Back to Journals » Clinical Ophthalmology » Volume 14
Diabetic Population-Based Model to Estimate Impact of Ranibizumab on Diabetic Retinopathy Severity in Patients with Diabetic Macular Edema
Authors Varma R, Bressler NM, Doan QV, Suñer IJ , Danese M, Dolan CM , Lee A, Ehrlich JS, Rajput Y
Received 1 November 2019
Accepted for publication 4 March 2020
Published 7 May 2020 Volume 2020:14 Pages 1249—1259
DOI https://doi.org/10.2147/OPTH.S236636
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Rohit Varma,1 Neil M Bressler,2 Quan V Doan,3 Ivan J Suñer,4 Mark Danese,3 Chantal M Dolan,5 Abraham Lee,3 Jason S Ehrlich,5 Yamina Rajput5
1Southern California Eye Institute, CHA Hollywood Presbyterian Medical Center, Los Angeles, CA, USA; 2Johns Hopkins University, Baltimore, MD, USA; 3Outcomes Insights, Inc., Westlake Village, CA, USA; 4Retina Associates of Florida, Tampa, FL, USA; 5Genentech, Inc., South San Francisco, CA, USA
Correspondence: Rohit Varma
Southern California Eye Institute, CHA Hollywood Presbyterian Medical Center, 1300 N. Vermont Avenue, Los Angeles, CA 90027, USA
Tel +1 833 270 3937
Email [email protected]
Purpose: Estimate effects of ranibizumab on diabetic retinopathy (DR) severity in US Hispanic and non-Hispanic white persons with center-involved diabetic macular edema (DME) causing vision impairment for whom ranibizumab treatment would be considered.
Patients and Methods: This model simulated DR severity outcomes over 2 years in the better-seeing eye using US census, National Health and Nutrition Examination Survey, Wisconsin Epidemiologic Study of Diabetic Retinopathy, and Los Angeles Latino Eye Study data. Baseline DR severity estimated from Diabetic Retinopathy Clinical Research Network trial data. Changes in DR severity after 2 years, with/without monthly ranibizumab (0.3 or 0.5 mg), were estimated from Phase III clinical trial data (RIDE/RISE) using a 2-dimensional Monte Carlo simulation model. Number of patients over a 2-year period for whom 1) DR severity worsening was avoided, 2) DR severity improved, and 3) selected clinical events related to proliferative DR (PDR) occurred, was estimated.
Results: An estimated 37,274 US Hispanic and non-Hispanic white persons were projected to have DR with center-involved DME and be eligible for ranibizumab treatment. The number of persons with moderately severe non-proliferative DR (NPDR) or less severe DR at baseline who would worsen to PDR and experience a PDR complication over 2 years would be reduced from 437 with no ranibizumab to 19 with ranibizumab (95% reduction; 95% simulation interval [SI], 79– 100%). The number of persons with severe NPDR or less severe DR at baseline who would be expected to improve by ≥ 2 DR severity levels over 2 years would increase from 1706 with no ranibizumab to 13,042 with ranibizumab (682% increase; 95% SI, 478– 967%).
Conclusion: This model estimates that ranibizumab treatment in US Hispanic and non-Hispanic white patients with center-involved DME causing vision impairment would potentially reduce the number of patients with worsening DR and potentially increase the number with DR improvements.
Keywords: diabetic macular edema, diabetic retinopathy, population-based model
Plain Language Summary
Diabetic retinopathy (DR) is a sight-threatening disease affecting millions of people with diabetes. Left untreated, it can slowly become more severe until vision is impaired or lost completely.
Ranibizumab is an effective treatment for diabetic macular edema (DME) and proliferative DR, producing improvements in vision and a decrease in DME and DR severity in many patients. An earlier study using a computer model estimated that monthly ranibizumab for 2 years could reduce the number of people with visual impairment or blindness due to DR by 45% and 75%, respectively.
When the same model was used to analyze the effect of monthly ranibizumab on the number of people with DR worsening or improvement, it estimated that the number of people who would worsen from a moderately severe to more severely sight-threatening form of DR would be reduced by 95%. This model also estimated that the number of people who would experience an improvement in DR severity would increase by nearly 7-fold (682%).
The results from this study build upon the results of earlier studies to suggest ranibizumab has the potential to provide meaningful benefits to large numbers of people with DR.
Introduction
Diabetic retinopathy (DR) is a vision-threatening microvascular complication of type 1 and type 2 diabetes mellitus.1 It affects ~93 million people worldwide and is a leading cause of new-onset blindness.2–5 Patients with either non-proliferative DR (NPDR) or proliferative DR (PDR) can develop diabetic macular edema (DME) which, if left untreated, is a major cause of vision impairment and legal blindness in patients with DR. In the United States from 2005 to 2008, 4.2 million adults with diabetes aged ≥40 years had DR; of these, 655,000 patients had advanced vision-threatening DR.5
Ranibizumab is a recombinant humanized monoclonal antibody fragment that binds and inhibits the biologic activity of all isoforms of human vascular endothelial growth factor (VEGF)-A. Ranibizumab 0.3 mg was approved by the US Food and Drug Administration for the treatment of DME in 2012 and for DR in patients with DME in 2015. In 2017, the US Food and Drug Administration broadened the DR indication, approving ranibizumab for the treatment of DR with or without DME. Evidence to support these approvals included the RIDE and RISE Phase III clinical trials (NCT00473382 and NCT00473330), which demonstrated that intravitreous ranibizumab every 4 weeks resulted in substantial visual acuity (VA) gains, on average, in patients with DME over 2 years, which were maintained through year 3.6,7 Additional supporting evidence for the broader DR indication was provided by the Diabetic Retinopathy Clinical Research (DRCR) Network Protocol S study (NCT01489189), which demonstrated the efficacy of ranibizumab for DR in patients with and without DME.8
A model created by Varma et al based on the RIDE/RISE data and DRCR Network trials estimated that, in Hispanic and non-Hispanic white individuals with center-involved DME, monthly ranibizumab 0.3 mg for 2 years potentially would reduce the number with visual impairment (VA worse than 20/40 in the better-seeing eye) by 45% (95% simulation interval [SI], 36–53%) and the number with legal blindness (VA 20/200 or worse in the better-seeing eye) by 75% (95% SI, 58–88%).9 The study reported here uses the same model developed by Varma et al9 to estimate the potential impact of monthly ranibizumab (0.3 or 0.5 mg) on improvement or worsening of DR severity, rather than the resultant vision loss, in Hispanic and non-Hispanic white persons in the United States with center-involved DME causing vision impairment. An assessment of the effect of treatment on DR severity is important because of the well-documented relationship between increasing DR severity and health-related quality of life scores.10–12 Increasing DR severity can have an impact on vision-related quality of life before changes in VA can be detected.12
Materials and Methods
Hispanic and non-Hispanic white persons in the United States in 2010 aged ≥45 years with center-involved DME with VA 20/32 or worse who would be eligible for ranibizumab treatment (Figure 1) were included based on the model previously described by Varma et al.9 The model included only Hispanic and non-Hispanic white persons with access to health insurance (and hence, presumably to ranibizumab treatment) because the incidence of DME was not available in other racial or ethnic groups. The present study simulated the DR severity levels of these persons over 2 years in the better-seeing eye only. Because this study was a population-based simulation, neither institutional review board nor ethics committee approval was required.
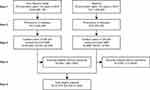 |
Figure 1 Estimated total number of Hispanic and non-Hispanic white persons aged ≥45 years with DR in the presence of center-involved DME causing vision of 20/32 or worse who would be considered for ranibizumab treatment in the United States in 2010. Numbers in parentheses represent the 95% SI. Reprinted from Ophthalmology, 122(5), Varma R, Bressler NM, Doan QV, et al. Visual impairment and blindness avoided with ranibizumab in Hispanic and non-Hispanic white persons with diabetic macular edema in the United States. 982–989, Copyright (2015), with permission from Elsevier.9Abbreviations: DME, diabetic macular edema; DR, diabetic retinopathy; SI, simulation interval. |
Estimate of Persons with Center-Involved DME for Whom Ranibizumab Treatment Would Be Considered
The total number of Hispanic and non-Hispanic white persons with DR in the presence of center-involved DME with vision impairment in the United States was estimated using a stepwise method (Figure 1).
First, 2010 US Census Bureau data were used to stratify non-Hispanic white and Hispanic persons aged ≥45 years into 10-year age groups.13,14 In step 2, the race/ethnic and age group–specific prevalence of self-reported diabetes mellitus was determined from National Health and Nutrition Examination Survey 2005–2008 data.15 In step 3, the 1-year incidence of center-involved DME for non-Hispanic white persons was determined from the Wisconsin Epidemiologic Study of Diabetic Retinopathy (WESDR)16 and for Hispanics from the Los Angeles Latino Eye Study (LALES).17 The proportion of incident eyes with center-involved DME with VA 20/32 or worse Snellen equivalent (best-corrected VA letter score ≤78) was estimated using data from LALES for both Hispanic and non-Hispanic white persons because equivalent data were not available from WESDR. Patients with DME that did not involve the center of the macula or with VA better than 20/32 Snellen equivalent in the incident eye were excluded because this was not the population evaluated in the studies used for this modeling (RIDE/RISE, Protocol I [NCT00444600], and Protocol S). In step 4, the percentage of uninsured persons by age and race/ethnic groups was determined from 2010 US Census Bureau data.18,19 Persons without health insurance were excluded, based on the assumption that they would not have access to ranibizumab. Lastly (step 5), the total number of persons eligible for treatment was derived by summing across all age and race/ethnic groups. This final number was used to simulate treatment with and without ranibizumab 0.3 and 0.5 mg.
Estimated Rates of DR Severity Level Change and Selected Clinical Events
The model was conducted as a 2-dimensional Monte Carlo simulation to account for various sources of patient-level variability and parameter uncertainty. To achieve stable rates, 300 averages of 215 simulated patients based on the size of the trial population were sampled using TreeAge Pro (TreeAge Software, Inc., Williamstown, MA, USA). The population-level simulation was carried out in @Risk 6.1.1 (Palisade Corporation, Ithaca, NY, USA) using 10,000 iterations. For each simulated patient, the DR severity level of the incident eye at baseline was sampled from the DR severity level distribution of patients in the DRCR Network (Protocol I; Table 1).6,13,15–22 The DR severity level change over 2 years was conditioned on the patient’s baseline DR severity level. Changes in DR severity level (scale is shown in Table 2)23,24 of the incident eye after 2 years were estimated, with and without monthly ranibizumab 0.3 or 0.5 mg, using patient-level data from the RIDE/RISE trials.6 The model also tracked any vitreous hemorrhage, panretinal photocoagulation (PRP), or vitrectomy for PDR, which are hereafter referred to as “clinical events related to PDR” because these events could represent a clinically relevant worsening of DR severity to the proliferative form in the absence of photographic documentation. DR severity in the fellow (non-study) eye was not evaluated.
 |
Table 1 Specification for Model Parameter Inputs |
 |
Table 2 Definition of DR Severity Level Based on the ETDRS-DRSS Scoring Using Fundus Photographya |
To determine the population-level impact of treatment for each outcome, the difference in the number of patients with and without ranibizumab was calculated along with the percentage change. The risk of death was applied using base US age-specific mortality rates adjusted for additional mortality risk for persons with diabetes.22 The simulation also accounted for the patient’s risk of treatment discontinuation every 4 weeks. While off treatment, it was assumed that there were no further changes in DR severity level; as such, the DR severity level remained constant from the time of discontinuation to month 24.
Outcomes Examined
The outcomes evaluated by the model are detailed in Table 3. The primary analysis was conducted with 0.3-mg treatment, with the results for 0.5-mg treatment provided in Table S1. As previously reported, the RIDE/RISE DR severity outcomes for the 2 doses were similar.24,25
 |
Table 3 DR Severity Outcomes for Ranibizumab 0.3 mg at 2 Years Based on Baseline DR Severity |
The 4 outcomes judged to be most clinically relevant were included in the analysis. The first and second outcomes were the number of persons with moderately severe NPDR or less severe DR (level ≤5) at baseline who were predicted to worsen to severe NPDR (level ≥6) or PDR (level ≥7) over 2 years. The third outcome was the number of persons without PDR (level ≤6) at baseline who were predicted to worsen to PDR (level ≥7) over 2 years. The fourth outcome was the estimated number of persons with severe NPDR or better (level ≤6) at baseline who were predicted to improve by ≥2 DR severity levels over 2 years. In addition to the specified level of DR severity worsening/improvement, all outcomes were considered in combination with the occurrence of the aforementioned clinical events related to PDR. Results for other outcomes are provided in Table S2.
Results
As previously reported, the model estimated that 37,274 Hispanic and non-Hispanic white persons in the United States in 2010 aged ≥45 years with DR and center-involved DME causing vision impairment could be eligible for ranibizumab treatment (Figure 1).9 Treatment with monthly ranibizumab 0.3 mg could potentially result in improvements for all DR-related outcomes examined in the model (Table 4, Table S2).
 |
Table 4 DR Progression-Related Outcomes for Ranibizumab 0.3 mg versus No Ranibizumab at 2 Years |
Without ranibizumab treatment, 469 persons (95% SI, 209–832) with moderately severe NPDR or less severe DR (level ≤5) at baseline were predicted to develop severe NPDR or more severe DR (level ≥6) and potentially experience a clinical event related to PDR over 2 years. Treatment with monthly ranibizumab 0.3 mg could potentially reduce this number to 20 (95% SI, 0–81), reducing the number of patients experiencing this level of DR severity worsening to 450 (95% SI, 187–807; value rounded up, see Table 4); equivalent to a 95% reduction over 2 years (95% SI, 80–100%; Table 4).
Worsening of DR severity from moderately severe NPDR or less severe DR (level ≤5) at baseline to PDR (level ≥7) plus a clinical event related to PDR over 2 years was predicted in 437 persons (95% SI, 197–778) who did not receive ranibizumab. Treatment with monthly ranibizumab 0.3 mg was predicted to reduce this number to 19 (95% SI, 0–81), potentially reducing the number of patients experiencing this level of DR severity worsening by 418 (95% SI, 173–758); equivalent to a 95% (95% SI, 79–100%) reduction over 2 years (Table 4).
The model predicted that 604 persons (95% SI, 289–1021) who did not receive ranibizumab would worsen from severe NPDR or less severe DR (level ≤6) at baseline to PDR (level ≥7) and potentially experience a clinical event related to PDR over 2 years. Treatment with monthly ranibizumab 0.3 mg could potentially reduce this number to 19 (95% SI, 0–80), potentially reducing the number of patients experiencing this level of DR severity worsening (95% SI, 271–997) by 585; equivalent to a 97% (95% SI, 85–100%) reduction over 2 years (Table 4).
With monthly ranibizumab 0.3 mg, 13,042 persons (95% SI, 8199–18,279) with severe NPDR or less severe DR (level ≤6) at baseline were expected to improve by ≥2 DR severity levels over 2 years. This is compared with 1706 persons (95% SI, 989–2617) without ranibizumab, such that ranibizumab treatment resulted in an estimated 6-fold (682%; 95% SI, 478–967%) improvement over 2 years (Table 4). Similar patterns were observed for monthly ranibizumab 0.5 mg across all levels of severity of DR that were examined (Table S1).
Discussion
This study, based on a previously developed population-based model,9 predicts that treatment with monthly ranibizumab 0.3 or 0.5 mg has the potential to substantially reduce the number of patients experiencing worsening DR severity within 2 years among the 37,274 Hispanic and non-Hispanic white persons with center-involved DME for whom ranibizumab treatment would be considered. Furthermore, the model predicts that treatment of these patients with ranibizumab could markedly increase the number of patients experiencing improvement in DR severity.
The results of this study are supported by an exploratory analysis of data from the RIDE/RISE trials, which demonstrated that ranibizumab reduced the risk of worsening of DR severity, with improvement in DR severity seen in many patients.24,25 Among patients who received ranibizumab 0.3 or 0.5 mg every 4 weeks (n=468), 13.2% and 14.5% of patients, respectively, had an ≥3 Early Treatment Diabetic Retinopathy Study severity level improvement from baseline at 2 years, compared with 1.3% of sham patients (n=239; P<0.001). Furthermore, 37.2% and 35.9% of patients, respectively, had an ≥2 Early Treatment Diabetic Retinopathy Study severity level improvement over the same period, compared with 5.4% of sham patients (P<0.001).24 Similar proportions of ranibizumab-treated patients maintained this level of improvement through to 3 years post baseline.25 Recently, the clinical relevance of these changes has been explored in a post hoc analysis of patients enrolled in the RIDE/RISE trials, showing that improvements in DR severity were associated with greater VA gains, improved contrast sensitivity, and increased likelihood of resolution of macular edema.26
In addition, an exploratory analysis by Bressler et al from the DRCR Network Protocol I study of 792 eyes from patients with center-involved DME causing VA impairment suggested that ranibizumab (0.5 mg every 4 weeks for 12 weeks, then as needed with either prompt or deferred [≥24 weeks] laser) was also associated with a reduced risk of DR worsening in eyes with and without PDR versus sham with prompt laser.27 Similar findings were noted in uncontrolled case series with ranibizumab28,29 as well as other anti-VEGF agents used for treatment of DME.30 Additionally, the 2-year results of the DRCR Network Protocol S study comparing PRP with ranibizumab treatment for PDR demonstrated non-inferiority of ranibizumab (0.5 mg up to every 4 weeks based on a structured re-treatment program) to PRP (completed in 1–3 visits, plus ranibizumab for DME); notably, 53% of patients in the PRP group received ranibizumab injections for DME.8 The findings of this study suggest that anti-VEGF therapy with ranibizumab may have additional utility as a treatment option for PDR when it has developed, including preservation of peripheral visual field that occurs following PRP and a marked reduction in the occurrence of macular edema in patients with PDR without concurrent macular edema at the time of diagnosis.
Results from LALES demonstrated that patients with more severe DR had worse health-related quality of life scores, based on results from the National Eye Institute Visual Function Questionnaire-25 and Medical Outcomes Study 12-Item Short Form Health Survey.12 A steep decline in patient-reported visual function (driving difficulty) and health-related quality of life was noted once both eyes were affected by moderate NPDR (level 4). Results from WESDR demonstrated that the National Eye Institute Visual Function Questionnaire-25 score was associated most closely with central vision.11 As such, these results indicate that DR impacts patient-reported visual function, which may be related, at least in part, to vision loss. A decline in vision-related functioning with increasing DR severity can be detected before changes in VA can be measured.12 A series of focus groups involving patients with mild, moderate, or severe NPDR, or PDR highlighted a substantial impact of DR-associated visual impairment on independence, mobility, leisure, and self-care activities (including diabetes management).10 As such, preserving visual function through reducing DR worsening or improving DR severity with ranibizumab for treatment of DME has the potential to greatly improve patient well-being and function (potentially reducing health care resource utilization).31
This study is subject to several limitations. The model only included data from Hispanic and non-Hispanic white persons with center-involving DME and estimated their response in the best-seeing eye only based on the results of randomized controlled clinical trials of ranibizumab. Consequently, it is not known whether the findings can be generalized to other patient groups, other anti-VEGF agents, other settings, or analyses based on the worse-seeing eye. It should be noted, however, that the groups included do comprise a large percentage of the overall population and there is no evidence of a difference in response to anti-VEGF therapy based on ethnicity32,33 or presence versus absence of DME.8 The results of the Protocol T study also suggest that the different anti-VEGF agents have similar effects on DR severity level.34 Secondly, the model did not differentiate between patients with type 1 versus type 2 diabetes. Patients with either type 1 or type 2 diabetes are commonly included in clinical trials, and there is no evidence of a differential response to treatment based on diabetes type. Thirdly, only incident (not prevalent) cases of DR were included; considering prevalent cases should increase the number of patients achieving the study outcomes substantially. The model only considered incident cases of center-involved DME with DR for 1 year (2010), meaning that cases that may have arisen in the second year were not included. Fourthly, only patients with health insurance were included in the study, which could have resulted in an underestimation of the number of patients who have access to ranibizumab treatment after introduction of the Affordable Care Act. Fifthly, although the model took into account the patients’ risk of ranibizumab discontinuation every 4 weeks, it was assumed that there were no further changes in DR severity level from the time of discontinuation through month 24. In Protocol I, Protocol S, and the RIDE/RISE open-label extension, DR severity improvements with ranibizumab were maintained with a marked reduction in treatment frequency for long periods of time (up to 5 years in Protocol I) after the end of monthly treatment, suggesting that the risk of progression may remain low for up to 18 months after treatment discontinuation.8,35,36 This may not be true for all patients, or among different anti-VEGF agents, however, and should be considered a limitation of the study. Sixthly, the model did not include the potential effect of adverse effects from the injections. In these studies, the adverse event rate was low and unlikely to have a substantial effect on DR severity. Finally, this study did not examine the quality of life or economic impacts of monthly ranibizumab injections for the patient versus the benefits of the effects of treatment on DR worsening.
In conclusion, the findings of this simulation model show that, in patients with center-involved DME causing vision impairment, ranibizumab has the potential to reduce the number of patients with worsening DR severity, and to increase the number of patients with improvements in DR severity, within the US Hispanic and non-Hispanic white populations. This compliments and builds upon our previous study that estimated that ranibizumab treatment could significantly reduce the number of these patients developing visual impairment or progressing to legal blindness.9 Together, these impacts have the potential to improve daily functioning and independence in patients, many of whom may be of working age. It might also reduce the need for PRP or other interventions required for the treatment of worsening DR severity and its complications.37 The potential economic impact and overall cost-effectiveness remain to be investigated.
Abbreviations
DME, diabetic macular edema; DR, diabetic retinopathy; DRCR, Diabetic Retinopathy Clinical Research; LALES, Los Angeles Latino Eye Study; NPDR, non-proliferative diabetic retinopathy; PDR, proliferative diabetic retinopathy; PRP, panretinal photocoagulation; SI, simulation interval; VA, visual acuity; VEGF, vascular endothelial growth factor; WESDR, Wisconsin Epidemiologic Study of Diabetic Retinopathy.
Previous Presentation
American Diabetes Association 72nd Annual Scientific Sessions, Philadelphia, PA, USA, June 8–12, 2012 (preliminary analysis).
Acknowledgments
Funding was provided by Genentech, Inc., a member of the Roche Group, for the study and third-party writing assistance, which was provided by Emily Howard, PhD, and Charlie Bellinger, BSc, of Envision Pharma Group.
Author Contributions
All authors contributed to study conception and design, or the acquisition, analysis, or interpretation of the data. All authors also contributed to the drafting and revision of the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
Funding was provided by Genentech, Inc., a member of the Roche Group, for the study and third-party writing assistance, which was provided by Emily Howard, PhD, and Charlie Bellinger, BSc, of Envision Pharma Group. The sponsor participated with investigators to address study design and conduct; data analysis; interpretation of results; and preparation, review, and approval of the manuscript.
Disclosure
RV is a consultant for Aerie (Irvine, CA, USA), Allergan (Irvine, CA, USA), Aquesys (Aliso Viejo, CA, USA), Bausch + Lomb (Rochester, NY, USA), and Genentech, Inc. (South San Francisco, CA, USA); and has received an unrestricted departmental grant from Research to Prevent Blindness (New York, NY, USA). NMB is principal investigator of grants at Johns Hopkins University; grants to investigators at Johns Hopkins University are negotiated and administered by the School of Medicine, which receives the grants through the Office of Research Administration. Individual investigators who participate in sponsored projects are not directly compensated by the sponsor, but may receive salary or other support from the institution to support their effort on the projects. He also has an independent contract agreement at Johns Hopkins University with the American Medical Association (Chicago, IL, USA). He is also sponsored by Bayer (Wayne, NJ, USA), Genentech, Inc./Roche (South San Francisco, CA, USA), the National Institutes of Health (Bethesda, MD, USA), Novartis Pharma AG (Basel, Switzerland), and Samsung Bioepis (Seoul, Korea). QVD is an employee of Outcomes Insights, Inc. (Westlake Village, CA, USA); and consultant for and reports grants from Genentech, Inc. (South San Francisco, CA, USA). IJS is a consultant for Genentech, Inc. (South San Francisco, CA, USA), Optos (Dunfermline, UK), Pfizer (New York, NY, USA), and Regeneron (Tarrytown, NY, USA); has received financial support from Genentech, Inc. (South San Francisco, CA, USA); and been on the medical advisory board for Optos (Dunfermline, Scotland). MD is an employee of Outcomes Insights, Inc. (Westlake Village, CA, USA); and consultant for and reports grants from Genentech, Inc. (South San Francisco, CA, USA). CMD is a consultant for Genentech, Inc. (South San Francisco, CA, USA), Gilead (Foster City, CA, USA), Myokardia, Inc., Iconic Therapeutics (South San Francisco, CA, USA), Portola Pharmaceuticals, Inc. (South San Francisco, CA, USA), Relypsa (Redwood City, CA, USA), Halozyme (San Diego, CA, USA), and Regenxbio (Rockville, MD, USA). AL is an employee of Outcomes Insights, Inc. (Westlake Village, CA, USA); and consultant for Genentech, Inc. (South San Francisco, CA, USA). JSE was an employee of Genentech, Inc. (South San Francisco, CA, USA) at the time of the study; and holds stock/stock options in the Roche Group (Basel, Switzerland); currently an employee of Kodiak Sciences Inc (Palo Alto, CA, USA). YR is an employee of Genentech, Inc. (South San Francisco, CA, USA); and holds stock/stock options in the Roche Group (Basel, Switzerland). The authors report no other conflicts of interest in this work.
References
1. Klein BE. Overview of epidemiologic studies of diabetic retinopathy. Ophthalmic Epidemiol. 2007;14(4):179–183. doi:10.1080/09286580701396720
2. World Health Organization. Prevention of blindness from diabetes mellitus: report of a WHO consultation in Geneva, Switzerland, 9–11 November 2005. Available from: http://www.who.int/blindness/Prevention%20of%20Blindness%20from%20Diabetes%20Mellitus-with-cover-small.pdf.
3. Yau JW, Rogers SL, Kawasaki R, et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012;35(3):556–564. doi:10.2337/dc11-1909
4. Zhang X, Saaddine JB, Chou CF, et al. Prevalence of diabetic retinopathy in the United States, 2005-2008. JAMA. 2010;304(6):649–656. doi:10.1001/jama.2010.1111
5. Centers for Disease Control and Prevention. National diabetes statistics report. 2014. Available from: https://www.cdc.gov/diabetes/pdfs/data/2014-report-estimates-of-diabetes-and-its-burden-in-the-united-states.pdf.
6. Nguyen QD, Brown DM, Marcus DM, et al. Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology. 2012;119(4):789–801. doi:10.1016/j.ophtha.2011.12.039
7. Brown DM, Nguyen QD, Marcus DM, et al. Long-term outcomes of ranibizumab therapy for diabetic macular edema: the 36-month results from two phase III trials: RISE and RIDE. Ophthalmology. 2013;120(10):2013–2022. doi:10.1016/j.ophtha.2013.02.034
8. Gross JG, Glassman AR, Jampol LM, et al. Panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy: a randomized clinical trial. JAMA. 2015;314(20):2137–2146. doi:10.1001/jama.2015.15217
9. Varma R, Bressler NM, Doan QV, et al. Visual impairment and blindness avoided with ranibizumab in Hispanic and non-Hispanic whites with diabetic macular edema in the United States. Ophthalmology. 2015;122(5):982–989. doi:10.1016/j.ophtha.2014.12.007
10. Coyne KS, Margolis MK, Kennedy-Martin T, et al. The impact of diabetic retinopathy: perspectives from patient focus groups. Fam Pract. 2004;21(4):447–453. doi:10.1093/fampra/cmh417
11. Klein R, Moss SE, Klein BE, Gutierrez P, Mangione CM. The NEI-VFQ-25 in people with long-term type 1 diabetes mellitus: the Wisconsin Epidemiologic Study of Diabetic Retinopathy. Arch Ophthalmol. 2001;119(5):733–740. doi:10.1001/archopht.119.5.733
12. Mazhar K, Varma R, Choudhury F, et al. Severity of diabetic retinopathy and health-related quality of life: the Los Angeles Latino Eye Study. Ophthalmology. 2011;118(4):649–655. doi:10.1016/j.ophtha.2010.08.003
13. US Census Bureau. 2010 Census summary file 1. 2010 Census of population and housing. 2012. Available from: http://www.census.gov/prod/cen2010/doc/sf1.pdf.
14. US Census Bureau. American fact finder. Available from: http://factfinder.census.gov/faces/nav/jsf/pages/searchresults.xhtml?refresh=t.
15. Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey (NHANES) data, 2005–2006 and 2007–2008. 2012. Available from: http://www.cdc.gov/nchs/nhanes.htm.
16. Klein R, Klein BE, Moss SE, Cruickshanks KJ. The Wisconsin Epidemiologic Study of Diabetic Retinopathy, XV: the long-term incidence of macular edema. Ophthalmology. 1995;102(1):7–16. doi:10.1016/S0161-6420(95)31052-4
17. Varma R, Choudhury F, Klein R, Chung J, Torres M, Azen SP. Four-year incidence and progression of diabetic retinopathy and macular edema: the Los Angeles Latino Eye Study. Am J Ophthalmol. 2010;149(5):
18. US Census Bureau. Current Population Survey (CPS) 2011 table creator. Health insurance coverage for “white alone & non-Hispanic,” and “white alone & Hispanic.” 2012. Available from: http://www.census.gov/cps/data/cpstablecreator.html.
19. US Census Bureau. Current Population Survey (CPS) 2011 Annual Social and Economic (ASEC) supplement. 2011. Available from: http://www.census.gov/prod/techdoc/cps/cpsmar11.pdf.
20. Elman MJ, Aiello LP, Beck RW, et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010;117(6):1064–1077.e35. doi:10.1016/j.ophtha.2010.02.031
21. Centers for Disease Control and Prevention. National Vital Statistics Reports: United States life tables, 2006. 2010. Available from: http://www.cdc.gov/nchs/products/life_tables.htm.
22. Gregg EW, Gu Q, Cheng YJ, Narayan KM, Cowie CC. Mortality trends in men and women with diabetes, 1971 to 2000. Ann Intern Med. 2007;147(3):149–155. doi:10.7326/0003-4819-147-3-200708070-00167
23. Early Treatment Diabetic Retinopathy Study Research Group. Grading diabetic retinopathy from stereoscopic color fundus photographs—an extension of the modified Airlie House classification. ETDRS report number 10. Ophthalmology. 1991;98(5 Suppl):786–806. doi:10.1016/S0161-6420(13)38012-9
24. Ip MS, Domalpally A, Hopkins JJ, Wong P, Ehrlich JS. Long-term effects of ranibizumab on diabetic retinopathy severity and progression. Arch Ophthalmol. 2012;130(9):1145–1152. doi:10.1001/archophthalmol.2012.1043
25. Ip MS, Domalpally A, Sun JK, Ehrlich JS. Long-term effects of therapy with ranibizumab on diabetic retinopathy severity and baseline risk factors for worsening retinopathy. Ophthalmology. 2015;122(2):367–374. doi:10.1016/j.ophtha.2014.08.048
26. Ip MS, Zhang J, Ehrlich JS. The clinical importance of changes in diabetic retinopathy severity score. Ophthalmology. 2017;124(5):596–603. doi:10.1016/j.ophtha.2017.01.003
27. Bressler SB, Qin H, Melia M, et al. Exploratory analysis of the effect of intravitreal ranibizumab or triamcinolone on worsening of diabetic retinopathy in a randomized clinical trial. JAMA Ophthalmol. 2013;131(8):1033–1040. doi:10.1001/jamaophthalmol.2013.4154
28. Kernt M, Cserhati S, Seidensticker F, et al. Improvement of diabetic retinopathy with intravitreal ranibizumab. Diabetes Res Clin Pract. 2013;100(1):e11–e13. doi:10.1016/j.diabres.2013.01.006
29. Messias A, Ramos Filho JA, Messias K, et al. Electroretinographic findings associated with panretinal photocoagulation (PRP) versus PRP plus intravitreal ranibizumab treatment for high-risk proliferative diabetic retinopathy. Doc Ophthalmol. 2012;124(3):225–236. doi:10.1007/s10633-012-9322-5
30. Simó R, Hernández C. Intravitreous anti-VEGF for diabetic retinopathy: hopes and fears for a new therapeutic strategy. Diabetologia. 2008;51(9):1574–1580. doi:10.1007/s00125-008-0989-9
31. Chen E, Looman M, Laouri M, et al. Burden of illness of diabetic macular edema: literature review. Curr Med Res Opin. 2010;26(7):1587–1597. doi:10.1185/03007995.2010.482503
32. Wong TY, Klein R, Islam FM, et al. Diabetic retinopathy in a multi-ethnic cohort in the United States. Am J Ophthalmol. 2006;141(3):446–455. doi:10.1016/j.ajo.2005.08.063
33. Roy MS, Klein R. Macular edema and retinal hard exudates in African Americans with type 1 diabetes: the New Jersey 725. Arch Ophthalmol. 2001;119(2):251–259.
34. Bressler SB, Liu D, Glassman AR, et al. Change in diabetic retinopathy through 2 years: secondary analysis of a randomized clinical trial comparing aflibercept, bevacizumab, and ranibizumab. JAMA Ophthalmol. 2017;135(6):558–568. doi:10.1001/jamaophthalmol.2017.0821
35. Boyer DS, Nguyen QD, Brown DM, et al. Outcomes with as-needed ranibizumab after initial monthly therapy: long-term outcomes of the phase III RIDE and RISE trials. Ophthalmology. 2015;122(12):2504–2513.e1. doi:10.1016/j.ophtha.2015.08.006
36. Bressler SB, Odia I, Glassman AR, et al. Changes in diabetic retinopathy severity when treating diabetic macular edema with ranibizumab: DRCR.net Protocol I 5-year report. Retina. 2018;38(10):1896–1904. doi:10.1097/IAE.0000000000002302
37. Kiss S, Chandwani HS, Cole AL, Patel VD, Lunacsek OE, Dugel PU. Comorbidity and health care visit burden in working-age commercially insured patients with diabetic macular edema. Clin Ophthalmol. 2016;10:2443–2453. doi:10.2147/OPTH.S114006
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
