicated that inhibition of the PI3K/Akt/mTOR pathway can be combined with a chemotherapeutic agent to increase treatment efficacy in colon cancer. However, only irinotecan was tested. 5-FU and other chemotherapeutic agents commonly used in colon cancer treatment were not tested.
A dual inhibitor of the PI3K/Akt/mTOR pathway, PI-103, has been shown to increase the efficacy of 5-FU in both in vitro and in vivo studies in gastric cancer.24 In comparison with single inhibitors of PI3K and mTOR, PI-103 produced an enhanced effect. PI-103 is the first-identified dual inhibitor of the PI3K and mTOR pathways. Although it has a strong antitumor effect, it is not suitable for clinical application, due to its high toxicity.47,48 In contrast, BEZ-235 has been extensively tested in clinical trials for many types of cancers, and has demonstrated high effectiveness and low toxicity.39,40,49–52 In the present study, we applied BEZ-235 in colon cancer HCT-116 in combination with LDH-5-FU to demonstrate their combination effect.
Our study has partially elucidated the mechanisms for the combination effect of BEZ-235 and LDH-5-FU. In cell cycle analysis, the combination treatment produced many more sub-G1 apoptotic cells than BEZ-235 or LDH-5-FU alone, indicating the additive effect on cell death of the two drugs. Cell cycle arrest was observed in the G2/M phase, with a remarkable decrease of the G1 phase. This differs from another study, which showed that BEZ-235 caused G1 arrest in PC3M cells.32 The difference could be caused by the different cell types and drug doses used. In that study, 10 nm and 50 nm of BEZ-235 were used. The combination treatment produced more reduction in Bcl-2. Bcl-2 is an antiapoptotic protein and its reduction could lead to increased apoptosis. Therefore, both cell proliferation and cell apoptosis are affected by the combination treatment. Further studies are warranted, so that the combination application can be used for clinical trials.
Acknowledgment
The authors would like to acknowledge the funding support of the Australian Research Council to Renfu Shao and Wenyi Gu (Project Id: DP120100240).
Disclosure
The authors report no conflicts of interest in this work.
References
Thakral NK, Ray AR, Bar-Shalom D, Eriksson AH, Majumdar DK. The quest for targeted delivery in colon cancer: mucoadhesive valdecoxib microspheres. Int J Nanomedicine. 2011;6:1057–1068. | ||
Thakral NK, Ray AR, Majumdar DK. Eudragit S-100 entrapped chitosan microspheres of valdecoxib for colon cancer. J Mater Science Mater Med. 2010;21(9):2691–2699. | ||
Tournigand C, André T, Achille E, et al. FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol. 2004;22(2):229–237. | ||
De Angelis PM, Svendsrud DH, Kravik KL, Stokke T. Cellular response to 5-fluorouracil (5-FU) in 5-FU-resistant colon cancer cell lines during treatment and recovery. Mol Cancer. 2006;5:20. | ||
Liu W, Li X, Wong YS, et al. Selenium nanoparticles as a carrier of 5-fluorouracil to achieve anti-cancer synergism. ACS Nano. 2012;6(8)6578–6591. | ||
Wang J-M, Xiao B-L, Zheng J-W, Chen H-B, Zou S-Q. Effect of targeted magnetic nanoparticles containing 5-FU on expression of bcl-2, bax and caspase 3 in nude mice with transplanted human liver cancer. World J Gastroenterol. 2007;13(23):3171–3175. | ||
Hu YX, Chang J, Guo Y, Yuan XB, Kang CS, Pu P. Preparation and evaluation of 5-FU/PLGA/gene nanoparticles. Key Eng Mater. 2005; 288–289:147–150. | ||
Lo CL, Lin KM, Hsiue GH. Preparation and characterization of intelligent core-shell nanoparticles based on poly(D,L-lactide)-g-poly(N-isopropyl acrylamide-co-methacrylic acid). J Control Release. 2005;104(3):477–488. | ||
Joondeph BC, Peyman GA, Khoobehi B, Yue BY. Liposome-encapsulated 5-fluorouracil in the treatment of proliferative vitreoretinopathy. Ophthalmic Surg. 1988;19(4):252–256. | ||
Choi SJ, Oh JM, Chung HE, Hong SH, Kim IH, Choy JH. In vivo anticancer activity of methotrexate-loaded layered double hydroxide nanoparticles. Curr Pharm Des. 2013;19(41):7196–7202. | ||
Zhu R, Wu X, Xiao Y, et al. Synergetic effect of SLN-curcumin and LDH-5-Fu on SMMC-7721 liver cancer cell line. Cancer Biother Radiopharm. 2013;28(8):579–587. | ||
Xu ZP, Niebert M, Porazik K, et al. Subcellular compartment targeting of layered double hydroxide nanoparticles. J Control Release. 2008; 130(1):86–94. | ||
Xu ZP, Lu GC. Layered double hydroxide nanomaterials as potential cellular drug delivery agents. Pure Appl Chem. 2006;78(9):1771–1779. | ||
Chen J, Shao R, Zhang XD, Chen C. Applications of nanotechnology for melanoma treatment, diagnosis, and theranostics. Int J Nanomedicine. 2013;8:2677–2688. | ||
Li L, Gu W, Chen J, Chen W, Xu ZP. Co-delivery of siRNAs and anti-cancer drugs using layered double hydroxide nanoparticles. Biomaterials. 2014;35(10):3331–3339. | ||
de Gramont A, Boni C, Navarro M, et al. Oxaliplatin/5FU/LV in adjuvant colon cancer: Updated efficacy results of the MOSAIC trial, including survival, with a median follow-up of six years. J Clin Oncol. 2007;25(18S):4007. | ||
de Gramont A, Vignoud J, Tournigand C, et al. Oxaliplatin with high-dose leucovorin and 5-fluorouracil 48-hour continuous infusion in pretreated metastatic colorectal cancer. European journal of Cancer. 1997;33(2):214–219. | ||
Mamounas E, Wieand S, Wolmark N, et al. Comparative efficacy of adjuvant chemotherapy in patients with Dukes’ B versus Dukes’ C colon cancer: results from four National Surgical Adjuvant Breast and Bowel Project adjuvant studies (C-01, C-02, C-03, and C-04). J Clin Oncol. 1999;17(5):1349–1355. | ||
Arbuck SG. Overview of clinical trials using 5-fluorouracil and leucovorin for the treatment of colorectal cancer. Cancer. 1989;63(S6): 1036–1044. | ||
Yin H, Xie F, Zhang J, et al. Combination of interferon-α and 5-fluorouracil induces apoptosis through mitochondrial pathway in hepatocellular carcinoma in vitro. Cancer Lett. 2011;306(1):34–42. | ||
Nakamura K, Abu Lila AS, Matsunaga M, Doi Y, Ishida T, Kiwada H. A double-modulation strategy in cancer treatment with a chemotherapeutic agent and siRNA. Mol Ther. 2011;19(11):2040–2047. | ||
Meng H, Mai WX, Zhang H, et al. Codelivery of an optimal drug/siRNA combination using mesoporous silica nanoparticles to overcome drug resistance in breast cancer in vitro and in vivo. ACS Nano. 2013;7(2): 994–1005. | ||
Van Cutsem E, Nowacki M, Lang I, et al. Randomized phase III study of irinotecan and 5-FU/FA with or without cetuximab in the first-line treatment of patients with metastatic colorectal cancer (mCRC): The CRYSTAL trial. J Clin Oncol. 2007;25(18S):4000. | ||
Bhattacharya B, Akram M, Balasubramanian I, et al. Pharmacologic synergy between dual phosphoinositide-3-kinase and mammalian target of rapamycin inhibition and 5-fluorouracil in PIK3CA mutant gastric cancer cells. Cancer Biol Ther. 2012;13(1):34–42. | ||
Chen J. Potential value and limitation of dual inhibitors of PI3K and mTOR in the treatment of cancer. Curr Cancer Drug Targets. 2013; 13(2):117–120. | ||
Bu Z, Ji J. Therapeutic implications of mTOR inhibitors in the treatment of gastric cancer. Curr Cancer Drug Targets. 2013;13(2):121–125. | ||
Cho DC, Mier JW. Dual inhibition of PI3-kinase and mTOR in renal cell carcinoma. Curr Cancer Drug Targets. 2013;13(2):126–142. | ||
Elfiky AA, Jiang Z. The PI3 kinase signaling pathway in prostate cancer. Curr Cancer Drug Targets. 2013;13(2):157–164. | ||
Page C, Lin HJ, Jin Y, et al. Overexpression of Akt/AKT can modulate chemotherapy-induced apoptosis. Anticancer Res 2000;20(1A):407–416. | ||
Brognard J, Clark AS, Ni Y, Dennis PA. Akt/protein kinase B is constitutively active in non-small cell lung cancer cells and promotes cellular survival and resistance to chemotherapy and radiation. Cancer Res. 2001;61(10):3986–3997. | ||
Roper J, Richardson MP, Wang WV, et al. The dual PI3K/mTOR inhibitor NVP-BEZ-235 induces tumor regression in a genetically engineered mouse model of PIK3CA wild-type colorectal cancer. PLoS One. 2011;6(9):e25132. | ||
Maira SM, Stauffer F, Brueggen J, et al. Identification and characterization of NVP-BEZ-235, a new orally available dual phosphatidylinositol 3-kinase/mammalian target of rapamycin inhibitor with potent in vivo antitumor activity. Mol Cancer Ther. 2008;7(7):1851–1863. | ||
Johnston PG, Lenz HJ, Leichman CG, et al. Thymidylate synthase gene and protein expression correlate and are associated with response to 5-fluorouracil in human colorectal and gastric tumors. Cancer Res. 1995;55(7):1407–1412. | ||
Copur S, Aiba K, Drake JC, Allegra CJ, Chu E. Thymidylate synthase gene amplification in human colon cancer cell lines resistant to 5-fluorouracil. Biochem Pharmacol. 1995;49(10):1419–1426. | ||
Gusella M, Frigo AC, Bolzonella C, et al. Predictors of survival and toxicity in patients on adjuvant therapy with 5-fluorouracil for colorectal cancer. Br J Cancer. 2009;100(10):1549–1557. | ||
Han R, Yang YM, Dietrich J, Luebke A, Mayer-Proschel M, Noble M. Systemic 5-fluorouracil treatment causes a syndrome of delayed myelin destruction in the central nervous system. J Biol. 2008;7(4):12. | ||
Choi SJ, Choy JH. Layered double hydroxide nanoparticles as target-specific delivery carriers: uptake mechanism and toxicity. Nanomedicine (Lond). Jul 2011;6(5):803–814. | ||
Chen J. Multiple signal pathways in obesity-associated cancer. Obes Rev. 2011;12(12):1063–1070. | ||
Manara MC, Nicoletti G, Zambelli D, et al. NVP-BEZ-235 as a new therapeutic option for sarcomas. Clin Cancer Res. 2010;16(2):530–540. | ||
Wu P, Hu YZ. PI3K/Akt/mTOR pathway inhibitors in cancer: a perspective on clinical progress. Curr Med Chem. 2010;17(35):4326–4341. | ||
Chen J, Huang XF, Qiao L, Katsifis A. Insulin caused drug resistance to oxaliplatin in colon cancer cell HT29. J Gastrointest Oncol. 2011;2(1): 27–33. | ||
Chen J, Katsifis A, Hu C, Huang XF. Insulin decreases therapeutic efficacy in colon cancer cell line HT29 via the activation of the PI3K/Akt pathway. Curr Drug Discov Technol. 2011;8(2):119–125. | ||
Bartucci M, Svensson S, Ricci-Vitiani L, et al. Obesity hormone leptin induces growth and interferes with the cytotoxic effects of 5-fluorouracil in colorectal tumor stem cells. Endocr Relat Cancer. 2010;17(3): 823–833. | ||
Chen Y, Wang Z, Chang P, et al. The effect of focal adhesion kinase gene silencing on 5-fluorouracil chemosensitivity involves an Akt/NF-kappaB signaling pathway in colorectal carcinomas. Int J Cancer. 2010;127(1):195–206. | ||
Liao X, Lochhead P, Nishihara R, et al. Aspirin use, tumor PIK3CA mutation, and colorectal-cancer survival. N Engl J Med. 2012;367(17): 1596–1606. | ||
Muller A, Bachmann E, Linning M, et al. Selective PI3K inhibition by BKMIZO and BEZ235 alone or in combination with chemotherapy in wild-type and mutated human gastro intestinal cancer cell lines. Cancer Chemther Pharmacol. 2012;69(6):1601–1615. | ||
Park S, Chapuis N, Bardet V, et al. PI-103, a dual inhibitor of Class IA phosphatidylinositide 3-kinase and mTOR, has antileukemic activity in AML. Leukemia. 2008;22(9):1698–1706. | ||
Schwab J, Antonescu C, Boland P, et al. Combination of PI3K/mTOR inhibition demonstrates efficacy in human chordoma. Anticancer research. 2009;29(6):1867–1871. | ||
Chapuis N, Tamburini J, Green AS, et al. Dual inhibition of PI3K and mTORC1/2 signaling by NVP-BEZ-235 as a new therapeutic strategy for acute myeloid leukemia. Clin Cancer Res. 2010;16(22):5424–5435. | ||
Santiskulvong C, Konecny GE, Fekete M, et al. Dual targeting of phosphoinositide 3-kinase and mammalian target of rapamycin using NVP-BEZ-235 as a novel therapeutic approach in human ovarian carcinoma. Clin Cancer Res. 2011;17(8):2373–2384. | ||
Wander SA, Hennessy BT, Slingerland JM. Next-generation mTOR inhibitors in clinical oncology: how pathway complexity informs therapeutic strategy. J Clin invest. 2011;121(4):1231–1241. | ||
Serra V, Scaltriti M, Prudkin L, et al. PI3K inhibition results in enhanced HER signaling and acquired ERK dependency in HER2-overexpressing breast cancer. Oncogene. 2011;30(22):2547–2557. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.