Back to Journals » Psoriasis: Targets and Therapy » Volume 12
Economic Burden of Generalized Pustular Psoriasis in Sweden: A Population-Based Register Study
Authors Löfvendahl S , Norlin JM, Schmitt-Egenolf M
Received 21 February 2022
Accepted for publication 13 April 2022
Published 5 May 2022 Volume 2022:12 Pages 89—98
DOI https://doi.org/10.2147/PTT.S359011
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Uwe Wollina
Sofia Löfvendahl,1 Jenny M Norlin,1 Marcus Schmitt-Egenolf2
1The Swedish Institute for Health Economics (IHE), Lund, Sweden; 2Department of Public Health and Clinical Medicine, Dermatology, Umeå University, Umeå, Sweden
Correspondence: Marcus Schmitt-Egenolf, Department of Public Health and Clinical Medicine, Dermatology, Umeå university, Umeå, SE-901 85, Sweden, Tel +46 90 785 2875, Email [email protected]
Background: Generalized pustular psoriasis (GPP), which can occur with or without psoriasis vulgaris (PV), is a severe form of pustular psoriasis with potentially life-threatening symptoms. GPP is also associated with several comorbidities, which further adds to the burden of disease. This study investigates the economic burden of disease in patients with GPP.
Methods: All-cause and GPP-specific healthcare resource use (inpatient stays, physician visits and drug use), as well as associated costs, were compared for year 2015 between GPP patients (n = 914) and two matched control groups representing the general population (n = 4047) and patients with PV but no GPP (n = 2556). Information on resource use for 2015 was obtained from the Swedish National Patient Register and Swedish Prescribed Drug Register, respectively.
Results: All-cause inpatient stays, physician visits, and use of psoriasis-related drugs were significantly more common among GPP patients compared to both control groups. This difference was reflected in total direct cost for GPP patients (5062 euros/year) which was 3.1 and 1.8 times higher (p < 0.001) compared to the general population and PV controls, respectively. For GPP patients, the share of total cost was 22% for all-cause physician outpatient visits and 40% for all-cause inpatient stays. However, only 6.3% and 11.3% of these costs, respectively, were due to GPP-specific problems. Psoriasis-related drugs constituted 27% of total costs for GPP patients of which a large fraction (86%) was represented by biologics.
Conclusion: This study demonstrates a higher economic burden for GPP patients compared to both the general population and patients with PV, with inpatient visits and use of biologic drugs as major cost driving factors. Only fractions of the costs for physician visits and inpatient stays were attributable to specific GPP problems, indicating a higher economic burden of GPP-consequences and complications.
Keywords: generalised pustular psoriasis, population-based controls, direct costs, incremental analysis
Introduction
Generalized pustular psoriasis (GPP), which can occur with or without psoriasis vulgaris, is a severe form of pustular psoriasis affecting large areas of the skin.1,2 The occurrence of acute flares is typical. The flares, that can be life-threatening, regularly require inpatient care.2 The patient is often severely ill with general symptoms including fever and severe systemic inflammation. This repeating pattern may continue episodically throughout life.3
Comorbidities may have a significant impact on treatment choices, drug interaction and the patients’ health-related quality of life. Like moderate-to-severe psoriasis, GPP is associated with an increased occurrence of comorbidities compared to the general population.4 Comorbidities that have been reported in GPP include obesity, hypertension, hyperlipidemia, diabetes, uveitis,4–7 cholestasis,8 joint involvement,4–7,9,10 allergic rhinoconjunctivitis and peptic ulcer disease.4,7
Although the prevalence of GPP is relatively low (ranging from 0.18 to 18/100,000),11–15 the condition may still contribute considerably to the economic burden of psoriasis. Due to the chronicity and risk for comorbidities, GPP has implications not only for the individuals but also for society in terms of healthcare costs. In a recent US cost study on GPP, based on claims data, it was found that total medical costs (direct medical use and drugs) in GPP was higher compared to matched cohorts of persons from the general population and persons with psoriasis vulgaris, respectively. Noteworthy was that the rate of inpatient stays was four times higher in persons with GPP compared to matched cohorts.16 These results are corroborated by a study from Japan, that also showed elevated healthcare resource and drug use in GPP patients compared to both population-based and psoriasis vulgaris controls.4 However, to our knowledge, there are no published studies on costs associated with the use of healthcare resources in GPP, nor on the cost burden of GPP compared to psoriasis vulgaris with no GPP manifestations. In this context, it is important to investigate the allocation of costs related to the diagnosis of GPP and potential comorbidities.
We investigated the economic burden of disease in patients with GPP in Sweden. Using longitudinal population-based register data and matched control groups, we estimated the all-cause healthcare resource use (including physician visits, inpatient stays and drugs), and associated costs, in patients with GPP compared to both the general population and to patients with psoriasis vulgaris. We also estimated the healthcare resource use and direct costs attributable to specific GPP problems.
Materials and Methods
GPP Population
During 2004–2015 (12-year period), we identified all patients with a primary- or secondary diagnosis for psoriasis in the Swedish National Patient Register (NPR). The NPR covers inpatient care and secondary outpatient physician visits with diagnostic codes (ICD-10) and admission/discharge dates from both private and public caregivers. The NPR has been described in detail elsewhere.17 From this national cohort of patients with physician-confirmed diagnosis of psoriasis (ICD-10 code L40.x), we selected patients (all ages) with a primary or secondary GPP diagnosis. The patients in the GPP sub-cohort were identified by at least one ICD-10 code (L40.1) of GPP irrespective of concomitant psoriasis diagnosis (base case). First year with registration of a diagnosis of L40.1 within the study period was identified as index year. The same GPP population has previously been described in a study reporting the prevalence and incidence of GPP in Sweden.15
Population-Based Controls
We created a population-based control group from the Total Population Register (TPR) by matching five controls on year of birth, sex and residential area for each included patient. The TPR is the civil registration of vital events (eg, births, deaths) of all Swedish inhabitants, administrated by Statistics Sweden. The register is continuously updated and used for a variety of purposes by healthcare providers and medical researchers.18 In TPR, all citizens are identified by their unique personal identification number (PIN). By law, all healthcare providers must be registered by the patient’s PIN, which is automatically assigned to all residents. The controls were required to have no history of registered physician visits or inpatient stays (registration of diagnostic code L40.x), or drug use (ATC codes: D05AX02, D05AX52), consistent with psoriasis in the NPR and the PDR, respectively, during the period 2004–2015.
Psoriasis Vulgaris Control Population
From the national psoriasis cohort identified in the NPR, we also created a psoriasis vulgaris control population. For each GPP patient, we matched three patients with a diagnosis of psoriasis vulgaris on year of birth, sex, and index year. To be defined as a psoriasis vulgaris patient, the requirement was at least one registered ICD-10 code of L40.0 or L40.9 as primary or secondary diagnosis 2004–2015. The rationale for using two different codes is that there is reason to believe that both codes are used for the diagnosis of psoriasis vulgaris in clinical practice. The psoriasis vulgaris controls were required to have no history of registered healthcare consistent with GPP or palmoplantar pustulosis. First year within the study period with registration of L40.0/L40.9 was identified as index year.
Analyses of Resource Use and Associated Costs
Individuals included in the healthcare resource use and cost analyses consisted of patients and controls who were still alive and living in Sweden at the end of 2014. Direct costs, usually representing costs associated with healthcare resource utilization (eg, inpatient stays, outpatient physician visits and pharmaceutical services), were analyzed. Data on physician visits and inpatient stays were collected from the NPR for the year of 2015. Data on any filled prescriptions were collected from the Swedish Prescribed Drug Register (PDR). The PDR is a national individual-level data register where all dispensed prescribed drugs to the entire Swedish population are registered since 1 July 2005, with estimated national coverage close to 100%. The PDR includes information on dispensed item according to the Anatomic Therapeutic Chemical Classification (ATC), dispensed amount, PIN, age, sex, date of prescribing and dispensing, and costs.
We investigated separately the healthcare resource use attributable to GPP. Resource use presumed to be attributable to GPP were physician outpatient visits and inpatient stays identified in the NPR with registration of the ICD-10 code L40.1 and prescriptions attributable to psoriasis (according to drug list in Supplementary Table 1). As PDR does not hold information about treatment indication,19 we assumed that the drugs classified as psoriasis-related drugs were indicated for GPP and not for any other disease subtype within psoriasis for the GPP population. In addition, the consequences of PDR lacking information on treatment indication is also that the general population, required to have no registration of a psoriasis diagnosis, can have use of psoriasis-related drugs, but for another indication than psoriasis. Healthcare resource use and associated direct costs, both all-cause and GPP-related, were calculated for the year of 2015. All costs were converted from Swedish kronor to euro using currency conversion rate for 2015 of 1 euro=11.7995 SEK obtained from Sveriges Riksbank (https://www.riksbank.se).
Alternative Analysis
In an alternative analysis, we excluded GPP patients with a registered psoriasis vulgaris diagnosis (one primary diagnosis of L40.0 or L40.9) in the NPR 2004–2015. The rationale for the analysis of GPP patients without the presence of psoriasis vulgaris was to exempt the analyses from possible effects of psoriasis vulgaris on costs within the GPP groups. The alternative analysis was performed only in comparison to psoriasis vulgaris controls.
Statistical Analysis
Descriptive statistics were used on demographic variables, healthcare resource use (proportion of individuals with use and mean/median number of visits/inpatients) and costs. Differences between groups were tested by chi-squared test for categorical data and for numerical data, Student’s t-test or Mann–Whitney U-test were used as appropriate. Analyses were performed using STATA Statistical software, version Stata/IC 14.2 (College Station, Texas, USA).
Ethical Approval and Data Protection
This study was conducted according to the Declaration of Helsinki and approved by the Regional Ethical Review Board at Umeå University, Sweden. The data accessed for the study comply with relevant data protection and privacy regulations.
Results
Characteristics of the Study Population
During the study period (2004–2015), we identified 1093 patients who fulfilled the inclusion criteria for GPP. Out of these patients, 914 (84%) were still alive and residents in Sweden at the start of 2015 and could be included in analyses of healthcare resource use and costs (Table 1). There were no major differences in the sex- or age distribution, or proportion of GPP patients with concomitant psoriasis vulgaris, between the total number of GPP cases during the study period and those remaining in the GPP population in 2015. For the 914 GPP patients analyzed, there were 4047 matched controls from the general population and 2556 matched psoriasis vulgaris controls in 2015 (Table 1). Age- and sex distribution was similar between GPP patients and control groups.
 |
Table 1 Characteristics of Study Populations Including GPP Patients, Matched Population-Based Controls and Matched Controls with Psoriasis Vulgaris |
Physician Outpatient Visits and Inpatient Stays
Analyses of healthcare resource use during 2015 showed that a significantly larger proportion of patients with GPP had all-cause physician outpatient visits and inpatient stays compared to both population-based and psoriasis vulgaris controls (Table 2). Among those with all-cause physician visits, patients with GPP did on average almost two more visits compared to population-based controls and one more visit compared to psoriasis vulgaris controls. Moreover, among those with inpatients stays, the mean (median) length of stay (LOS) for patients with GPP was approximately 7 (2) and 6 (1) days longer compared to population-based and psoriasis vulgaris controls, respectively. GPP-specific physician visits and inpatient stays were defined as those with diagnostic code L40.1 for primary or secondary diagnosis. Among patients with GPP, 16% had a GPP-specific secondary outpatient physician visit and 3.2% had a GPP-specific inpatient stay.
 |
Table 2 Physician Visits and Inpatient Stays During 2015 for Patients with GPP Compared to Population-Based and Psoriasis Vulgaris Matched Controls, Respectively |
Drug Use
The proportion of persons using any prescribed drug during 2015 was significantly higher among patients with GPP compared to population-based controls, but not compared to psoriasis vulgaris controls (Table 3). Not surprisingly, patients with GPP had significantly higher rate of psoriasis-related drug use compared to population-based controls. Psoriasis-related drug use was also somewhat higher compared to psoriasis vulgaris controls and the difference was significant. Dermatological corticosteroids and emollients were the most commonly used psoriasis-related drugs among both patients with GPP and psoriasis vulgaris controls.
 |
Table 3 Drug Use During 2015 for Patients with GPP Compared to Matched Population-Based and Psoriasis Vulgaris Controls, Respectively |
Direct Costs
For patients with GPP, the mean total annual direct cost per patient was estimated to 5062 euros (95% CI=4471–5653), which was more than three times higher compared to population-based controls (mean=1610 euros; 95% CI=1436–1786) and nearly double compared to psoriasis vulgaris controls (mean=2826 euros; 95% CI=2597–3054) (Supplementary Table 2). Total costs and costs across the main cost components (ie, physician visits, inpatients stays and drugs) were all significantly higher for patients with GPP compared to both control groups (Figure 1, Supplementary Table 2). Costs due to all-cause inpatient stays represented the largest share of the total costs for both patients with GPP (40%) and population-based controls (51%), whereas for psoriasis vulgaris controls, all-cause drug treatment was the largest share (61%). The annual mean psoriasis-related drug cost represented 27% (biologics 23%) for the GPP cases. The corresponding values for population-based controls and psoriasis vulgaris controls were 1% (biologics 0.8%) and 15% (biologics 11%), respectively.
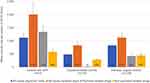 |
Figure 1 Mean annual direct costs over different cost components during 2015 for patients with GPP compared to population-based and psoriasis vulgaris-matched controls. All costs are in euros. GPP, generalized pustular psoriasis (L40.1 as primary or secondary diagnosis). For all numbers from costs analyses including confidence intervals and p-value, see Supplementary Table 2. |
For patients with GPP, the total estimated cost attributable to GPP problems, including GPP-cause physician visits, GPP-cause inpatients stays and psoriasis-related drugs, was 1646 euros and represented 33% of total healthcare cost (Table 4). Drug treatment was the cost component with the highest proportion (70%) of costs attributable to GPP. In contrast, out of the total cost for physician outpatient visits and inpatient care, respectively, costs attributed to GPP accounted for only 6.3% and 11.3%, respectively.
 |
Table 4 Costs Due to All-Cause Healthcare Use for Patients with GPP in 2015 and Costs Attributable to Resource Use Associated Specifically with GPP. All Costs are in Euros |
Alternative Analysis
Out of the 914 GPP patients included in the base case, we identified 430 (47%) with no registration of a psoriasis vulgaris primary diagnosis of L40.0 or L40.9 in the NPR 2004–2015. For these patients, mean age at index year was 56 year and 65% were women. To this population, we matched 1195 psoriasis vulgaris controls (mean age at index year=55; 67% women) for year of birth, sex, and index year (Supplementary Table 3).
Excluding GPP cases without psoriasis vulgaris in alternative analyses reduced the total direct cost by nearly 1148 euros, but it remained significantly higher compared to psoriasis vulgaris controls (3878 vs 2777 euros, p=0.004) (Supplementary Table 6). There were no major changes with regards to physician visits, inpatient stays or associated costs (Supplementary Tables 4, 6 and 7); compared to psoriasis vulgaris controls, the significantly higher costs for GPP patients remained for all-cause physician visits (1115 vs 831 euros, p=0.010) and inpatient stays (1856 vs 1093, p=0.010) (Supplementary Table 6). However, for drug use, the alternative analyses showed that removing GPP patients with psoriasis vulgaris reduced the percentage of GPP patients using psoriasis-related drugs from 67% to 51% (12.5–4% for biologics) (Table 3, Supplementary Table 5). This difference was also reflected in the costs of psoriasis-related drugs which decreased from 1348 to 436 euros (Table 4, Supplementary Table 7). Particularly evident was the reduction in the cost of biologics after removing GPP patients with psoriasis vulgaris (1153 vs 343 euros) which was no longer significantly higher compared to psoriasis vulgaris controls (Supplementary Tables 2 and 6).
Discussion
In this large nationwide population-based register study, we found a higher economic disease burden in GPP patients compared to both controls from the general population and patients with psoriasis vulgaris.
Results from the cost calculations showed that a pronounced proportion of the economic burden of disease in GPP patients was represented by costs attributable to inpatients stays and use of psoriasis-related drugs, biologics in particular. The results also showed that only a minor part of costs of physician visits and inpatients stay (6.3% and 11.3%, respectively) was attributable to specific GPP problems indicating a high economic burden due to complications and consequences of GPP or comorbidities in these patients. This finding is in line with results from a separate study using the same GPP patients and controls, which demonstrated an increased risk of comorbidities in patients with GPP compared to both the general population and patients with psoriasis vulgaris (manuscript in progress). Notably, the present study showed that 22% of GPP patients had an all-cause inpatient stay during the one-year follow-up but only 3.2% had an GPP-cause inpatient stay. Factors that may trigger GPP flares include corticosteroid use and withdrawal, pregnancy, upper respiratory tract infections, stress and certain medication.20 A GPP flare, which regularly require inpatient care, may include a number of symptoms, such as fever, chills, malaise, anorexia, nausea, and severe pain.20 Our results support previous findings demonstrating an elevated healthcare need in GPP patients who often involve hospitalization,2,4,5,7 but also suggest that the hospitalization in this patient group may be related to consequences and complications of GPP registered in the NPR with diagnostic codes other than the one for GPP (L40.1).
We also found that the greater proportion (70%) of cost due to psoriasis-related drug use was attributable to GPP problems. Interestingly, the alternative analyses showed that removing GPP patients with psoriasis vulgaris reduced the percentage of GPP patients using psoriasis-related drugs from 67% to 51% (12.5–4% for biologics) indicating that a proportion of psoriasis-related drugs used in this patient group may be prescribed for treatment of symptoms related to psoriasis vulgaris rather than GPP. This was also reflected by a reduction in the relative costs of psoriasis-related drugs from 70% to 48% of total drug costs. In the context of availability of treatment options for patients with GPP, this is not an unexpected finding. As there are no approved treatment alternatives for GPP patients without concomitant plaques psoriasis at the market, there are no labelled products, ie, biologics at relatively high prices, to be exclusively prescribed to this patient group. However, regardless of whether GPP with or without psoriasis vulgaris is analyzed, the proportion of psoriasis-related drugs may be overestimated since the PDR does not hold information about the indication for treatment. Several drugs, in particular systemic agents, classified as psoriasis-related in this paper, are also indicated for treatment in rheumatic diseases, a category of conditions with an occurrence of around 20% in our population of GPP patients (manuscript in progress).
The alternative analyses showed that the mean annual direct cost remained significantly elevated also in GPP patients with no psoriasis vulgaris compared to psoriasis vulgaris controls. Worth noticing is that the mean annual cost per GPP patient decreased by nearly 1150 euros when excluding GPP patients with a concomitant psoriasis vulgaris diagnosis. This was primarily due to a decreased proportion of patients using biologics. The proportion of GPP patients with inpatient stays, however, did not change substantially.
Because of differences in disease geno- and phenotype expressions between different ethnicities, cost perspective, methodologies, and healthcare systems, comparison between resource use and cost studies should be done cautiously. Until recently, there were no studies on the healthcare burden of GPP.2 However, in addition to comorbidities, the study by Morita et al4 presented all-cause resource use for GPP patients (N=614) in Japan of all ages compared to both matched population-based controls and matched psoriasis vulgaris controls during a 12-month follow-up period. In accordance with our study, the resource use was elevated in GPP patients compared to both population-based and psoriasis vulgaris controls. In the Japanese study, around 36% of the GPP patients experienced an all-cause inpatient stay with a mean LOS of 25 days during follow-up. The corresponding figure in the present study was 22% and a mean LOS of 17 days. Morita et al also observed that 74% of the GPP patients received systemic treatment compared to 38% for the psoriasis vulgaris controls and the authors suggested that this finding probably indicates a more severe disease in GPP patients compared to controls. This is supported by our study, which showed that 38% of the GPP patients and 15% of the psoriasis vulgaris controls were prescribed a systemic drug (data not shown). Comparison of other use of medication was not possible due to differences in presentation of data between the studies. Results from the Morita study4 indicate an overall higher resource use and economic burden in GPP patients compared to that of our study. One reason for this might be that in the Morita study, the patients were recruited from mostly larger hospitals with acute care, which means that a large proportion of included patients likely were in poor health.
An apparent strength of this study is the large population-based register used for analyses, which facilitate generalization beyond the study population. Of particular strength was the use of the NPR for inpatient and outpatient specialist care visits covering the whole Swedish population for an extensive period. Information about resource use was collected from a large register source consisting of routinely collected data from clinical practice. This decreases the risk of recall bias that might occur in studies based on self-registration. Another strength is the comparison of direct costs in GPP to both the general population and psoriasis vulgaris population, which was lacking in most previous studies.
Our study has also some weaknesses. Firstly, the validity of the GPP diagnosis has not been examined. As there is no standard case definition of the diagnosis of GPP in Sweden, our case definitions were based on coded diagnoses of GPP and not on classification criteria or validation through medical record review, and thus, could potentially be subject to misclassification bias. In the NPR, we required one visit in specialized care with a diagnostic code of L40.1 to be classified as a GPP case, which is a similar method used in the studies by Morita et al and Okubo et al.4,7 The rationale behind this is that, as GPP is considered a severe and potentially life-threatening disease that frequently leads to hospitalization, hence we consider that if a person has been in specialized care and received a physician confirmed GPP diagnosis, it is likely to indicate genuine GPP disease. Moreover, the GPP disease is usually described based on three phases: pre-pustular, flare and post flare phase. A cost calculation stratified on these stages, differing in disease severity, would have added valuable information on the economic burden in GPP patients. Unfortunately, our data do not allow for such analysis. Furthermore, this study has a healthcare payer perspective including neither out-of-pocket expenses of the patients nor indirect costs due to work loss. Thus, the results may underestimate the costs. Also, the study lacks data on potential confounding such as smoking, drinking and treatment effects.
This study is part of a larger research project aiming to increase the understanding of the burden of disease of GPP. In a previous study, we have used the same nationwide GPP population reported here to calculate a GPP point prevalence of 9/100 000.15 Here, we continued the research on the Swedish GPP population to increase the knowledge regarding the economic burden. Other relevant aspects for understanding of the full burden of disease are, eg, the comorbidity profile of GPP and how the disease affects the patients’ health-related quality of life.
In conclusion, our study indicates a higher economic burden for GPP patients compared to both the general population and patients with psoriasis vulgaris and no GPP. Cost driving components were primarily inpatient care followed by use of biologics. Only about a third of the costs was directly attributable to GPP problems, indicating a high economic burden of GPP-consequences and complications. The severity of GPP in combination with the economic burden described here highlights the need for better therapeutical options for GPP patients.
Acknowledgments
The authors would like to thank Gunnar Brådvik, data analyst, for valuable data management, and Karin Wahlberg, Medical Writer, for writing and editorial support, both at The Swedish Institute for Health Economics (IHE).
Funding
This research is the result of a research platform, which has received financial support from AbbVie, Boehringer Ingelheim, Eli Lilly, Janssen Cilag, Leo Pharma and Novartis. This study’s design and analysis was funded by Boehringer Ingelheim. Sponsors had no access to data. None of the authors has any conflict of interest in connection to the article. The authors had full independence regarding study design, data collection, analysis, result interpretation and decision to publish.
Disclosure
MSE is responsible for dermatology in the project management for the national guidelines for psoriasis at the Swedish Board of Health and Welfare. JMN and SL have been involved in the health economic analyses of the national guidelines for psoriasis at the Swedish Board of Health and Welfare, both report grants from Boehringer Ingelheim, during the conduct of the study. The authors have no other conflicts of interest to declare in this work.
References
1. Twelves S, Mostafa A, Dand N, et al. Clinical and genetic differences between pustular psoriasis subtypes. J Allergy Clin Immunol. 2019;143:1021–1026. doi:10.1016/j.jaci.2018.06.038
2. Kharawala S, Golembesky AK, Bohn RL, et al. The clinical, humanistic, and economic burden of generalized pustular psoriasis: a structured review. Expert Rev Clin Immunol. 2020;16(3):239–252. doi:10.1080/1744666X.2019.1708193
3. Hoegler KM, John AM, Handler MZ, et al. Generalized pustular psoriasis: a review and update on treatment. J Eur Acad Dermatol Venereol. 2018;32(10):1645–1651. doi:10.1111/jdv.14949
4. Morita A, Kotowsky N, Gao R, et al. Patient characteristics and burden of disease in Japanese patients with generalized pustular psoriasis: results from the Medical Data Vision claims database. J Dermatol. 2021;48(10):1463–1473. doi:10.1111/1346-8138.16022
5. Choon SE, Lai NM, Mohammad NA, et al. Clinical profile, morbidity, and outcome of adult-onset generalized pustular psoriasis: analysis of 102 cases seen in a tertiary hospital in Johor, Malaysia. Int J Dermatol. 2014;53(6):676–684. doi:10.1111/ijd.12070
6. Jin H, Cho HH, Kim WJ, et al. Clinical features and course of generalized pustular psoriasis in Korea. J Dermatol. 2015;42:674–678. doi:10.1111/1346-8138.12863
7. Okubo Y, Kotowsky N, Gao R, et al. Clinical characteristics and health-care resource utilization in patients with generalized pustular psoriasis using real-world evidence from the Japanese Medical Data Center database. J Dermatol. 2021;48(11):1675–1687. doi:10.1111/1346-8138.16084
8. Viguier M, Allez M, Zagdanski AM, et al. High frequency of cholestasis in generalized pustular psoriasis: evidence for neutrophilic involvement of the biliary tract. Hepatology. 2004;40:452–458. doi:10.1002/hep.20305
9. Umezawa Y, Ozawa A, Kawasima T, et al. Therapeutic guidelines for the treatment of generalized pustular psoriasis (GPP) based on a proposed classification of disease severity. Arch Dermatol Res. 2003;295(Suppl 1):S43–S54. doi:10.1007/s00403-002-0371-6
10. Zheng J, Chen W, Gao Y, et al. Clinical analysis of generalized pustular psoriasis in Chinese patients: a retrospective study of 110 patients. J Dermatol. 2021;48(9):1336–1342. doi:10.1111/1346-8138.15958
11. Augey F, Renaudier P, Nicolas JF. Generalized pustular psoriasis (Zumbusch): a French epidemiological survey. Eur J Dermatol. 2006;16:669–673.
12. Fabbri P. Psoriasis. From clinical diagnosis to new therapies. Florence, Italy; 2004.
13. Lee JY, Kang S, Park JS, et al. Prevalence of psoriasis in Korea: a population-based epidemiological study using the Korean National Health Insurance Database. Ann Dermatol. 2017;29(6):761–767. doi:10.5021/ad.2017.29.6.761
14. Ohkawara A, Yasuda H, Kobayashi H, et al. Generalized pustular psoriasis in Japan: two distinct groups formed by differences in symptoms and genetic background. Acta Derm Venereol. 1996;76:68–71. doi:10.2340/00015555766871
15. Löfvendahl S, Norlin JM, Schmitt-Egenolf M. Prevalence and incidence of generalised pustular psoriasis in Sweden - a population-based register study. Br J Dermatol. 2022. doi:10.1111/bjd.20966
16. Hanna ML, Singer D, Valdecantos WC. Economic burden of generalized pustular psoriasis and palmoplantar pustulosis in the United States. Curr Med Res Opin. 2021;37:735–742. doi:10.1080/03007995.2021.1894108
17. Ludvigsson JF, Andersson E, Ekbom A, et al. External review and validation of the Swedish national inpatient register. BMC Public Health. 2011;11:450. doi:10.1186/1471-2458-11-450
18. Ludvigsson JF, Almqvist C, Bonamy AK, et al. Registers of the Swedish total population and their use in medical research. Eur J Epidemiol. 2016;31:125–136. doi:10.1007/s10654-016-0117-y
19. Wettermark B, Hammar N, Fored CM, et al. The new Swedish Prescribed Drug Register–opportunities for pharmacoepidemiological research and experience from the first six months. Pharmacoepidemiol Drug Saf. 2007;16:726–735. doi:10.1002/pds.1294
20. Benjegerdes KE, Hyde K, Kivelevitch D, et al. Pustular psoriasis: pathophysiology and current treatment perspectives. Psoriasis. 2016;6:131–144. doi:10.2147/PTT.S98954
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
