Back to Journals » International Journal of General Medicine » Volume 14
Dual Growth Factor (rhTPO + G-CSF) and Chemotherapy Combination Regimen for Elderly Patients with Acute Myeloid Leukemia: A Phase II Single-Arm Multicenter Study
Authors Liu X, Shi H, Shen J, Li Y, Yan W , Sun Y, Liao A, Tan Y, Yang W, Wang H
Received 7 June 2021
Accepted for publication 20 September 2021
Published 27 September 2021 Volume 2021:14 Pages 6093—6099
DOI https://doi.org/10.2147/IJGM.S323699
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Xiaoyu Liu,1 Hua Shi,2 Jing Shen,1 Yang Li,1 Wei Yan,1 Ying Sun,1 Aijun Liao,1 Yehui Tan,3 Wei Yang,1,* Huihan Wang1,*
1Haematology Department of Shengjing Hospital, China Medical University, Shenyang, Liaoning, 110004, People’s Republic of China; 2Haematology Department of Sun Yat-sen Memorial Hospital, Sun Yat-sen University Shen Shan Central Hospital, Guangzhou, People’s Republic of China; 3Haematology Department of The First Hospital of Jilin University, Changchun, Jilin, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Wei Yang; Huihan Wang
Haematology Department of Shengjing Hospital, China Medical University, Shenyang, Liaoning, 110004, People’s Republic of China
Tel +86-18940251012
; +86-18940256966
Email [email protected]; [email protected]
Abstract: Acute myeloid leukemia (AML) is a disease affecting older adults, although optimal strategies for treating such patients remain unclear. This prospective phase II, open-label, multicenter study was designed to assess the efficacy and safety of two hematologic growth factors, recombinant human thrombopoietin (rhTPO) and granulocyte colony-stimulating factor (G-CSF), in combination with decitabine, cytarabine, and aclarubicin (D-CTAG regimen) to treat older adults with newly diagnosed AML (Identifier: NCT04168138). The above agents were administered as follows: decitabine (15 mg/m2 daily, days 1– 5); low-dose cytarabine (10 mg/m2 q12 h, days 3– 9); rhTPO (15,000 U daily, days 2, 4, 6, 8, 10– 24 or until > 50× 109/L platelets); aclarubicin (14 mg/m2 daily, days 3– 6); and G-CSF (300 μg daily, days 2– 9). We concurrently monitored historic controls treated with decitabine followed by cytarabine, aclarubicin, and G-CSF (D-CAG) only. After the first D-CTAG cycle, the overall response rate (ORR) was 84.2% (16/19), including 13 (73.7%) complete remissions (CRs) and three (15.8%) partial remissions. This CR rate surpassed that of the D-CAG treatment (p < 0.05). Median overall survival (OS) time in the D-CTAG group was 20.2 months (range, 4– 31 months), compared with 14 months in the D-CAG group, and 1-year OS was 78%. The proportion of those experiencing grade III–IV thrombocytopenia was significantly lower for D-CTAG (57.9%) than for D-CAG (88.4%; p < 0.05). Ultimately, the curative effect of adding rhTPO was not inferior to that of D-CAG, and D-CTAG proved safer for elderly patients, especially in terms of hematologic toxicity. A prospective phase III randomized study is warranted to confirm these observations.
Keywords: rhTPO, elderly, acute myeloid leukemia, CAG, decitabine
Introduction
Acute myeloid leukemia (AML) is one of the most common hematologic malignancies and affects older adults. The median age at diagnosis is 67 years. As the elderly increasingly account for a greater population percentage, AML is becoming more problematic. During the past 30 years, outcomes have improved for younger patients, whereas the prospects for older adults (> 60 years) have remained poor.1 The response rate in older adults given standard induction regimens is < 50%, and median overall survival (OS) is < 1 year.2–4 Older adults elderly are also inordinately burdened by unfavorable cytogenetic defects, medical comorbidities, and reduced tolerability to intensive chemotherapeutic protocols. These vulnerabilities predispose older adults to poorer outcomes than their younger counterparts, conferring lower response rates and shortened survival times (progression-free and overall survival [OS]).5 Safe and effective treatments for elderly patients with AML are thus urgently needed.
In 2000, Saito et al introduced a regimen of granulocyte colony-stimulating factor (G-CSF) plus low-dose cytarabine (ara-C) and aclarubicin (ACR) for use in this setting (CAG regimen),6 in an attempt to incorporate a hematologic growth factor in AML induction therapy, rather than relying on supportive care. The addition of decitabine, a demethylation agent, further improved the prognosis, yielding a 10-month median OS.7 This successful use of G-CSF in an induction regimen for AML confirmed its utility, helping to increase efficacy and reduce side effects in elderly patients.
Thrombopoietin (TPO) is a major factor in regulating megakaryocytic proliferation, maturation, and platelet formation.8 Recombinant human TPO (rhTPO) has been approved by China’s State Food and Drug Administration to treat thrombocytopenia after chemotherapy. TPO and c-MPL receptors are also involved in various physiologic processes, such as mitigating myocardial injury, nerve repair, vascular regeneration, sex hormone secretion, and immune regulation.9 However, the role of rhTPO in an induction regimen for AML is unreported as yet.
To determine if adding rhTPO to D-CAG (G-CSF) will increase the overall response rate (ORR) while decreasing the side effect of toxic agents, we designed a regimen of rhTPO and G-CSF in combination with decitabine, cytarabine, and aclarubicin (D-CTAG regimen). This trial aimed to determine the safety and efficacy of this D-CTAG regimen as a treatment for older adult patients with newly diagnosed AML.
Materials and Methods
Patients and Treatment
This was an investigator-initiated, prospective, nonrandomized, single-arm phase II clinical trial in elderly patients with AML registered at www.clinicaltrial.org (Identifier: NCT04168138). Candidates for the test regimen were elderly patients (> 60 years) newly diagnosed with AML, excluding those with acute promyelocytic leukemia or a chronic myeloid leukemic blast crisis. Eastern Cooperative Oncology Group performance status of 0–3 was further required.
This study was approved by the Human Ethics Committee of the China Medical University at Shengjing Hospital, and written informed consent was granted by all participants. This study was conducted in accordance with the Declaration of Helsinki.
The D-CTAG chemotherapeutic induction regimen consisted of decitabine (15 mg/m2 daily, days 1–5); low-dose cytarabine (10 mg/m2 q12 h, days 3–9); rhTPO (15,000 U daily, days 2, 4, 6, 8, and 10–24 or until a platelet count > 50 × 109/L was observed); aclarubicin (14 mg/m2 daily, days 3–6); and G-CSF (300 μg daily, days 2–9; Figure 1). We also collected data on a comparable patient group given traditional D-CAG chemotherapy without rhTPO (decitabine 15 mg/m2 daily, days 1–5; low-dose cytarabine 10 mg/m2 q12 h, days 3–9; aclarubicin 14 mg/m2 daily, days 3–6; G-CSF 300 μg daily, days 2–9) at the same time clinically to compare therapeutic outcomes. We evaluated the response after the first course of treatment. If the patient can tolerate and reach PR or above, continue D-CTAG chemotherapy and up to six courses. After this, the patient received decitabine monotherapy to maintain the disease until its progression. If patients stop the D-CTAG regimen for > 6 months and relapse, they can restart the D-CTAG regimen, enter clinical trials, or consider treatment with azacitidine or B-cell leukemia/lymphoma-2 inhibitors.
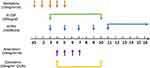 |
Figure 1 Schematic of the D-CTAG treatment regimen for AML. |
Treatment responses were assessed according to the National Comprehensive Cancer Network clinical practice guidelines of AML (version 2.2019). The ORR was defined as the rates of complete and partial remissions (CR; PR). Toxicities were determined according to the National Cancer Institute Common Toxicity Criteria.
Statistical Analysis
Differences in continuous variables were analyzed using t-tests and chi-squared tests. Kaplan-Meier analysis was performed to estimate the survival probabilities. The statistical analysis of all study data was driven by standard software (SPSS 22.0; IBM Corp, Armonk, NY, USA), setting significance at p < 0.05.
Results
Patient Characteristics
Between January 2017 and May 2020, 19 willing patient participants (age range, 61–81 years) with newly diagnosed AML were recruited for this trial of D-CTAG. A summary of patient characteristics is provided in Table 1. Six of these patients had also developed secondary malignant tumors. To assess the potential merit of added rhTPO, we concurrently monitored 38 patients treated with D-CAG only as controls. The baseline characteristics of the two therapeutic groups (D-CTAG vs D-CAG) were similar (Table 1).
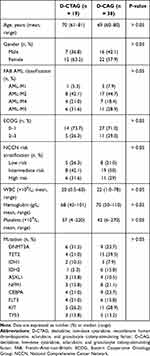 |
Table 1 Clinical Characteristics of the Patient Population |
Therapeutic Responses and Survival
After the first cycle, the D-CTAG induction regimen yielded an ORR of 84.2% (16/19), including 13 (73.7%) CRs and three (15.8%) PRs. In controls receiving D-CAG, the ORR was 78.9% (30/38), with 23 (60.5%) CRs and seven (18.4%) PRs. Although the CR rate for D-CTAG was significantly higher than D-CAG (p < 0.05), broader patient samplings are needed for verification.
During a median follow-up of 31 months, the median overall survival (OS) time was 20.2 months (range, 4–31 months) in the D-CTAG group (Figure 2). All patients were followed for at least 1 year, with an OS of 78%. Again, OS in the D-CTAG group exceeded that of the D-CAG group (median OS, 14 months; Figure 3).
 |
Figure 2 Efficacy and survival in patients given the D-CTAG regimen. |
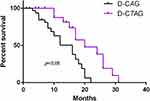 |
Figure 3 Survival curves of the D-CTAG and D-CAG regimens. |
Treatment-Related Toxicity
All recipients of D-CTAG or D-CAG were observed for thrombocytopenia and neutropenia. The proportion of those experiencing grade III–IV thrombocytopenia was significantly lower for D-CTAG (57.9%) than for D-CAG (88.4%; p < 0.05; Table 2). The D-CTAG group also exhibited earlier hematologic recovery than the D-CAG group. Median times to recovery of neutrophils were 9.6 days and 11.1 days, respectively (p < 0.05). In terms of platelet recovery, median times were 8.9 days and 13.2 days, respectively (p < 0.05), and the mean number of required platelet transfusions was significantly less for those receiving D-CTAG than D-CAG (3.2 ± 0.3 vs 9.9 ± 1.8; p < 0.05). Hence, it appears that rhTPO reduces hematologic toxicity.
 |
Table 2 Adverse Events During Induction Therapy |
Discussion
The CAG priming regimen launched by Saito et al in 2000 constituted the first use of a cellular growth factor (G-CSF) in AML treatment.6 The incorporation of G-CSF served to shorten neutropenic intervals, reduce the incidence of serious infection, and lower mortality. Leukemic cells were driven to enter the cell cycle, thus enhancing the efficacy of S-phase-specific cytotoxic drugs (eg, ara-C). Despite concerns that G-CSF might promote leukemic cell growth, many years of clinical practice have since proven that CAG-based chemotherapy is safe and effective for elderly patients with leukemia or relapsed refractory leukemia.10
This successful application of G-CSF in treating AML encouraged us to explore the addition of other hematopoietic growth factors. Once bound to its specific c-MPL receptor, TPO activates a series of downstream signaling pathways, such as Janus kinase/signal transducer and activator of transcription phosphatidylinositol-3-kinase/protein kinase B, and Ras/mitogen-activated protein kinase. Aside from megakaryocytic maturation and differentiation,11 TPO is implicated in various physiologic events, including mitigation of myocardial injury, nerve repair, angiogenesis, sex hormone secretion, and immune regulation;9 and it is generally acknowledged that rhTPO may alleviate chemotherapy-induced thrombocytopenia in patients with solid tumors. However, there has been no broad usage of rhTPO in AML chemotherapy, perhaps because c-MPL receptors are primarily limited to surfaces of hematopoietic stem cells, progenitor cells, megakaryocytes, and platelets.
Whether rhTPO is truly capable of AML propagation has always been in doubt. However, pertinent publications on c-MPL receptors have shown no evidence that leukemic disease is intensified. Chelvatheebam et al9 stratified patients with AML by age, gender, FAB type, karyotype, and long-term survival rate, finding no correlation with c-MPL receptor expression. In the context of AML, a positive correlation between c-MPL receptor expression and high-level expression of CD34+ has been documented by Ayala et al,12 but long-term and disease-free survival were both unrelated. Hence, c-MPL receptor expression in patients with AML has no clear bearing on prognosis.
In observing 108 patients with AML (no M3 or M7 variants), Archimbaud et al13 determined that leukemic stimulatory effect due to Pegylated recombinant human megakaryocyte growth and development factor (PEG-rHuMGDF), a c-MPL receptor agonist, was lacking. Schiffer et al14 also studied 57 patients with relapsed AML (no M3 or M7 variants), recording similar CR rates and survival times in patients given PEG-rHuMGDF or a placebo. These results indicate that PEG-rHuMGDF has no obvious in vivo proliferative effect on leukemic cells and provide an indirect theoretical basis for the use of rhTPO, another c-MPL receptor agonist.
With the emergence of molecularly targeted drugs, senile myeloid leukemia has also made some progress recently. Phase 1/2 studies of venetoclax combined with AZA, DEC, or LDAC in newly diagnosed patients unfit for intensive induction chemotherapy showed very respectable CR/CRi rates of 54–67%. Side effects were notable for significant myelosuppression, neutropenic fevers and other infections.15 In this study, the application of dual growth factors is of great significance for reducing the side effects of neutropenia and thrombocytopenia, enabling older adults to better tolerate the treatment and achieve a more favorable response.
We designed the D-CTAG regimen to assess rhTPO as an added treatment for AML in older adults. Ultimately, D-CTAG recipients achieved an ORR of 84.2%, showing a significantly higher CR rate (73.7%) than those given D-CAG only (60.5%; p < 0.05). The OS in the D-CTAG group (median, 20.2 months) also exceeded that in the D-CAG group (median, 14 months) and was far greater than the even lower OS (median, 10 months) reported by others after D-CAG therapy.16–19 The D-CTAG regimen should thus be quite effective in this patient demographic, proving non-inferior to D-CAG alone. Greater safety was afforded these elderly patients as well. Compared with our standard D-CAG protocol, D-CTAG therapy was less fraught with grade III–IV thrombocytopenia and brought earlier hematologic recovery, resulting in fewer required platelet transfusions. In the future, a prospective phase III randomized study is warranted to gauge the efficacy and safety of D-CTAG in a larger patient sample and validate this novel therapeutic modification for older adults with AML.
Conclusion
The curative effect of adding rhTPO was not inferior to that of D-CAG, and D-CTAG proved safer for older adults, especially in terms of hematologic toxicity. D-CTAG is a safe and effective treatment plan for older adults with AML.
Ethics Approval and Consent to Participate
The studies involving patients were approved by The Ethics Committee of Shengjing Hospital of China Medical University. The participants provided their written informed consent to participate in this study. This study was conducted in accordance with the Declaration of Helsinki.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This study was supported by the Natural Science Foundation of Liaoning Province (201800875).
Disclosure
The authors declare that they have no competing interests.
References
1. Webster JA, Pratz KW. Acute myeloid leukemia in the elderly: therapeutic options and choice. Leuk Lymphoma. 2018;59(2):274–287. doi:10.1080/10428194.2017.1330956
2. Podoltsev NA, Stahl M, Zeidan AM, Gore SD. Selecting initial treatment of acute myeloid leukaemia in older adults. Blood Rev. 2017;31(2):43–62. doi:10.1016/j.blre.2016.09.005
3. Klepin HD, Rao AV, Pardee TS. Acute myeloid leukemia and myelodysplastic syndromes in older adults. J Clin Oncol. 2014;32(24):2541–2552. doi:10.1200/JCO.2014.55.1564
4. Soura EN, Karikas GA. Acute myeloid leukaemia: recent data on prognostic gene mutations, in relation to stratified therapies for elderly patients. J BUON. 2019;24(4):1326–1339.
5. Klepin HD. Elderly acute myeloid leukemia: assessing risk. Curr Hematol Malig Rep. 2015;10(2):118–125. doi:10.1007/s11899-015-0257-2
6. Saito K, Nakamura Y, Aoyagi M, et al. Low-dose cytarabine and aclarubicin in combination with granulocyte colony-stimulating factor (CAG regimen) for previously treated patients with relapsed or primary resistant acute myelogenous leukemia (AML) and previously untreated elderly patients with AML, secondary AML, and refractory anemia with excess blasts in transformation. Int J Hematol. 2000;71(3):238–244.
7. Liu J, Jia JS, Gong LZ, et al. Efficacy and safety of decitabine in combination with G-CSF, low-dose cytarabine and aclarubicin in MDS-EB and AML-MRC. Zhonghua Xue Ye Xue Za Zhi. 2018;39(9):734–738.
8. Ebbo M, Riviere E, Godeau B. Adult immune thrombocytopenia and thrombopoietin receptor agonist: ten years later. Rev Med Interne. 2020;S0248-8663:30208.
9. Chelvatheebam S, Langabeer SE, Linch DC, Hills RK, Greenwell P. Incidence and prognostic significance of C-MPL expression in acute myeloid leukemia. Leuk Res. 2003;27(9):869–870. doi:10.1016/S0145-2126(03)00016-X
10. Qian SX, Li JY, Tian T, et al. Effect of low-dose cytarabine and aclarubicin in combination with granulocyte colony-stimulating factor priming (CAG regimen) on the outcome of elderly patients with acute myeloid leukemia. Leuk Res. 2007;31(10):1383–1388.
11. Kaushansky K. Thrombopoietin and its receptor in normal and neoplastic hematopoiesis. Thromb J. 2016;14(Suppl 1):40. doi:10.1186/s12959-016-0095-z
12. Ayala RM, Martinez-Lopez J, Albizua E, Diez A, Gilsanz F. Clinical significance of Gata-1, Gata-2, EKLF, and c-MPL expression in acute myeloid leukemia. Am J Hematol. 2009;84(2):79–86. doi:10.1002/ajh.21332
13. Archimbaud E, Ottmann OG, Yin JA, et al. A randomized, double-blind, placebo-controlled study with pegylated recombinant human megakaryocyte growth and development factor (PEG-rHuMGDF) as an adjunct to chemotherapy for adults with de novo acute myeloid leukemia. Blood. 1999;94(11):3694–3701. doi:10.1182/blood.V94.11.3694
14. Schiffer CA, Miller K, Larson RA, et al. A double-blind, placebo-controlled trial of pegylated recombinant human megakaryocyte growth and development factor as an adjunct to induction and consolidation therapy for patients with acute myeloid leukemia. Blood. 2000;9(8):2530–2535. doi:10.1182/blood.V95.8.2530
15. Perl AE. Which novel agents will have a clinically meaningful impact in AML at diagnosis? Best Pract Res Clin Haematol. 2021;34(1):101257. doi:10.1016/j.beha.2021.101257
16. Li J, Chen Y, Zhu Y, et al. Efficacy and safety of decitabine in combination with G-CSF, low-dose cytarabine and aclarubicin in newly diagnosed elderly patients with acute myeloid leukemia. Oncotarget. 2015;6(8):6448–6458. doi:10.18632/oncotarget.3361
17. Minakata D, Fujiwara S, Ito S, et al. A low-dose cytarabine, aclarubicin and granulocyte colony-stimulating factor priming regimen versus a daunorubicin plus cytarabine regimen as induction therapy for older patients with acute myeloid leukemia: a propensity score analysis. Leuk Res. 2016;42:82–87. doi:10.1016/j.leukres.2015.12.013
18. Suzushima H, Wada N, Yamasaki H, et al. Low-dose cytarabine and aclarubicin in combination with granulocyte colony-stimulating factor for elderly patients with previously untreated acute myeloid leukemia. Leuk Res. 2010;34(5):610–614. doi:10.1016/j.leukres.2009.08.010
19. Hirayama Y, Sakamaki S, Takayanagi N, et al. [Chemotherapy with ubenimex corresponding to patient age and organ disorder for 18 cases of acute myelogeneous leukemia in elderly patients–effects, complications and long-term survival]. Gan To Kagaku Ryoho. 2003;30(8):1113–1118. Japanese
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
