Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 15
Dupilumab for Chronic Prurigo in Different Backgrounds: A Case Series
Authors Luo N , Wang Q , Lei M, Li T, Hao P
Received 21 June 2022
Accepted for publication 5 September 2022
Published 12 September 2022 Volume 2022:15 Pages 1863—1867
DOI https://doi.org/10.2147/CCID.S379231
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Dr Jeffrey Weinberg
Nana Luo,1,2 Qiuyue Wang,2 Min Lei,2 Tianhao Li,1 Pingsheng Hao1
1Department of Dermatology, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu, People’s Republic of China; 2School of Clinical Medicine, Chengdu University of Traditional Chinese Medicine, Chengdu, People’s Republic of China
Correspondence: Tianhao Li; Pingsheng Hao, Hospital of Chengdu University of Traditional Chinese Medicine, No. 39 Shi-er-qiao Road, Chengdu, People’s Republic of China, Tel +86-13880986337 ; +86-13881965024, Fax +86-28-87732407, Email [email protected]; [email protected]
Abstract: Chronic prurigo (CPG) is a chronic inflammatory disease characterized by intense pruritus, and therapy is often challenging due to its unclear pathogenesis. Here, we report four patients with refractory CPG from different backgrounds who experienced failure of conventional therapy and were controlled with dupilumab, accompanied by a significant decrease in pruritus numerical rating scale, dermatology life quality index, and investigator global assessment, without adverse effects. Furthermore, dupilumab improved the condition of asthma in CPG patients along with an increase in asthma control test scores.
Keywords: dupilumab, chronic prurigo, therapy, asthma
Introduction
Chronic prurigo (CPG) is a chronic disease characterized by intensely pruritic hyperkeratotic nodules, which seriously affect the quality of life of patients and cause a heavy financial burden.1,2 Chronicity of CPG is caused by a pathological itch-scratch cycle and neuronal sensitization.3 The Food and Drug Administration has not yet approved any medications for the treatment of CPG, which makes the treatment for CPG diverse and challenging. Conventional treatments (eg, topical steroid ointments, antihistamines, immunosuppressants, antidepressants) have variable efficacy and may have potentially toxic side effects. Fortunately, emerging therapies include biologics (eg, dupilumab, lebrikizumab, tralokinumab, nemolizumab, omalizumab), small molecules (eg, neurokinin-1 receptor antagonists, janus kinase inhibitors), and nalbuphine (a μ-antagonist/κ-agonist) are being tried for the treatment of CPG.4,5
As T-helper 2 (Th2) inflammatory factors, interleukin (IL)-13, IL-4, and IL-31 have been demonstrated for their roles in the pathogenesis and maintenance of CPG, an increasing number of cases recommend dupilumab as an effective agent for CPG.6 Therefore, when we encountered 4 patients with refractory CPG in different backgrounds, we initiated a dupilumab subcutaneous injection regimen with a 600mg loading and 300mg/2 weeks maintenance dose, and the condition was evaluated using the itch Numerical Rating Scale (NRS), Dermatology Life Quality Index (DLQI) and Investigator Global Assessment for chronic prurigo (IGA-CPG). The IGA-CPG7 is a new assessment for objective evaluation of CPG lesions with high reliability: a score of 0 means no pruriginous lesions (0 lesions); a score of 1 means rare palpable pruriginous lesions (approximately 1–5 lesions); a score of 2 means few palpable pruriginous lesions (approximately 6–19 lesions); a score of 3 means many palpable pruriginous lesions (approximately 20–100 lesions); a score of 4 means abundant palpable pruriginous lesions (over 100 lesions).
Case Presentation
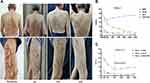
Patient 1; A 72-year-old male with a history of asthma, who had a CPG diagnosed 10 years. The history of atopic dermatitis (AD) was denied. Antihistamines, gabapentin, hydroxychloroquine, and steroid ointments were used (Table 1). The lesions were typically nodular on the extremities and trunk (Figure 1A) with an NRS score of 10, a DLQI score of 26, and an IGA-CPG score of 4 (Figure 1B). Laboratory tests showed normal eosinophils, elevated total immunoglobulin (Ig) E to 457 IU/mL. Due to the patient’s age and history of asthma, dupilumab was recommended. Two weeks after injection, the patient’s pruritus began to resolve, and at week 8, the NRS dropped to 2, the DLQI dropped to 9, and the IGA-CPG dropped to 3. At week 16, the patient’s NRS dropped to 0 and the DLQI dropped to 5, as well as the IgE returned to the normal range with relief of the clinical lesions (Figure 1A). At week 20, the IGA-CPG score dropped to 2, until week 24, the patient reached IGA-CPG 1 (Figure 1B), and we adjusted the injection regimen to 300 mg/4 weeks, based on experience with dupilumab in AD. After 3 months of treatment with such a regimen, the IGA-CPG score reached 0, and the patient discontinued dupilumab with the use of antihistamines and steroid creams for maintenance treatment. Meanwhile, we evaluated the patient’s asthma using the asthma control test (ACT), a score below 20 indicates uncontrolled asthma, a score of 20–24 indicates well controlled, and a score of 25 indicates complete control. The patient’s ACT increased from 16 at baseline to 20 at week 8. Until week 20, the ACT reached 23 (Figure 1B), and the frequency of salbutamol was reduced under the respiratory physician’s direction. We followed the patients for 6 months after discontinuation of dupilumab and no recurrence of lesions or pruritus was observed. In addition, asthma is well controlled. This suggests that dupilumab is beneficial for both CPG and asthma.
 |
Table 1 Basic Information of 4 Patients of CPG Treated with Dupilumab |
Patient 2; A 59-year-old male with a 7-year history of CPG, with a record of chronic actinic dermatitis, and hypertension (Table 1). His lesions presented as multiple nodules on the forehead, neck, and extremities of the trunk, with the exposure site being the most prominent. He was treated with hydroxychloroquine, tripterygium, glycosides, steroid ointments, and liquid nitrogen cryotherapy, but received poor feedback. His eosinophils and IgE were normal. Cyclosporine was excluded due to the underlying disease of hypertension. So we initiated hydroxychloroquine combined with dupilumab at a baseline of 10 on the NRS, 26 on the DLQI, and 4 on the IGA-CPG. At week 4, his pruritus was significantly relieved, NRS reduced to 3 and DLQI dropped to 13. Satisfactory efficacy was obtained at week 8, with an NRS score of 1, a DLQI score of 8, and an IGA-CPG score of 3. Meanwhile, the patient discontinued dupilumab for financial reasons and used hydroxychloroquine in combination with the antihistamine to control his disease. Interestingly, at week 12, which was 1 month after stopping the dupilumab, the patient’s NRS dropped to 0 and IGA-CPG dropped to 2.
Patient 3; A 44-year-old male with chronic hepatitis B and post-splenectomy presented with CPG for 3 years. The lesions were scattered on the back and extremities, and nodules on the back fused to plaques, and his work was severely affected by the recurrent condition. Despite trying various treatments, the itching continued to recur (Table 1). Laboratory tests showed normal hepatic function and eosinophils, and the IgE rose to 430 IU/mL. The NRS score was 10, the DLQI score was 25, and the IGA-CPG score was 3. We initiated treatment with dupilumab and the NRS decreased to 3 and the DLQI decreased to 12 at week 4, the itching was completely relieved at week 8, with an NRS score of 0, a DLQI score of 8, and an IGA-CPG score of 2. The IgE returned to normal, and the liver function was also within the normal range on recheck. However, the patient discontinued dupilumab treatment for financial reasons and used hydroxychloroquine in combination with antihistamines to control the condition, at the first month after discontinuation, the patient’s NRS remained at 0 with high satisfaction.
Patient 4; A 53-year-old female with a 2-year history of CPG with no underlying disease. Multiple treatments had been applied to her hospitalizations (Table 1). However, there was still unsatisfactory feedback. At the time of presentation, the patient presented with intense pruritic nodules and plaques on the extremities, back, and neck, accompanied by stinging pain. The condition affected the quality of life seriously, with a DLQI score of 27, an NRS score of 10, and an IGA-CPG score of 3. The laboratory tests showed eosinophils and IgE in the normal range. The pruritus and tingling resolved significantly by the 4th week after the patient received dupilumab injections, with a DLQI score of 13, and an NRS score of 2. At week 8, NRS dropped to 0, IGA-CPG dropped to 2 and DLQI dropped to 6. The patient also discontinued dupilumab treatment for financial reasons and used methotrexate in combination with antihistamines to control the condition. At the third month of discontinuation, the patient was followed up with an NRS score of 2, a DLQI score of 8, and an IGA-CPG score of 1, without emerging lesions.
Discussion
Neuroimmune dysregulation is central to the pathogenesis of CPG, triggering and maintaining a vicious itch-scratch cycle through complex interactions between inflammatory cells, pro-inflammatory cytokines, neuropeptides and skin sensitizing neurons.3 The main purpose of CPG treatment is to break the vicious itch-scratch cycle and to cure pruritic lesions by reducing itching. Recently, CPG receives increasing attention as new therapeutic targets have been identified, thereby allowing the use of targeted approaches. While Th2-type inflammation is core to the immune imbalance, the upregulation of lL-4 and lL-13 in CPG patients is confirmed.4 Dupilumab, the first off-label drug for CPG, has comparable efficacy to AD in the treatment of CPG by blocking the IL-4 and IL-13 pathways to inhibit the Th2-type immune response and pruritic neural pathway.8 Tralokinumab9 and lebrikizumab10 are targeted biologics against IL-13 that are effective in controlling moderate-to-severe AD. However, both tralokinumab and lebrikizumab have not been reported in the treatment of CPG.
IL-31, an important cytokine in Th2-type inflammation, was also found to infiltrate CPG lesions, and IL-31 messenger RNA was upregulated 50-fold in CPG lesions compared to healthy skin biopsy specimens.11 Nemolizumab, a humanized monoclonal antibody to the IL-31 receptor a, reduced pruritus and lesion severity to a greater extent compared to placebo in a randomized, double-blind Phase 2 trial.12 Omalizumab is a recombinant humanized anti-IgE monoclonal antibody for chronic spontaneous urticaria,13 there were two case reports of successful treatment of CPG with omalizumab.14,15 For janus kinase inhibitors, we reported a case of an elderly CPG patient with complete relief of pruritus after 6 weeks of tofacitinib therapy, and the lesions did not recur after 5 months of follow-up after discontinuation of tofacitinib.16 However, more real-world data are needed to demonstrate the effectiveness and safety of these emerging treatments for CPG in the future.
Our patients with CPG were treated with dupilumab with different underlying diseases and different histories of treatment, and they all benefited from it. Patient 1, an elderly patient who has asthma with elevated IgE, was considered to be a special AD patient. The change in IGA-CPG, NRS, and ACT confirmed dupilumab’s excellent control of asthma and CPG. It is consistent with the previous reports.17,18
Patient 2–4 remained under control 12 weeks after discontinuation of dupilumab with a mean NRS score of 2.3, a mean IGA-CPG score of 1.7, and a mean DLQI score of 9.3 (Figure 1C), for which feedback was satisfactory. For the underlying conditions, dupilumab reduced the severity of photosensitivity and had no adverse effects on hepatitis and hypertension. In contrast, Patient 1, 2 with a background of atopic disease and a longer duration of disease took longer to achieve an NRS score of 0 than patient 3.4 without an atopic background and a shorter history of the disease (Table 1).
Conclusion
Dupilumab relieves pruritus quickly and effectively. Our case series supports dupilumab as a potential therapy for CPG, especially for patients accompanied by allergic diseases. Off-label and high costs are problems for physicians and patients, and more real-world evidence is required to support dupilumab for CPG.
Consent Statement
Informed consent was obtained from the patient to publication of the report details and associated images. Institutional approval was not required to publish the case details.
Funding
Program of Health Commission of Sichuan Province (Grant No. ChuanGanYan 2021-505) and Hospital of Chengdu University of TCM Scientific Research Capacity Enhancement “Hundred Talents Program” (Grant No.20B04) supported publication fees.
Disclosure
Tianhao Li and Pingsheng Hao are co-correspondence authors for this study. All authors have no conflicts of interest in this work.
References
1. Kwatra SG. Prurigo nodularis. JAMA Dermatol. 2022;158(3):336. doi:10.1001/jamadermatol.2021.5307
2. Huang AH, Canner JK, Khanna R, et al. Real-world prevalence of prurigo nodularis and burden of associated diseases. J Invest Dermatol. 2020;140(2):480–483.e484. doi:10.1016/j.jid.2019.07.697
3. Williams KA, Huang AH, Belzberg M, et al. Prurigo nodularis: pathogenesis and management. J Am Acad Dermatol. 2020;83(6):1567–1575. doi:10.1016/j.jaad.2020.04.182
4. Müller S, Bieber T, Ständer S. Therapeutic potential of biologics in prurigo nodularis. Expert Opin Biol Ther. 2022;22(1):47–58. doi:10.1080/14712598.2021.1958777
5. Maredia H, Kwatra SG. Emerging novel therapeutic agents for the treatment of patients with prurigo nodularis. J Dermatolog Treat. 2021;32(7):741–744. doi:10.1080/09546634.2019.1707469
6. Williams KA, Roh YS, Brown I, et al. Pathophysiology, diagnosis, and pharmacological treatment of prurigo nodularis. Expert Rev Clin Pharmacol. 2021;14(1):67–77. doi:10.1080/17512433.2021.1852080
7. Zeidler C, Pereira MP, Augustin M, et al. Investigator’s global assessment of chronic prurigo: a new instrument for use in clinical trials. Acta Derm Venereol. 2021;101(2):adv00401. doi:10.2340/00015555-3701
8. Garcovich S, Maurelli M, Gisondi P, et al. Girolomoni G. Pruritus as a distinctive feature of type 2 inflammation. Vaccines. 2021;9(3):303. doi:10.3390/vaccines9030303
9. Wollenberg A, Blauvelt A, Guttman-Yassky E, et al. Tralokinumab for moderate-to-severe atopic dermatitis: results from two 52-week, randomized, double-blind, multicentre, placebo-controlled Phase III trials (ECZTRA 1 and ECZTRA 2). Br J Dermatol. 2021;184(3):437–449. doi:10.1111/bjd.19574
10. Guttman-Yassky E, Blauvelt A, Eichenfield LF, et al. Efficacy and safety of lebrikizumab, a high-affinity interleukin 13 inhibitor, in adults with moderate to severe atopic dermatitis: a Phase 2b randomized clinical trial. JAMA dermatol. 2020;156(4):411–420. doi:10.1001/jamadermatol.2020.0079
11. Meng J, Moriyama M, Feld M, et al. New mechanism underlying IL-31-induced atopic dermatitis. J Allergy Clin Immunol. 2018;141(5):1677–1689.e1678. doi:10.1016/j.jaci.2017.12.1002
12. Ständer S, Yosipovitch G, Legat FJ, et al. Trial of nemolizumab in moderate-to-severe prurigo nodularis. N Engl J Med. 2020;382(8):706–716. doi:10.1056/NEJMoa1908316
13. Metz M, Vadasz Z, Kocatürk E, et al. Omalizumab updosing in chronic spontaneous urticaria: an overview of real-world evidence. Clin Rev Allergy Immunol. 2020;59(1):38–45. doi:10.1007/s12016-020-08794-6
14. Ugajin T, Inazawa M, Inui K, et al. A case of chronic prurigo successfully treated with omalizumab. Eur j dermatol. 2018;28(5):691–692. doi:10.1684/ejd.2018.3351
15. Grieco T, Chello C, Faina V, et al. Omalizumab therapy in a patient with cronic spontaneous urticaria, ulcerative colitis, hypereosinophilia and prurigo Besnier: a case report. J Dermatolog Treat. 2018;29(sup3):10–13. doi:10.1080/09546634.2018.1527990
16. Peng C, Li C, Zhou Y, et al. Tofacitinib for prurigo nodularis: a case report. Clin Cosmet Investig Dermatol. 2022;15:503–506. doi:10.2147/CCID.S354025
17. Wechsler ME, Ford LB, Maspero JF, et al. Long-term safety and efficacy of dupilumab in patients with moderate-to-severe asthma (TRAVERSE): an open-label extension study. Lancet Respir med. 2022;10(1):11–25. doi:10.1016/S2213-2600(21)00322-2
18. Muñoz-Bellido FJ, Moreno E, Dávila I. Dupilumab: a review of present indications and off-label uses. J Invest Allergol Clin Immunol. 2022;32(2):97–115. doi:10.18176/jiaci.0682
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.