Back to Journals » Journal of Multidisciplinary Healthcare » Volume 15
Diagnostic Value of Color Doppler Ultrasound and Contrast-Enhanced Ultrasound in the Artery Steal Syndrome After Orthotopic Liver Transplantation
Authors Li W, Gao N, Pan YP, Ren XY
Received 19 August 2022
Accepted for publication 18 October 2022
Published 6 November 2022 Volume 2022:15 Pages 2563—2569
DOI https://doi.org/10.2147/JMDH.S386820
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Wei Li,1,2,* Nong Gao,2,* Yi-Peng Pan,1 Xiu-Yun Ren2,3
1Department of Liver Transplantation, the Second Affiliated Hospital of Hainan Medical University, SanYa, People’s Republic of China; 2Department of Organ Transplantation, the Third Medical Center of General Hospital of People’s Liberation Army, Beijing, People’s Republic of China; 3Department of Ultrasound Examination, Hainan Hospital of General Hospital of People’s Liberation Army, SanYa, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Xiu-Yun Ren, Department of Organ Transplantation, the Third Medical Center of General Hospital of People’s Liberation Army, Beijing, People’s Republic of China, Email [email protected]
Purpose: This study aimed to investigate the application of color Doppler ultrasound (CDU) and contrast-enhanced ultrasound (CEU) in the early diagnosis of arterial steal syndrome (ASS) after orthotopic liver transplantation (OLT).
Patients and Methods: A total of 1827 patients received OLT in our department between January 2007 and December 2021, and CDU and CEU were performed after surgery. Ultrasonographic data were collected and further analyzed. ASS was confirmed by digital subtraction angiography (DSA), and the use of splenic artery embolization was dependent on the DSA findings and clinical conditions. The ultrasonographic data were collected before and after embolization.
Results: ASS was found in 23 patients (23/1827; 1.26%). CDU: the portal vein velocity was higher than normal in 17 patients (52.83± 21.74 cm/s); the hepatic artery flow signals disappeared in 7 patients; the hepatic artery velocity was 13.57± 5.85 cm/s in 16 patients. In 23 patients, the spleen artery velocity was 170.12± 32.04 cm/s. CEU: the hepatic artery was observed in 7 patients without hepatic artery flow signals on CDU; in 23 patients, the contrast agent reached the portal vein at the presence of contrast in the hepatic artery or earlier (difference: 2.21± 1.09 s). Splenic artery embolization was done in 17 of 23 patients with ASS. The hepatic artery flow velocity, hepatic artery resistance index, splenic artery velocity and time to the presence of contrast were significantly improved after embolization (P< 0.05).
Conclusion: CDU and CEU are the preferred, effective tools in the diagnosis of ASS. Reduced peak hepatic artery velocity, increased spleen artery velocity and reduced time difference in the contrast reaching the portal vein and hepatic artery are indicative of ASS.
Keywords: contrast-enhanced ultrasound, color Doppler ultrasound, liver transplantation, arterial steal syndrome
Introduction
Orthotopic liver transplantation (OLT) is an effective treatment for the end-stage liver disease. Arterial steal syndrome (ASS) is a rare and serious complication after liver transplantation, and the cause of ASS is still unclear. Generally, it is related to following factors: the splenic artery, left gastric artery and gastroduodenal artery are too large to cause active blood stealing, and splenic artery blood stealing is the most common; the blood flow is high before surgery or some factors cause portal vein hyperperfusion after transplantation; factors related to the donor liver, ischemia-reperfusion injury or increased peripheral resistance of the arterial bed in the donor liver lead to hepatic artery hypoperfusion, resulting in passive blood stealing. Arterial blood stealing can lead to arterial hypoperfusion, hypoxia, dysfunction of the donor liver and biliary ischemic injury.1,2 If it is not diagnosed in time, a second liver transplantation will be required due to the loss of graft function.1 Thus, early diagnosis and timely treatment can avoid serious complications after OLT. Color Doppler ultrasound (CDU) is a preferred tool for detecting artery flow after liver transplantation, but it is insensitive to the slow blood flow, and it may be easily misdiagnosed as hepatic artery thrombosis. Ultrasound angiography can dynamically display the hepatic artery and portal vein perfusion, which can effectively compensate for the disadvantages of CDU. This study was to investigate the diagnostic value of CDU combined with contrast-enhanced ultrasound (CEU) in the ASS after OLT.
Materials and Methods
This study was approved by the Ethics Committee of the Third Medical Center of General Hospital of People’s Liberation Army, and conducted according to the Declaration of Helsinki. Written informed consent was obtained from all participants before the study. All donor livers were from donors with brain death or heart death, and their relatives had signed informed consent for organ donation. This was conducted in accordance with the Declaration of Istanbul.
Patients and Methods
From January 2007 to December 2021, 23 patients with ASS after OLT in our organ transplantation center were recruited into present study. The final diagnosis of above patients was confirmed by DSA: 17 patients were diagnosed as splenic artery steal, and 6 patients were diagnosed as gastroduodenal artery steal. All the recipients underwent routine CDU after surgery once daily for 1 week, and thereafter once weekly for 3 weeks. Then, patients received routine follow up. If ASS was suspected, CEU was performed. If necessary, DSA was employed to confirm the diagnosis of ASS. The Mylab Twice (Esaote, Italy) ultrasound system with (CnTI) ultrasound imaging software and the contrast (SonoVue; Bracco, Milan, Italy) were used.
The portal vein velocity, peak hepatic artery velocity and resistance index (RI), and peak splenic artery velocity were measured on CDU. During the CEU, a bolus of contrast agent was injected into the peripheral vein (0.5–1.5 mL), and then the catheter was flushed with 5 mL of normal saline. The dynamic images were stored. The main observations included the time to contrast reaching the hepatic artery and portal vein, vascular perfusion, and liver perfusion. The difference in the time to contrast reaching the hepatic artery and portal vein was calculated as follow: time difference = time to contrast reaching the portal vein - time to contrast reaching the hepatic artery (second, s). ASS after OLT is characterized by arterial hypoperfusion of the graft, which is caused by a shift in blood flow into the splenic or gastroduodenal arteries. In the present study, the threshold for the portal vein blood flow velocity was defined as 40 cm/s because the portal vein blood flow velocity is below 40 cm/s under normal conditions after OLT.
For patients diagnosed with ASS, splenic artery embolization was performed when digital subtraction angiography (DSA) showed the splenic artery/hepatic artery diameter was >1.5. On the following day, CDU and CEU were employed to assess the efficacy of embolization.
Statistical Analysis
Statistical analysis was done with Statistical Package for Social Sciences (SPSS) version 18.0 (IBM Corporation, Armonk, NY). Quantitative data are expressed as mean ± standard deviation (SD). Wilcoxon paired rank test was used for the comparisons of data before and after splenic artery embolization. A value of P<0.05 was considered statistically significant.
Results
General Characteristics
Among 1827 patients, ASS was noted in 23 patients and the incidence of ASS was 1.26% (23/1827). There were 17 males and 6 females with the mean age of 41±5.6 years. The primary liver diseases included hepatitis B cirrhosis (n=6), primary carcinoma of liver (n=12), alcoholic cirrhosis (n=3) and drug-induced liver failure (n=2). The mean time to diagnostic ultrasonography was 2.5±1.0 days and the time to DSA was 7.3±1.6 days.
Clinical Treatments and Prognosis
Four patients received coagulation therapy, the hepatic artery flow returned to normal 3 months after surgery in 3 patients and 1 patient had low hepatic artery flow and normal liver function. Two patients did not receive treatment due to the normal liver function, but biliary complications were observed 3 months after surgery. The remaining patients received splenic artery embolization and the hepatic artery flow returned to normal.
CDU Findings
Among 23 patients with ASS, the portal vein velocity was higher than normal in 17 patients, and the average portal vein velocity was 52.83±21.74 cm/s; the remaining 6 patients had normal portal vein velocity. Hepatic artery blood flow was abnormal in all the 23 patients: disappearance of hepatic artery blood flow was found in 7 patients and reduced hepatic artery velocity in 15 patients (13.57±5.85 cm/s). The splenic artery velocity ranged from 98.12 cm/s to 211.93 cm/s in 23 patients (mean: 178.76±36.81 cm/s).
CEU Findings
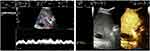
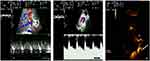
The contrast agent reached the portal vein and hepatic artery at the same time, or the contrast reached the portal vein slightly earlier than it reached the hepatic artery. The mean time difference was 2.21±1.09 s. The peak contrast intensity was lower in the artery than in the vein (Figure 1). In 7 patients with disappearance of hepatic artery blood flow, hepatic artery thrombosis was not identified (Figure 2). The findings from CDU and CEU are shown in Table 1.
 |
Table 1 Findings from CDU and CEU Before and After Splenic Artery Embolization |
Splenic Artery Embolization
A total of 17 patients underwent splenic artery embolization. The clinical prognosis of 17 patients is shown in Table 2. After splenic artery embolization, the hepatic artery blood flow was significantly improved in 16 patients, but the hepatic artery thrombosis was not improved in 1 patient. The hepatic artery velocity, RI, splenic artery velocity and time difference were significantly improved after splenic artery embolization (P=0.005, 0.012, 0.005 and 0.005, respectively). In patients without splenic artery embolization, hepatic artery blood flow returned to normal in 4 patients, and 2 patients had hepatic artery thrombosis (1 had concomitant intrahepatic infarction and underwent a second liver transplantation).
 |
Table 2 Clinical Prognosis of 17 Patients with Splenic Artery Embolism |
Discussion
OLT is an effective treatment for the end-stage liver disease. Postoperative arterial complications are the main cause of graft failure and recipient death. ASS is a rare, severe arterial complication after OLT with the reported incidence of 0.6%-10.1%, and the splenic artery is a main site of ASS.3,4 The presence of ASS will decrease the hepatic arterial perfusion, resulting in a series of diseases such as liver injury and biliary ischemic injury, which might be life-threatening.
The gold standard has a high diagnostic sensitivity for the active splenic ASS due to large splenic artery, but it is insensitive to passive splenic ASS caused by donor liver related factors, ischemia-reperfusion injury, portal hyperperfusion, and increased vascular resistance around the artery. In the passive splenic ASS, the diameter of splenic artery is not large, and the reduced hepatic artery velocity and slightly increased splenic artery velocity are the characteristics under this condition.5 In this study, CDU and CEU showed passive ASS in 6 patients, therefore, splenic artery embolization was not performed, and anticoagulation and microcirculation improving treatments were used. However, 2 patients still developed thrombosis, and one of them received a second liver transplantation. Therefore, the treatment of passive ASS is more difficult, and the prognosis is often poor once arterial thrombosis occurs.
In the present study, CDU provided information on the blood flow velocity, spectral morphology, and RI, but it is difficult to display information in case of slow blood flow. However, contrast-enhanced ultrasonography compensates the disadvantages of CDU and can display the real-time perfusion, especially the information about the time difference. The performance of CEU is similar to that of angiography under this condition, and the combination of CDU and CEU provides an effective tool for the diagnosis of ASS. Moreover, the bedside CEU is feasible, and the contrast agent can be metabolized by the lungs and has no nephrotoxicity. Thus, this examination is more non-invasive and more convenient than computer tomography angiography (CTA) and DSA.6
It has been reported that the main manifestations of ASS on CDU include evident hepatic artery, reduced blood flow spectrum, increased RI, and elevated splenic artery velocity.7,8 In the present study, the reduced hepatic artery flow velocity and increased spleen artery flow velocity were more valuable for the diagnosis of ASS. In the early stage after liver transplantation, the arterial RI is unstable, and largely increases. Therefore, RI elevation has a poor sensitivity in the diagnosis of ASS, but RI can be used to evaluate the efficacy of splenic artery embolization. In the present study, after splenic artery embolization, RI significantly reduced when the hepatic artery velocity returned to normal.
CDU has certain difficulties in identifying low blood flow. When the hepatic artery flow disappears, it is often misdiagnosed as hepatic artery thrombosis.9,10 CEU is superior to the CDU in identifying arterial patency, and can clearly identify whether the hepatic artery is thrombotic.11 In our study, the hepatic artery blood flow disappeared in 7 patients, which was difficult to distinguish from the arterial thrombosis. However, CEU showed the hepatic arterial blood flow was smooth, which excluded the false positive diagnosis of hepatic artery thrombosis. To date, few studies have reported the application of CEU in the diagnosis of ASS, especially with regard to the time difference. Our study showed the difference of time to portal vein and hepatic artery perfusion was shortened, which was important for the diagnosis of ASS. Under normal conditions, the contrast reaches the hepatic artery 10–20 s after the contrast is injected and arrived at the portal vein 30–45 s after the contrast is injected; the time difference is generally more than 10s. In our patients with ASS, the time difference was 2.21±1.09 s, which was significantly shorter than that under normal condition. In some cases, the contrast arrived at portal vein and hepatic artery at the same time, which is consistent with the criteria for the diagnosis of ASS (contrast arrives at portal vein and hepatic artery at the same time or contrast agent reaches the portal vein slightly earlier than it reaches the hepatic artery). Therefore, when ASS is suspected on CDU, the bedside CEU can be used as an effective tool for the diagnosis of ASS, which compensates the disadvantages of DSA as a gold standard. Thus, it may become a most convenient diagnostic tool. In our center, CEU has become the preferred tool in the diagnosis of vascular complications, especially in the identification of hepatic artery patency, hepatic artery thrombosis and ASS.
The normal liver is supplied by both portal vein and hepatic artery, the portal vein accounts for 75% of blood supply to the liver and the hepatic artery accounts for 25%. In the early stage after liver implantation, the hemodynamics of transplanted liver is different from that of normal liver, and becomes unstable due to the influence of primary liver disease, liver texture and ischemia-reperfusion injury. Under this condition, the portal vein is the main vessel for blood supply to the liver and accounts for more than 90% of blood supply. The more the blood supply from the portal vein, the less the blood supply from the hepatic artery.12 This will affect the diagnosis of ASS to a certain extent. The portal vein blood flow reduces and the hepatic artery blood flow increases gradually over time after liver transplantation. Thus, the findings of examination cannot be used for the diagnosis of ASS, and the combined use of CDU and CEU for close observation is needed to avoid missed diagnosis and misdiagnosis.
CDU and CEU also play an important role in the evaluation of therapeutic efficacy after splenic artery embolization in ASS patients. Splenic artery embolization is an effective strategy for the treatment of ASS after liver transplantation.13 The diameter assessment is often employed to evaluate the efficacy of embolization.14 The important parameters used to evaluate the recovery of hepatic artery blood flow after splenic artery embolization are as follows: increased hepatic artery velocity, decreased RI, decreased splenic artery velocity, and time difference returning to normal. These parameters change significantly after embolization. When these parameters related to hemodynamics remain unchanged after splenic artery embolization, therapeutic failure should be suspected. In our study, the blood flow velocity failed to return to normal after splenic artery embolization in patients, and hepatic artery thrombosis was confirmed.
The incidence of ASS in this study was 1.33%, which was significantly lower than previously reported, and the low incidence is related to the surgical strategy used in our center. In our center, the recipients routinely underwent CTA after liver transplantation. When the splenic artery is large and the splenic artery/hepatic artery diameter ratio is >1.5, intraoperative ultrasonography should be performed in the liver implanted; when the hepatic artery velocity is <25 cm/s, the splenic ASS may be suspected, and routine splenic artery circumcision or ligation may be performed to prevent splenic ASS. It has been reported that the potential incidence of ASS is 11.9% in the absence of splenic artery ligation and 1.3–5.1% after ligation of the splenic artery,15,16 which is close to the results in our study.
Conclusion
In summary, CDU combined with CEU is a preferred strategy for the diagnosis of ASS. The reduced hepatic artery velocity, increased splenic artery velocity, and shortened time difference are important parameters in the diagnosis of ASS. However, the portal vein velocity and arterial RI may be used as references, but not the dominant parameters.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Grieser C, Denecke T, Steffen IG, et al. Multidetector computed tomography for preoperative assessment of hepatic vasculature and prediction of splenic artery steal syndrome in patients with liver cirrhosis before transplantation. Eur Radiol. 2010;20(1):108–117. doi:10.1007/s00330-009-1535-y
2. Pinto S, Reddy SN, Horrow MM, Ortiz J. Splenic artery syndrome after orthotopic liver transplantation: a review. Int J Surg. 2014;12(11):1228–1234. doi:10.1016/j.ijsu.2014.09.012
3. Li C, Kapoor B, Moon E, Quintini C, Wang W. Current understanding and management of splenic steal syndrome after liver transplant: a systematic review. Transplant Rev. 2017;31:188–192.
4. Saad WE. Nonocclusive hepatic artery hypoperfusion syndrome (splenic steal syndrome) in liver transplant recipients. Semin Intervent Radiol. 2012;29:140–146.
5. Kelly DM, Shiba H, Nakagawa S, et al. Hepatic blood flow plays an important role in ischemia-reperfusion injury. Liver Transpl. 2011;17:1448–1456.
6. Durur Karakaya A, Çil BE, Kanmaz T, Oğuzkurt L. Non-occlusive hepatic artery hypoperfusion syndrome and imaging findings: a systematic review. Abdom Radiol. 2021;46:2467–2473.
7. Li C, Quintini C, Hashimoto K, et al. Role of doppler sonography in early detection of splenic steal syndrome. J Ultrasound Med. 2016;35:1393–1400.
8. Teegen EM, Denecke T, Schmuck RB, et al. Impact of Doppler ultrasound on diagnosis and therapy control of lienalis steal syndrome after liver transplantation. Ann Transplant. 2017;22:440–445.
9. Breguet R, Dondero F, Pupulim L, et al. Endovascular treatment of arterial complications after liver transplantation: long-term follow-up evaluated on doppler ultrasound and magnetic resonance cholangiopancreatography. Cardiovasc Intervent Radiol. 2019;42:381–388.
10. Chiang PL, Cheng YF, Huang TL, et al. Intensive Doppler ultrasonography for early detection of hepatic artery thrombosis after adult living donor liver transplantation. Ann Transplant. 2020;25:e924336.
11. García-Criado A, Gilabert R, Bianchi L, et al. Impact of contrast-enhanced ultrasound in the study of hepatic artery hypoperfusion shortly after liver transplantation: contribution to the diagnosis of artery steal syndrome. Eur Radiol. 2015;25:196–202.
12. Ricci K, Asharf EH. The use of splenic artery embolization to maintain adequate hepatic arterial inflow after hepatic artery thrombosis in a split liver transplant recipient. Int J Surg Case Rep. 2018;51:241–243.
13. Guan YS, Hu Y. Clinical application of partial splenic embolization. Sci World J. 2014;2014:961345.
14. Kim JH, Kim KW, Gwon DI, et al. Effect of splenic artery embolization for splenic artery steal syndrome in liver transplant recipients: estimation at computed tomography based on changes in caliber of related arteries. Transplant Proc. 2011;43:1790–1793.
15. Obmann VC, Chalian M, Mansoori B, Sanchez E, Gulani V. Advantages of time-resolved contrast-enhanced 4D MR angiography in splenic arterial steal syndrome. Clin Imaging. 2018;49:169–173.
16. Song JY, Shi BY, Zhu ZD, et al. New strategies for prevention and treatment of splenic artery steal syndrome after liver transplantation. World J Gastroenterol. 2014;20:15367–15373.
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.