Back to Journals » ClinicoEconomics and Outcomes Research » Volume 12
Economic Burden of Malaria and Associated Factors Among Rural Households in Chewaka District, Western Ethiopia
Authors Tefera DR , Sinkie SO , Daka DW
Received 24 December 2019
Accepted for publication 16 February 2020
Published 12 March 2020 Volume 2020:12 Pages 141—152
DOI https://doi.org/10.2147/CEOR.S241590
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Samer Hamidi
Dufera Rikitu Tefera,1 Shimeles Ololo Sinkie,2 Dawit Wolde Daka2
1Public Health Department, Institute of Health Sciences, Wollega University, Nekemte, Ethiopia; 2Department of Health Economics, Management and Policy, Faculty of Public Health, Jimma University, Jimma, Ethiopia
Correspondence: Dawit Wolde Daka
Faculty of Public Health; Department of Health Economics, Management and Policy, Jimma University, P.O. Box 378, Jimma Ethiopia
Tel +251-966763913
Email [email protected]
Background: There has been a remarkable decline in the burden of malaria in the past few decades in Ethiopia. However, malaria remains a major impediment to both health and economic development in Ethiopia, with 60% of the population at risk of contracting malaria. Hence, this study aimed to estimate the economic burden of malaria among rural households in Chewaka district, Buno Bedele zone, Oromia regional state, Western Ethiopia.
Methods: Community-based cross-sectional study design was employed to estimate the economic burden of malaria at the household level from August 13 to September 2, 2018. A retrospective costing approach was employed, and cost was estimated from the perspective of households. The study included malaria expenditure of households during a one-year period (July 9, 2017 to July 9, 2018). Data were collected from 765 randomly selected households and analyzed using SPSS version 20. Multivariate logistic regression analysis was performed to identify predictors of the economic burden of malaria among rural households and all variables with P-value < 0.05 were considered as statistically significant at 95% CI.
Results: On average, each household comprised 2 malaria cases (SD 1.1) in the past one-year period and the prevalence of malaria in the study setting was 32% (95% CI, 30.5– 33.2). The average annual income of households was US$626.7 (95% CI, 590.4– 663.0). The mean annual cost of malaria illness to households was US$16 (95% CI, 14.8– 17.2), and most of this cost (78%) was contributed by the indirect costs. In every household, on average, patients and companions or caregivers lost 3.4 productive workdays due to malaria illness, respectively. Fourteen households out of 100 spent more than 5% of their annual income on malaria treatment and hence, they were prone to high economic burden or catastrophic costs. Household level economic burden of malaria was determined by the sex and educational status of household head, means of transportation to treatment center, the episodes of malaria, the number of malaria “ill days“ and type of malaria diagnosis.
Conclusion: Malaria continues to significantly impose an economic burden on the rural households of Ethiopia. Hence, the national malaria program needs to recognize and address the catastrophic costs associated with malaria illness. Efforts should be made to ensure universal access to and utilization of malaria prevention, diagnosis, and treatment services.
Keywords: economic burden, cost of illness, household, malaria, Ethiopia
Plain Language Summary
Malaria remains a main public health problem worldwide. Malaria has health, social, and economic impact on a nation. The prevalence of malaria is high in Ethiopia, where nearly sixty percent of the population is at risk of malaria infection. Hence, this study aimed to estimate the economic burden of malaria and to identify the predictors of economic burden of malaria in rural households of Chewaka district, Oromia region, Western Ethiopia. The study findings revealed that the prevalence of malaria was high and an unacceptable number of households were at risk of catastrophic health expenditure. These households spent more than five percent of their annual income for malaria illness. The majority (over three quarters) of the total cost of malaria illness was comprised of the indirect costs, the productive loss borne by patients and their companion. Malaria continued to significantly impose economic burden to the rural households. The study finding may help policy makers and program designers to consider interventions that minimize the catastrophic costs associated with malaria illness.
Introduction
Despite a decline in the morbidity and mortality of malaria worldwide, the current level is unacceptably high and the burden of malaria varies across different parts of the world. In the period between 2010 and 2018, the number of malaria cases declined by about 9.2% from the level of 251 million cases in 2010 to 228 million cases in 2018. The incidence of malaria declined from 71 cases/1000 population at risk to 57 cases/1000 population at risk. The burden of malaria was higher in the African region, where around 93% or 213 million cases of malaria were observed in 2018 alone. Worldwide, around 85% of the global burden of malaria was located in 19 sub-Saharan African (SSA) countries and India alone. While six SSA countries (Nigeria, Democratic Republic of Congo, Uganda, Côte d’Ivoire, Mozambique and Niger) alone shared more than half of all malaria cases worldwide.1,2
Malaria imposes substantial costs to individuals, society, and the government. It affects the health and wealth of nations alike hence; it was understood as both a disease of poverty and cause of poverty. The direct costs of malaria include both personal and public expenditures related to prevention and treatment of the disease. Whereas, the indirect costs include lost productivity or income due to illness or death.3–5 The high prevalence of malaria combined with the economic burden it imposes on society are the major challenges of health system in SSA region. Particularly the poorer and vulnerable households are at increased risk of the burdens of malaria and the cost of malaria is far worse for complicated cases.6–10 The environmental and socio-demographic factors predominantly affect the burden of malaria at household level.11
Ethiopia has made notable progress in decreasing the prevalence and burden of malaria through public health measures taken during the millennium development goals (MDGs) era.12 However, malaria remains one of the public health problems with a high level of mortality and disability adjusted life years (DALYs).13 The economic burden of malaria is high in Ethiopia, leading to catastrophic costs for society.14 The lost productive personnel due to malaria illness, school absenteeism, direct and indirect costs are the major economic burdens of malaria in Ethiopia.15 The seasonal transmission of malaria in Ethiopia has complicated the burden of malaria because the peak malaria transmission season and the major planting and harvesting period coincide. In Ethiopia high malaria transmission occurs bi-annually from September to December and April to May.16
Nearly sixty percent of the population was at risk to malaria in Ethiopia and 54 or 6.4% of the Woredas have high malaria transmission rates with annual parasite incidence (API) of ≥100 cases per 1000 population per year.17 With this rate, the economic burden of the society also increases.
Therefore, in this study we aimed to estimate the economic burden of malaria to households in the high malaria transmission district of Ethiopia. Both direct and indirect costs of malaria illness were estimated. Additionally, we also identified the potential predictors of economic burden of malaria to households.
Methods and Materials
Study Setting
The study was conducted in Chewaka districts, Buno Bedele zone, Oromia regional state, Western Ethiopia. Chewaka was one of the districts located in Buno Bedele zone. The district was in the geographic coordinates of approximately 7°40ʹ N latitude and 36°50ʹ E longitude. It was found 72 km in the Eastern direction of the big zonal town, Bedele and 403 km Western direction of the capital city of Ethiopia, Addis Ababa. The district has a land mass area of 52.2 km2 and an altitude of 900–1400 m above sea level. The annual temperature ranges from 37–40°C and it has average annual rainfall of 1000–1200 mm3.18
The total population of the district in 2018 as projected from 2007 was 75,111 with male to female ratio of 1.2:1. The estimated number of households in the district were 15,649. Administratively the district had a total of 28 kebeles, out of which two were urban kebeles. All of the kebeles were malarious and hence, all of the district population were at risk of malaria infection.18
Kebeles of the district were further divided into Gots (“Zooni”), “Gare” and one-to-five networks. “Zooni” is an Afan Oromo word implying the sub-section of kebeles. In the district each kebele has three zones. Likewise, “Gare” is an Afan Oromo word implying the sub-section of each zone and each “Gare” comprised 25–30 households. While one-to-five networks referred to the network of 5 households in one group. Accordingly, there were a total of 84 “Zoonis” or Gots, 494 “Gare” or groups and 2248 one-to-five networks.18
In Chewaka district, there were 3 public health centers, 28 health posts, 9 private clinics, and 2 drug stores providing promotive, preventive, and curative health services to the population. Malaria diagnosis and management services were provided at health centers, health posts, and private clinics. At health posts level, the health extension workers diagnose malaria using rapid diagnostic tests (RDT) and provide coartem for free for those who tested positive. At health center and private clinics level the diagnosis of malaria was based on microscopic examinations.18
The district had not yet implemented community-based health insurance (CBHI) services. The livelihood of households were dependent on agriculture and the main agricultural products were maize, sorghum, fruits, vegetables, “Khat”, coffee, and animal husbandry.18
Study Design and Participants
Study Design
Community based cross-sectional study design was conducted in selected kebeles of the district from 13 August to 2 September, 2018. A retrospective costing approach was used to estimate household level economic burden of malaria.
Sample Size
All households who had at least one confirmed malaria case in the period between July 9, 2017 to July 9, 2018 and those who had lived in the study district for at least more than one-year period were eligible for this study. Single population proportion formula was used to determine the study sample size. By assuming 50% of households would have had at least one confirmed malaria case in the past one-year period with 5% of margin of error at 95% confidence interval and a design effect of 2, the calculated sample size yielded 768 households. The calculated sample size was proportionally allocated to each kebele based on the number of households in each kebele.
Sampling Procedure
Stratified multistage sampling strategy was employed to select study participants. In the first stage, 30% of the kebeles (8 kebeles) were selected using simple random sampling strategy through computer-generated random numbers.
Following this, the health records of malaria patients were reviewed from health posts located under the catchments of the selected kebeles. All households that fulfilled the inclusion criteria were retrieved from the family folders of the community health information system (CHIS) and the household numbers were listed down. A family folder is a file folder that captures information about the environment, socio-demographic profile of the family and a file for each individual member of a family. Each family folder comprised integrated cards and health cards.19 The integrated card was used to record information related to the antenatal care, delivery, postnatal care, immunizations and growth monitoring and promotion services provided to mother and their children by the health extension workers. While, the health cards were used to record information about the diagnosis and management of illnesses, family planning, tuberculosis and HIV/AIDS care and support services, respectively. Family folders are kept at health posts and maintained by health extension workers.19
The list of household numbers in each kebele indicated the number of households that would have had at least one confirmed case of malaria in the past one year. The sample size was proportionally allocated to each kebele based on the number of households in every kebele.
By following these procedures, the number of households and sample size allocated for each kebele were presented as follows: K1 (N=1166, n=92), K2 (N=1027, n=81), K3 (N=1410, n=112), K4 (N=1216, n=96), K5 (N=1258, n=100), K6 (N=1016, n=80), K7 (N=1404, n=112) and K8 (N=1398, n=104). K, N and n referred to Kebeles, the source population and the sample size, respectively. Every selected household was approached and interviewed after obtaining consent.
Data Collection and Procedures
A semi-structured, interviewer administered questionnaire was employed in the study. The contents of the data collection questionnaire were developed based on the household costing tool that was primarily developed and used by Hansen and Yeung20 and other related literature.14 The questionnaire was comprised of two main sections. The first section presented the information about the characteristics of households and the second section was about the costs related to malaria diagnosis and treatments. Data collection questionnaires were translated into local language (Afan Oromo) by an experienced translator and back translated to English by an independent translator for consistency.
Data were collected by three experienced health professionals who had a qualification of diploma and were proficient in local language (Afan Oromo). The overall data collection processes were monitored and supervised by another health professional who had a qualification of bachelor's degree and was proficient in local language (Afan Oromo). Both data collectors and supervisors were trained for two days on the data collection questionnaires, the study procedures, and research ethics.
The data collection processes were implemented in such a way that data collectors approached every household by using household numbers and additionally they were supported by guides whenever needed. These guides were local people who knew the location of each household. Data collection was conducted at the household after obtaining consent. When there was no one at the household, data collectors revisited their homes three times. If the household was not available at the final visit, the case was recorded as absent. The supervisors closely monitored the field level data collection process on a daily basis and when errors were noticed, they approached data collectors and corrected errors at field level.
Techniques and Approaches of Cost Estimation
The outcome variable was economic burden of malaria. The independent variables were the socio-demographic and socio-economic characteristics of households, disease severity and duration of illness, malaria episodes, distance to health facility and availability of anti-malarial drugs.
In this study the economic burden of malaria referred to the level of expenditure that threatens households’ livelihoods due to malaria. All households that spent more than 5% of their annual income to pay for the diagnosis and treatment of malaria were classified as catastrophic payment or high economic burden and those households who paid less than this were classified as low economic burden.10
The cost of illness approach was used to estimate the economic burden of malaria in the study area. The costs per episode of malaria to the patient and to the household were estimated by using the prevalence-based retrospective costing approach. In this study total costs, direct costs, and indirect costs associated with malaria illnesses were estimated.
Operational Definition
Total cost: referred to the sum of the direct and indirect costs of malaria illness.
Direct cost: was a cost or expenditure borne by patients and their families for the diagnosis and treatment of malaria. Direct cost included both medical costs and non-medical costs.
Medical cost: was amount spent to purchase medical goods or services such as registration, drugs, diagnosis or testing and consultation.
Non-medical costs: referred to amount spent to purchase non-medical goods or services such as food, transportation, other non-medical supplies and services while seeking malaria treatment.
Indirect costs: referred to the productive loss borne by patients and their families. It included income losses due to lost workdays resulting from seeking treatment.
Malaria case: referred to a person with complaints of one or more symptoms such as fever, sweating, shivering, headache and vomiting in addition to those microscopically confirmed at health facility labelled as malaria cases.
Severity of disease: referred to the degree of severity of malaria for patients. The degree of severity can be mild, moderate or severe. Mild - when there was loss of appetite and headache. Moderate - when there was sitting down and tiredness. Severe - when there was fitting and convulsions.
The ingredient-based bottom-up approach was used to identify and measure all costs at patient and household level. All cost information was collected in Ethiopian birr (ETB) and then converted to US$ (US$1=27.5 ETB as of August 2018).
The direct cost was estimated in such a way that, primarily the direct medical and the direct non-medical costs were estimated separately. Following this, the direct cost which equals the sum of direct medical and direct non-medical cost was estimated.
The indirect cost was measured in terms of number of forgone working days of the patients and caregivers (companion) due to malaria illnesses. The indirect cost was estimated using the human capital approach.21 The daily wage rates of patients and their companion were used to convert the lost workdays to a monetary value. The daily wage rate of patients and companion who were paid on monthly basis was computed by dividing the monthly net salary by 30. The daily wage rate of patients who had no regular payments such as agricultural workers or farmers was estimated based on the 2013 National Labor Force Survey (NLFS) report of Ethiopia. The average monthly wage rate for agricultural workers in Ethiopia was ETB 697, which was divided by 20 in order to obtain the daily wage rate of ETB 35.22
Data Processing and Analysis
Collected data were checked for completeness and consistency. Data were entered into Epidata 3.1 and exported to SPSS version 20. Data cleaning was also conducted after data entry by running frequencies. Duplicated records were removed from SPSS. Coding and recoding of variables was performed in the preparation for analysis.
The data analysis was conducted in a step wise procedure in which first the characteristics of study participants were analyzed and described. Following this, the direct, indirect and total costs of malaria illness were analyzed. To simplify the cost analysis, a spreadsheet was employed, and the information was exported to SPSS. All cost information was converted to US$. The sum, mean, median and standard deviation of each cost category were calculated. Moreover, 95% confidence interval was also constructed for the mean.
The outcome variable, the economic burden of malaria, was a categorical variable created from the total cost of malaria illness and annual income of each household. Households who spent more than 5% of their annual income on malaria illness were categorized as high economic burden and those who spent less than 5% were categorized as low economic burden.
Multivariate logistic regression was performed to identify the potential predictors of the economic burden of malaria. All variables with P-value<0.05 in the bivariate analysis were entered into the multivariate model and in the multivariate model those variables with P<0.05 were considered as statistically significant and interpreted.
Ethical Consideration
Appropriate research ethical clearance was obtained from the ethical review committee of Jimma University institute of health (reference number: IHRPGD/435/2018, July 2018) and Oromia regional health bureau. Study permission or support letter was obtained from Oromia regional health bureau and Buno Bedele zone health department. This study was conducted in accordance with the Declaration of Helsinki. All study participants were well informed about the aim of the study, benefits and risks. Following this, informed written consent was secured from study participants. Study participants’ confidentiality was maintained. No personal identifiers were used in the data collection tools and codes were used in place of it. Data were kept in protected and safe locations. Paper based data were kept in a locked cabinet and computer-based data were password secured. The recorded data were not accessed by a third person, except the research team. Data sharing will be enacted based on the consent and permission of research participants and the ethical and legal rules of data sharing.
Results
Out of the total study households (n=768), 765 participated in the study with a response rate of 99.6%. In the study households, there were a total of 1477 malaria patients.
Characteristics of Study Participants
The mean age of households was 34 years (SD 10.7), with household age ranging from 18 years to 85 years. Majority of the household heads were male (98%), married (96%), Muslim religion followers (97%) and farmers (96%). While, over half of the household heads had no formal education (59%). The average family size of households was 6 (SD 2.7) and the mean monthly income was US$61.8 (95% CI, 58.2–65.4). A total of 68 (9%) households were in a village that experienced malaria epidemic in the previous year (Table 1).
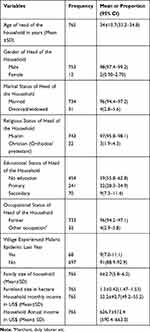 |
Table 1 Characteristics of Households in Chewaka District, Oromia Regional State, Western Ethiopia. August 2018 |
Overall, 1477 cases of malaria were identified in the study households. The total population of the study households were 4642 and hence, the estimated prevalence of malaria in the study area was 32% (95% CI, 30.5–33.2). On average there were 2 (SD 1.1) malaria cases in each household. Over half of patients were male (58%; 95% CI, 55.4–60.5) and in the age range of 15–64 years (56%; 95% CI, 53.7–58.8), respectively. A bit over four in ten of the patients had moderate fever (44%; 95% CI, 41.8–46.8) and initiated treatment on the days following onset of fever (42%; 95% CI, 39.1–44.2). Over two-thirds (69%; 95% CI, 66.3–70.9) of the patients had one episode of malaria in the year considered (Table 2).
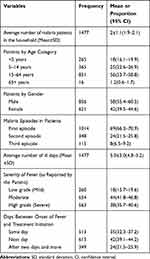 |
Table 2 Characteristics of Sick Member of the Household in Chewaka District, Oromia Regional State, Western Ethiopia. August 2018 |
Patients located in 195 (26%) of the households or a total of 437 patients had taken self-treatment measures before visiting formal treatment centers (health facility). Out of these, 103 (53%) used anti-malarial drugs or medications (37% chloroquine, 54% coartem, and 9% other medications). The sources of medication for these patients were reserved drug from previous treatments (46%), purchased drug from private drug store (48%), and other sources (6%). The total cost of self-treatment was US$915.5 and on average each household spent US$4.6±6.6 (median cost= US$2.4).
Generally, patients located in 753 (98%) of the households or a total of 1442 patients received treatment from formal sources that included hospital, health center, health post or private clinics. The means of transportation for patients included motor bike or car (1284 or 87%) and on foot (193 or 13%).
For majority of patients the location of the first (1440 (98%) out of 1477) and second (564 (71%) out of 800) formal malaria treatments were at public health facilities (hospital, health center and health posts), respectively. Whereas, most patients who had third treatment (162 (67%) out of 241) visited private health facilities. Of the public health facilities, most patients received malaria treatment from health centers in successive visits; first (1358 or 92%), second (517 or 65%) and third (40 or 17%) visits, respectively.
Regarding the accessibility of drugs at the health facilities, variation was observed over the successive visits. In the first formal treatment, patients located in 297 (39%) of the total households (n=765) received drugs. Moreover, patients located in 270 (35%) households and in 198 (26%) households received prescriptions only and both drugs and prescriptions, respectively. When this was translated to the number of patients (n=1477), a total of 738 (50%) patients received drugs and another 697 (47%) patients received prescriptions only. Patients located only in a bit over half (52% or 395) of households procured drugs from public pharmacies for the first malaria treatment.
In the second formal treatment, patients located in 208 (55%) households out of 380 households or a total of 324 (41%) patients out of 800 patients received anti-malarial drugs. While patients located in one hundred and twenty-seven (33%) households received prescriptions only, patients located in 45 (12%) households received both anti-malarial drugs and prescriptions. The number of patients who had received prescriptions only were 226 (26%). Patients located only in 28% or 106 households procured anti-malarial drugs from public health facilities and patients located in most households (72% or 271) procured drugs from private pharmacies.
In the third formal treatment, patients located in 77 (75%) households (out of 103) or a total of 85 (35%) patients (out of 241) received anti-malarial drugs. Patients located in 26 (25%) households or a total of 22 (11%) patients received prescriptions only. Patients located in majority of the households (86 households or 84%) procured drugs from private pharmacy and only patients located in 16% of the households procured drugs from public pharmacies (ie, pharmacies at hospital, health center or health posts).
Economic Burden of Malaria: Direct, Indirect and Total Costs of Malaria Illness
The total cost of malaria illness to households in the past one year was US$12,242.65. The mean annual cost of malaria illness to households was US$16 (95% CI, 14.8–17.2). Majority of the total cost of malaria illness (78% or US $9512.65) was comprised of indirect costs. The mean direct cost of malaria illness was US$14.5 (95% CI, 13.6–15.5) and most of the direct cost of malaria illness (55%) was comprised of medical costs (Table 3).
 |
Table 3 Direct, Indirect and Total Costs of Malaria Illness to Households at Chewaka District, Oromia Regional State, Western Ethiopia. August 2018 (US$) |
Predictors of Economic Burden of Malaria
Table 4 presented the bivariate logistic regression analysis results and Table 5 presented the multivariate logistic regression analysis results. Overall, 106 (13.9%) (95% CI, 11.5–16.4) of households had a high economic burden of malaria. The economic burden of malaria was high in female, no education status and married household heads, respectively. Further, the economic burden of malaria was higher among households with three and more malaria episodes, severe fever illnesses and patients with >5 days of illness, respectively. Higher proportion of households that were located >10 km away from malaria treatment center had higher economic burden of malaria (Tables 4 and 5).
 |
Table 4 Bivariate Analysis Results of the Economic Burden of Malaria to Households in Chewaka District, Oromia Regional State, Western Ethiopia. August 2018 |
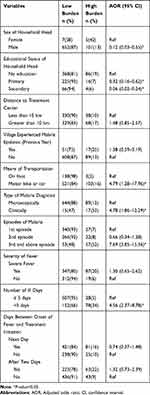 |
Table 5 Multivariate Analysis Results of the Economic Burden of Malaria to Households in Chewaka District, Oromia Regional State, Western Ethiopia. August 2018 |
In the bivariate analysis, socio-demographic characteristics of the household such as sex and educational status of household head were associated with high economic burden of malaria. Additionally, distance to treatment center, means of transportation, the village experience of malaria epidemic in the previous year, episodes of malaria, severity of fever illness, number of ill days, days between onset of fever and treatment initiation and the type of malaria diagnosis were associated with high economic burden of malaria, respectively (Table 4).
After adjusting for other variables, sex and educational status of household heads, means of transportation to treatment center, episodes of malaria, number of ill days and type of malaria diagnosis showed statistically significant association with the economic burden of malaria (Table 5).
High economic burden of malaria was 88% less likely in male household heads than female household heads (AOR=0.12; 95% CI, 0.03–0.55). The high economic burden of malaria was 69% (AOR=0.32; 95% CI, 0.16–0.62) and 94% (AOR=0.06; 95% CI, 0.02–0.24) less likely among household heads with a primary and secondary educational status compared to those with no educational status, respectively.
High economic burden of malaria was five times more likely among patients who used motor bike or car transportation to reach treatment center than those who traveled on foot (AOR=4.79; 95% CI, 1.28–17.96).
The odds of high economic burden of malaria were five times more likely among patients who were diagnosed clinically than patients who were diagnosed microscopically (AOR=4.78; 95% CI, 1.86–12.29).
Moreover, the high economic burden of malaria was eight times more likely among households with patients with three and more episodes (AOR=7.69; 95% CI, 3.85–15.36) than households with only one episode of malaria. The odds of high economic burden of malaria were also more likely in households with more than 5 ill days (AOR= 4.56; 95% CI, 2.37–8.78) compared to less than 5 ill days.
Discussion
In this study we aimed to analyze the economic burden of malaria and the predictors of the cost of malaria illness among households in the high malaria transmission district of Ethiopia for the year 2018. Our study indicated that the prevalence of malaria was high (32%) and a bit over one in ten households had a high economic burden of malaria in the study area. The total annual cost of malaria illness was US$12,242.7 and on average every household spent US$16 due to malaria illness. In this study sex and educational status of household heads, the means of transportation, the episodes of malaria, number of ill days of malaria and type of malaria diagnosis were predictors of the economic burden of malaria.
Fourteen households out of hundreds suffered catastrophic health expenditure in the study setting. These households spent more than 5% of their annual income due to malaria illness. A similar catastrophic health expenditure to the society was also reported by other studies in SSA region.7,9,10,23
In our study households had a higher mean annual cost of malaria illness (mean annual total cost=16) compared to another study report in South central Ethiopia (mean annual total cost=6.15).14 Whereas, the mean annual cost of malaria illness was slightly lower than a study report in Mozambique (mean annual total cost=17.41).23 The reason for the variation in the mean cost of illness might be the different period and method of cost analysis and difference in settings. In the latter report, transportation cost for visiting traditional healers was considered.
Most of the cost of malaria illness was attributed to indirect costs such as patients' lost workdays and costs of companion or caregivers. This was comparable to a study done in other settings elsewhere.6,7,9,10,14,23 The higher indirect costs of malaria illness may be due to the fact that the costs of anti-malarial drugs and diagnostics or testing were subsidized or free at public health sector of Ethiopia.17
Nonetheless, a significant proportion of households' total costs of malaria illness (22.3%) was also attributed to direct costs. Out of the total direct costs of malaria illness, the majority (70%) accounted for cost of drugs (32%), transportation (25%) and food (14%), respectively. This finding was comparable to other reports elsewhere.14,23 Households in the study setting paid out of their pockets (OOP) and were not insured. Hence, they were at risk of catastrophic costs.
In the study setting, most of the patients procured anti-malarial drugs from pharmacies located at public health facilities. However, it was a common that health workers provided prescriptions only to a significant proportion of patients due to unavailability of anti-malarial drugs. This may lead patients to procure drugs from private pharmacies, consequently leading to risk of catastrophic costs to patients.
The economic burden of malaria was associated with sex and educational level of household heads, means of transportation, type of malaria diagnosis, episodes of malaria and the number of ill days. The odds of high economic burden of malaria were less likely among male household heads and household heads at the primary and secondary educational level. Household heads with no education spent more than those with primary and secondary education. Likewise, the odds of high economic burden of malaria were more likely among households with patients who traveled with motor bike or car, diagnosed clinically, had three or more episodes of malaria and who had longer duration of malaria illness. The cost related to traveling via motor bike or car could add to the cost of transportation of both patients and companion. Additionally, more episodes of malaria and longer duration of illness were associated with increased costs to both patients and companion. This finding was supported by another study elsewhere.14
Despite knowledge of high impact interventions, the burden of malaria remains unacceptably high in Ethiopia.12,13,15 The incidence rate of malaria is high and this is partly attributed to a combination of factors that include lack of access to treatment, low utilization of available interventions, climate, physiography and economic activity.15 Ethiopia has made notable progress in increasing the coverage of insecticide treated nets (ITNs), indoor residual spraying (IRS) and malaria diagnosis and treatment services. However, the ownership and utilization of services are still low.16,17 In Ethiopia the high malaria transmission season coincides with the harvesting and planting season, and the livelihood of the majority of the population is dependent on agricultural activities.15,17 This might escalate the economic burden of malaria at the society level. This demands universal access to and utilization of malaria prevention, diagnosis and treatment services, and accelerating effort toward elimination and attainment of malaria-free status.
In this study most of the cost data were estimated based on self-reports of households. This may partly lead to over or under estimation of cost due to recall bias. This study only assessed the cost of medications, diagnosis and indirect costs related to malaria illness. It did not address other economic costs of malaria such as the costs of mortality and other manifestations and complications of malaria. This may also underestimate the economic burden of malaria.
Conclusion
The study findings showed that the total costs of malaria illness are high and a significant proportion of households, mainly the low socio-economic status ones, have a high economic burden due to malaria illness. Therefore, our findings call for serious attention to be paid to preventing catastrophic costs of the society attributed to malaria illness. Public health measures that include both malaria prevention and control strategies should be strengthened. Access to quality malaria services needs to be improved at the primary health care level of Ethiopia in terms of malaria diagnostics and antimalarial drugs.
Acknowledgments
The researchers would like to acknowledge all participants of the study and administrative structure at different hierarchy of the study setting for their due cooperation and involvement.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
All authors declared that they have no conflicts of interest related to this work.
References
1. World Health Organization. The World Malaria Report 2018. Geneva: World Health Organization; 2018. Licence: CC BY-NC-SA 3.0 IGO. 22. Available from: https://apps.who.int/iris/bitstream/handle/10665/275867/9789241565653-eng.pdf.
2. World Health Organization. World malaria report 2019. Licence: CC BY-NC-SA 3.0 IGO. Geneva; 2019. Available from: https://www.who.int/publications-detail/world-malaria-report-2019.
3. Gallup JLSJ. The economic burden of malaria. The intolerable burden of malaria: a new look at the numbers. Am Soc Trop Med Hyg Northbrook. 2001;64(1):43.
4. CDC. Malaria’s impact worldwide. 1–3. Available from: https://www.cdc.gov/malaria/malaria_worldwide/impact.html.
5. Chuma J, Okungu V, Molyneux C Economic Costs of Malaria. 2001–2010 United Nations Decade to Roll Back Malaria. Vol. 13. World Health Organization; 2010:1298–1307. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2890678&tool=pmcentrez&rendertype=abstract.
6. Hennessee I, Chinkhumba J, Hagen MB, et al. Household costs among patients hospitalized with malaria: evidence from a national survey in Malawi, 2012. Malar J. 2017;16:395. doi:10.1186/s12936-017-2038-y
7. Sicuri E, Vieta A, Lindner L, Constenla D, Sauboin C. The economic costs of malaria in children in three sub-Saharan countries: Ghana, Tanzania and Kenya. Malar J. 2013;12:307. doi:10.1186/1475-2875-12-307
8. Goodman C, Coleman P, Mills A, et al. Economic Analysis of Malaria Control in Sub-Saharan Africa. Geneva: Global Forum for Health Research; May2000.
9. Chimbari MJ. Economic burden of malaria on rural households in Gwanda district, Zimbabwe. Afr J Prim Health Care Fam Med. 2017;9(1):a1317.
10. Dalaba MA, Welaga P, Oduro A, Danchaka LL, Matsubara C. Cost of malaria treatment and health seeking behaviour of children under-five years in the upper west region of Ghana. PLoS One. 2018;13(4):e0195533.
11. Ngatu NR, Kanbara S, Renzaho A, et al. Environmental and sociodemographic factors associated with household malaria burden in the Congo. Malar J. 2019;18:53. doi:10.1186/s12936-019-2679-0
12. Deribew A, Dejene T, Kebede B, Tessema GA, Melaku YA. Incidence, prevalence and mortality rates of malaria in Ethiopia from 1990 to 2015: analysis of the global burden of diseases 2015. Malar J. 2017;16:271. doi:10.1186/s12936-017-1919-4
13. Girum T, Shumbej T, Shewangizaw M. Burden of malaria in Ethiopia, 2000–2016: findings from the global health estimates. Trop Dis Travel Med Vaccines. 2019;5:5–11. doi:10.1186/s40794-019-0090-z
14. Hailu A, Lindtjørn B, Deressa W, Gari T, Loha E, Robberstad B. Economic burden of malaria and predictors of cost variability to rural households in south-central Ethiopia. PLoS One. 2017;12(10):e0185315. doi:10.1371/journal.pone.0185315
15. Alelign A, Dejene T. Current status of malaria in Ethiopia: evaluation of the Burden, factors for transmission and prevention methods. Acta Parasitol Glob. 2016;7(1):01–6.
16. Taffese HS, Hemming-schroeder E, Koepfli C, et al. Malaria epidemiology and interventions in Ethiopia from 2001 to 2016. Infect Dis Poverty. 2018;7:103. doi:10.1186/s40249-018-0487-3
17. USAID. President’s Malaria Initiative Ethiopia. Malaria Operational Plan FY 2019; 2019.
18. Chewaka District Health Office. Annual health indicators performance report of the year 2017/2018. Unpublished; 2018.
19. Federal Ministry of Health of Ethiopia. Community health information system data recording and reporting. User’s Manual; 2011. Available from: https://www.measureevaluation.org/resources/publications/ms-13-69.
20. Hansen KS, Yeung S. ACT Consortium Guidance on Collecting Household Costs ; 2009. Available from: https://globalhealthtrials.tghn.org/site_media/media/articles/Collecting-household-costs-ACT-Consortium-guidance.pdf.
21. Jo C. Cost-of-illness studies: concepts, scopes, and methods. Clin Mol Hepatol. 2014;20:327–337. doi:10.3350/cmh.2014.20.4.327
22. Federal Democratic Republic of Ethiopia Centeral Statistical Agency. Key findings on the 2013 national labour force survey; 2014. Available from: http://www.csa.gov.et/survey-report/category/34-nlfs-2013?download=765:nlfs-latest-key-findings-2013.
23. Alonso S, Chaccour CJ, Elobolobo E, et al. The economic burden of malaria on households and the health system in a high transmission district of Mozambique. Malar J. 2019;18(1):1–10. doi:10.1186/s12936-019-2995-4
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
