Back to Journals » Clinical Pharmacology: Advances and Applications » Volume 9
Effect of food on the pharmacokinetics of dronabinol oral solution versus dronabinol capsules in healthy volunteers
Authors Oh DA, Parikh N, Khurana V, Cognata Smith C, Vetticaden S
Received 13 August 2016
Accepted for publication 22 October 2016
Published 11 January 2017 Volume 2017:9 Pages 9—17
DOI https://doi.org/10.2147/CPAA.S119676
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Arthur E. Frankel
D Alexander Oh, Neha Parikh, Varun Khurana, Christina Cognata Smith, Santosh Vetticaden
INSYS Therapeutics, Inc., Chandler, AZ, USA
Abstract: Dronabinol is a pharmaceutical tetrahydrocannabinol originally developed as an oral capsule. A dronabinol oral solution was recently approved, and the effects of food on absorption and bioavailability of the oral solution versus capsules were compared in an open-label, single-dose, 3-period crossover study. Healthy volunteers were randomized to either dronabinol oral solution 4.25 mg (fed) or dronabinol capsule 5 mg (fed or fasted). Dosing was separated by a 7-day washout period. Plasma pharmacokinetics were evaluated for dronabinol and its major metabolite, 11-hydroxy-delta-9-tetrahydrocannabinol (11-OH-∆9-THC). Pharmacokinetic data were available for analysis in 54 volunteers. In the fed state, initial dronabinol absorption was faster with oral solution versus capsule (mean time to the first measurable concentration, 0.15 vs 2.02 hours, respectively), with 100% and 15% of volunteers, respectively, having detectable plasma dronabinol levels 30 minutes postdose. There was less interindividual variability in plasma dronabinol concentration during early absorption with oral solution versus capsule. Compared with the fasted state, mean area under the plasma concentration–time curve from time zero to the last measurable concentration (AUC0-t) increased by 2.1- and 2.4-fold for dronabinol oral solution and capsule, respectively, when taken with food. Mean time to maximum plasma concentration was similarly delayed for dronabinol oral solution with food (7.7 hours) and capsule with food (5.6 hours) versus capsule with fasting (1.7 hours). Under fed conditions, AUC0–t and area under the plasma concentration–time curve from time zero to infinity were similar for the oral solution versus capsule based on 11-OH-∆9-THC levels. An appreciable food effect was observed for dronabinol oral solution and capsules. Dronabinol oral solution may offer therapeutic benefit to patients, given its rapid and lower interindividual absorption variability versus dronabinol capsule.
Keywords: bioavailability, dronabinol, Marinol, pharmacokinetics, observer variation
Introduction
Dronabinol is an orally active, synthetic delta-9-tetrahydrocannabinol that has complex effects on the central nervous system. Dronabinol in capsule formulation has been approved in the US since 1985 and is indicated for the treatment of anorexia associated with weight loss in patients with acquired immune deficiency syndrome (AIDS) and for nausea and vomiting associated with cancer chemotherapy in patients who have failed to respond adequately to conventional antiemetic treatments. The efficacy and safety of oral dronabinol in patients with AIDS have been reported in several clinical studies (reviewed in Badowski and Perez1). In a large, randomized, double-blind, placebo-controlled study, oral dronabinol 2.5 mg twice daily for 6 weeks was shown to stabilize patient weight and resulted in statistically significant improvement in appetite compared with placebo in 139 individuals with AIDS who had lost ≥2.3 kg of their normal body weight.2 In an open-label extension phase of oral dronabinol 2.5 mg twice daily, beneficial effects on weight, assessed at month 7, were maintained or increased in patients continuing dronabinol and were maintained or slightly decreased in patients previously switched from placebo; dronabinol also showed consistent improvement in mean appetite from baseline through month 12.3 Clinical benefits of oral dronabinol have also been reported in patients with chemotherapy-induced nausea and vomiting.4–11 Reduction in the incidence or intensity of nausea, incidence of vomiting, and improvement in appetite were generally comparable with those of other studied antiemetics such as metoclopramide, prochlorperazine, and ondansetron.4–11
Dronabinol is almost completely absorbed (90%–95%) after a single oral dose of the capsule formulation.12 However, 10%–20% of the administered dose reaches the systemic circulation due to extensive first-pass hepatic metabolism and high lipid solubility.12 Dronabinol and its principal active metabolite, 11-hydroxy-delta-9-tetrahydrocannabinol (11-OH-∆9-THC), are present in plasma in about equal concentrations, peaking up to 4 hours after oral dosing and declining over several days.12 The onset of action is ~0.5–1 hour with peak effects from 2–4 hours.12 However, the onset of action (up to 1 hour) is less than ideal, with highly variable interindividual pharmacokinetics and pharmacodynamics and a lack of flexibility with dosing.12 Thus, certain patient populations may benefit from or prefer an alternative oral delivery method.
Dronabinol oral solution was approved in July 2016 to offer patients an easy-to-swallow formulation of dronabinol as an alternative to dronabinol capsules.13 A recent study reported that dronabinol oral solution 4.25 mg was bioequivalent to dronabinol capsule 5 mg under fasted conditions in healthy individuals.13 Further, dronabinol oral solution had lower intraindividual variability for area under the plasma concentration–time curve (AUC) from time zero to infinity (AUC0-∞) compared with dronabinol capsule (13.5% vs 36.8%, respectively). All individuals receiving dronabinol oral solution had detectable concentrations of plasma dronabinol 15 minutes after administration of dronabinol oral solution compared with 16.8% of individuals receiving dronabinol capsule.
The primary objective of the current study was to evaluate the comparative bioavailability of dronabinol oral solution 4.25 mg administered under fed conditions to that of dronabinol capsule 5 mg under fed or fasted conditions. The secondary objective was to compare the onset of detectable dronabinol concentrations for dronabinol oral solution 4.25 mg and dronabinol capsule 5 mg.
Methods
Population
The study was conducted from November 30, 2015, through December 18, 2015 (ClinicalTrials.gov identifier: NCT02604992), in accordance with the International Conference on Harmonization principles of Good Clinical Practice and the Declaration of Helsinki. The protocol was approved by IntegReview Institutional Review Board (Austin, TX, USA). All participants provided written informed consent. Healthy male or female volunteers aged 18–50 years with body weight ≥60 kg and body mass index (BMI) of 18–30 kg/m2 were eligible for the study. To be included, individuals had no known medical conditions and (females) were not pregnant or lactating. Individuals had not used any prescription medication (except for female hormonal contraceptives) or over-the-counter products (including vitamins and herbal supplements) within 7 days. Individuals also did not have a recent (within 1 year) history or strong potential for alcohol abuse (ie, >14 drinks/week) or substance abuse.
Study design
This was a Phase I, open-label, randomized, single-dose, 3-period crossover study to evaluate the bioavailability of dronabinol delivered as an oral solution, as compared with capsule under the fed condition. The study was not intended to be a bioequivalence study but intended to assess the comparative bioavailability of dronabinol oral solution 4.25 mg to that of dronabinol capsule 5 mg under fed and fasted conditions. Doses selected were based on a prior study that demonstrated the bioequivalence of dronabinol oral solution 4.25 mg and dronabinol capsule 5 mg under fasted conditions.13 A computerized randomization schedule was generated to assign volunteers into 1 of 6 treatment sequences via SAS® (Version 9.3; SAS Institute Inc., Cary, NC, USA). Individuals received a single oral dose of dronabinol oral solution 4.25 mg (Syndros, INSYS Therapeutics, Inc., Chandler, AZ, USA) under fed conditions, a single oral dose of dronabinol capsule 5 mg (Marinol®; AbbVie, Inc., North Chicago, IL, USA) under fed conditions, or a single oral dose of dronabinol capsule 5 mg under fasted conditions. Each dose was administered with 240 mL of water with additional water permitted as needed during the study, except during the period of time between 1 hour pre- and 1 hour postdose. After an overnight fast of at least 10 hours for all treatment groups, individuals being evaluated under fed conditions received dronabinol oral solution or capsules after consumption of a US Food and Drug Administration standard high-fat, high-calorie meal beginning 30 minutes prior to dose administration and ending (ie, last bite of meal taken) within 5 minutes of study drug administration. All individuals fasted for an additional 4 hours postdose. Standard meals were provided at ~4 and 10 hours after study treatment administration and at appropriate times thereafter. Each treatment was separated by a minimum washout period of 7 days. Dronabinol capsule is insoluble in water and is formulated in sesame oil.12 Dronabinol oral solution contains 50% (w/w) dehydrated alcohol and 5.5% (w/w) propylene glycol.14
Assessments
Blood samples were collected at 0 (predose), 0.08 (5 minutes), 0.17 (10 minutes), 0.25 (15 minutes), 0.5 (30 minutes), 0.75 (45 minutes), 1, 1.5, 2, 2.5, 3, 3.5, 4, 6, 8, 12, 16, 24, 36, and 48 hours postdose in each study period. Samples were centrifuged, and plasma was harvested and frozen at approximately –20°C within 1 hour of collection for shipment to a central laboratory for analysis. Plasma samples were analyzed for dronabinol and its active metabolite, 11-OH-∆9-THC, according to validated liquid chromatography-tandem mass spectrometry methods developed by Worldwide Clinical Trials Drug Development Solutions (Austin, TX, USA), with a range of 0.025–10.0 ng/mL for each analyte, using a 0.500 mL aliquot of plasma. Pharmacokinetic parameters were determined by noncompartmental methods using the Phoenix™ WinNonlin® simulation software (Version 6.3; Certara, Princeton, NJ, USA). Both maximum plasma concentration (Cmax) and time to maximum plasma concentration (Tmax) values were determined from observed data, and AUC data were determined by the logarithmic-linear trapezoidal method.
Adverse events (AEs) were monitored throughout the study. Vital signs were measured at screening, predose (within 1 hour of dose administration), at 2, 4, 8, 12, 24, and 48 hours postdose during each treatment period, at early study discontinuation, and at any other time deemed by the investigators to be medically necessary. A 12-lead electrocardiogram was conducted at screening and on the day of study discharge. Individuals were also observed for a psychotic episode (ie, hallucinations, paranoid delusions, personality changes, and disorganized thinking) and monitored until mental status returned to normal.
Statistical analysis
The safety population included all individuals who received at least 1 dose of study drug. The pharmacokinetic population included all individuals in the safety population who completed at least 1 treatment period. Concentration–time data were summarized using descriptive statistics. Cross-treatment comparisons of log-transformed pharmacokinetic parameters such as Cmax, AUC from time zero to the last measurable concentration (AUC0–t), and AUC0–∞ for dronabinol and 11-OH-Δ9-THC were performed using an analysis of variance (ANOVA) model and the two 1-sided t-tests procedure. The ANOVA model included factors for sequence, subject within sequence, treatment, and period. The ratios of the geometric means and 90% confidence intervals (CIs) were calculated for comparisons of dronabinol capsule 5 mg in a fed versus fasted condition. Comparative bioavailability was evaluated by the point estimates and the corresponding 90% CIs for dronabinol Cmax, AUC0–t, and AUC0–∞. Data are mean ± standard deviation unless otherwise indicated. Statistical analyses were performed using SAS, Phoenix WinNonlin, or Microsoft Excel® 2013 (Microsoft Corp, Redmond, WA, USA). A sample size of 54 individuals was selected to ensure that 52 individuals would complete the study to provide 80% power with an α of 0.05 to demonstrate bioequivalence if the value of the true difference between the formulations was <5%.
Results
A total of 54 individuals were randomized and included in the safety and pharmacokinetic populations, of whom 50% were females and 50% were white, 48.1% were black/African-American, and 1.9% were Asian. The mean age was 34.9±7.1 years (range, 23–49 years) with a mean BMI of 26.4±2.5 kg/m2 (range, 20.1–30 kg/m2). Overall, 52 of the 54 individuals (96.3%) completed all 3 study periods. One individual withdrew because of a protocol violation (use of concomitant restricted medication) and 1 withdrew due to an AE of vomiting, which was not considered by investigators to be related to treatment.
Pharmacokinetics
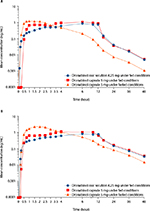
Pharmacokinetic parameters (Tables 1 and 2) and mean concentration–time data indicated a food effect on the pharmacokinetics of dronabinol (Figure 1A) and its active metabolite, 11-OH-∆9-THC (Figure 1B). Compared with dronabinol capsule in fasted state, mean AUC0-t increased by 2.1- and 2.4-fold for dronabinol oral solution and dronabinol capsule, respectively, when taken with food. Dronabinol capsule in the fed state resulted in an 18.7% higher mean dronabinol Cmax and an ~4-hour delay in Tmax compared with dronabinol capsule in the fasted state. Geometric least-squares mean ratio for fed/fasted condition (90% CI) was 106.73% (91.36–124.69) for Cmax; values were 246.43% (219.31–276.90) for AUC0–t and 288.78% (248.06–336.20) for AUC0–∞ (Table S1).
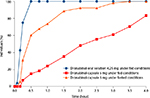
In the fed state, initial dronabinol absorption was faster with dronabinol oral solution than dronabinol capsule (Table 1), with a difference of 1.87 hours in the mean time to the first measurable concentration (Tlag) between the 2 treatment groups. Furthermore, in the fed state, a greater percentage of individuals had detectable plasma dronabinol concentrations earlier with dronabinol oral solution versus dronabinol capsule (Figure 2). At 30 minutes postdose, 100% of the dronabinol oral solution (fed state) group had detectable plasma dronabinol levels versus 15% of the dronabinol capsule (fed state) group. Dronabinol capsule in the fasted state did not reach 100% of individuals with detectable plasma dronabinol concentrations until 3.5 hours postdose. Under fed conditions, AUC0–t and AUC0–∞ were similar but slightly lower for the oral solution versus capsule based on 11-OH-Δ9-THC levels (Table 2).
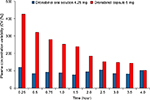
In addition, under fed conditions, the interindividual variability in the dronabinol plasma concentrations (coefficient of variation) was lower with dronabinol oral solution versus dronabinol capsule for up to 4 hours postdose (Figure 3). Regarding statistical analysis of pharmacokinetic parameters under fed conditions, the Cmax of dronabinol was lower after the administration of dronabinol oral solution 4.25 mg compared with dronabinol capsule 5 mg, while the AUC0–t and AUC0–∞ with dronabinol oral solution and dronabinol capsule were comparable (Table S2). In addition, Cmax of dronabinol was lower following the administration of dronabinol oral solution in the fed state compared with dronabinol capsule in the fasted state, while the AUC0–t and AUC0–∞ were both higher after the administration of dronabinol oral solution in the fed state compared with dronabinol capsule in the fasted state (Table S3).
Adverse events
Single-dose administration of all treatments was well tolerated. The most commonly reported AE was headache, reported by 2 (3.8%) individuals after the administration of dronabinol oral solution in the fed state, 1 (1.9%) individual after dronabinol capsule in the fed state and 2 (3.8%) individuals after dronabinol capsule in the fasted state. The second most commonly reported AE was euphoric mood, reported by 2 (3.8%) individuals after the administration of dronabinol oral solution (fed state) and 1 (1.9%) after the administration of dronabinol capsule (fasted state). All AEs were mild in intensity and no serious AEs were reported. No AEs were related to abnormal laboratory evaluations, and no clinically significant abnormalities in vital signs were observed.
Discussion
In a previous report, dronabinol oral solution formulation was shown to be bioequivalent to dronabinol capsule under fasted conditions in healthy individuals.13 Because the current study was designed to provide a general comparison of the pharmacokinetics and bioavailability of dronabinol oral solution 4.25 mg versus dronabinol capsule 5 mg under fed conditions, this study also included a treatment arm of dronabinol capsule 5 mg under fasted conditions, but not a dronabinol oral solution fasted treatment arm.
Comparing dronabinol oral solution 4.25 mg with dronabinol capsule 5 mg under fed conditions, the mean Cmax was 42% lower and mean dronabinol total exposure (AUC0–t and AUC0–∞) was 13%–16% lower with dronabinol oral solution. Mean Tmax was generally similar between these 2 treatment arms. However, the Tlag under fed conditions was 92.6% faster for dronabinol oral solution 4.25 mg compared with dronabinol capsule 5 mg, indicating a 1.87-hour faster time to measurable dronabinol plasma levels in the fed state. This was also supported by the larger percentage of individuals with plasma concentrations of dronabinol above the lower limit of quantification up to 4 hours postdose for dronabinol oral solution 4.25 mg compared with dronabinol capsule 5 mg under fed conditions: 100% versus 15% of individuals, respectively, had measurable plasma dronabinol concentrations at 0.5 hour postdose.
Interestingly, dronabinol oral solution 4.25 mg showed lower interindividual variability in plasma concentration than dronabinol capsule 5 mg under fed conditions. This may, in part, be due to the different drug delivery systems used by the capsule and oral solution. Dronabinol capsules utilize a sesame-oil-based system, while dronabinol oral solution is a hydroalcoholic formulation. Oil-based drug delivery systems undergo lipolysis prior to absorption,15 which may affect pharmacokinetic parameters and interindividual variability. Hydroalcoholic formulations do not undergo lipolysis; thus, interindividual variability may be lower than what is observed with oil-based drug formulations. It has been previously noted with other formulations that Δ9-THC or dronabinol has a concentration-dependent antiemetic effect; any inherent variability of absorption may cause prescribers to either increase a dose unnecessarily or abandon the drug prematurely.16 Consequently, it is reasonable to consider that both intra- and interindividual variability are likely to impact both efficacy and safety. Indeed, the importance of this variability has been recognized with previous formulations, as stated by Lucas and Laszlo7 in 1980: “Δ9-tetrahydrocannabinol is erratically absorbed from the gastrointestinal tract, and dosage individualization may be necessary to control these patients.” Intra- and interindividual variability are likely to have important implications for safety and efficacy of dronabinol products, as well as patient adherence with these drugs. Consequently, the lower interindividual pharmacokinetic variability observed with the oral solution of dronabinol relative to dronabinol capsule may have implications with regard to providing more consistent drug delivery for patients and may be an important consideration in the choice of dronabinol product for the care of patients.
There are several potential limitations to the study. Because the study was designed to assess comparative bioavailability of dronabinol oral solution and dronabinol capsules under fed conditions, the study was conducted in a healthy population. In addition, the effects of gender on the pharmacokinetic differences observed between formulations and/or fed and fasted states were not explored. Consequently, additional studies are warranted to understand whether the findings of the current study can be generalized to the indicated patient populations, namely patients with AIDS who have anorexia associated with weight loss and patients with chemotherapy-induced nausea and vomiting without adequate response to conventional antiemetic therapy.12,14
Conclusion
An appreciable food effect was observed for both the dronabinol oral solution and dronabinol capsule formulations. Under fed conditions, dronabinol oral solution exhibited a similar pharmacokinetic profile for key parameters, such as Cmax and AUC. However, initial dronabinol absorption was faster, with detectable plasma dronabinol concentrations within 30 minutes in all individuals tested. Dronabinol as an easy-to-swallow oral solution also showed lower interindividual absorption variability versus the capsule formulation; this may be an important consideration in the selection of an appropriate dronabinol product for patients.
Acknowledgments
This study was supported by INSYS Therapeutics, Inc., which had a role in study design, supervision of data collection, analysis and interpretation of data, and reporting of results. Technical editorial and medical writing assistance was provided, under the direction of the authors, by Mary Beth Moncrief, PhD, and Gregory Scott, PharmD, Synchrony Medical Communications, LLC, West Chester, PA. Funding for this support was provided by INSYS Therapeutics, Inc. This research was published in abstract form (Oh DA, Parikh N, Khurana V, Cognata Smith C, Vetticaden S. J Clin Oncol. 2016;34(suppl; abstr e21593), available at http://meetinglibrary.asco.org/content/170535-176).
Disclosure
All authors are full-time employees of INSYS Therapeutics, Inc. The authors report no other conflicts of interest in this work.
References
Badowski ME, Perez SE. Clinical utility of dronabinol in the treatment of weight loss associated with HIV and AIDS. HIV AIDS (Auckl). 2016;8:37–45. | ||
Beal JE, Olson R, Laubenstein L, et al. Dronabinol as a treatment for anorexia associated with weight loss in patients with AIDS. J Pain Symptom Manage. 1995;10(2):89–97. | ||
Beal JE, Olson R, Lefkowitz L, et al. Long-term efficacy and safety of dronabinol for acquired immunodeficiency syndrome-associated anorexia. J Pain Symptom Manage. 1997;14(1):7–14. | ||
Plasse TF, Gorter RW, Krasnow SH, Lane M, Shepard KV, Wadleigh RG. Recent clinical experience with dronabinol. Pharmacol Biochem Behav. 1991;40(3):695–700. | ||
Ekert H, Waters KD, Jurk IH, Mobilia J, Loughnan P. Amelioration of cancer chemotherapy-induced nausea and vomiting by delta-9-tetrahydrocannabinol. Med J Aust. 1979;2(12):657–659. | ||
Kluin-Neleman JC, Neleman FA, Meuwissen OJ, Maes RA. Delta 9-tetrahydrocannabinol (THC) as an antiemetic in patients treated with cancer chemotherapy; a double-blind cross-over trial against placebo. Vet Hum Toxicol. 1979;21(5):338–340. | ||
Lucas VS Jr, Laszlo J. Delta 9-tetrahydrocannabinol for refractory vomiting induced by cancer chemotherapy. JAMA. 1980;243(12):1241–1243. | ||
Orr LE, McKernan JF, Bloome B. Antiemetic effect of tetrahydrocannabinol. Compared with placebo and prochlorperazine in chemotherapy-associated nausea and emesis. Arch Intern Med. 1980;140(11):1431–1433. | ||
Sallan SE, Cronin C, Zelen M, Zinberg NE. Antiemetics in patients receiving chemotherapy for cancer: a randomized comparison of delta-9-tetrahydrocannabinol and prochlorperazine. N Engl J Med. 1980;302(3):135–138. | ||
Lane M, Vogel CL, Ferguson J, et al. Dronabinol and prochlorperazine in combination for treatment of cancer chemotherapy-induced nausea and vomiting. J Pain Symptom Manage. 1991;6(6):352–359. | ||
Meiri E, Jhangiani H, Vredenburgh JJ, et al. Efficacy of dronabinol alone and in combination with ondansetron versus ondansetron alone for delayed chemotherapy-induced nausea and vomiting. Curr Med Res Opin. 2007;23(3):533–543. | ||
Marinol® (dronabinol) capsules [package insert]. North Chicago, IL: AbbVie, Inc.; 2015. | ||
Parikh N, Kramer WG, Khurana V, Cognata Smith C, Vetticaden S. Bioavailability study of dronabinol oral solution versus dronabinol capsules in healthy volunteers. Clin Pharmacol. 2016;8:155–162. | ||
SYNDROS (dronabinol) oral solution, CX [package insert]. Chandler, AZ: Insys Therapeutics, Inc; 2016. | ||
MacGregor KJ, Embleton JK, Lacy JE, et al. Influence of lipolysis on drug absorption from the gastro-intestinal tract. Adv Drug Deliv Rev. 1997;25(1):33–46. | ||
Chang AE, Shiling DJ, Stillman RC, et al. Delta-9-tetrahydrocannabinol as an antiemetic in cancer patients receiving high-dose methotrexate: a prospective, randomized evaluation. Ann Intern Med. 1979;91(6):819–824. |
Supplementary materials
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.