Back to Journals » Drug Design, Development and Therapy » Volume 15
Effect of Evodiamine on Rat Colonic Hypermotility Induced by Water Avoidance Stress and the Underlying Mechanism
Authors Ren H, Yuan F, Tan W, Ding Y, An P, Luo H
Received 24 December 2020
Accepted for publication 28 January 2021
Published 10 February 2021 Volume 2021:15 Pages 441—452
DOI https://doi.org/10.2147/DDDT.S298954
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Tuo Deng
HaiXia Ren,1,* FangTing Yuan,1,2,* Wei Tan,1 YiJuan Ding,1 Ping An,1 HeSheng Luo1
1Department of Gastroenterology, Renmin Hospital of Wuhan University, Wuhan, Hubei Province, People’s Republic of China; 2Key Laboratory of Hubei Province for Digestive System Diseases, Renmin Hospital of Wuhan University, Wuhan, Hubei Province, People’s Republic of China
*These authors contributed equally to this work
Correspondence: HeSheng Luo Email [email protected]
Background and Aim: EVO is a natural alkaloid that reportedly has potential value in regulating gastrointestinal motility, but this conclusion remains controversial, and the molecular mechanism is unclear. In this study, we aimed to explore the effect of short-chain fatty acids on rat colonic hypermotility induced by water avoidance stress and the underlying mechanism.
Methods: We constructed a hypermotile rat model by chronic water avoidance stress, and Western blot was used to detect the protein level of nNOS in colon tissue. The organ bath and multichannel physiological signal acquisition systems were used to examine the spontaneous contractions of smooth muscle strips. The whole‐cell patch‐clamp technique was used to investigate L‐type voltage‐dependent calcium and BKCa channel currents in colonic smooth muscle cells.
Results: EVO inhibited the spontaneous contractions of colonic smooth muscle strips in a dose-dependent manner. Moreover, EVO decreased the fecal output induced by chronic water avoidance stress. TTX did not block the inhibitory effect of EVO on spontaneous colon contractions, while L-NNA, a selective nNOS synthase inhibitor, did partially abolish this inhibitory effect. The protein expression of nNOS in the colon tissues of rats administered EVO was significantly increased compared to that in control rats. EVO reversibly inhibited the L-type calcium channel current without changing the steady-state activation or inactivation in colonic smooth muscle cells. EVO significantly inhibited the BKCa current but did not change the shape of the I‐V curves.
Conclusion: EVO inhibits gastrointestinal motility by inhibiting L-type calcium and BKCa channels in colonic smooth muscle cells and indirectly interacting with nNOS.
Keywords: evodiamine, gastrointestinal motility, nitric oxide, L‐type voltage‐dependent calcium channels, BKCa channel
Introduction
Evodiamine (EVO), the major alkaloid component isolated from dry Evodiae fructus, has a variety of biological activities and has been recommended for the treatment of ailments such as abdominal pain, nausea, diarrhea.1 In recent years, studies have demonstrated that EVO can regulate gastrointestinal motility, but the evidence is insufficient, and the potential mechanisms remain unknown.
Both neurotransmitters and ion channels play important roles in regulating gastrointestinal motility. Nitric oxide (NO) is a small and reactive molecule that has various physiological and biological properties. In the gastrointestinal tract, NO is recognized as a negative neurotransmitter that mediates a variety of motility patterns.2–4 Animal studies5–7 have proven that both basal fundic tone and relaxation of the proximal stomach induced by vagal stimulation and meal ingestion are mediated by NO. Data from human studies8 indicate that NO inhibits esophageal smooth muscle function. Studies have also found that NO decreases the frequency of gastric contractions and inhibits gastric emptying,9 while NO donors slow gastric emptying and improve accommodation of the proximal stomach.10 In terms of intestinal motility, NO induces the relaxation of smooth muscles in the distal colon and rectum11,12 While there is no evidence of EVO affecting the production of NO in the gastrointestinal tract, studies have reported that EVO can increase NO production in cardiomyocytes and umbilical vein endothelial cells.13,14
Three types of NO synthases exist in the gastrointestinal tract, neuronal, endothelial, and inducible NOS (nNOS, eNOS, and iNOS, respectively), and the influence of NO on gastrointestinal motility is largely controlled by nNOS.
Calcium is a fundamental second messenger in smooth muscle cells (SMCs) that directly or indirectly controls the contractile activity of smooth muscles.15 The entry of Ca2+ through L-type calcium channels is the primary mechanism for excitation–contraction coupling in gut smooth muscles, and L-type calcium channels play a critical role in the amplitude of gut contractions.16,17 Furthermore, the combination of depolarization and an increase in the [Ca2+]I contributes to BKCa channel opening, and BKCa channel repolarization functions as an ideal negative feedback regulator by decreasing the voltage‐dependent calcium influx via membrane hyperpolarization. However, whether the relaxant effect of EVO on colonic contraction is associated with L-type calcium and BKCa channels in SMCs remains unknown. In this study, we wanted to investigate 1) the effects of EVO on colonic motility and 2) the mechanism by which EVO regulates gastrointestinal motility.
Materials and Methods
Animals
Male Wistar rats weighing 180–200 g were obtained from Vital River Laboratories (Beijing, China), housed in an environmentally controlled room (22±1°C, 65% humidity, 12 h light/dark cycle) and fed standard laboratory chow with free access to water. All animal experimental procedures were approved by the Institutional Animal Care and Use Committee of Wuhan University (approval ID: WHU 20110312) and adhered to the Declaration of the National Institutes of Health Guide for Care and Use of Laboratory Animals and the People’s Republic of China animal welfare legislations.
Water Avoidance Stress Protocol
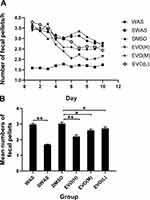
Referring to our previous experimental methods,18 the rats were placed on a block (8×8×10 cm) affixed to the center of a plexiglass cage (45×25×25 cm) for 1 h daily for 10 consecutive days. The cage was filled with room temperature water (25 °C), and the top of the block was ensured to be higher than the liquid level, which was less than 1 cm. The sham water avoidance stress (SWAS) rats were placed on the same block, but the container was empty. The procedures were performed at 9:00 am every day. The number of fecal pellets within 24 h was counted for each rat throughout the entire experiment.
Drug Administration
The rats were randomly divided into the SWAS, WAS, DMSO, EVO (L), EVO (M) and EVO (H) groups. After 1 week of acclimation, the rats in the SWAS group were subjected to sham water avoidance stress, and the other groups were subjected to water avoidance stress for 10 consecutive days. Ten days later, rats in DMSO, EVO (L), EVO (M) and EVO (H) groups were intraperitoneally injected with 0.5 mL of DMSO containing 0, 0.67, 2.0, or 6.0 mg/kg EVO daily for 7 consecutive days. Notably, the water avoidance stress continued to be administered on these days, and the total numbers of fecal pellets within 24 h were still counted on days of medication administration.
Western Blotting
Total protein was extracted using RIPA lysis buffer composed of 20 mM Tris–HCl, 0.1 mM PMSF, and 5 ll/mL protease inhibitor cocktail. The protein concentration was determined by a Bio-Rad protein assay (Bio-Rad Laboratories, Hercules, CA, USA). Protein lysate from each sample was separated electrophoretically on a 10% sodium dodecyl sulfate-polyacrylamide gel and then transferred to polyvinylidene fluoride membranes. Then, the membranes were blocked with 5% BSA in TBS-T (Tris-buffered saline, pH 7.6 plus 0.1% Tween-20) for 1 h and incubated with primary antibodies overnight at 4°C. The membranes were then washed and incubated with a secondary antibody (goat anti-rabbit, 1:5000). The results were visualized using an electrogenerated chemiluminescence (ECL) system, and the data are expressed as the band intensity ratios of the target proteins to GAPDH. The following primary antibodies were utilized: a rabbit polyclonal antibody for GAPDH (1:1000, China, AB-P-R001) and a rabbit monoclonal antibody for nNOS (1:1000, Cell Signaling, 4231).
Colonic Motility Tests in vitro
The rats were sacrificed by cervical dislocation, and their colons were carefully removed and cleaned in Ca2+-free physiological saline solution and cut along the mesenteric border. The proximal colon was clipped, and the tissue was pinned with the mucosal side up in a dish containing Tyrode’s buffer (147.0 mM NaCl, 4.0 mM KCl, 2.0 mM CaCl2, 0.42 mM NaH2PO4, 2.0 mM Na2HPO4, 1.05 mM MgCl2, 5.5 mM glucose). In our experiments, two kinds of muscle strips were prepared: smooth muscle comprising all layers and denuded mucosa and submucosa. The circular muscle (CM) and longitudinal muscle (LM) strips (3×10 mm; width× length) were cut along the direction of the circular or longitudinal axis, respectively. Each smooth muscle strip was fixed in a tissue chamber containing 7 mL of Tyrode’s buffer (pH 7.4, temperature 37°C, bubbled with a mixture of 97% O2 and 3% CO2). One end of the strip was fixed to a hook on the bottom of the chamber, while the other end was fixed to an isometric force transducer (JZJOIH, Chengdu, China). The muscle strips were incubated for 60 min under a resting preload (CM 1.0 g, LM 1.5 g) and washed every 20 min with Tyrode’s buffer during the equilibration period. The contractile amplitude of the colonic strips was recorded by a RM6240 multichannel physiological signal system (Chengdu, China).
Preparation of Isolated Colonic Smooth Muscle Cells
Referring to previous experimental methods,19 colonic SMCs were isolated by enzymatic digestion. The rats were sacrificed by cervical dislocation, and their colons were carefully removed, after which the fresh colon tissue was pinned in a Petri dish lined with Sylgard. The mucosal and submucosal layers were removed under a dissection microscope (Olympus, Japan). The muscle layer was cut carefully into small pieces (3×4 mm), placed in a Ca2+-free physiological saline solution (Ca2+-free PSS) containing 135 mM NaCl, 5 mM KCl, 10 mM HEPES, 1.2 mM MgCl2, and 10 mM glucose (pH adjusted to 7.35–7.45), and bubbled with oxygen. The segments were incubated for 20–35 min at 36.5°C in digestion medium comprised of Ca2+-free PSS containing 0.12% (w/v) collagen II, 0.2% soybean trypsin inhibitor and 0.2% bovine serum albumin (BSA). After the digestion was complete, the segments were removed from the digestion medium and washed 4 times with Ca2+-free PSS to terminate the digestion; the tissue was then stored at 4°C in conservation medium containing 10% BSA. Single SMCs were dispersed via gentle trituration with a fire-polished Pasteur pipette.
Whole-Cell Patch-Clamp Recording
A few drops of cell suspension were placed into a perfusion chamber that was mounted on an inverted microscope (IX70, Olympus, Japan). After the cells adhered to the bottom of the chamber (approximately 10 min), they were infused with oxygen-saturated Tyrode’s buffer (1.2 mL/min) using a pump (DHL-B, Shanghai, China). Borosilicate pipettes (inside diameter of 0.86 mm, outside diameter of 1.5 mm, Sutter, Novato, CA, USA) were constructed using a micropipette puller (P-97; Sutter, Novato, CA, USA) and had a resistance of 4–6 MΩ. Whole-cell currents were recorded with an Axopatch 700B amplifier (Axon Instruments, Burlingame, CA, USA), and the data were digitized at 1 kHz and filtered at 800 Hz. Acquisition and analysis of the physiological signals were carried out using pClamp 10.2 software (Axon Instruments, Burlingame, CA, USA). The experiment was performed at room temperature (approximately 25°C). The pipette solution for recording L-type calcium currents (ICa,L) contained 135 mM CsCl, 3 mM MgCl2, 2 mM Na2ATP, 10 mM ethylene glycol-bis (2-aminoethyl-ether)-N,N,N’,N’-tetraacetic acid (EGTA), 10 mM HEPES, and 20 mM tetraethylammonium (TEA) (pH adjusted to 7.3 with CsOH).
Statistical Analysis
The data are expressed as the mean ± SEM and were analyzed using pClamp 10.2, Origin 6.0, SPSS 20.0 and SigmaPlot 12.5 software. Significant differences between groups were evaluated using paired Student’s t-tests and one-way ANOVA followed by the LSD post hoc test. The mean value of the average tension was the 5-min period before or after treatment with EVO, CCK-8 or devazepide. The significance level was set to P<0.05.
Results
Effect of EVO on Gastrointestinal Motor Function in Rats Subjected to Water Avoidance Stress
Rats were randomly divided into 6 groups: SWAS, WAS, DMSO, EVO (L), EVO (M), and EVO (H). A rat model of chronic stress was constructed according to previously described methods. On the fourth day after model establishment, the rats in the DMSO, EVO (L), EVO (M), EVO (H) groups were intraperitoneally injected with DMSO (0.5 mL) and EVO (0.67 mg/kg, 2.0 mg/kg, 6 mg/kg), respectively, and fecal pellet expulsion was used to evaluate intestinal motor function. Figure 1A shows fecal pellet expulsion during the whole experimental protocol. Figure 1B shows the mean numbers of fecal expulsion of 6 groups for the last 7 days. As shown above, the fecal pellets per hour for the WAS is higher than SWAS group (WAS:2.98±0.06 VS SWAS: 1.70±0.02, P=0.0000). Further, compared with WAS group, the fecal output in DMSO group did not change significantly (DMSO:3.03±0.09 vs WAS:2.98±0.06, P=0.47).However, the fecal pellets in DMSO group were higher than that of three EVO group (DMSO:3.03±0.09 vs EVO (H): 2.21±0.01, P=0.0006; DMSO:3.03±0.09 vs EVO (M): 2.60±0.06, P=0.01; DMSO:3.03±0.09 vs EVO (L): 2.73±0.09, P=0.03). These data demonstrated that DMSO had no effect on intestinal motility.
Evodiamine Inhibits the Spontaneous Contractile Activity of the Proximal Colon in vitro
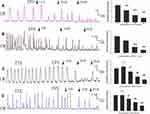
EVO dose-dependently inhibited the spontaneous contraction of the LM and CM strips comprising all layers. In the LM strips (Figure 2A), the mean contractile amplitude before adding EVO was1.11±0.12g, in the presence of EVO 10,20,40uM, it was dropped to 0.77±0.06 g (p=0.000 VS Control),0.45±0.07 g (p=0.006 VS Control),0.28±0.06 g (p=0.003 VS Control), respectively. Similarly, before the addition of EVO, the mean amplitude of CM strips (Figure 2B) was 0.74±0.09 g, after adding EVO 5,10,20uM, it was decreased to 0.55±0.03g (p=0.097 VS Control),0.26±0.02g (p=0.005 VS Control), 0.13±0.01g (p=0.004 VS Control), respectively.
What’s more, we observed that spontaneous contractions of colonic strips and in vitro induced by EVO in the presence of TTX (1uM) was still existed. After preincubation with TTX, the contractile amplitudes of both LM and CM strips were changed, with the mean contractile amplitude of LM strips (Figure 2C) increasing from 1.63±0.33 to 1.83±0.37 (P=0.783, compared to normal, n=5), and the average amplitude of CM strips (Figure 2D) changing from 0.90±0.13 g to 0.90±0.11 g (P=0.987, compared to normal, n=5). When EVO 10,20,40 µM was added to the organ bath after TTX incubation, the mean contractile amplitude of LM strips (Figure 2C) decreased to 1.30±0.22 g (P=0.037 vs TTX), 0.76±0.16 g (P=0.008 vs TTX), 0.45±0.18 g (P=0.006 vs TTX), respectively. When EVO 5,10,20 µM was added to the organ bath after TTX incubation, the mean contractile amplitude of CM strips (Figure 2D) decreased to 0.70±0.10 g (P=0.06 vs TTX), 0.50±0.05 g (P=0.006 vs TTX), 0.29±0.04 g (P=0.004 vs TTX), respectively. From what has been discussed above, these inhibitory effects of EVO cannot be blocked by TTX (1 μM).
Determination of Whether NO is Involved in the Inhibitory Effect of EVO on Colonic Strip Contractions
To investigate whether EVO inhibits colonic muscle strips via the NO pathway, L-NNA (a selective nNOS synthase inhibitor) was used to block the generation of NO. After preincubation with L-NNA, the contractile amplitudes of both LM and CM strips were significantly increased, with the mean contractile amplitude of LM strips (Figure 3C) increasing from 1.06±0.06 to 1.23±0.07 (P=0.098, compared to normal, n=5), and the average amplitude of CM strips (Figure 3D) increasing from 0.80±0.15 g to 0.96±0.22 g (P=0.26, compared to normal, n=5). When 40 µM EVO was added to the organ bath after L-NNA incubation, the mean contractile amplitude of LM strips (Figure 3A) decreased to 0.86±0.10 g (P=0.01 vs control), and when 20 µM EVO was added to the organ bath after L-NNA incubation, the mean contractile amplitude of CM strips (Figure 3B) decreased to 0.66±0.20 g (P=0.022 vs control). The percent inhibition of EVO on CM strips without L-NNA preincubation was 0.80±0.05, and the percent inhibition of EVO on CM strips with L-NNA preincubation was 0.35±0.07 (P=0.001 vs without L-NNA incubation). The percent inhibition of EVO on LM strips without L-NNA was 0.76±0.09, and the percent inhibition of EVO on LM strips with L-NNA preincubation was 0.30±0.04 (P=0.011 vs without L-NNA incubation).
nNOS Protein Expression in Proximal Colon Tissue
According to the Western blot results in Figure 4A and B, nNOS expression was dose-dependently upregulated in EVO-treated rats compared with rats in the DMSO group (EVO (L) vs DMSO: P=0.011; EVO (M) vs DMSO: P=0.000; EVO (H) vs DMSO: P=0.000). Chronic water avoidance stress did not significantly change the nNOS protein level in the colon tissue compared with that in SWAS rats.
EVO Continues to Inhibit Colonic Strips After Removal of the Mucosa and Submucosa
To investigate whether the inhibitory effect of EVO on colonic muscle strips is mucosal dependent, we removed the mucosa and submucosa, and the inhibitory effect of EVO on colonic strips remained (Figure 5A and B). In Figure 5E, prior to the addition of EVO, the mean amplitude of LM strip contraction was 0.82± 0.12 g, and the amplitude was reduced to 0.49± 0.14 g after the addition of EVO at a concentration of 40μM (P =0.006 vs normal, n = 5). Prior to the addition of EVO, the mean amplitude of CM strip contractions was 0.47 ± 0.02 g, and the amplitude was reduced to 0.29±0.05 g after the addition of EVO at a concentration of 20 um (P=0.01 vs normal, n = 5).
Figure 5C and D showed the spontaneous contractions of colonic strips after removal of the mucosa and submucosa induced by EVO in the presence of TTX (1uM). In Figure 5F, prior to the preincubation of TTX, the mean amplitude of LM strips was 0.93± 0.16 g, and the amplitude was changed to 0.96± 0.16 g in the presence of TTX (P =0.655 vs normal, n = 3). When EVO was added at a concentration of 40μM, the amplitude was reduced to 0.48± 0.17 g (P =0.009 vs TTX, n = 3). Prior to the preincubation of TTX, the mean amplitude of CM strips was 1.37± 0.08 g, and the amplitude was changed to 1.39± 0.20 g in the presence of TTX (P =0.930 vs normal, n = 3). When EVO was added at a concentration of 20uM, the amplitude was reduced to 0.98± 0.18 g (P =0.018 vs TTX, n = 3). In conclusion, TTX (1μm) did not block these inhibitory effects of EVO, even when mucosa and submucosa were removed.
Effects of EVO on L-Type Calcium Channels
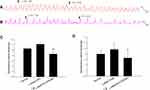
The cell membrane potential was held at −50 mV, and ICa,L was evoked by the single-pulse protocol in whole-cell voltage-clamp mode as follows: 10 mV depolarizing steps from a constant holding potential of −50 mV to +20 mV for 300 ms. I–V curves were obtained by plotting the peak current density (Imax/Cm) in response to voltage pulses at potentials between −50 and +20 mV, and the peak current value appeared at 0 mV. To examine the direct effects of EVO on L-type calcium channels, we incubated the cells with 5, 10 and 20 µM EVO. Figure 6A and C shows the representative traces before (normal) and after the administration of EVO (5, 10 and 20 μM) and DMSO (1, 2 µL and 4 µL). In Figure 6B, EVO at concentrations of 5 μM, 10 µM and 20 μM exerted inhibitory effects on the peak ICa,L, with the current density decreasing from −5.17 ± 0.35 pA/pF to −3.10 ± 0.16 pA/pF (P=0.002, vs normal, n = 6), −2.98 ±0.26 pA/pF (P=0.000, vs normal, n = 6) and −1.39 ±0.22 pA/pF (P=0.000, vs normal, n = 6) at 0 mV. We did not observe any significant inhibitory effect on the peak ICa,L when the same dose of DMSO was added (Figure 6D). The inhibitory effect reached a steady level after the application of EVO for 1‐2 min and was partially reversed after the removal of EVO.
Figure 7A and B shows the raw traces recorded before (normal) and after the addition of EVO (5, 10 µM and 20 μM) and DMSO (1, 2 µL and 4 µL). I–V curves were obtained by plotting the peak current density (Imax/Cm) in response to voltage pulses at potentials between −50 and 20 mV. From the I–V curve, we can see that EVO inhibited I Ca,L at every depolarizing voltage pulse from −50 mV to +20 mV, but the shape of the I–V curves was not changed (Figure 7C). We did not observe any significant change in I Ca,L when the same dose of DMSO was added (Figure 7D).
Figure 8 shows that effect of EVO (10uM) on the steady-state activation of I Ca,L and the steady-state inactivation of I Ca, L. The curve was fitted by the Boltzmann equation G/Gmax = 1/[1 + exp (VT − V1/2/κ)], where I/Imax was used instead of G/Gmax. I/Imax represents the ratio of an individual current to the maximum current, VT represents the depolarizing pulse value, V1/2 represents the half-maximum activation voltage, and k is the slope factor. It is worth noting that V1/2 and k together determine the shape of the steady-state activation of I Ca, L. Figure 8A shows that EVO evoked no significant shifts in the steady-state activation curve of L-type calcium channels. The V1/2 values were −18.78 ± 2.42 mV in the normal group and −19.73± 2.30 mV in the EVO (10 μM)‐treated group, and the values for the slope factor κ were 3.94 ± 2.53 and 3.78 ± 2.71 in the normal and EVO (10 μM)‐treated groups, respectively (R2=0.95, vs normal, n = 5).
To analyze the effect of EVO (10 μM) on the steady-state inactivation of L-type calcium channels, the following double-pulse protocol was used: 1000 ms of a conditioning pulse ranging from −50 mV to +20 mV in 10 mV increments, followed by 200 ms of a test pulse at 0 mV. Then, the data were fitted to the Boltzmann equation, G/Gmax = 1/[1 +exp (VT - V1/2/k)], where G/Gmax was replaced by I/Imax, which represents the ratio of an individual current to the maximum current, VT represents the membrane potential value, V1/2 represents the half-maximum inactivation voltage, and κ is the slope factor, and V1/2 and k together determine the shape of the steady-state inactivation curve of I Ca, L. Figure 8B shows the effect of EVO (10 μM) evoked no significant shifts in the steady-state inactivation curve of ICa.L. In the normal and EVO (10 µM)-treated groups, the V1/2 values were −30.96 ± 0.48 mV and −32.24 ± 0.48 mV, and the κ values were 5.56 ± 0.43 and 5.19 ± 0.41, respectively (R2=1.0 vs normal, n=5).
Effects of EVO on BKCa Channels
The cell membrane potential was held at −80 mV, and the BKCa current was evoked by the following single‐pulse protocol: 20 mV depolarizing steps from a constant holding potential of −80 mV to +60 mV. As shown in Figure 9A, EVO at concentrations of 5 µM and 10 µM significantly inhibited the BKCa current. Figure 9B shows the representative traces of BKCa channels at 60 mV before and after application of EVO (5 and 10μM).In Figure 9C, the current density at 60 mV decreased from 17.93 ±1.49 pA/pF to 12.13 ± 1.23 pA/pF (P=0.000, vs normal, n = 5) and 5.73 ± 1.56 pA/pF (P=0.000, vs normal, n = 5). The I‐V curves indicate that EVO inhibited IBK,Ca at every pulse from a holding potential of −80 mV to +60 mV, but the shape of the I‐V curves did not change (Figure 9D).
Discussion
We herein investigated the effect of EVO on gastrointestinal motility and the underlying mechanism in isolated colon tissue. These data provide the first evidence that both NO and L-type calcium channels play important roles in the inhibitory effect of EVO on gastrointestinal motility.
EVO, a natural alkaloid isolated from dry Evodiae fructus that has several well-defined physiological effects, has the potential to regulate gastrointestinal motility and has aroused our interest. Studies have reported that EVO inhibits both gastric emptying and gastrointestinal transit,20 but this conclusion has been verified in only animal experiments in vivo, and the specific mechanism has not been discussed. In addition, there are also reports of EVO exerting stimulatory effects on jejunal contractility, and this effect is Ca2+-dependent.21 Because different approaches have led to different conclusions, the regulatory effect of EVO on gastrointestinal motility and the mechanism need to be further explored.
Our results showed that EVO significantly reversed the hypermotility induced by water avoidance stress and slowed gastrointestinal transit. Our observations were consistent with those of a previous study reporting that EVO inhibited both gastric emptying and gastrointestinal transit.19 To further explore the mechanism, we chose the colon as the research object of the in vitro experiment.
NO is a small and reactive molecule that has been recognized as a negative neurotransmitter that mediates a variety of motility patterns in the gastrointestinal tract. Three types of NO synthase exist in the gastrointestinal tract, with the main type being nNOS. Studies have reported that EVO can increase NO production in cardiomyocytes and umbilical vein endothelial cells, but there are no reports on the relationship between EVO and colonic NO production. In our experiment, the relaxant effects of EVO on both LM and CM strips were significantly reduced by L‐NNA, and the protein level of nNOS in the colon tissue of rats administered EVO was significantly increased compared to that in control rats, indicating that the inhibitory effect of EVO on the rat colon was partially mediated by neural NO release. However, unexpectedly, L-NNA did not completely block the inhibitory effect of EVO on colonic muscle strips, and further experiments were thus designed to determine whether EVO can act directly on colon smooth muscles.
We next removed the mucosal and submucosal layers from the colon smooth muscle strips, and the relaxant effect of EVO remained. Moreover, this effect could not be blocked by TTX, a neuronal voltage‐dependent Na+ channel inhibitor, indicating that neurons within the intramural plexuses are not responsible for the action of EVO. We inferred that EVO most likely acts directly on SMCs. To investigate this possibility, further studies on the EVO mechanism of action were performed in colonic SMCs with the whole‐cell patch‐clamp technique. Calcium is a principal second messenger in SMCs, and it is well known that increasing the [Ca2+]I, which leads to binding to calmodulin and to the activation of myosin light chain kinase, is the primary contraction stimulant.22 The entry of Ca2+ through L‐type voltage‐dependent calcium channels opened by depolarization or during an action potential is the main factor contributing to increases in intracellular Ca2+ concentrations.14–16 One study reported that the increased intracellular Ca2+ induced by high glucose in human umbilical vein endothelial cells was significantly reversed by EVO,14 but no similar studies on gastrointestinal SMCs have been performed until now. In our study, EVO inhibited ICa,L in a concentration-dependent manner, but it did not change the shape of the ICa,L I–V curve, suggesting that EVO did not modify the voltage‐gated property of L‐type calcium channels. Moreover, the steady‐state activation and inactivation of L‐type calcium channels were not significantly altered by EVO. Depolarization and an increase in the [Ca2+]I in combination contributes to BKCa channel opening, and BKCa channel repolarization also functions as an ideal negative feedback regulator by decreasing voltage‐dependent calcium influx via membrane hyperpolarization. In the present study, IBK,Ca was significantly inhibited by EVO in colonic SMCs. The reduction in IBK,Ca may be related to the EVO‐induced decrease in Ca2+ influx, but the evidence herein does not rule out that EVO directly acts on BKCa channels and inhibits the BKCa current. Taken together, our results indicate that EVO inhibits L‐type calcium and BKCa channel currents, and these reactions lead to the subsequent EVO-induced relaxation of spontaneous smooth muscle strip contractions in the rat colon.
Conclusion
In summary, EVO can significantly reverse the hypermotility induced by chronic water avoidance stress, potentially by promoting the release of neuronal NO and inhibiting L-type calcium channels, BKCa channels and colonic SMCs. Many patients with functional gastrointestinal disorders, such as diarrhea and predominant irritable bowel syndrome (IBS), have increased gastrointestinal motility to some degree. Due to the similar characteristics of the animal model used in the study of IBS, these findings may offer interesting clinical therapeutic perspectives.
Acknowledgments
Thanks to all who contributed to this research. This work was supported by a grant from Special funds for basic scientific research operating expenses of central universities (2042020kf0115) and by the National Natural Science Foundation of China [NSFC: 81600423].
Disclosure
The authors report no conflicts of interest in this work.
References
1. Tan Q, Zhang J. Evodiamine and its role in chronic diseases. Adv Exp Med Biol. 2016;929:315–328.
2. Russo A, Fraser R, Adachi K, et al. Evidence that nitric oxide mechanisms regulate small intestinal motility in humans. Gut. 1999;44(1):72–76. doi:10.1136/gut.44.1.72
3. Hirsch DP, Holloway RH, Tytgat GN, et al. Involvement of nitric oxide in human transient lower esophageal sphincter relaxations and esophageal primary peristalsis. Gastroenterology. 1998;115(6):1374–1380. doi:10.1016/S0016-5085(98)70015-0
4. Tonini M, De Giorgio R, De Ponti F, et al. Role of nitric oxide- and vasoactive intestinal polypeptide-containing neurones in human gastric fundus strip relaxations. Br J Pharmacol. 2000;129(1):12–20. doi:10.1038/sj.bjp.0702977
5. Coulie B, Tack J, Sifrim D, et al. Role of nitric oxide in fasting gastric fundus tone and in 5-HT1 receptor-mediated relaxation of gastric fundus. Am J Physiol. 1999;276(2 Pt 1):G373–G377. doi:10.1152/ajpgi.1999.276.2.G373
6. Meulemans AL, Eelen JG, Schuurkes JA. NO mediates gastric relaxation after brief vagal stimulation in anesthetized dogs. Am J Physiol. 1995;269(2 Pt 1):G255–G261. doi:10.1152/ajpgi.1995.269.2.G255
7. Boeckxstaens GE, Pelckmans PA, Bogers JJ, et al. Release of nitric oxide upon stimulation of nonadrenergic noncholinergic nerves in the rat gastric fundus. J Pharmacol Exp Ther. 1991;256(2):441–447.
8. Murray JA, Ledlow A, Launspach J, et al. The effects of recombinant human hemoglobin on esophageal motor functions in humans. Gastroenterology. 1995;109(4):1241–1248. doi:10.1016/0016-5085(95)90584-7
9. Tack J, Demedts I, Meulemans A, et al. Role of nitric oxide in the gastric accommodation reflex and in meal induced satiety in humans. Gut. 2002;51(2):219–224. doi:10.1136/gut.51.2.219
10. Konturek JW, Thor P, Domschke W. Effects of nitric oxide on antral motility and gastric emptying in humans. Eur J Gastroenterol Hepatol. 1995;7(2):97–102.
11. Smith TK, Spencer NJ, Hennig GW, et al. Recent advances in enteric neurobiology: mechanosensitive interneurons. Neurogastroenterol Motil. 2007;19(11):869–878. doi:10.1111/j.1365-2982.2007.01019.x
12. Jing H, Qin J, Feng M, et al. Nitric oxide in enteric nervous system mediated the inhibitory effect of vasopressin on the contraction of circular muscle strips from colon in male rats. Neurogastroenterol Motil. 2011;23(3):e125–e135. doi:10.1111/j.1365-2982.2010.01646.x
13. He N, Gong QH, Zhang F, et al. Evodiamine inhibits angiotensin II-induced rat cardiomyocyte hypertrophy. Chin J Integr Med. 2018;24(5):359–365. doi:10.1007/s11655-017-2818-9
14. Lv Q, Xue Y, Li G, et al. Beneficial effects of evodiamine on P2X(4)-mediated inflammatory injury of human umbilical vein endothelial cells due to high glucose. Int Immunopharmacol. 2015;28(2):1044–1049. doi:10.1016/j.intimp.2015.08.020
15. Murthy KS. Signaling for contraction and relaxation in smooth muscle of the gut. Annu Rev Physiol. 2006;68:345–374. doi:10.1146/annurev.physiol.68.040504.094707
16. Bolton TB, Prestwich SA, Zholos AV, et al. Excitation-contraction coupling in gastrointestinal and other smooth muscles. Annu Rev Physiol. 1999;61:85–115. doi:10.1146/annurev.physiol.61.1.85
17. Wegener JW, Schulla V, Koller A, et al. Control of intestinal motility by the Ca(v)1.2 L-type calcium channel in mice. FASEB J. 2006;20(8):1260–1262. doi:10.1096/fj.05-5292fje
18. Quan X, Luo H, Fan H, et al. Brain-derived neurotrophic factor contributes to colonic hypermotility in a chronic stress rat model. Dig Dis Sci. 2015;60(8):2316–2326. doi:10.1007/s10620-015-3695-8
19. Quan X, Luo H, Liu Y, et al. Hydrogen sulfide regulates the colonic motility by inhibiting both L-type calcium channels and BKCa channels in smooth muscle cells of rat colon. PLoS One. 2015;10(3):e121331. doi:10.1371/journal.pone.0121331
20. Wu CL, Hung CR, Chang FY, et al. Effects of evodiamine on gastrointestinal motility in male rats. Eur J Pharmacol. 2002;457(2–3):169–176. doi:10.1016/S0014-2999(02)02687-0
21. Xiong YJ, Chen DP, Peng JY, et al. Characteristics of evodiamine-exerted stimulatory effects on rat jejunal contractility. Nat Prod Res. 2015;29(4):388–391. doi:10.1080/14786419.2014.947485
22. Sanders KM. Invited review: mechanisms of calcium handling in smooth muscles. J Appl Physiol. 2001;91(3):1438–1449. doi:10.1152/jappl.2001.91.3.1438
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.