Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Drug Attitude, Insight, and Patient’s Knowledge About Prescribed Antipsychotics in Schizophrenia: A Cross-Sectional Survey
Authors Nagai N , Tani H, Yoshida K , Gerretsen P, Suzuki T , Ikai-Tani S , Mimura M , Uchida H
Received 28 November 2019
Accepted for publication 26 February 2020
Published 23 March 2020 Volume 2020:16 Pages 781—787
DOI https://doi.org/10.2147/NDT.S240377
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Taro Kishi
Nobuhiro Nagai, 1–3 Hideaki Tani, 1, 4 Kazunari Yoshida, 1, 5 Philip Gerretsen, 6, 7 Takefumi Suzuki, 8 Saeko Ikai-Tani, 1, 9 Masaru Mimura, 1 Hiroyuki Uchida 1
1Department of Neuropsychiatry, Keio University School of Medicine, Tokyo, Japan; 2Department of Psychiatry, Minami-Hanno Hospital, Saitama, Japan; 3Department of Psychiatry, Tokyo-Kaido Hospital, Tokyo, Japan; 4Kimel Family Translational Imaging-Genetics Laboratory, Centre for Addiction and Mental Health, Toronto, ON, Canada; 5Pharmacogenetics Research Clinic, Campbell Family Mental Health Research Institute, Centre for Addiction and Mental Health, Toronto, ON, Canada; 6Multimodal Imaging Group - Research Imaging Centre, Campbell Family Mental Health Research Institute, Centre for Addiction and Mental Health, Toronto, ON, Canada; 7Geriatric Psychiatry Division, Centre for Addiction and Mental Health, Toronto, ON, Canada; 8Department of Neuropsychiatry, University of Yamanashi Faculty of Medicine, Yamanashi, Japan; 9Physical Activity and Mental Health, Faculty of Kinesiology & Physical Education, University of Toronto, Toronto, ON, Canada
Correspondence: Hiroyuki Uchida
Department of Neuropsychiatry, Keio University School of Medicine, 35, Shinanomachi, Shinjuku-Ku, Tokyo 160-8582, Japan
Tel +81353633829
Fax +81353790187
Email [email protected]
Introduction: While patients’ perspectives toward pharmacotherapy are expected to be directly influenced by their motivation and understanding of the treatment that they are currently receiving, no study has comprehensively investigated the impact of insight into illness and knowledge for the ongoing pharmacotherapy on the attitude towards drug treatment among patients with schizophrenia.
Materials and Methods: One hundred forty-eight Japanese outpatients diagnosed with schizophrenia, according to the International Classification of Diseases 10th edition, were included (mean±SD age, 47.3± 12.4 years; 90 men (60.8%)). Attitudes toward antipsychotic treatment and insight into illness were assessed with the Drug Attitude Inventory-10 (DAI-10) and the VAGUS, respectively. In addition, a multiple-choice questionnaire that was designed to examine patients’ knowledge about therapeutic effects, types, and implicated neurotransmitters of antipsychotic drugs they were receiving was utilized.
Results: The mean±SD of DAI-10 score was 4.7± 4.2. The multiple regression analysis found that lower Positive and Negative Syndrome Scale (PANSS) scores, higher VAGUS scores, and longer illness duration were significantly associated with higher DAI-10 scores (β=− 0.226, P=0.009; β=0.250, P=0.008; β=0.203, P=0.034, respectively). There was a significant difference in the DAI-10 scores between the subjects who gave more accurate answers regarding the effects of their primary antipsychotic and those who did not (mean±SD, 5.57± 4.38 vs 4.13± 4.04, P=0.043); however, this finding failed to survive the multiple regression analysis.
Conclusion: Better insight into illness and treatment, lower illness severity, longer illness duration, and possibly greater knowledge about the therapeutic effects of medications may lead to better attitudes towards pharmacotherapy among patients with schizophrenia, which has an important implication for this typically chronic mental condition requiring long-term antipsychotic treatment to sustain stability.
Keywords: drug attitude, adherence, insight, knowledge, schizophrenia, antipsychotic
Introduction
Schizophrenia is typically a life-long chronic illness, often requiring long-term antipsychotic treatment to avert negative consequences such as relapse and hospitalization.1–3 However, adherence to antipsychotic treatment has been reported to be notoriously low. For example, the proportion of patients who are not sufficiently adherent to antipsychotic treatment declined approximately by half within 1 year and 75% within 2 years after discharge.4 Such suboptimal medication adherence is of serious concern since it unequivocally increases the risk of unwanted outcomes.5,6
Patient’s perspective on psychopharmacology has consistently been reported to be related to actual treatment adherence in the management of schizophrenia.7–12 These findings clearly emphasize the need to enhance the patient’s drug attitudes among patients with schizophrenia and improve our understanding as to which factors influence such attitudes. While patient’s attitude toward psychopharmacology is expected to be directly influenced by motivation and understanding of the treatment that the patient is currently receiving, no study has comprehensively investigated the impact of patient’s insight into illness and their knowledge for ongoing pharmacotherapy on their attitudes towards drug treatment among patients with schizophrenia.
This is critically important in light of generally poor knowledge about pharmacotherapy on the side of patients as we have reported elsewhere.13 The percentages of subjects with schizophrenia who chose correct answers regarding effects, types, and implicated neurotransmitters were as low as 30.9%, 30.9% and 7.4%, respectively. To fill in the gap in the literature, we set out to conduct a cross-sectional study to further evaluate these issues among Japanese outpatients with schizophrenia. In this study, we aimed to test the hypothesis of the positive correlation between the attitude towards prescribed medications and patients’ knowledge of their medications or insight into illness.
Materials and Methods
Subjects
We attempted to enroll a wide variety of patients to reflect the real-world clinical settings and made the inclusion criteria as broad as possible. Therefore, we included outpatients with a diagnosis of schizophrenia or schizoaffective disorder according to the International Classification of Diseases, 10th edition (ICD-10) (World Health Organization, 1992) who were 18 years of age or older and capable of providing informed consent. This study was conducted at the following eight sites in Tokyo, Saitama, and Chiba, Japan: Asakadai Mental Clinic, Ohizumi Hospital, Ohizumi Mental Clinic, Kinosaki Hospital, Minami-Hanno Hospital, National Hospital Organization Shimofusa Psychiatric Medical Center, Tokyo-Kaido Hospital, and Umesato Mental Clinic. The study was approved by the institutional review board at each site and conducted in accordance with the Declaration of Helsinki. Prior to study entry, all subjects provided written informed consent after receiving detailed information about the protocol. The patients were explained about the aim of this study in the informed consent form as follows:
The main goal of this study is to explore the knowledge of prescribed medications and illnesses among people who are receiving psychiatric treatment. Through this research we wish to find the importance of the illness knowledge as well as the patients’ medication knowledge in the treatment.
Cross-Sectional Assessments
The primary measure was the Japanese version of the Drug Attitude Inventory-10 (DAI-10).14,15 A total score ranges from −10 to 10, with a higher score indicating a greater degree of positive attitudes toward psychiatric medications. We set out the DAI-10 as a primary outcome because it is a simple and good psychometric property as well as it is used as a proxy of the medication adherence in previous studies.7–12,16 The following assessments were also performed: the Positive and Negative Syndrome Scale (PANSS),17 Global Assessment of Functioning (GAF),18 and VAGUS for insight into illness.19 The VAGUS measures four core dimensions of clinical insight into psychosis, including general illness awareness, symptom attribution, awareness of need for treatment, and awareness of negative consequences attributable to the illness. We translated the VAGUS into Japanese and carried out a back-translation to ensure consistency in its meaning. The VAGUS has clinician-rated and self-report scales. The latter version was used for this study. A total score in the VAGUS ranges from 0 to 10, with a higher score indicating a greater insight into illness. Because some of the subjects were unaware of their diagnosis, the VAGUS was only performed by those patients who were already informed on their diagnosis by their psychiatrists.
A multiple-choice questionnaire that was specifically designed to examine patients’ knowledge about therapeutic effects, types, and implicated neurotransmitters of their primary antipsychotic drug, was also used. This questionnaire has been described in detail elsewhere.13 In cases where the subjects were concomitantly receiving two or more antipsychotics, they were asked to respond on a basis of the primary antipsychotic drug that accounted for the greatest chlorpromazine equivalent relative dose (CPZE).20 In the questionnaire, the participants were asked to choose from one of the following eight options regarding three categories: (1) effects of the drug: (i) delusions and/or hallucinations, (ii) depression, (iii) mania, (iv) anxiety, (v) insomnia, (vi) relieving adverse effect, (vii) others, and (viii) I do not know; (2) types of the drug: (i) antidepressant, (ii) antipsychotic, (iii) drug for side effects, (iv) hypnotic, (v) anxiolytic, (vi) mood stabilizer, (vii) others, and (viii) I do not know; and (3) neurotransmitters that they thought were related to the drug’s mechanism of action: (i) benzodiazepine, (ii) serotonin, (iii) dopamine, (iv) noradrenalin, (v) histamine, (vi) acetylcholine, (vii) others, and (viii) I do not know.13 Those who chose (viii) in any instances were classified as those who did not answer correctly. Since target symptoms varied among the subjects, correct responses regarding the effects of the drug were determined jointly by the subject’s psychiatrist and one of the investigators (N.N.). The following information was also collected age, sex, ethnicity, educational background, current medications, and duration of illness. Daily doses of antipsychotics were converted to CPZE.20
Statistical Analyses
The Shapiro–Wilk test was used to examine the normality of the continuous variables. Correlation analysis was conducted between the DAI-10 score and VAGUS score using the Spearman correlation coefficient because DAI-10 scores did not show a normal distribution (P<0.001). Secondly, the independent t-test was applied to compare the average DAI-10 scores between two groups. Thirdly, multiple linear regression analysis (forced entry model) was conducted to examine the association between the DAI-10 total score (dependent variable) and the following independent variables: gender, age (ie, <50 years or ≥50), duration of illness (ie, <20 years or ≥20), years of education (ie, <13 years or ≥13), number of prescribed psychotropics (ie, 1 or ≥2), PANSS total score, VAGUS score, and correct or incorrect response about effects, types, and implicated neurotransmitters of their primary antipsychotic drugs. The cut-offs for age (50 years) and duration of illness (20 years) were set according to their rounded mean values. Another multiple linear regression analysis (forced entry model) was also performed to examine the association between the DAI-10 total score (dependent variable) and the four core dimension scores of the VAGUS. SPSS Statistics Version 25 (IBM Corp., Armonk, NY) was used for statistical analyses. A P-value of <0.05 was considered statistically significant (two-tailed).
Results
Study Sample
This study was conducted between September 2015 and June 2018. A total of 159 outpatients agreed to participate in this study. Of these, 148 subjects (93.1%) completed the questionnaire, and the data from these completers were used for the following analysis. One hundred twenty-six (85.1%) and 22 (14.9%) subjects were recruited from psychiatric hospitals and psychiatric clinics, respectively. Table 1 summarizes the demographic and clinical characteristics of the subjects. The most frequently used primary antipsychotic drug was olanzapine (n=48, 32.4%), followed by risperidone (n=40, 27.0%) and aripiprazole (n=33, 22.3%). Eighty-two subjects (55.4%) were treated with antipsychotic monotherapy. Mean±SD of DAI-10 score was 4.7±4.2, indicating a relatively positive attitude toward medications.
 |
Table 1 Demographic and Clinical Characteristics of Subjects (N=148) |
DAI-10 and VAGUS Score
A significant correlation was observed between the DAI-10 score and the VAGUS score (r=0.234, P=0.005).
DAI-10 and Knowledge About Medications
Fifty-six (37.8%), 48 (32.4%), and 14 (9.5%) subjects gave right answers about effects, types, and implicated neurotransmitters of the primary antipsychotic drug, respectively. Among 148 subjects, 22.3% (n=33) correctly answered for both effects and types, and only 7.4% (n=11) correctly responded to all three questions.
There was a significant difference in the DAI-10 scores between the subjects who gave correct answers regarding the effects of the primary antipsychotic and those who did not (mean±SD, 5.57±4.38 vs 4.13±4.04, P=0.043). On the other hand, no significant differences were found in the DAI-10 scores between the subjects who chose correct answers about types or related neurotransmitters and those who did not (mean±SD, 5.25±3.99 vs 4.40±4.31, P=0.25; and 5.29±5.18 vs 4.61±4.12, P=0.57, respectively).
Factors Associated with DAI-10
A lower PANSS score, higher VAGUS score, and the longer duration of illness were significantly associated with a higher DAI-10 score (β=−0.226, P=0.009; β=0.250, P=0.008; β=0.203, P=0.034, respectively) (Table 2). Other clinical variables including education level failed to show any statistically significant association with the DAI-10 score. “Awareness of need for treatment” score in the VAGUS was significantly correlated with the DAI-10 score in a positive manner (β=0.552, P=0.001) (Table 3).
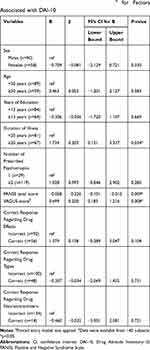 |
Table 2 Multiple Linear Regression Analysisa for Factors Associated with DAI-10 |
 |
Table 3 Multiple Linear Regression Analysisa for Four Dimensions in VAGUS Associated with DAI-10 (n=140b) |
With regard to the comparisons between the DAI-10 scores and the demographic variables, the average DAI-10 score of patients receiving CPZE of 1000 or more mg/day was higher than that of patients receiving lower than 1000 mg/day: 6.1 (n=29) and 4.3 (n=119) (P=0.037). A Spearman’s rank correlation coefficient found a significant correlation between CPZE and the duration of illness (r=0.205, P=0.012). Therefore, this difference may be explained by the duration of illness.
Average DAI-10 scores were 4.9 in 96 patients who took benzodiazepine and 4.3 in 52 patients who did (P=0.486).
Discussion
The present study showed favorable insight into illness and lower illness severity was associated with better attitudes towards pharmacotherapy among patients with schizophrenia. Moreover, longer illness duration was also related to better attitudes on drug treatment. A significant difference was found in the DAI-10 scores between the subjects who gave correct answers regarding the effects of the primary antipsychotic in use and those who did not; however, this finding was not replicated in the multiple regression analysis. While drug attitudes as assessed with the DAI-10 was relatively favorable overall, a large SD indicated interindividual differences. The percentages of participants with the right answers on effects, types, and implicated neurotransmitters were not high.
While there was a positive association between the DAI-10 scores and the VAGUS total scores as well as the dimension score of “awareness of need for treatment”, there was no association between the other dimension scores of “general illness awareness”, “symptom attribution” and “awareness of negative consequences”. Previous literatures have been consistent in showing that the DAI-10 score can be considered to be a proxy for medication adherence.7–12 Moreover, it is unequivocal that good adherence to treatment reduces risks of relapse and associated negative outcomes in the management of schizophrenia.1–6 Therefore, our finding emphasizes the need for interventions to improve insight into illness as well as treatment so as to enhance their commitment to pharmacotherapy, which would consequently lead to better treatment outcomes.
A significant difference in the DAI-10 scores between the subjects who gave correct answers regarding the effects of their prescribed primary antipsychotics and those who did not was found when the independent t-test was used. On the other hand, multiple linear regression analysis failed to confirm this correlation, which requires a conservative interpretation.
Furthermore, neither independent t-test nor multiple linear regression analysis found any significant association in the DAI-10 scores between correct and incorrect responses about types or related neurotransmitters. These preliminary findings might suggest a possibility that detailed knowledge about medications may not always be necessary in terms of improving their attitudes towards pharmacotherapy; it might be sufficient to understand the therapeutic effects of their medications, which directly speaks to the needs of the patients.
This study replicated the low percentages of the subjects who gave the correct answers to the effects of their prescribed medications. Given that previous studies have reported a wide range of percentages (10–80%) of subjects who gave correct answers regarding the effects of the drugs,21,22 there is no consensus on the optimal percentage of correct answers. However, physicians need to be cautious to that their patients may have insufficient knowledge on the prescribed medications.
There are several limitations to the present study. First, due to the nature of a cross-sectional study design, the associations that we found in the present study do not necessarily indicate causality. Actual treatment outcomes and direct adherence data were not evaluated in this study. Some of the assessments were subjectively completed, leaving a room for desirability bias. Moreover, our questionnaire on antipsychotic properties was arbitrary and quite a few participants were on psychotropic polypharmacy. Thus, prospective studies that examine the effects of psychoeducation, or the change of prescribed medication, on patients’ knowledge, illness insight, illness severity, and adherence as well as how they will affect the treatment outcomes in this population are clearly warranted to replicate the preliminary findings of this study. Second, the subjects included in this study were Japanese outpatients with mild symptomatology, which may limit the generalizability of the data to other populations. Third, we were not able to classify respondents by other potential confounders such as family input or history of psychoeducation because we did not collect them in this study. Fourth, we did not calculate a target sample size as this was a preliminary study and there were no previous reports that matched the scope of this study. Lastly, the sample size was relatively small, which may have resulted in type-II errors.
Conclusion
Better insight into illness and treatment, lower illness severity, longer illness duration, and possibly greater knowledge about therapeutic effects of medications appear to be associated with more favorable attitudes towards pharmacotherapy among patients with schizophrenia. Although this preliminary contention should be confirmed in prospective studies, psychoeducation specifically targeting the insight into illness and treatment may enhance commitment to antipsychotic treatment and therefore reduce the risk of relapse in patients with schizophrenia.
Abbreviations
DAI-10, Drug Attitude Inventory-10; GAF, Global Assessment of Functioning; PANSS, Positive and Negative Syndrome Scale.
Acknowledgments
The authors thank Ms. Gounaridis for her administrative support.
Funding
This research was funded by the Inokashira Hospital Grant for Psychiatry Research.
Disclosure
Dr Nagai has received personal/manuscript fees from Sumitomo Dainippon Pharma within the past 3 years. Dr Tani has received grants from Eli Lilly and the Japanese Society of Clinical Neuropsychopharmacology; manuscript or personal/speaker’s fees from Otsuka, Sumitomo Dainippon Pharma, Wiley, and Yoshitomi Yakuhin, outside the submitted work. Dr Yoshida has received manuscript/personal fees from Sumitomo Dainippon Pharma, fellowship grants from the Japan Research Foundation for Clinical Pharmacology, Azrieli Centre Postdoctoral Fellowship at CAMH, and personal/consultant fees from Signant Health and VeraSci within the past 3 years. Dr Gerretsen has received research supports/grants from the Canadian Institutes of Health Research (CIHR), Ontario Mental Health Foundation, and Centre for Addiction and Mental Health Foundation. He has also received Academic Scholar Award from the Department of Psychiatry, University of Toronto, grants from Alternative Funding Program Innovation Fund, Ministry of Health and Long-term Care, outside the submitted work; In addition, Dr Gerretsen reports a patent VAGUS Scale. Dr Suzuki has received manuscript or personal/speaker’s fees from Astellas, Dainippon Sumitomo Pharma, Eisai, Eli Lilly, Elsevier Japan, Janssen Pharmaceuticals, Kyowas Yakuhin, Meiji Seika Pharma, Mitsubishi Tanabe Pharma, MSD, Novartis, Otsuka Pharmaceutical, Shionogi Tsumura, Wiley Japan, and Yoshitomi Yakuhin within the past 3 years; grants from Eisai, Mochida Pharmaceutical, and Meiji Seika Pharma, outside the submitted work. Dr Ikai-Tani has received a manuscript/personal fees from Sumitomo Dainippon Pharma for the past 3 years. Dr Uchida has received grants and/or personal fees from Astellas Pharmaceutical, Eisai, Otsuka Pharmaceutical, Shionogi, Dainippon-Sumitomo Pharma, Eli Lilly, Mochida Pharmaceutical, Meiji-Seika Pharmaceutical, and Novartis; speaker’s honoraria from Otsuka Pharmaceutical, Eli Lilly, Shionogi, Pfizer, Yoshitomi Yakuhin, Dainippon-Sumitomo Pharma, Meiji-Seika Pharma, MSD, and Janssen Pharmaceutical; and advisory panel payments from Dainippon-Sumitomo Pharma within the past 3 years. Dr Mimura has received speaker’s honoraria and/or grants/personal fees from Daiichi Sankyo, Dainippon-Sumitomo Pharma, Eisai, Eli Lilly, Fuji Film RI Pharma, Janssen Pharmaceutical, Mochida Pharmaceutical, MSD, Nippon Chemipher, Novartis Pharma, Ono Yakuhin, Otsuka Pharmaceutical, Pfizer, Shionogi, Takeda Yakuhin, Tanabe Mitsubishi, Tsumura, and Yoshitomi Yakuhin within the past 3 years. The authors report no other conflicts of interest in this work.
References
1. Leucht S, Barnes TR, Kissling W, et al. Relapse prevention in schizophrenia with new-generation antipsychotics: a systematic review and exploratory meta-analysis of randomized, controlled trials. Am J Psychiatry. 2003;160(7):1209–1222. doi:10.1176/appi.ajp.160.7.1209
2. Uchida H, Suzuki T, Takeuchi H, et al. Low dose vs standard dose of antipsychotics for relapse prevention in schizophrenia: meta-analysis. Schizophr Bull. 2011;37(4):788–799. doi:10.1093/schbul/sbp149
3. Suzuki T, Uchida H, Takeuchi H, et al. A review on schizophrenia and relapse–a quest for user-friendly psychopharmacotherapy. Hum Psychopharmacol. 2014;29(5):414–426. doi:10.1002/hup.2421
4. Keith SJ, Kane JM. Partial compliance and patient consequences in schizophrenia: our patients can do better. J Clin Psychiatry. 2003;64(11):1308–1315. doi:10.4088/JCP.v64n1105
5. Thieda P, Beard S, Richter A, et al. An economic review of compliance with medication therapy in the treatment of schizophrenia. Psychiatr Serv. 2003;54(4):508–516. doi:10.1176/appi.ps.54.4.508
6. Gilmer TP, Dolder CR, Lacro JP, et al. Adherence to treatment with antipsychotic medication and health care costs among Medicaid beneficiaries with schizophrenia. Am J Psychiatry. 2004;161(4):692–699. doi:10.1176/appi.ajp.161.4.692
7. Brain C, Allerby K, Sameby B, et al. Drug attitude and other predictors of medication adherence in schizophrenia: 12 months of electronic monitoring (MEMS(R)) in the Swedish COAST-study. Eur Neuropsychopharmacol. 2013;23(12):1754–1762. doi:10.1016/j.euroneuro.2013.09.001
8. Yang J, Ko YH, Paik JW, et al. Symptom severity and attitudes toward medication: impacts on adherence in outpatients with schizophrenia. Schizophr Res. 2012;134(2–3):226–231. doi:10.1016/j.schres.2011.11.008
9. Lee Y, Lee MS, Jeong HG, et al. Medication adherence using electronic monitoring in severe psychiatric illness: 4 and 24 weeks after discharge. Clin Psychopharmacol Neurosci. 2019;17(2):288–296. doi:10.9758/cpn.2019.17.2.288
10. Yilmaz E, Okanli A. The effect of internalized stigma on the adherence to treatment in patients with schizophrenia. Arch Psychiatr Nurs. 2015;29(5):297–301. doi:10.1016/j.apnu.2015.05.006
11. Meier J, Becker T, Patel A, et al. Effect of medication-related factors on adherence in people with schizophrenia: a European multi-centre study. Epidemiol Psichiatr Soc. 2010;19(3):251–259. doi:10.1017/S1121189X00001184
12. Chandra IS, Kumar KL, Reddy MP, et al. Attitudes toward medication and reasons for non-compliance in patients with schizophrenia. Indian J Psychol Med. 2014;36(3):294–298. doi:10.4103/0253-7176.135383
13. Nagai N, Tani H, Suzuki T, et al. Patients’ knowledge about prescribed antipsychotics and medication adherence in schizophrenia: a cross-sectional survey. Pharmacopsychiatry. 2017;50(6):264–269. doi:10.1055/s-0043-113828
14. Hogan TP, Awad AG, Eastwood R. A self-report scale predictive of drug compliance in schizophrenics: reliability and discriminative validity. Psychol Med. 1983;13(1):177–183. doi:10.1017/S0033291700050182
15. Miyata R. Drug compliance in schizophrenia. Jpn J Clin Psychiatry. 1999;28:265–275. Japanese.
16. Nielsen RE, Lindstrom E, Nielsen J, et al. DAI-10 is as good as DAI-30 in schizophrenia. Eur Neuropsychopharmacol. 2012;22(10):747–750. doi:10.1016/j.euroneuro.2012.02.008
17. Kay SR, Fiszbein A, Opler LA. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13(2):261–276. doi:10.1093/schbul/13.2.261
18. Endicott J, Spitzer RL, Fleiss JL, et al. The global assessment scale. A procedure for measuring overall severity of psychiatric disturbance. Arch Gen Psychiatry. 1976;33(6):766–771. doi:10.1001/archpsyc.1976.01770060086012
19. Gerretsen P, Remington G, Borlido C, et al. The VAGUS insight into psychosis scale–self-report and clinician-rated versions. Psychiatry Res. 2014;220(3):1084–1089. doi:10.1016/j.psychres.2014.08.005
20. Inada T, Inagaki A. Psychotropic dose equivalence in Japan. Psychiatry Clin Neurosci. 2015;69(8):440–447. doi:10.1111/pcn.2015.69.issue-8
21. Macpherson R, Double DB, Rowlands RP, et al. Long-term psychiatric patients’ understanding of neuroleptic medication. Hosp Community Psychiatry. 1993;44:71–73. doi:10.1176/ps.44.1.71
22. Tempier R. Long-term psychiatric patients’ knowledge about their medication. Psychiatr Serv. 1996;47:1385–1387.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
