Back to Journals » Clinical Interventions in Aging » Volume 9
Effect of whole-body vibration for 3 months on arterial stiffness in the middle-aged and elderly
Authors Lai C , Chen H, Tseng S, Liao W , Liu B, Lee M, Chen H
Received 7 January 2014
Accepted for publication 11 February 2014
Published 12 May 2014 Volume 2014:9 Pages 821—828
DOI https://doi.org/10.2147/CIA.S60029
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Chung-Liang Lai,1,2 Han-Yu Chen,3 Shiuan-Yu Tseng,1,2 Wan-Chun Liao,2 Bing-Tang Liu,2 Meng-Chih Lee,1,4,* Hsin-Shui Chen5,6,*
1Institute of Medicine, Chung Shan Medical University, Taichung, Taiwan; 2Department of Physical Medicine and Rehabilitation, Taichung Hospital, Ministry of Health and Welfare, Taichung, Taiwan; 3Department of Physical Therapy, Hungkuang University, Taichung, Taiwan; 4Department of Family Medicine, Taichung Hospital, Ministry of Health and Welfare, Taichung, Taiwan; 5Department of Physical Medicine and Rehabilitation, China Medical University Beigang Hospital, Yunlin, Taiwan; 6School of Medicine, College of Medicine, China Medical University, Taichung, Taiwan
*These authors contributed equally to this work
Background: Cardiovascular disease (CVD) is a common problem of middle-aged and older adults. Increased arterial stiffness is a CVD risk factor. Whole-body vibration (WBV) is a simple and convenient exercise for middle-aged and older adults; however, there have been few studies investigating the effect of WBV on arterial stiffness. This study mainly investigated the effect of WBV on arterial stiffness in middle-aged and older adults.
Methods: A total of 38 (21 women and 17 men) middle-aged and elderly subjects (average age, 61.9 years) were randomly divided into the WBV group and the control group for a 3-month trial. The WBV group received an intervention of 30 Hz and 3.2 g WBV in a natural full standing posture at a sports center. The brachial–ankle pulse wave velocity (baPWV), a marker of systemic arterial stiffness, and blood pressure and heart rate were measured before and after the intervention.
Results: After 3 months, there were no significant changes in blood pressure or heart rate in both groups. However, the bilateral baPWV was significantly reduced in the WBV group (decreased by 0.65 m/second [P=0.014]; 0.63 m/second [P=0.041] in either side), but not in the control group. The comparison between the two groups was not statistically significant.
Conclusion: This study found that 3 months of WBV had a positive effect on arterial stiffness in middle-aged and older adults and could therefore be regarded as a supplementary exercise. Larger-scale studies are needed to confirm the effects of WBV in the future.
Keywords: whole-body vibration, arterial stiffness, middle-aged, elderly
Introduction
Cardiovascular disease is one of the major causes of death worldwide. In the United States, the average annual rate for having a first cardiovascular event rose from three per 1,000 men aged 35–44 years to 74 per 1,000 men aged 85–94 years from 1980–2003.1 Increased aortic stiffness is an independent predictor of cardiovascular mortality in hypertensive patients,2 and increased arterial stiffness is an independent risk factor for cardiovascular disease.3
Arterial stiffness can be measured by the following three kinds of noninvasive methods: the relationship between changes in vascular caliber and changes in blood pressure, pulse wave velocity (PWV), and augmentation index (AI), which is calculated from the blood pressure waveform. PWV is regarded as the standard for arterial stiffness measurement.4 PWV can be measured in different ways, the most commonly studied of which include brachial–ankle pulse wave velocity (baPWV), leg PWV, and carotid–femoral pulse wave velocity (cfPWV). Some studies have proposed that arterial stiffness may contribute to increased cardiac afterload and left heart failure.5,6 Chow et al compared various methods of measuring PWV (baPWV, leg PWV, and cfPWV), AI, and ankle–brachial index with left ventricular failure. They found that only baPWV and the cardiac ultrasound early-late filling rate ratio were significantly correlated.7 The validity and reproducibility of baPWV measurements are high, and it seems to be an acceptable marker reflecting vascular damage and systemic arterial stiffness.8 baPWV correlates well with arterial stiffness and is a useful index to assess arteriosclerosis.9 Furthermore, it is relatively low-cost and quick for the assessment of arteriosclerosis and atherosclerosis in a community.10,11 Therefore, this study used baPWV as a tool to measure arterial stiffness.
Arterial stiffness is mainly treated with antihypertensive drugs, especially renin–angiotensin–aldosterone system inhibitors. According to the findings of previous studies, in addition to their blood pressure-lowering effect, these inhibitors also inhibit inflammation caused by peroxide, thereby reducing vascular stiffness.12 The drugs involved in arterial stiffness include aminoguanide, pyridoxamine, and OPB-9195. Aminoguanide increases blood vessel dilation and reduces PWV. However, it is clinically believed that a high dosage of aminoguanide will increase the risk for glomerulonephritis.13–15 The other two drugs are still involved in animal experiments.16 Exercise is also a method of reducing arterial stiffness other than medicine. In 2007, Tabara et al conducted a 6-month trial in which elderly subjects engaged in 30 minutes of moderate- to high-intensity aerobic exercises twice per week. The results discovered that such exercise can reduce arterial stiffness and blood pressure.17 However, the relationship between resistance exercise and arterial stiffness has been debated. Miyachi reviewed eight articles on random clinical trials, finding that resistance training in middle-aged subjects was not associated with changes in arterial stiffness, that high-intensity resistance training was significantly associated with an increase in stiffness, and that moderate-intensity resistance training showed no such association.18
Whole-body vibration (WBV) has become a popular exercise. WBV generates an oscillating movement on a plate and transmits vertical acceleration to muscle and bone.19 Its mechanism of stimulating muscle spindles and alpha motor neurons initiates reflex muscle contractions.20 WBV is beneficial for enhancing leg muscle strength and improving muscle power and balance in older people.21,22 Furthermore, it may reduce bone loss in postmenopausal women.23,24
There are a number of published studies on the effects of WBV training on cardiovascular systems. Increased blood flow in healthy people after WBV training has been found in some studies,25–27 and there were similar results in Friedreich’s ataxia and spinal cord injury patients.28,29 However, regarding arterial stiffness, studies on WBV are relatively few. In 2008, Otsuki et al proposed that baPWV is significantly reduced 40 minutes after WBV, but that it returns to the baseline 60 minutes after.30 Figueroa et al pointed out that arterial stiffness can be reduced in young overweight and obese women after 6 weeks of WBV and in postmenopausal women with prehypertension and hypertension after 12 weeks of WBV.31,32 However, there have been no studies focusing on middle-aged and older adults. This study aimed to investigate the effects of 3 months of WBV on arterial stiffness in middle-aged and older adults. This study hypothesized that 3 months of WBV training would result in a significant decrease of arterial stiffness in middle-aged and older adults.
Methods
Subjects
This study was a randomized controlled trial. The subjects who met the inclusion criteria as community-dwelling middle-aged and older adults were recruited through advertisements from February 2013 to June 2013. As shown in Figure 1, a total of 50 middle-aged and older adults were recruited, of whom 43 met the inclusion criteria. In the end, a total of 38 subjects completed the study. The inclusion criteria were age 50 years and older, the ability to follow the protocol, a lack of regular exercise at least three times per week, and blood pressure  140/90 mmHg with or without drugs. The exclusion criteria were acute hernias or thrombosis, kidney stones or bladder stones, a history of seizures, arrhythmia, use of a pacemaker, severe cardiovascular or lung disease, dizziness, and being hospitalized or undergoing surgery in the last 6 months. The subjects were all notified of the research purpose and relevant information, and they all consented with understanding. This study was approved by the Institutional Review Board of Jianan Mental Hospital (Tainan, Taiwan).
140/90 mmHg with or without drugs. The exclusion criteria were acute hernias or thrombosis, kidney stones or bladder stones, a history of seizures, arrhythmia, use of a pacemaker, severe cardiovascular or lung disease, dizziness, and being hospitalized or undergoing surgery in the last 6 months. The subjects were all notified of the research purpose and relevant information, and they all consented with understanding. This study was approved by the Institutional Review Board of Jianan Mental Hospital (Tainan, Taiwan).
  | Figure 1 Flowchart of this trial. |
Study design
A total of 43 subjects met the inclusion criteria. They were randomly assigned to either the WBV group or the control group (CON group) by computer-generated numbers. During the trial, the two groups of subjects were required to follow their original living habits and to continue taking their medication. The WBV group received vibration trainings three times per week in the hospital’s sports center. The subjects stood barefoot on the platform of the machine in a natural, full standing position. The source of vibration was a WBV device (LV-1000; X-trend Fitness Equipment, Luntai Enterprise Co, Ltd, Taichung, Taiwan). The vibration was horizontal at a frequency of 30 Hz (1 Hz =1 oscillation/second), and the magnitude (acceleration) was 3.2 g (gravity, 1 g =9.81 m/second2). A well-trained physical therapist was responsible for executing the WBV training and for monitoring the safety of the subjects. The baPWV, systolic blood pressure, diastolic blood pressure, mean artery pressure, and resting heart rate before the training and 3 months after training were measured using a PWV/ABI automatic device (VP-2000; Omron Healthcare Inc., Vernon Hills, IL, USA). The measurement was implemented by an experienced technician who had no knowledge of the subjects. In the measurement, appropriate cuffs were selected to wrap the elbow joint (brachial artery) and ankle (posterior tibial artery). The brachial–ankle distance was automatically inputted after entering each subject’s height into the machine. The transit time was the time gap between the start of the electrocardiogram’s R-wave and the reception of the waveform in four limbs. The baPWV can be automatically computed in the instrument according to the transit time and distance. To avoid any immediate effect after a single training, the second measurement was conducted 2 days after the last training.
Statistical analysis
The statistical analysis software used in this study included Statistical Package for the Social Sciences, Windows version 14 (SPSS Inc., Chicago, IL, USA) for data analysis. Descriptive statistics included the mean and standard deviation, the chi-square test, and the independent t-test for the baseline characteristics. This study used a paired-samples t-test to compare the change in baPWV, blood pressure, mean artery pressure, and heart rate of the two groups before and after the intervention. The effects between the two groups were tested using an independent t-test. This study adopted P<0.05 as the level of statistical significance in the two-tailed analysis.
Results
Among 43 middle-aged and elderly subjects, five subjects dropped out because of lack of interest during the trial. Therefore, a total of 38 subjects (88%) completed the program. During the entire program, none of the subjects experienced adverse effects such as dizziness, pain, or falls.
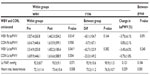
The basic data and parameters before the intervention can be seen in Table 1. A comparison showed there were no significant differences in sex, age, body mass index, bilateral baPWV, bilateral systolic blood pressure, bilateral diastolic blood pressure, bilateral mean artery pressure, or resting heart rate, suggesting the subjects were properly randomized.
After the 3 month trial, the WBV and CON groups had no significant intragroup or intergroup differences in bilateral systolic pressure, bilateral diastolic pressure, bilateral mean artery pressure, or resting heat rate (Table 2).
As shown in Table 3, the bilateral baPWV of the WBV group and the CON group after 3 months were 1,462.3±204.2, 1,452.2±180.4 (compared with the pretest, P=0.014 and P=0.041), and 1,615.9±399.1 and 1,616.6±388.9 (compared with the pretest, P=0.958 and P=0.504), respectively. There was a significant decrease in bilateral baPWV of the WBV group, although there was no obvious change in that of the CON group. However, the difference of posttest and pretest between the two groups did not reach statistical significance in either side (P=0.164 and P=0.382) (Figure 2). The change between the two groups indicated that the bilateral baPWV in the WBV group decreased by 3.7%±6.1% and 3.4%±6.5%, whereas that of the CON group increased by 0.8%±2.1% and decreased by 0.7%±1.8%.
Discussion
The loss in muscle mass over time, particularly the loss in leg muscle mass in women and men, is related to increased baPWV.33,34 Exercises, especially resistance exercises, have been proven to increase muscle mass.35 Some studies also have shown that aerobic exercise can improve arterial stiffness in healthy adults and older adults.17,36 However, aerobic exercises are not suitable for patients with disabilities; therefore, WBV training can be provided as an alternative or supplementary exercise. WBV has some adverse effects, including dizziness, headaches, and falls; however, these can be minimized when exposure is of low intensity and for a short duration.37 Thus, it is important to select the vibration model and the duration of the intervention.
This study was pilot research on the effect of WBV on arterial stiffness in middle-aged and older adults. The most important finding in this study is that after 3 months of 30 Hz and 3.2 g horizontal WBV training, the bilateral baPWV were significantly reduced (P=0.014 and P=0.041). However, compared with the control group, there was no significant difference; meanwhile, the two groups had no significant changes in bilateral blood pressure or heart rate.
The mechanism of how WBV reduces arterial stiffness is still not clear. In addition, WBV may have various effects on the human body’s physiological functions when different frequencies, durations, postures, and exercise modes are applied.38 It was found that short-term WBV may reduce leg PWV by enhancing the release of local contraction-related vasodilator factors and nitric oxide.39,40 The baPWV consists of the aortic PWV and leg PWV. Repeated exposure to local vasodilator substances may explain the long-term reduction in baPWV after WBV training.32
Regarding the immediate effects of WBV on arterial stiffness, Otsuki et al30 used 20 Hz WBV on young healthy men in a static squat position with knee bending and found a reduction of baPWV 40 minutes after WBV. However, the baPWV returned to baseline 60 minutes after vibration. Figueroa et al38 used 30 Hz WBV on young men in a static squat posture and found that WBV attenuated the increase of baPWV in response to sympathetic overactivity by postexercise muscle ischemia.
In a review of the effect of WBV for a period of time on arterial stiffness, Figueroa et al31 studied young overweight and obese women for 6 weeks, using 25–30 Hz WBV with a set of dynamic and static exercises. They found that arterial stiffness and blood pressure could be apparently improved. Similarly, they studied postmenopausal women with prehypertension and hypertension for 12 weeks, using 25–40 Hz of WBV with a set of dynamic and static exercises and found that arterial stiffness and blood pressure could be significantly improved.32 These two studies by Figueroa et al produced different baPWV decreases (0.4 m/second and 1.3 m/second) because of differences in the subjects’ ages, vibration frequencies, and intervention period. Our research mainly studied middle-aged and adults older than 50 years, and unlike the two studies mentioned earlier,31,32 the vibration frequency was fixed on 30 Hz, and subjects used a natural full standing position on the vibrating plate. We found that bilateral baPWV of the WBV group decreased by 0.65 m/second and 0.63 m/second, respectively. The degree of decrease is between that in the previous two studies. Thus, it can be seen that WBV contributes to the decrease of arterial stiffness of middle-aged and older adults, as well as women, as in the previous finding. We also found that 3 months of WBV could not cause blood pressure changes. Unlike in the previous two studies, this may be caused by the selection of different groups of people. The subjects of the previous two studies were overweight and obese women or postmenopausal women with prehypertension and hypertension who all were at a high risk of high blood pressure. The subjects in our study were middle-aged and older adults with normal blood pressure during the trial.
There were some limitations to this study. First, the number of cases was insufficient, and the intervention was short. As a result, it may not have properly displayed the comparison between the WBV group and the CON group. Second, blank WBV was not provided to the CON group, and a double-blind design could not be implemented in this study. Third, this study could not engage in stratified analysis because of its small sample size. The ability to display which group of people can benefit from WBV will make a difference in the judgment and suggestions of physicians. Fourth, the subjects were not told to refrain from smoking and drinking beverages containing caffeine before assessments, which could interfere with the measurements. Moreover, different WBV characteristics have varying physiological effects.41 The findings of this study are not applicable to the WBV training of different models. In the future study, a big sample size with appropriate controlled and layered analyses is required to confirm the effectiveness of WBV on arterial stiffness.
Overall, this study suggested that 3 months of WBV had a trend to improve the arterial stiffness of middle-aged and older adults. The findings suggest that WBV, for a certain period of time, may have an effect on arterial stiffness. However, the period of effectiveness and the reduction in the incidence of cardiovascular disease must be confirmed by more comprehensive, large-scale studies.
Conclusion
This study concluded that 3 months of WBV may reduce arterial stiffness in middle-aged and older adults and, thus, can be used as a supplementary exercise. However, its effectiveness needs to be confirmed by future studies.
Acknowledgments
This research was supported by the foundation of the Taichung Hospital and the Ministry of Health and Welfare, Taichung, Taiwan.
Disclosure
The authors report no conflicts of interest in this work.
References
Go AS, Mozaffarian D, Roger VL, et al; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics – 2013 update: a report from the American Heart Association. Circulation. 2013;127(1):e6–e245. | |
Laurent S, Boutouyrie P, Asmar R, et al. Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension. 2001;37(5):1236–1241. | |
Sutton-Tyrrell K, Najjar SS, Boudreau RM, et al; Health ABC Study. Elevated aortic pulse wave velocity, a marker of arterial stiffness, predicts cardiovascular events in well-functioning older adults. Circulation. 2005;111(25):3384–3390. | |
Sakuragi S, Abhayaratna WP. Arterial stiffness: methods of measurement, physiologic determinants and prediction of cardiovascular outcomes. Int J Cardiol. 2010;138(2):112–118. | |
Weber T, O’Rourke MF, Ammer M, Kvas E, Punzengruber C, Eber B. Arterial stiffness and arterial wave reflections are associated with systolic and diastolic function in patients with normal ejection fraction. Am J Hypertens. 2008;21(11):1194–1202. | |
Borlaug BA, Melenovsky V, Redfield MM, et al. Impact of arterial load and loading sequence on left ventricular tissue velocities in humans. J Am Coll Cardiol. 2007;50(16):1570–1577. | |
Chow B, Rabkin SW. Brachial-ankle pulse wave velocity is the only index of arterial stiffness that correlates with a mitral valve indices of diastolic dysfunction, but no index correlates with left atrial size. Cardiol Res Pract. 2013;2013:986847. | |
Yamashina A, Tomiyama H, Takeda K, et al. Validity, reproducibility, and clinical significance of noninvasive brachial-ankle pulse wave velocity measurement. Hypertens Res. 2002;25(3):359–364. | |
Shiotani A, Motoyama M, Matsuda T, Miyanish T. Brachial-ankle pulse wave velocity in Japanese university students. Internal Medicine. 2005:44(7):696–701. | |
Chuang SY, Chen CH, Cheng CM, Chou P. Combined use of brachial-ankle pulse wave velocity and ankle-brachial index for fast assessment of arteriosclerosis and atherosclerosis in a community. Int J Cardiol. 2005;98(1):99–105. | |
Oliver JJ, Webb DJ. Noninvasive assessment of arterial stiffness and risk of atherosclerotic events. Arterioscler Thromb Vasc Biol. 2003;23(4):554–566. | |
Mahmud A, Feely J. Effect of angiotensin ii receptor blockade on arterial stiffness: beyond blood pressure reduction. Am J Hypertens. 2002;15(12):1092–1095. | |
Corman B, Duriez M, Poitevin P, et al. Aminoguanidine prevents age-related arterial stiffening and cardiac hypertrophy. Proc Natl Acad Sci U S A. 1998;95(3):1301–1306. | |
Cantini C, Kieffer P, Corman B, Limiñana P, Atkinson J, Lartaud-Idjouadiene I. Aminoguanidine and aortic wall mechanics, structure, and composition in aged rats. Hypertension. 2001;38(4):943–948. | |
Bolton WK, Cattran DC, Williams ME, et al; ACTION I Investigator Group. Randomized trial of an inhibitor of formation of advanced glycation end products in diabetic nephropathy. Am J Nephrol. 2004;24(1):32–40. | |
Pase MP, Grima NA, Sarris J. The effects of dietary and nutrient interventions on arterial stiffness: a systematic review. Am J Clin Nutr. 2011;93(2):446–454. | |
Tabara Y, Yuasa T, Oshiumi A, et al. Effect of acute and long-term aerobic exercise on arterial stiffness in the elderly. Hypertens Res. 2007;30(10):895–902. | |
Miyachi M. Effects of resistance training on arterial stiffness: a meta-analysis. Br J Sports Med. 2013;47(6):393–396. | |
Rauch F, Sievanen H, Boonen S, et al; International Society of Musculoskeletal and Neuronal Interactions. Reporting whole-body vibration intervention studies: recommendations of the International Society of Musculoskeletal and Neuronal Interactions. J Musculoskelet Neuronal Interact. 2010;10(3):193–198. | |
Rauch F. Vibration therapy. Dev Med Child Neurol. 2009;51 Suppl 4:166–168. | |
Lau RW, Liao LR, Yu F, Teo T, Chung RC, Pang MY. The effects of whole body vibration therapy on bone mineral density and leg muscle strength in older adults: a systematic review and meta-analysis. Clin Rehabil. 2011;25(11):975–988. | |
Sitjà-Rabert M, Rigau D, Fort Vanmeerghaeghe A, Romero-Rodríguez D, Bonastre Subirana M, Bonfill X. Efficacy of whole body vibration exercise in older people: a systematic review. Disabil Rehabil. 2012;34(11):883–893. | |
Lai CL, Tseng SY, Chen CN, et al. Effect of 6 months of whole body vibration on lumbar spine bone density in postmenopausal women: a randomized controlled trial. Clin Interv Aging. 2013;8:1603–1609. | |
Beck BR, Norling TL. The effect of 8 mos of twice-weekly low- or higher intensity whole body vibration on risk factors for postmenopausal hip fracture. Am J Phys Med Rehabil. 2010;89(12):997–1009. | |
Kerschan-Schindl K, Grampp S, Henk C, et al. Whole-body vibration exercise leads to alterations in muscle blood volume. Clin Physiol. 2001;21(3):377–382. | |
Lohman EB 3rd, Petrofsky JS, Maloney-Hinds C, Betts-Schwab H, Thorpe D. The effect of whole body vibration on lower extremity skin blood flow in normal subjects. Med Sci Monit. 2007;13(2):CR71–CR76. | |
Lythgo N, Eser P, de Groot P, Galea M. Whole-body vibration dosage alters leg blood flow. Clin Physiol Funct Imaging. 2009;29(1):53–59. | |
Herrero AJ, Martín J, Martín T, et al. Whole-body vibration alters blood flow velocity and neuromuscular activity in Friedreich’s ataxia. Clin Physiol Funct Imaging. 2011;31(2):139–144. | |
Herrero AJ, Menéndez H, Gil L, et al. Effects of whole-body vibration on blood flow and neuromuscular activity in spinal cord injury. Spinal Cord. 2011;49(4):554–559. | |
Otsuki T, Takanami Y, Aoi W, Kawai Y, Ichikawa H, Yoshikawa T. Arterial stiffness acutely decreases after whole-body vibration in humans. Acta Physiol (Oxf). 2008;194(3):189–194. | |
Figueroa A, Gil R, Wong A, et al. Whole-body vibration training reduces arterial stiffness, blood pressure and sympathovagal balance in young overweight/obese women. Hypertens Res. 2012;35(6):667–672. | |
Figueroa A, Kalfon R, Madzima TA, Wong A. Whole-body vibration exercise training reduces arterial stiffness in postmenopausal women with prehypertension and hypertension. Menopause. 2014;21(2):131–136. | |
Sanada K, Miyachi M, Tanimoto M, et al. A cross-sectional study of sarcopenia in Japanese men and women: reference values and association with cardiovascular risk factors. Eur J Appl Physiol. 2010;110(1):57–65. | |
Ochi M, Kohara K, Tabara Y, et al. Arterial stiffness is associated with low thigh muscle mass in middle-aged to elderly men. Atherosclerosis. 2010;212(1):327–332. | |
Landi F, Marzetti E, Martone AM, Bernabei R, Onder G. Exercise as a remedy for sarcopenia. Curr Opin Clin Nutr Metab Care. 2014;17(1):25–31. | |
Vaitkevicius PV, Fleg JL, Engel JH, et al. Effects of age and aerobic capacity on arterial stiffness in healthy adults. Circulation. 1993; 88(4 Pt 1):1456–1462. | |
Totosy de Zepetnek JO, Giangregorio LM, Craven BC. Whole-body vibration as potential intervention for people with low bone mineral density and osteoporosis: a review. J Rehabil Res Dev. 2009;46(4):529–542. | |
Figueroa A, Gil R, Sanchez-Gonzalez MA. Whole-body vibration attenuates the increase in leg arterial stiffness and aortic systolic blood pressure during post-exercise muscle ischemia. Eur J Appl Physiol. 2011;111(7):1261–1268. | |
Figueroa A, Vicil F, Sanchez-Gonzalez MA. Acute exercise with whole-body vibration decreases wave reflection and leg arterial stiffness. Am J Cardiovasc Dis. 2011;1(1):60–67. | |
Maloney-Hinds C, Petrofsky JS, Zimmerman G, Hessinger DA. The role of nitric oxide in skin blood flow increases due to vibration in healthy adults and adults with type 2 diabetes. Diabetes Technol Ther. 2009;11(1):39–43. | |
Petit PD, Pensini M, Tessaro J, Desnuelle C, Legros P, Colson SS. Optimal whole-body vibration settings for muscle strength and power enhancement in human knee extensors. J Electromyogr Kinesiol. 2010;20(6):1186–1195. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.