Back to Journals » OncoTargets and Therapy » Volume 13
Down-Regulation of ZEB1 by miR-199a-3p Overexpression Restrains Tumor Stem-Like Properties and Mitochondrial Function of Non-Small Cell Lung Cancer
Received 2 January 2020
Accepted for publication 13 April 2020
Published 22 May 2020 Volume 2020:13 Pages 4607—4616
DOI https://doi.org/10.2147/OTT.S244525
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Sanjay Singh
Juan Bai,1 Wen-Yu Jiao2
1Department of Oncology, Affiliated Hospital of Chengdu University, Chengdu 610081, People’s Republic of China; 2Department of Respiratory and Critical Care Medicine, Xi’an Daxing Hospital, Xi’an 710016, People’s Republic of China
Correspondence: Wen-Yu Jiao
Email [email protected]
Objective: MicroRNA-199a-3p (miR-199a-3p or miR-199b-3p) targeting of 3ʹ-UTR of ZEB1 was characterized as an important way to inhibit invasion and metastases in non-small cell lung cancer (NSCLC), one of the most common cancers around the world. Here we aimed to investigate the tumor-suppressive role of miR-199a-3p targeted ZEB1.
Materials and Methods: A549 cells were transfected with ZEB1 and/or miR-199a-3p. Then, tumor growth was investigated in xenograft mice. Stem-like property, proliferation and mitochondria injury were further validated in vitro.
Results: Overexpression of miR-199a-3p with premiRNAs significantly reduced tumor growth inhibited CD44 and Ki67 and increased Caspase-3 in A549 xenograft mice. Sphere formation and protein expression of stem-like markers showed that miR-199a-3p inhibited stemness of A549 cell. miR-199a-3p reduced proliferation of A549 cells, as showed with EdU staining and reduced expression of Ki67. Transfection of miR-199a-3p also promoted apoptosis, as indicated with increased apoptotic cells with flow cytometry, and increased cleaved Caspase-3/Caspase3 and Bcl-2/Bax. Apoptosis was further validated to be induced with mitochondria dysfunction, which indicated with JC-1 labeled loss of mitochondrial membrane potential, reduced activity of SOD, and increased MDA and LDH. All these effects were inverted with overexpression of ZEB1.
Conclusion: Altogether, the findings suggested that the up-regulation of miR-199a-3p significantly inhibited NSCLC growth in vivo, and reduced A549 cell proliferation and promoted mitochondrial-mediated apoptosis, through down-regulation of ZEB1. The findings supported ZEB1 down-expression with miR-199a-3p as a novel therapeutic target for NSCLC treatment.
Keywords: lung cancer, miR-199a-3p, miR-199b-3p, ZEB1
Introduction
Non-small cell lung cancer (NSCLC) is one of the most common tumors around the world, predicted representing more than 1.6 million incidences and 1.4 million deaths in 2018.1,2 Individualized management based on the molecular characteristics of the patients and the specific cancer subtypes brings about new advances in lung cancer treatment. However, surgery, chemotherapy, and radiation therapy at early stages are not enough for an effective management.3 Metastases and drug resistance remain the most fatal events.
Zinc Finger E-box Binding Homeobox 1 (ZEB1), an E-box transcriptional repressor, has been suggested to induce epithelial–mesenchymal transition (EMT) and enable metastases. ZEB1 was indicated to be the pivotal inducer of metastases in a series of tumors including lung cancer.4,5 Overexpressed ZEB1 dramatically promoted the invasive and migratory lung cancer cells and increased the number and sites of metastases in vivo.6–8 ZEB1 was regulated by a series of regulatory RNA, including circular RNA, long non-coding RNA and microRNA.9–11 To a great extent, ZEB1 overexpression was positively related with severity of lung cancer. ZEB1 overexpression also indicated a poor prognosis. ZEB1 was elevated in lung cancer tissues as well as most lung cancer cell lines, especially A549 cells.12 ZEB1 expression was strictly controlled. There are lots of reports that ZEB1 promoted EMT or metastases, and ZEB1 also affects fundamental intracellular decision-making processes, including stemness, differentiation, proliferation, senescence, survival, apoptosis, immune response and drug resistance.13 Obviously, ZEB1 has a pivotal role in cell fate determination in many aspects of NSCLC.
Another interesting regulator in NSCLC is miR-199a-3p (or miR-199b-3p). miR199a-3p and miR-199b-3p are equivalent sequences of 22 nucleotides. Serum microRNAs including miR-199a-3p and other 4 microRNAs improved the diagnostic accuracy in lung cancer presenting with pulmonary nodules with a creditable specificity, indicating miR-199a-3p as an important player in lung cancer.14 Indeed, miR-199a-3p was proved to regulate Axl, a receptor that induces proliferation, migration and invasion of cancer.15,16 And most importantly, miR-199a-3p was predicted to silence ZEB1 mRNA, and miR-199a-3p down-regulated ZEB1 in NSCLC to inhibit cell proliferation, migration and invasion.17 On the other hand, the role of miR-199a-3p and ZEB1 is possibly varied considering the functional diversity of ZEB1. For example, miR-199a-3p targeted ZEB1 to induce toxicity of Gambogic acid to melanoma cells.18 Interaction of miR-199a-3p and ZEB1 was also indicated to be involved in diabetic nephropathy and development of human fetal lung.19,20 The target relation of miR-199a-3p and ZEB1 was evident. However, other possible roles of miR-199a-3p and ZEB1 like stemness, apoptosis and drug resistance regulation still need to be studied.
In this paper, we investigated the role of miR-199a-3p and ZEB1 in regulating proliferation, stemness and apoptosis of NSCLC in vivo and in vitro. A549 cells were used; pcDNA-ZEB1 and premiR-199a-39p were transfected to investigate their effects.
Materials and Methods
Cell Culture
Lung cancer cell line A549 was purchased from the American Type Culture Collection (ATCC; Manassas, VA, USA). Cells were cultured in RPMI-1640 medium (HyClone Company, Logan, UT, USA) with 10% FBS (ThermoFisher Scientific, Waltham, MA, USA) and 1% Penicillin-Streptomycin solution (ThermoFisher Scientific, Waltham, MA, USA) in 37°C with 5% CO2, digested with 0.25% trypsin (Beyotime Biotechnology, Shanghai, China), stored at −80°C in FBS with 10% DMSO (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany).
Cell Transfection
The premiR-199a-3p and control vector used in the experiment were designed and synthesized by Landm Biotech (Guangzhou, China). The pcDNA-ZEB1 constructed by plasmids was constructed by cloning the sequence of ZEB1 into pcDNA-3.1 vector from Invitrogen (Carlsbad, CA, USA). The A549 cells were seeded in a 6 well plate at a density of 1×106 cells/mL each well. After incubation for 24 h, 50nM of pcDNA-ZEB1, pcDNA-Control, premiR-199a-3p or microRNA control were transfected into each well using Lipofectamine 3000 (Invitrogen, Carlsbad, CA, USA) according to the instructions, respectively, or in combination. The scrambled control miRNA and empty vector (named vector and pcDNA) were used as control.
Xenograft Experiments
The animal experiments were approved by the Ethics Committee of Affiliated Hospital of Chengdu University. All animal experiments were carried out in strict accordance with the Guidelines for Animal Use in the National Institutes of Health. Six-week-old male BALB/c null nude mice (bought from Guangdong Medical Laboratory Animal Center, Guangdong, China) were used for the xenograft experiment. About 1 × 106 vector or premiR-199a-3p transfected A549 cells with matrigel (BD Biosciences, San Jose, CA, USA) at 1:1 dilution were injected subcutaneously into the right flank region of mice (6 mice × 2 groups). Thirty days later, the nude mice were anesthetized and sacrificed, and the tumors were removed for measurement and photographing.
Quantitative Real-Time Polymerase Chain Reaction
After washed with cold PBS (Solarbio, Beijing, China), cultured cells were directly provided and tissues were homogenized so that 1 mL of Trizol (TransGen Biotech, Beijing, China) were added to each sample to collect raw RNA. The collected solution was purified with 200 μL of 1-bromo-3-chloropropane, 500 μL of Isopropanol and 70% ethanol (Beyotime Biotechnology, Shanghai, China). DNase/RNase-free water was used to dissolve the purified RNA pellets and the RNA concentration was tested. Then, first-strand cDNA was synthesized using a cDNA synthesis kit (TaKaRa, Dalian, China) according to manufacturer’s instructions. All primers were synthesized by Ribobio (Guangzhou, China). Primers used were:
GAPDH: 5ʹ-GAGT CAAC GGAT TTGG TCGT-3ʹ and 5ʹ-TTGA TTTT GGAG GGAT CTCG-3`; snRNA U6: 5ʹ-CTCG CTTC GGCA GCAC A-3ʹ and 5ʹ-AACG CTTC ACGA ATTTG CGT-3ʹ; miR-199a-3p: 5ʹ-GGAG GTTT GTTT GTAA GCAT GAA-3` and 5ʹ-AATG AAGA GAGC ACCT ATGA CAA-3ʹ; ZEB1, 5ʹ-GTAC GAAT TCFG CCAA TAAG CAAA CGA-3ʹ, 5ʹ-GTCA GAGC TCAA GGTC TTCA TCAT GTG-3ʹ.
The synthesized cDNA (2 μL), 10× PCR Buffer (with Mg2+ 2 μL), dNTP (2.5 mM each, 1.6 μL), primer mix (10 μM each, 1.6 μL), Taq DNA Polymerase (5U/μL, 0.1 μL) and pure water (12.7 μL) (Beyotime Biotechnology, Shanghai, China) with 10 μL SYBR Green qPCR Master Mix (Bio-Rad, Hercules, California) were mixed and incubated at 94°C for 3 min. Then, 30 cycles of incubation were applied as 94°C for 30 sec, 55°C for 30 sec, and 72°C 60 sec on an Applied Biosystems 7500 Fast Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). At the end of the cycle, keep the mixture in 72°C for extra 10 min. The relative expression of specific genes was semi-quantified using the relative Cq to U6 or GAPDH.
Immunohistochemical Analysis
Immunohistochemical staining (IHC) was performed (Dako Envision plus System, Dako, Carpinteria, CA, USA) according to the manufacturer’s instructions. Briefly, properly diced tumor tissues were fixed in 4% paraformaldehyde overnight and then embedded in paraffin wax. 5 μm thick sections were sliced and wax was washed out and rehydrated with a series of xylene and ethanol solutions. Antigen retrieval was performed by heat mediation in citrate buffer. Samples were blocked with 10% goat serum before incubation with primary antibody. Then, CD44, Ki67 and Caspase-3 primary antibodies were incubated overnight before secondary antibody was incubated for 30 min. The staining positive cells were evaluated by digital image analyzed with Image J software.
Western Blotting
A549 cells or tissues were lysed and collected, then balanced with PBS according to Bradford assay. About 40 μg of proteins were used in SDS-PAGE for each sample. Primary and secondary antibodies were incubated according to the manufacturers’ protocols. The proteins of interest were visualized by enhanced chemiluminescence reagents with ChemiDoc XRS. Anti-CD44 (ab157107), anti-Ki67 (ab15580), and anti-Caspase-3 (ab32351) were from Abcam (Cambridge, UK). And anti-ZEB1 (#3396), anti-SOX-2 (#2748), anti-ALDH-1 (#12035), anti-OCT4 (#4286), anti-Bax (#2772), anti-Bcl-2 (#4223), anti-GAPDH (#51332) and all secondary antibodies were from Cell Signaling Technology (Danvers, MA, USA).
Sphere Formation Assay
A549 cells were seeded in low adherent 24-well culture plates at 2 × 103 cells per well, and incubated under serum-free conditions in RPMI 1640 containing 20 μL/mL of B27, 20 ng/mL of EGF, 20 ng/mL of bFGF and 1% of the penicillin-streptomycin. After incubation at 37°C in a 5% CO2 incubator for 10 days, spheres were recorded under a microscope in three fields.
Flow Cytometry
Flow Cytometry analysis was applied according to the manufacturer’s protocol. In brief, cells differently transfected were collected and re-suspended in PBS, incubated with Annexin V-FITC (BestBio, Shanghai, China) for 15 min and with PI for 10 min, and analyzed with a FACScan flow cytometer (BD Biosciences, Franklin Lakes, NJ, USA). For analysis of mitochondrial potential, living cells were re-suspended with JC-1 working solution, incubated in 37°C, 5% CO2 for 15 min, and re-suspended again in PBS, analyzed with FACScan.
Tests of SOD, MDA and LDH Activity
For test of SOD (superoxide dismutase), cells were washed, scratched and collected in cold PBS. Then, homogenized and centrifuged under 600 g for 15 min at 4°C and supernatants were collected. Bradford assay was applied to test protein concentrations. About 50 μg of proteins were used for each sample. The samples, Wst-8/enzyme working solution, reaction initiating solution (Beyotime Biotechnology, Shanghai, China) and PBS were added according to protocol, incubated at 37°C for 30 min, and optical density was tested under 450 nm.
For MDA (malondialdehyde) activity assay, cell lysates were harvested and tested using BCA KIT as in Western blotting. According to the protocol, MDA testing working solution (Beyotime Biotechnology, Shanghai, China) was prepared, optical density under 532 nm was tested and MDA concentration was corrected using curve of the standard.
For test of LDH (Layered Double Hydroxide), LDH Assay Kit (Beyotime Biotechnology, Shanghai, China) was used. Transfected A549 cells were seeded in 96 well plates at a density of 1 × 105 cells per well in 200 μL culture medium with 1% FBS for 24 h. Then, the plate was centrifuged at 400g for 5 min, 120 μL supernate was transferred to a new well, and 60 μL LDH test working solution was added. Optical density at 490 nm was read after incubated in the dark for another 30 min, LDH concentrations were normalized with absorbance of standard solutions.
EdU
The proliferation of transfected A549 cells was assayed by EdU (5-ethynyl-2ʹ-deoxyuridine) kit (Cell-Light EdU Apollo 567 In Vitro Imaging Kit, Ribobio, Guangzhou, China). Briefly, 5 × 103 transfected A549 cells were seeded into each well of 96-well plates. 24 h after seeded, 100 μL medium with 50 μM EdU was added into each well for 2 h at 37°C. Then, cells were fixed with 4% paraformaldehyde (Beyotime Biotechnology, Shanghai, China), stained with Hoechst 33342 and Apollo reaction mix. Images were captured using a fluorescence microscopy (Nikon). EdU-positive cells and total cells were counted within each field.
Statistics
Graphpad Prism 6.0 was used in statistics. Analysis was done with one way ANOVA or Student’s t-test, and data were shown as mean ± standard deviation (SD). P<0.05 was taken as statistically significant.
Results
Overexpression of miR-199a-3p Suppressed Tumor Growth
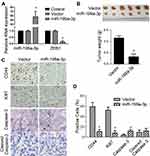
The overall tumor-suppressive role of miR-199a-3p in lung cancer in vivo was investigated. Related empty vector and premiR-199a-3p transfected A549 cells were established and then RNA expression of miR-199a-3p and its proposed target gene ZEB1 were validated with qPCR. Transfection of miR-199a-3p greatly elevated miR-199a-3p while significantly inhibited ZEB1 mRNA expression (Figure 1A, p<0.05), which implicated successful transfection of miR-199a-3p and inhibition of the indicated target gene ZEB1. Xenograft tumors in BALB/c nude mice were inoculated with the established cell lines and raised for 30 days. As expected, overexpression of miR-199a-3p significantly suppressed tumor growth of xenograft mice (Figure 1B, p<0.05). IHC of these tumor tissues showed decrease of CD44 and Ki67, while showed increase of caspase-3 by miR-199a-3p transfection (Figure 1C and D, p<0.05). These results indicated that the tumor-suppressive role of miR-199a-3p could be related to down-regulated ZEB1, inhibited stemness and proliferation, and promoted apoptosis. Then, in vitro gain- and loss-of-function assays with pcDNA-ZEB1 transfection were introduced.
Overexpression of miR-199a-3p Suppressed Stemness of A549
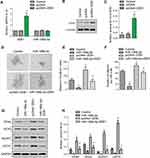
Transfection of pcDNA-ZEB1 was used to construct stable ZEB1 overexpression A549 cell lines. Then, elevated RNA (Figure 2A, p<0.05) and protein (Figure 2B and C, p<0.05) expression of ZEB1 was validated. Stem-like property of A549 was assessed with miR-199a-3p and pcDNA-ZEB1 transfection. As a result, miR-199a-3p significantly inhibited, while pcDNA-ZEB1 promoted sphere formation (Figure 2D). Both diameter of spheres and number of spheres per 100 cells (Figure 2E and F, respectively, *p<0.05 versus control and #p<0.05 versus miR-199a-3p or pcDNA-ZEB1) were decreased by miR-199a-3p and increased by pcDNA-ZEB1, co-transfection of both genes counteracted their effects. Relative expression of stemness marker proteins CD44, SOX-2, ALDH1 and OCT4 was evaluated (Figure 2G and H, *p<0.05 versus control, and # p<0.05 versus miR-199a-3p or pcDNA-ZEB1). Consequently, overexpressed miR-199a-3p significantly reduced protein expression of CD44, SOX-2, ALDH1 and OCT4, while ZEB1 overexpression elevated these proteins. Similarly, co-transfection of miR-199a-3p and ZEB1 eliminated up-regulation by ZEB1 and down-regulation by miR-199a-3p of these stemness markers.
Overexpression of miR-199a-3p Suppressed Proliferation and Promoted Apoptosis of A549
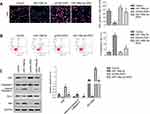
The effects of miR-199a-3p on A549 cell proliferation and apoptosis were evaluated with EDU staining and FITC Annexin V labeled flow cytometry, respectively. As indicated in Figure 3A (*p<0.05 versus control, and # p<0.05 versus miR-199a-3p or pcDNA-ZEB1), overexpression of miR-199a-3p significantly reduced, while ZEB1 overexpression increased EDU-positive cells per 100 cells. Co-transfection of both miR-199a-3p and ZEB1 counteracted their effects. These trends were further validated by the relative protein expression of proliferation marker Ki67 (Figure 3C, *p<0.05 versus control, and # p<0.05 versus miR-199a-3p or pcDNA-ZEB1). Apoptosis was induced with miR199a-3p while partly counteracted by ZEB1 overexpression (Figure 3B, *p<0.05 versus control, and # p<0.05 versus miR-199a-3p or pcDNA-ZEB1). Apoptosis in transfected cells was further validated by the ratio of cleaved Caspase-3 to Caspase-3 and the ratio of Bcl-2 to Bax as indicated in Figure 3C, in which miR-199a-3p decreased the ratio of cleaved Caspase-3 to Caspase-3, and increased the ratio of Bcl-2 to Bax. Opposite trend was observed in pcDNA-ZEB1 treated cells and counteracted with co-transfection.
Overexpression of miR-199a-3p Induced Mitochondria Injury of A549
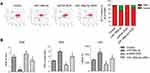
As apoptosis was induced by miR-199a-3p, mitochondria injury was assessed. JC-1 labeled flow cytometry indicated that overexpressed miR-199a-3p increased green fluorescence in A549 cells and co-transfection of ZEB-1 partly restrained this increase (Figure 4A). Mitochondria injury was further validated with mitochondria oxidative stress marker SOD and MDA, and membrane damage indicator LDH in transfected A549 cells.21,22 As expected, activity of SOD, MDA and LDH exhibited the same trend with JC-1 labeled flow cytometry (Figure 4B, *p<0.05 versus control, and # p<0.05 versus miR-199a-3p or pcDNA-ZEB1). Overexpression of miR-199a-3p decreased activity of SOD, and this was partly alleviated with co-transfection of ZEB1. PremiR-199a-3p transfection led to significant increase of MDA and LDH, which were partly inhibited by pcDNA-ZEB1 co-transfection.
Discussion
Targeting of ZEB1 by miR-199a-3p was validated in a series of cells.17–20 Generally, ZEB1 was over-expressed and miR-199a-3p was low-expressed in tumors, and the inadequate of miR-199a-3p was shown to be negatively related with ZEB1 expression, further up-regulation of miR-199a-3p significantly inhibited ZEB1 expression.17,18 As ZEB1 was largely considered as an EMT promoting gene, miR-199a-3p targeted ZEB1 was mainly shown to inhibit proliferation, migration and invasion including in lung cancer cells.17 Here we investigated the down-regulation of ZEB1 with miR-199a-3p concerning stemness and proliferation, and mitochondria-dependent apoptosis in A549 cells and xenograft mice.
Both up-regulation of miR-199a-3p and down-regulation of ZEB1 have directly or indirectly been proven to inhibit tumors in vivo. For example, miR-199-dependent oncolytic adenovirus exhibited a therapeutic potential against liver cancer without causing significant hepatotoxicity,23 and premiR-199a-3p overexpressing suppressed tumor growth of HCC cell xenograft NOD/SCID mice.24 In NSCLC cell line H460 xenograft model, ZEB1 knockdown with polypeptide cationic micelles inhibits metastasis and drug resistance.25 More evidently, gambogic acid inhibition of melanoma was mir-199a-3p and ZEB1 dependent. Up-regulation of miR-199a-3p and down-regulation of ZEB1 obviously reduced tumor weight and increased cisplatin sensitivity in CDDP melanoma cell xenograft mice.18 Consistence with these studies, here we showed that tumors were smaller in premiR-199a-3p transfected A549 cells xenograft mice. The elevated miR-199a-3p inhibited ZEB1 expression, decreased CD44 and Ki67 while increased Caspase-3 positive cells of tumor tissues. Implicating inhibited proliferation and stemness and promoted apoptosis with miR-199a-3p transfection.
ZEB1 is a classic EMT promoter that enables stemness in several dimensions. After EMT, transformed tumor cells possibly gain stem-like property, and ZEB1 plays a major role in the dynamic conversion between stem-like cells and non-stem-like cells.13 On the other hand, role of miR-199a-3p in the suppression of stemness was reported in several cells through multiple molecules including Wnt, KDM6A, Sema3A and PTPN3.26–29 Though exactly how miR-199a-3p inhibited stemness still needs to be investigated, miR-199a-3p targeted ZEB1 should be a pivotal mechanism in EMT as well as stemness in NSCLC.
ZEB1 and miR-199a-3p were also indicated in regulating proliferation and apoptosis. miR-199 loci on chromosomes 1 and 19 were heavily methylated in normal tissues. De-methylation of chromosomes or adding of miR-199a-3p significantly induced apoptosis in A549 cells.30 Inhibition of miR-199a-3p promoter methylation also reduced tumor growth and inhibited metastasis in vivo.15 Overexpressed miR-199a/b-5p inhibited cell proliferation, migration and invasion across several tumors both in vitro and in vivo, especially liver and lung cancers.31 ZEB1, on the other hand, promoted proliferation and inhibited apoptosis in lung cancer. For example, ZEB1 was inhibited by long non-coding (lnc) RNA ZEB1-AS1 to promote cell proliferation and inhibit apoptosis of NSCLC.32 Knockdown of ZEB1suppressed NSCLC cell proliferation through apoptosis induction but not senescence.4 These indicated miR-199a-3p inhibited proliferation could be apoptosis dependent. At the same time, different signaling pathways, miRNAs, lncRNAs, and Circular RNAs were indicated to be related to ZEB1 and miR-199a-3p in regulating EMT, proliferation and apoptosis.10,17,32-35 This highlighted the complexity of miR-199a-3p regulation.
As apoptosis was induced with miR-199a-3p overexpression, involvement of mitochondria in apoptosis induced with miR-199a-3p was further investigated in this research. Overexpression of miR-199a-3p induced mitochondria dysfunction as indicated with decreased mitochondrial membrane potential and SOD activity, and increased MDA and LDH. In fact, mitochondria dysfunction was also indicated in ZEB1 inhibited conditions. For example, knockdown of 2-oxoglutarate dehydrogenase induced mitochondria dysfunction and apoptosis, and inhibited ZEB1 at the same time in GC cells.36 In another research, ZEB1 was necessary for ROS generation and maintenance of mitochondrial membrane potential.37 For miR-199a-3p, targets for regulation of apoptosis could be varied. Other than ZEB1 as characterized by us and others, miR-199a-3p may also target 3ʹ-UTR of HIF-alpha, which also would lead to mitochondria dysfunction and apoptosis.38
Generally, the transcription of miR-199a-3p was strictly controlled, as miR-199 locus was heavily methylated in normal tissues. Activation of miR-199a-3p possibly would activate other neighbor genes such as miR-199a-5p or mir-199b-5p. miR-199a-5p has opposite functions with miR-199a-3p in many aspects. It was proved to target and inhibit Zinc-fingers and homeoboxes 1 (ZHX1) to promote the growth and proliferation of cancer cells.31 ZEB1 down-expression with miR-199a-3p was able to inhibit the proliferation, migration and invasion of NSCLC cells.17 We also showed that ZEB1 down-expression with miR-199a-3p inhibited the viability, stemness and apoptosis in A549 cells. Considering the strict transcriptional control of miR-199-a3-3p, and the many tumor promotional roles of ZEB1, the targeting of ZEB1 with miR-199a-3p may be a useful focus to better understanding of NSCLC, and may provide a novel therapeutic target for the disease.
Altogether, the present findings suggested that the up-regulation of miR-199a-3p with premiRNA significantly inhibited NSCLC growth in vivo, and reduced A549 cell proliferation and promoted mitochondrial-mediated apoptosis, through down-regulation of ZEB1. The findings supported ZEB1 down-expression with miR-199a-3p as a novel therapeutic target for NSCLC treatment.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.21492
2. Osmani L, Askin F, Gabrielson E, Li QK. Current WHO guidelines and the critical role of immunohistochemical markers in the subclassification of non-small cell lung carcinoma (NSCLC): moving from targeted therapy to immunotherapy. Semin Cancer Biol. 2018;52(Pt 1):103–109. doi:10.1016/j.semcancer.2017.11.019
3. Mott TF. Lung cancer: management. FP Essent. 2018;464:27–30.
4. Takeyama Y, Sato M, Horio M, et al. Knockdown of ZEB1, a master epithelial-to-mesenchymal transition (EMT) gene, suppresses anchorage-independent cell growth of lung cancer cells. Cancer Lett. 2010;296(2):216–224. doi:10.1016/j.canlet.2010.04.008
5. Manshouri R, Coyaud E, Kundu ST, et al. ZEB1/NuRD complex suppresses TBC1D2b to stimulate E-cadherin internalization and promote metastasis in lung cancer. Nat Commun. 2019;10(1):5125. doi:10.1038/s41467-019-12832-z
6. Liu Y, Zhang N, Wang Y, et al. Zinc finger E-box binding homeobox 1 promotes invasion and bone metastasis of small cell lung cancer in vitro and in vivo. Cancer Sci. 2012;103(8):1420–1428. doi:10.1111/j.1349-7006.2012.02347.x
7. Argast GM, Krueger JS, Thomson S, et al. Inducible expression of TGFbeta, snail and Zeb1 recapitulates EMT in vitro and in vivo in a NSCLC model. Clin Exp Metastasis. 2011;28(7):593–614. doi:10.1007/s10585-011-9394-8
8. Larsen JE, Nathan V, Osborne JK, et al. ZEB1 drives epithelial-to-mesenchymal transition in lung cancer. J Clin Invest. 2016;126(9):3219–3235. doi:10.1172/JCI76725
9. Hu X, Mu Y, Wang J, Zhao Y. LncRNA TDRG1 promotes the metastasis of NSCLC cell through regulating miR-873-5p/ZEB1 axis. J Cell Biochem. 2019. doi:10.1002/jcb.29559
10. Liu C, Zhang Z, Qi D. Circular RNA hsa_circ_0023404 promotes proliferation, migration and invasion in non-small cell lung cancer by regulating miR-217/ZEB1 axis. Onco Targets Ther. 2019;12:6181–6189. doi:10.2147/OTT.S201834
11. Nourmohammadi B, Tafsiri E, Rahimi A, et al. Expression of miR-9 and miR-200c, ZEB1, ZEB2 and E-cadherin in non-small cell lung cancers in Iran. Asian Pac J Cancer Prev. 2019;20(6):1633–1639. doi:10.31557/APJCP.2019.20.6.1633
12. Ma DJ, Liu HS, Li SQ, et al. Correlations of the ZEB1 expression with the incidence and prognosis of non-small cell lung cancer. Eur Rev Med Pharmacol Sci. 2019;23(4):1528–1535. doi:10.26355/eurrev_201902_17111
13. Caramel J, Ligier M, Puisieux A. Pleiotropic roles for ZEB1 in cancer. Cancer Res. 2018;78(1):30–35. doi:10.1158/0008-5472.CAN-17-2476
14. He Y, Ren S, Wang Y, Li X, Zhou C, Hirsch FR. Serum microRNAs improving the diagnostic accuracy in lung cancer presenting with pulmonary nodules. J Thorac Dis. 2018;10(8):5080–5085. doi:10.21037/jtd.2018.07.138
15. Mudduluru G, Ceppi P, Kumarswamy R, Scagliotti GV, Papotti M, Allgayer H. Regulation of Axl receptor tyrosine kinase expression by miR-34a and miR-199a/b in solid cancer. Oncogene. 2011;30(25):2888–2899. doi:10.1038/onc.2011.13
16. Tian R, Xie X, Han J, et al. miR-199a-3p negatively regulates the progression of osteosarcoma through targeting AXL. Am J Cancer Res. 2014;4(6):738–750.
17. Wang J, Zhou F, Yin L, Zhao L, Zhang Y, Wang J. MicroRNA-199b targets the regulation of ZEB1 expression to inhibit cell proliferation, migration and invasion in nonsmall cell lung cancer. Mol Med Rep. 2017;16(4):5007–5014. doi:10.3892/mmr.2017.7195
18. Liang L, Zhang Z, Qin X, et al. Gambogic acid inhibits melanoma through regulation of miR-199a-3p/ZEB1 signalling. Basic Clin Pharmacol Toxicol. 2018;123(6):692–703. doi:10.1111/bcpt.13090
19. Meng L, Li G, Liu X, Jiang J, Zhu M, Sun Y. Decreased urine miR-199-3p may be a potential biomarker for diabetic nephropathy via targeting zinc finger E-box-binding protein 1. Clin Lab. 2018;64(7):1177–1182. doi:10.7754/Clin.Lab.2018.180126
20. Mishra R, Benlhabib H, Guo W, Lerma Cervantes CB, Mendelson CR. Developmental decline in the MicroRNA 199a (miR-199a)/miR-214 cluster in human fetal lung promotes Type II cell differentiation by upregulating key transcription factors. Mol Cell Biol. 2018;38(11). doi:10.1128/MCB.00037-18
21. Ek B, Hallberg C, Sjogren KG, Hjalmarson A. Reoxygenation-induced cell damage of isolated neonatal rat ventricular myocytes can be reduced by chain-breaking antioxidants. Free Radic Biol Med. 1994;16(1):117–121. doi:10.1016/0891-5849(94)90248-8
22. Zhao Z, Liu M, Zhang Y, et al. Cardioprotective effect of monoammonium glycyrrhizinate injection against myocardial ischemic injury in vivo and in vitro: involvement of inhibiting oxidative stress and regulating Ca(2+) homeostasis by L-Type calcium channels. Drug Des Devel Ther. 2020;14:331–346. doi:10.2147/DDDT.S232130
23. Callegari E, Elamin BK, D’Abundo L, et al. Anti-tumor activity of a miR-199-dependent oncolytic adenovirus. PLoS One. 2013;8(9):e73964. doi:10.1371/journal.pone.0073964
24. Ghosh A, Dasgupta D, Ghosh A, et al. MiRNA199a-3p suppresses tumor growth, migration, invasion and angiogenesis in hepatocellular carcinoma by targeting VEGFA, VEGFR1, VEGFR2, HGF and MMP2. Cell Death Dis. 2017;8(3):e2706. doi:10.1038/cddis.2017.123
25. Fang S, Wu L, Li M, et al. ZEB1 knockdown mediated using polypeptide cationic micelles inhibits metastasis and effects sensitization to a chemotherapeutic drug for cancer therapy. Nanoscale. 2014;6(17):10084–10094. doi:10.1039/C4NR01518E
26. Chen B, Zhang D, Kuai J, Cheng M, Fang X, Li G. Upregulation of miR-199a/b contributes to cisplatin resistance via Wnt/beta-catenin-ABCG2 signaling pathway in ALDHA1(+) colorectal cancer stem cells. Tumour Biol. 2017;39(6):1010428317715155. doi:10.1177/1010428317715155
27. Shuai Y, Yang R, Mu R, Yu Y, Rong L, Jin L. MiR-199a-3p mediates the adipogenic differentiation of bone marrow-derived mesenchymal stem cells by regulating KDM6A/WNT signaling. Life Sci. 2019;220:84–91. doi:10.1016/j.lfs.2019.01.051
28. Zhu G, Pei L, Lin F, et al. Exosomes from human-bone-marrow-derived mesenchymal stem cells protect against renal ischemia/reperfusion injury via transferring miR-199a-3p. J Cell Physiol. 2019;234(12):23736–23749. doi:10.1002/jcp.28941
29. Li S, Cao J, Zhang W, et al. Protein tyrosine phosphatase PTPN3 promotes drug resistance and stem cell-like characteristics in ovarian cancer. Sci Rep. 2016;6:36873. doi:10.1038/srep36873
30. Kim S, Lee UJ, Kim MN, et al. MicroRNA miR-199a* regulates the MET proto-oncogene and the downstream extracellular signal-regulated kinase 2 (ERK2). J Biol Chem. 2008;283(26):18158–18166. doi:10.1074/jbc.M800186200
31. Wang Q, Ye B, Wang P, Yao F, Zhang C, Yu G. Overview of microRNA-199a regulation in cancer. Cancer Manag Res. 2019;11:10327–10335. doi:10.2147/CMAR.S231971
32. Qu R, Chen X, Zhang C. LncRNA ZEB1-AS1/miR-409-3p/ZEB1 feedback loop is involved in the progression of non-small cell lung cancer. Biochem Biophys Res Commun. 2018;507(1–4):450–456. doi:10.1016/j.bbrc.2018.11.059
33. Gemmill RM, Roche J, Potiron VA, et al. ZEB1-responsive genes in non-small cell lung cancer. Cancer Lett. 2011;300(1):66–78. doi:10.1016/j.canlet.2010.09.007
34. Chiu L-Y, Hsin I-L, Yang T-Y, et al. The ERK–ZEB1 pathway mediates epithelial–mesenchymal transition in pemetrexed resistant lung cancer cells with suppression by vinca alkaloids. Oncogene. 2017;36(2):242–253. doi:10.1038/onc.2016.195
35. Gao Y, Cao Y, Cui X, et al. miR-199a-3p regulates brown adipocyte differentiation through mTOR signaling pathway. Mol Cell Endocrinol. 2018;476:155–164. doi:10.1016/j.mce.2018.05.005
36. Lu X, Wu N, Yang W, Sun J, Yan K, Wu J. OGDH promotes the progression of gastric cancer by regulating mitochondrial bioenergetics and Wnt/beta-catenin signal pathway. Onco Targets Ther. 2019;12:7489–7500. doi:10.2147/OTT.S208848
37. Xu WY, Hu QS, Qin Y, et al. Zinc finger E-box-binding homeobox 1 mediates aerobic glycolysis via suppression of sirtuin 3 in pancreatic cancer. World J Gastroenterol. 2018;24(43):4893–4905. doi:10.3748/wjg.v24.i43.4893
38. Liu DW, Zhang YN, Hu HJ, Zhang PQ, Cui W. Downregulation of microRNA199a5p attenuates hypoxia/reoxygenation induced cytotoxicity in cardiomyocytes by targeting the HIF1alphaGSK3betamPTP axis. Mol Med Rep. 2019;19(6):5335–5344. doi:10.3892/mmr.2019.10197
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.