Back to Journals » International Journal of Nanomedicine » Volume 16
EDTA-K2 Improves the Detection Sensitivity of SARS-CoV-2 IgM and IgG Antibodies by Chelating Colloidal Gold in the Immunochromatographic Assay
Authors Hu X , Wu C, Situ B, Tian P, An T, Li Q, Pan W, Zhang R, Yang B, Sun D, Hu Y, Wang Q, Zheng L
Received 17 September 2020
Accepted for publication 24 December 2020
Published 29 January 2021 Volume 2021:16 Pages 715—724
DOI https://doi.org/10.2147/IJN.S281594
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Farooq A. Shiekh
Xiumei Hu,1,* Changmeng Wu,1,* Bo Situ,1,* Peifu Tian,2,* Taixue An,1 Qiang Li,1 Weilun Pan,1 Ruyi Zhang,1 Biao Yang,3 Dehua Sun,1 Yuhai Hu,2 Qian Wang,1,3 Lei Zheng1
1Department of Laboratory Medicine, Nanfang Hospital, Southern Medical University, Guangzhou 510515, People’s Republic of China; 2Department of Medicine Laboratory, Hankou Hospital of Wuhan, Wuhan 430010, People’s Republic of China; 3Department of Laboratory Medicine, Zhujiang Hospital, Southern Medical University, Guangzhou, Guangdong 510280, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Lei Zheng; Qian Wang Email [email protected]; [email protected]
Objective: The coronavirus disease (COVID-19) pandemic, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is now rapidly spreading globally. Serological tests are an important method to assist in the diagnosis of COVID-19, used for epidemiological investigations. In this study, we aimed to investigate the impact of different types of vacuum collection tubes on the detection of SARS-CoV-2 IgM and IgG antibodies, using the colloidal gold immunochromatographic assay (GICA).
Patients and Methods: A total of 112 patients with COVID-19 and 200 healthy control subjects with no infection were enrolled in this study. Their serum and plasma were collected into four different types of vacuum blood collection tubes. SARS-CoV-2 IgM and IgG specific antibodies in the plasma and serum were then detected by GICA and chemiluminescence assay (CA), respectively. In addition, the particle sizes of different colloidal gold solutions in the presence of different anticoagulants and coagulants were evaluated by both laser diffraction (Malvern) and confocal laser microscope, respectively.
Results: Our results revealed that anticoagulated plasma with EDTA-K2 improved the positive detection rate of SARS-CoV-2 IgM antibodies. Furthermore, our results shown that the detection results by GICA and CA were highly consistent, especially, the results of EDTA-K2 anticoagulated plasma detected by GICA was more consistent with CA results. We confirmed that EDTA-K2 could improve the detection sensitivity of SARS-CoV-2 IgG antibodies by chelating excessive colloidal gold compared with sodium citrate or lithium heparin, these methodologies did not appear to cause false positives. Colloidal gold particles could be chelated and aggregated by EDTA-K2, but not by sodium citrate, lithium heparin and coagulants.
Conclusion: GICA is widely used to detect antibodies for the advantages of convenient, fast, low cost, suitable for screening large sample and require minimal equipment. In this study, we found that EDTA-K2 amplified the positive antibody signal by chelating colloidal gold and improved the detection sensitivity of SARS-CoV-2 IgM and IgG antibodies when using the GICA. Therefore, we suggested that EDTA-K2 anticoagulated plasma was more suitable for the detection of SARS-CoV-2 antibodies.
Keywords: EDTA-K2, SARS-CoV-2, antibodies, gold immunochromatographic assay; GICA
Introduction
Currently, the widespread threat to human health from SARS-CoV-2 urgently requires the development of fast and accurate analytical methods for early diagnosis. At present, pathogen detection and nucleic acid testing are commonly used to identify COVID-19 in the clinic. In addition, infected patients are mostly positive for SARS-CoV-2-specific IgM antibodies after seven to eight days from disease onset and their IgG titers during the recovery period, are more than four times higher than those in the acute phase,1 indicating that serological detection of specific antibodies is crucial for the rapid identification of SARS-CoV-2. Specifically, this study confirmed that the gold immunochromatographic assay (GICA) could serve as a rapid diagnostic test for RT-PCR-negative highly suspected patients and screening of SARS-CoV-2 carries in limited-resource countries.2,3 Recently, GICA has been widely used to detect antibodies, because the test trips based on colloidal gold particles were fast, convenient, and require minimal equipment. Therefore, we feel that this method is a good candidate for the large scale, rapid screening, of both symptomatic and asymptomatic COVID-19 carriers and the screening of healthy workers following isolation.4
Early in 2014, a study found that different additives in the blood collection tubes, were able to influenced the detection of alcohol concentration.5 Moreover, a study found that DNA samples extracted from blood using two different anticoagulants (heparin tubes and EDTA-K2), gave similar results, especially for PCR based applications.6 These studies revealed that different additives in the vacuum blood collection tubes, showed clear confounding effects clinically. However, it is not clear whether these vacuum blood collection tubes with different additives affect the detection of SARS-CoV-2 IgM and IgG antibodies. Therefore, this study investigated the impact of different vacuum blood collection tubes, when detecting SARS-CoV-2 IgM and IgG antibodies in the plasma and serum using GICA. These results are expected to provide a theoretical basis for the future accurate detection of SARS-CoV-2 IgM and IgG antibodies.
Patient Samples
A total of 112 patients diagnosed as having COVID-19 by nucleic acid testing were enrolled from the Respiratory Department of Hankou Hospital of Wuhan, China, from March 6 to March 18, 2020. Nine of the 112 patients had their samples collected into four different types of collection tube. Among the patients, seven of the nine patients diagnosed with COVID-19 were used to quantify the concentration of SARS-CoV-2 IgM and IgG antibodies. Thirty-two of the 112 patients were used to detect SARS-CoV-2 IgM and IgG antibodies using EDTA-K2 as plasma and whole blood anticoagulants. Next, 200 healthy control subjects diagnosed as noninfectious for COVID-19 based on nucleic acid testing, were enrolled from the Department of Clinical Laboratory Medicine of Nanfang Hospital of Guangdong, China, from April 4 to April 5, 2020. Serum and plasma specimens were isolated from 2 mL of blood and placed into four different types of vacuum blood collection tube, which contained different additional reagents, such that the serum was collected into tubes with coagulants present and plasma with different anticoagulants (EDTA-K2, sodium citrate or lithium heparin).
Materials
IgG and IgM specific antibodies were detected in blood samples using recombinant coronavirus N protein labeled, and the results were seen using the naked eye. The new coronavirus antibody detection kit (colloidal gold based) and the colloidal gold solution were developed by Innotek (Tangshan) Biotechnology, and the chemiluminescence detection reagents were developed by Mairui Biotechnology Co., Ltd (Guangdong, China). The vacuum blood collection tubes with coagulants and EDTA-K2 were produced by Zhiyuan Biotechnology, Co., Ltd (Guangdong and Hubei, China), and those with sodium citrate and lithium heparin were from Jinxing Biotechnology Co., Ltd (Guangdong and Hubei, China). Blood samples from healthy subjects were collected into EDTA-K2 coated test tubes, and the samples from COVID-19 patients were collected into the test tubes as stated above. They were centrifuged at 1000×g at 4°C for 15 min and the serum and plasma collected. Finally, the samples were subjected to the GICA, or tested on an ARCHITECT i2000 (Mairui Biotechnology, Shenzhen, China) for the detection of IgM and IgG as specified by the manufacturer.
Detection Principle
Immunocapture methodology was used here to detect the SARS-CoV-2 IgM and IgG antibodies. The nitrocellulose membrane was coated with mouse anti-human IgM monoclonal antibodies, mouse anti-human IgG monoclonal antibodies, and goat anti-mouse IgG antibodies. Colloidal gold-labeled recombinant coronavirus antigen and mouse IgG antibodies were then used as a tracer. When the sample was added to the well in the presence of the IgM or IgG detection reagent, it is able to combine with the corresponding antigen to form a complex and then the coated mouse anti-human antibodies will react with this complex, forming a purple-red band, as detected on the card. The colloidal gold-labeled mouse IgG antibodies will form a purple-red band with the goat anti-mouse IgG antibodies.
Determination of Particle Sizes
Here, our aim was to determine the effect of different additives in the collection test tubes on their ability to chelate the colloidal gold. The size of the particle was determined by a Zetasizer Nano ZS90 analysis system (Malvern Instruments, UK, Zetasizer version 7.12). A size polydispersity Index (PDI), for number particle size distribution (PSD), with the x-axis showing the distribution of estimated particle diameter (nm) and the y-axis showing the relative percentage, was created.
Statistical Analysis
Data were analyzed using origin (version 2018), IBM SPSS Statistical software for Windows, version 22.0 (IBM Corporation Armonk, NY, USA) and GraphPad software (GraphPad Software, Inc., La Jolla, CA, USA). Data are presented as the mean ± standard deviation or median (interquartile range), unless otherwise indicated. The results from the concentration of SARS-CoV-2 antibodies detected by chemiluminescence assay (CA) were assessed by one-way analysis of variance followed by Student-Newman-Keuls test. A two-tailed probability (P) value of < 0.05 was considered statistically significant.
Results
EDTA-K2 Increased the Rate of Detection of SARS-CoV-2 IgM Antibodies in Anticoagulated Plasma
We collected serum and plasma with EDTA-K2 from 112 patients with COVID-19 to detect SARS-CoV-2 IgM and IgG antibodies using GICA. The vacuum collection tubes with coagulants and a separator were used to collect serum. Our results revealed that EDTA-K2 anticoagulated plasma had a higher positive detection rate for detecting SARS-CoV-2 IgM antibodies when compared to the serum group. Specifically, seventeen patients were found to be weakly positive, with an additional fifteen patients testing positive but were negative based on serum testing. Five patients were weakly positive, thirteen patients were positive and three patients were strongly positive, but very weak positive using serum testing. Twenty-one patients were detected as positive and two patient was strongly positive but weakly positive based on serum testing. Similarly, for IgG antibody testing, forty samples were interpreted as strongly positive but were detected as only positive when serum tested (Table 1).
 |
Table 1 The Number of SARS-CoV-2 IgG and IgM Antibodies Detected in 112 Patients with Serum and Anticoagulated Plasma with EDTA-K2 |
In addition, we analyzed the consistency of the results in detecting SARS-CoV-2 IgM and IgG antibodies when comparing serum and EDTA-K2 anticoagulated plasma. We classified very weakly positive, weakly positive and strongly positive, as positive, and kappa values were used to evaluate the consistency of the positive and negative groups. We found the total coincidence rate of IgG antibodies were all 100% in these two groups (weighted kappa value = 1.0), while the kappa value for the IgM antibody was 0.409 between the two groups (Table 2). Taken together, these findings demonstrated that EDTA-K2 increased the positive detection rate of SARS-CoV-2 IgM antibodies.
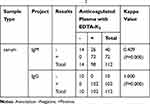 |
Table 2 Analysis of the Consistency of Detecting SARS-CoV-2 IgG and IgM Antibodies in 112 Patients with Serum and Anticoagulated Plasma with EDTA-K2 |
EDTA-K2 Improves the Detection of SARS-CoV-2 IgM Antibodies
Based on our hypothesis above, in order to investigate the impact of different types of vacuum collection tubes on the detection of SARS-CoV-2 antibodies, we collected blood specimens from nine COVID-19 patients with four different vacuum blood collection tubes (serum and anticoagulated plasma with lithium heparin, sodium citrate and EDTA-K2). Then, SARS-CoV-2 IgM and IgG antibodies were detected by GICA. Our results showed that EDTA-K2 anticoagulated plasma had a higher positive detection rate for IgM antibodies, with a darker color being present on the IgG card. However, there were no significant differences among the serum and anticoagulated plasma, when lithium heparin or sodium citrate were used (Figure 1).
In addition, we aimed to further verify that this highly positive effect was attributable to EDTA-K2. Therefore, thirty-two of the 112 patients with COVID-19 had their plasma collected into EDTA-K2 tubes and the whole blood from these patients was tested. Our data showed that there was no difference in the ability to detect SARS-CoV-2 IgM or IgG antibodies when both the anticoagulated blood plasma or whole blood with EDTA-K2 was used (Table 3 and Supplemental Figure 1). These results strongly demonstrated that anticoagulated blood plasma in the presence of EDTA-K2 improved the detection rates of SARS-CoV-2 IgM and IgG antibodies, and this was likely to be due to EDTA-K2, rather than the use of blood serum, plasma, or whole blood. Hence, these findings indicated a marked improvement when EDTA-K2 was used in the ability to enhance the detection of SARS-CoV-2 IgM antibodies.
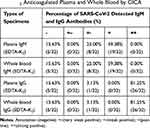 |
Table 3 Percentage of SARS-CoV-2 Antibodies Detected in EDTA-K2 Anticoagulated Plasma and Whole Blood by GICA |
EDTA-K2 Improves the Sensitivity of GICA for the Detection of SARS-CoV-2 IgM and IgG Antibodies
To explore the ability of EDTA-K2 to improve the detection sensitivity of SARS-CoV-2 IgM and IgG antibodies, four different kinds of blood specimens from seven COVID-19 patients with four different vacuum blood collection tubes (serum and anticoagulated plasma with lithium heparin, sodium citrate and EDTA-K2), were then subjected to the GICA and CA, respectively. The latter assay has a higher sensitivity than the former.7 Our results revealed that when CA was used to detect SARS-CoV-2 IgM and IgG antibodies, the presence of different anticoagulants had no effect on the measurements for serum or plasma (Figure 2A and B). However, when the same samples were detected by GICA, the results became significantly variable (Figure 1). Compared with other anticoagulated plasma and serum specimens, the results using EDTA-K2 anticoagulated plasma detected by GICA were more consistent with those results using CA. For example, in patients from 36 to 39, the concentration of IgM antibodies was found to be 2.71±0.03, 2.84±0.09, 1.15±0.08, and 5.24±0.11 Au/mL respectively when using CA (the reference interval was 0.0–1.0 Au/mL). These patients would be expected to test positive or strongly positive using the GICA. However, only the results from plasma treated with EDTA-K2 were consistent with those from the CA. The other anticoagulated plasma samples were judged as negative or weakly positive by GICA (Figure 1 and Table 4).
 |
Table 4 The Detection Results of SARS-CoV-2 IgM and IgG Antibodies in seven Patients by Colloidal Gold Assay and Chemiluminescence Assay |
EDTA-K2 Did Not Increase the False Positive of SARS-CoV-2 Specific Antibody Detection Using GICA
We found that the anticoagulant EDTA-K2, increased detection sensitivity of the GICA. Next, we determined whether it induced the false positive for the detection of SARS-CoV-2 specific antibodies. We set up a blank group containing simply diluent and EDTA-K2. Surprisingly, our results showed that there was no visible purple-red color on the test line. Additionally, we collected anticoagulated plasma with EDTA-K2 from 200 healthy control patients with no COVID-19 infection, and all 200 healthy individuals were negative for both SARS-CoV-2 IgM and IgG antibodies, providing evidence that EDTA-K2 did not increase the false positive rate.
EDTA-K2 Amplifies Positive Signals by Chelating Colloidal Gold to Form High Molecular Weight Polymers
EDTA-K2 is commonly used in blood testing, as it can chelate calcium and reduce the anticoagulation reaction, However, it is unknown whether EDTA-K2 can also chelate colloidal gold and participate in antigen-antibody reactions. Therefore, we set up four groups: the control group was treated with the colloidal gold solution and coagulants, the test group was treated with EDTA-K2, sodium citrate or lithium heparin and colloidal gold solution. Next, Malvern apparatus was used to evaluate the particle size in the four groups. As expected, our results showed that there were two peaks in the EDTA-K2 group, but only one peak in other three groups.
Specifically, the particle sizes were 47.58 ± 1.67 nm (PDI = 0.247 ± 0.02), 49.62 ± 2.20 nm (PDI = 0.250 ± 0.04) and 48.27 ± 2.65 nm (PDI = 0.209 ± 0.07) in the Au, Au+sodium citrate and Au+lithium heparin groups respectively. However, for the Au+EDTA-K2 group, the particle size of first peak was 164 ± 30 nm, which was larger than in the other three groups by approximately four times, and the second peak was 5399 ± 143 nm (PDI = 0.275 ± 0.03) (Figure 3A). Next, they were observed under a confocal laser microscope. We found that larger particle sizes in the test group with EDTA-K2 (Figure 3E), but no significant differences were seen between the Au group and the test groups containing lithium heparin, or sodium citrate (Figure 3B–D). These results provided solid evidence in support of our hypothesis that EDTA-K2 could amplify the positive signal by chelating colloidal gold.
Discussion
The incubation period of various SARS-CoV-2 infections in humans, is from one to fourteen days, and its transmission channels are diverse. In addition to the main routes of transmission through respiratory droplets and contact, it can also be transmitted through aerosols, the digestive tract, and vertical transmission.8 It has a high infection rate due to its multiple channels of transmission and strong infectivity. Therefore, early recognition of suspected patients, and identification of those with severe illness and the early isolation of those with clinical symptoms, are essential for disease control and prevention. At present, the nucleic acid SARS-CoV-2 RT-PCR tests have become the gold standard assay for the diagnosis of COVID-19 with a high sensitivity and specificity.9 However, this method is a relatively time-consuming and complicated and therefore, is not conducive to the screening of large numbers of samples.
GICA based on colloidal gold particles is widely used to screen for antibodies in the blood against SARS-CoV-2. It has the advantages of convenient, fast detection, low cost and suitable for rapid screening of large numbers of samples, the results are intuitive and there is no requirement for specialized equipment, etc.10–12 Early in 2011, a study confirmed that GICA could be combine with the enrichment technique of immunomagnetic nanoparticles, to detect Escherichia coli O157: H7 with speed and sensitivity,13 this study provide powerful evidence for the conclusion that EDTA-K2 could chelate colloidal gold particles and increase the sensitivity of detection on SARS-CoV-2 IgM and IgG antibodies. However, some limitations exist for these assays, such as individual differences in the interpretation of results, which may lead to inconsistent results. In addition, these assays could be affected by several factors, including pH, where it was shown to influence the diagnosis of syphilis using GICA.14 Other studies reported a matrix effect when looking at five kinds of meat, using a colloidal GICA to detect sulfamethazine.15
EDTA-K2 has anticoagulant effects because it can chelate large amounts of calcium ions in the blood.16 Therefore, EDTA-K2 is commonly applied to blood samples as an anticoagulant in the clinical setting, but it can lead to the aggregation of platelets in the blood of individual patients, leading to pseudothrombocytopenia.17 EDTA-K2 has been found to influence the test results of Ca, Mg and Fe compared with the collection tubes excluding anticoagulant.18 Others have reported that EDTA-K2 can impact on the extent of detection of coagulation by PLT.19 However, researchers have found that EDTA-K2 no significant effects on the detection of divalent cation chelators containing anticoagulant detected ITP platelet specific antibodies (GP IIb/IIIa and GPI bα) using multi-antigen printing immunoassays.20 Whether EDTA-K2 could affect the detection of antibodies based on GICA remains unknown. In this study, we confirmed that EDTA-K2 could improve the detection sensitivity of anti-SARS-CoV-2 specific antibodies. Previous study found that colloidal selenium melamine test strips could rapidly detect colloidal selenium melamine test strips in contaminated milk products or animal feed, with high sensitivity.21 This finding provided powerful evidence for our results that the method of EDTA-K2 based on colloidal gold could be used in the detection of SARS-CoV-2. Therefore, we concluded that there is a significant effect of EDTA-K2 on colloidal gold, but these mechanisms remain largely unknown, and more studies are needed to clarify this phenomenon.
IgM antibodies are rapidly produced in patients in response to acute infections with pathogens, therefore, it is important to accurately detect IgM antibodies at the time of initial diagnosis. In addition, IgM antibody tilters are important for monitoring recovering and relapsed patients. We found that EDTA-K2 could increase the positive detection rate of SARS-CoV-2 IgM antibodies when using GICAs. In this study, we designed a series of experiments to determine how EDTA-K2 affected the detection of anti-SARS-CoV-2 antibodies. We used confocal laser microscope and a Zetasizer Nano ZS90 analysis system to evaluate the particle sizes of our colloidal gold. The results revealed that EDTA-K2 could amplify the positive signal by chelating the colloidal gold. This suggested that, even if there was only a small amount of antibody in EDTA-K2 anticoagulated plasma, the color of the T line was darker. A deeper understanding of the reaction principle suggested that EDTA-K2 could amplify a weak signal by chelating colloidal gold and this can explain why the SARS-CoV-2 IgM antibodies could not be detected in other anticoagulated blood specimens such as sodium citrate and lithium heparin but could work in EDTA-K2 anticoagulated plasma.
Although its sensitivity is not as high as CA, GICA can easily be used to screen large numbers of samples and is suitable for use in primary hospitals. Currently, with the new coronaviruses spreading worldwide,22,23 it is urgent to improve the sensitivity and speed of detection of SARS-CoV-2 IgM and IgG antibodies. These are major priorities to help curb the spread of the virus, so it is important to ensure that any diagnosis is made promptly and accurately.
Overall, the results of this study provided solid evidence for the use of EDTA-K2 to improve the sensitivity of detection of SARS-CoV-2 IgM and IgG antibodies. However, it should be noted that when results between serum and anticoagulated plasma were inconsistent, we recommend that multiple methods should be used to confirm results, such as nucleic acid detection, CA, ELISA, and imaging examinations, combined with the patient’s clinical symptoms. These findings provided new insights for improving the detection of SARS-CoV-2 by GICA. However, several problems need to be addressed. First, the samples we used were from patients during the middle and late stages of treatment and rehabilitation. There 112 patients with COVID-19 and 200 healthy people involved in the project, but only seven patients with COVID-19 were enrolled to detect four different tubes. Since there are few patients infection SARS-CoV-2 in China, it is difficult to collect samples from the early stage of infection for verification, which is the limitation of this study. Second, other effects influencing the detection of COVID-19 by GICA remains unclear. Third, only one commercial kit was used for comparison in this study, and whether the pore size of the chromatography membrane had an effect on the test results remains largely unknown.
Conclusion
GICA and equipment-dependent CA were usually used to detect antibodies in clinic, GICA is widely used to detect antibodies for the advantages of convenient, fast, low cost, suitable for screening large sample and require minimal equipment. In this study, we found that EDTA-K2 anticoagulated plasma has higher positive rate than other anticoagulated plasma (sodium citrate and lithium heparin) or serum, especially, the results of EDTA-K2 anticoagulated plasma detected by GICA was high consistent with CA results. Further study shown that EDTA-K2 could amplify positive signal by chelating colloidal gold. Therefore, we suggested that EDTA-K2 anticoagulated plasma maybe more suitable for the detection of SARS-CoV-2 antibodies. However, more positive samples with SARS-CoV-2 were needed to further verify this results.
Ethical Approval
This study was approved by the Hankou Hospital Ethics Committee (No. HKYY-2020-028), and according to the Ethics Committee review, patients informed consent was not required. It is noteworthy that these specimens taken were part of routine hospital procedure, rather than being taken specifically for this study. There was no additional manipulation or injury performed on the participants. Everything was done to ensure that the patient’s personal information was maintained under strict privacy. This study complied with the Declaration of Helsinki.
Funding
This work was supported by funds from the National Natural Science Foundation of China (81601819), the Outstanding Youths Development Scheme of Nanfang Hospital, Southern Medical University (2016J013), the Medical Science and Technology Research Foundation of Guangdong Province (A2016280) and Funds for prevention and control of major infectious diseases from China government in 2020.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Diagnosis and treatment plan for new coronavirus pneumonia (trial version seventh); 2020. Available from: https://www.cma.org.cn/art/2020/2023/2024/art_2928_33242.html.
2. Sman SEG, Ahmed EMEMM, Hussain NE. Sensitivity and specificity of gold chromatography immuno-assays IgM/IgG antibodies test for COVID-19: review of the current literature. Sudan J Med Sci. 2020. doi:10.18502/sjms.v15i5.6969.
3. Linda H, Jen K, Ayla G, et al. The antibodies response to SARS-CoV-2 infection. Open Forum Infect Dis. 2020. doi:10.1093/ofid/ofaa387.
4. Liu C, Mao B, Martinez V, et al. A facile assay for rapid detection of COVID-19 antibodies. RSC Adv. 2020;10(47):28041–28048. doi:10.1039/D0RA04107F
5. Liu D-X, He J-N. [Effects of additives in blood collection tubes on testing the alcohol concentration in blood samples].. Fa Yi Xue Za Zhi. 2014;30(6):452–455.
6. Kotikalapudi R, Patel RK. Comparative study of the influence of EDTA and sodium heparin on long term storage of cattle DNA.. Cell J. 2015;17(1):181–186. doi:10.22074/cellj.2015.526
7. Kaczur V, Vereb G, Molnar I, et al. Effect of anti-thyroid peroxidase (TPO) antibodies on TPO activity measured by chemiluminescence assay. Clin Chem. 1997;43(8):1392–1396. doi:10.1093/clinchem/43.8.1392
8. Chen HJ, Guo JJ, Wang C, et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: a retrospective review of medical records. Lancet. 2020;395(10226):809–815. doi:10.1016/S0140-6736(20)30360-3
9. Corman VM, Landt O, Kaiser M, et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 2020;25(3):2000045. doi:10.2807/1560-7917.ES.2020.25.3.2000045
10. Ray M, Achary KG, Nayak S, et al. Development of a colloidal gold strip-based immunochromatographic assay for rapid detection of Fusarium oxysporum in ginger. J Sci Food Agric. 2019;99(14):6155–6166. doi:10.1002/jsfa.9859
11. Li R, Zhang X, Yang C, et al. Development of a Colloidal Gold-Based Immunochromatographic Assay for the Rapid Detection of Edwardsiella Ictaluri. J Nanosci Nanotechnol. 2018;18(6):3800–3805. doi:10.1166/jnn.2018.15257
12. Liu C, Mao B, Martinez V, et al. A facile assay for rapid detection of COVID-19 antibodies. RSC Adv. 2020;170:112673.
13. Qi H, Zhong Z, Zhou H-X, et al. A rapid and highly sensitive protocol for the detection of Escherichia coli O157:H7 based on immunochromatography assay combined with the enrichment technique of immunomagnetic nanoparticles.. International Journal of Nanomedicine. 2011;6:3033–3039. doi:10.2147/IJN.S25684
14. Wang ZF, Li GJ. Value of colloidal gold assay on syphilis diagnosis and influence factors of its weakly positive. Chin J Dermatovenereology. 2012.
15. Liu NM, Xing KY, Wang C, et al. Matrix effect of five kinds of meat on colloidal gold immunochromatographic assay for sulfamethazine detection. Analytical Methods. 2018;390(6):1619–1627.
16. Pullar JM, Bayer S, Carr AC. Appropriate handling, processing and analysis of blood samples is essential to avoid oxidation of vitamin C to dehydroascorbic acid. Antioxidants. 2018;7(2):68–81.
17. Lin JH, Luo YL, Yao SY, et al. Discovery and correction of spurious low platelet counts due to EDTA-dependent pseudothrombocytopenia. J Clin Lab Anal. 2015;29(5):419–426. doi:10.1002/jcla.21818
18. Chen H, Jiang MJ, Deng TY, et al. Effects of vacuum blood collection tubes containing different anticoagulants on test results of five elements in whole blood. Chin J Health Lab Tech. 2013;1:124.
19. Liu QP. Correction of false PLT results affected by anticoagulants EDTA-K2. J China Med Univ. 2013;42(7):636–637.
20. Zhu LY. The influence of the anticoagulant in detection of the ITP anti-platelet GPIIb/шa and GPIbα antibodies on MAPIA. Anhui Med J. 2011;1:24.
21. Wang ZZ, Zhi DJ, Zhao Y, et al. Lateral flow test strip based on colloidal selenium immunoassay for rapid detection of melamine in milk, milk powder, and animal feed. Int J Nanomed. 2014;9:1699–1707. doi:10.2147/IJN.S58942
22. Verity R, Okell LC, Dorigatti I, et al. Estimates of the severity of coronavirus disease 2019: a model-based analysis. Lancet. 2020;20(6):669–677. doi:10.1016/S1473-3099(20)30243-7
23. Hellewell J, Abbott S, Gimma A, et al. Feasibility of controlling COVID-19 outbreaks by isolation of cases and contacts. Lancet. 2020;8(4):e488–e96.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



