Back to Journals » Drug Design, Development and Therapy » Volume 13
Dexmedetomidine Attenuates Neuroinflammation In LPS-Stimulated BV2 Microglia Cells Through Upregulation Of miR-340
Authors Bao Y, Zhu Y, He G, Ni H, Liu C, Ma L, Zhang L, Shi D
Received 29 March 2019
Accepted for publication 30 August 2019
Published 3 October 2019 Volume 2019:13 Pages 3465—3475
DOI https://doi.org/10.2147/DDDT.S210511
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Qiongyu Guo
Yang Bao,1,* Yijun Zhu,1,* Guangbao He,1 Hongwei Ni,1 Chenxia Liu,1 Limin Ma,1 Lifeng Zhang,2 Dongping Shi1
1Department of Anesthesiology, Jiading District Central Hospital Affiliated Shanghai University of Medicine & Health Sciences, Shanghai 201800, People’s Republic of China; 2Department of Anesthesiology, Jiading Maternal and Child Health Hospital, Shanghai 201821, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Dongping Shi
Department of Anesthesiology, Jiading District Central Hospital Affiliated Shanghai University of Medicine & Health Sciences, No.1 Chengbei Road, Jiading District, Shanghai, Shanghai 201800, People’s Republic of China
Email [email protected]
Lifeng Zhang
Department of Anesthesiology, Jiading Maternal and Child Health Hospital, No 1216 Gaotai Road, Jiading District, Shanghai, Shanghai 201821, People’s Republic of China
Email [email protected]
Background: Dexmedetomidine (Dex) was reported to exhibit anti-inflammatory effect in the nervous system. However, the mechanism by which Dex exhibits anti-inflammation effects on LPS-stimulated BV2 microglia cells remains unclear. Thus, this study aimed to investigate the role of Dex in LPS-stimulated BV2 cells.
Methods: The BV2 cells were stimulated by lipopolysaccharides (LPS). BV2 cells were infected with short-hairpin RNAs targeting NF-κB (NF-κB-shRNAs) and NF-κB overexpression lentivirus, respectively. In addition, miR-340 mimics or miR-340 inhibitor was transfected into BV2 cells, respectively. Meanwhile, the dual-luciferase reporter system assay was used to explore the interaction of miR-340 and NF-κB in BV2 cells. CCK-8 was used to detect the viability of BV2 cells. In addition, Western blotting was used to detect the level of NF-κB in LPS-stimulated BV2 cells. The levels of TNF-α, IL-6, IL-1β, IL-2, IL-12, IL-10 and MCP-1 in LPS-stimulated BV2 cells were measured with ELISA.
Results: The level of miR-340 was significantly upregulated in Dex-treated BV2 cells. Meanwhile, the level of NF-κB was significantly increased in BV2 cells following infection with lenti-NF-κB, which was markedly reversed by Dex. LPS markedly increased the expression of NF-κB and proinflammatory cytokines in BV2 cells, which were reversed in the presence of Dex. Moreover, miR-340 mimics enhanced the anti-inflammatory effects of Dex in LPS-stimulated BV2 cells via inhibiting NF-κB and proinflammatory cytokines. Furthermore, Dex obviously inhibited LPS-induced phagocytosis in BV2 cells.
Conclusion: Taken together, our results suggested that Dex might exert anti-inflammatory effects in LPS-stimulated BV2 cells via upregulation of miR-340. Therefore, Dex might serve as a potential agent for the treatment of neuroinflammation.
Keywords: NF-κB, dexmedetomidine, BV2 microglia cells, postoperative cognitive dysfunction
Introduction
Postoperative cognitive dysfunction (POCD), a common clinical complication, was generated in patients who suffer the anesthesia and hypoxic intervals during surgery.1 The morbidity of POCD was 16–40% at 7 days after surgery in middle‐aged and elderly patients.2 POCD could influence information processing speed, memory and executive function in patients, especially in middle‐aged and elderly patients.3,4 In addition, POCD severely reduced the quality of life and increased death rate in patients.5 Thus, it is imminently for us to take preventive measures to decrease the incidence.
It has been reported that POCD was related to an elevated level of neuroinflammation.3 Neuroinflammation induced by surgical trauma leads to POCD.6 Microglia, a resident macrophage in the central nervous system, plays an important role in regulating immune response and neuronal homeostasis.7 Microglia was activated and then secreted several pro-inflammatory cytokines during the occurrence of inflammation.7 In addition, nuclear factor (NF)-κB is a transcription factor, which plays an important role in the inflammatory response of microglia cells.8 NF-κB could regulate multiple inflammatory cytokines including IL-2, IL-6, IL-1β and TNF-α.9,10 Zhang et al indicated that surgery could increase the levels of NF-κB, IL-6 and IL-1β in rats.11 However, inhibition of NF-κB could attenuate neuroinflammation and succedent POCD.12 Previous study indicated that suppression of NF-κB signaling pathway could alleviate POCD after nerve anesthesia.13
Dexmedetomidine (Dex) is a selective α2-adrenergic agonist, which exhibits a neuroprotective role.14 Dex was used as an auxiliary sedative or an agent from mechanical ventilation.15 Previous study found that Dex could decrease the duration of mechanical ventilation and the morbidity of POCD.16,17 In addition, Dex could alleviate POCD in rats via regulating cAMP signaling pathway.18 Moreover, Li et al found that Dex decreased the production of inflammatory factors and reduced postoperative recurrent rate in patients with POCD.19
In spite of many reports indicated that Dex exerted a protective effect of the nervous system via inhibiting inflammatory response,20,21 little is known about the relationship of Dex and NF-κB pathway. In addition, previous study reported that POCD was associated with microRNAs (miRNAs).22 Therefore, the present study aimed to investigate the neuroprotective effect of Dex in LPS-stimulated BV2 cells from the perspective of miRNAs.
Materials And Methods
Cell Culture
Due to the low cell number and time-consuming techniques needed to cultivate primary microglia cultures, the immortalized microglia BV2 cell line has been used extensively in the study associated with neurodegenerative disorders.23 We used lipopolysaccharide (LPS) to simulate BV2 microglia cells to mimic the inflammatory environment in the brain.24 The BV2 cell line was purchased from Conservation Genetics CAS Kunming Cell Bank (Kunming, China). BV2 cells were cultured in DMEM medium with 10% fetal bovine serum (FBS, Thermo Fisher Scientific, Waltham, MA, USA) and penicillin-streptomycin (100 μg/mL, Thermo Fisher Scientific) at 37°C with 5% CO2. Dex was obtained from Sigma-Aldrich (St. Louis, MO, USA).
CCK-8 Assay Of Cell Viability
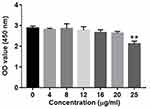
Cell viability was measured by cell counting kit-8 (CCK-8, Sigma) according to the specification. BV2 cells were seed into 96-well plates at a density of 5000 cells per well overnight at 37°C. Then, cells were treated with Dex (0, 4, 8, 12, 16, 20 or 25 μg/mL) for 48 hrs at 37°C. Later on, after incubation with CCK-8 reagent (10 μL) for 2 hrs, microplate reader (Bio-Rad Laboratories, Hercules, CA, USA) was used to measure the absorbance of BV2 cells at a wavelength of 450 nm.
Lentiviral Construction And Cell Infection
The NF-κB sequence was synthesized by GenePharma (Shanghai, China). The NF-κB-shRNA1, NF-κB-shRNA2 and NF-κB-shRNA3 plasmids were purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA) and then sub-cloned into the pWPXL (lentiviral expression vector). 293T cells were co-transfected with pWPXL-NF-κB, NF-κB-shRNAs plasmids or pSuper-puro-GFP vector, respectively. Forty-eight hours later, the supernatant of cells was collected.
BV2 cells were infected with 1×107 NF-κB lentivirus or NF-κB lentivirus-shRNAs transducing units containing 5 µg/mL polybrene (Sigma) for 48 hrs, respectively. For stably infected cells, BV2 cells were treated with puromycin (2.5 µg/mL, Invitrogen) for 72 hrs. Real-time qPCR and Western blotting assays were used to measure the expression of NF-κB in the BV2 cells.
Cell Transfection
BV2 cells (4×105 cells per well) were seed onto 6-well plates overnight to reach 70% confluence for transfection. Then, miR-340 mimic or miR-34 inhibitor was transfected into cells for 24 hrs using Lipofectamine 2000 (Thermo Fisher Scientific) according to the manufacturer’s instructions. After that, transfected cells were treated with 20 μg/mL Dex for another 24 hrs at 37°C.
Real-Time qPCR
Trizol kit (Thermo Fisher Scientific) was used to obtain the total RNA according to the manufacturer’s procedure. Then, cDNA was synthesized by using a cDNA synthesis kit (Thermo Fisher Scientific). After that, SYBR premix Ex Taq II kit (TaKaRa, Dalian, China) was used to perform the RT-qPCR reaction on an ABI 7900HT instrument (ABI, NY, USA) according to the manufacturer’s procedure. The qPCR reaction conditions were as follows: 94°C for 3 mins followed by 45 cycles of 94°C for 45 s, 56°C for 30 s, finally, 72°C for 45 s. NF-κB: F: 5ʹ-ACGATCTGTTTCCCCTCATCT-3ʹ; R: 5ʹ-TGCTTCTCTCCCCAGGAATA-3ʹ, miR-572: F: 5ʹ-GCCAGATCTCTGAGGAAAGCAGGAGGAGG-3ʹ; R: 5ʹ-GCCGAATTCTCGGCACAAATCTTCAGAGC-3ʹ, miR-155: F: 5ʹ-GTAACCCGTTGAACCCCATT-3ʹ; R: 5ʹ-CCATCCAATCGGTAGTAGCG-3ʹ, miR-340: F: 5ʹ-GTGGGGTTATAAAGCAATGAGA-3ʹ; R: 5ʹ-GTGCAGGGTCCGAGGT-3ʹ, GAPDH: F: 5ʹ-CTGGGGACGACATGGAGAAAA-3ʹ; R: 5ʹ-AAGGAAGGCTGGAAGAGTGC-3ʹ. U6: F: 5ʹ-CTCGCTTCGGCAGCACAT-3ʹ; R: 5ʹ-AACGCTTCACGAATTTGCGT-3ʹ. The primers were obtained from GenePharma (Shanghai, China). The 2−ΔΔCt method was used to analyze the data (Supplementary figure 1).25 The level of NF-κB was normalized by internal control GAPDH.26,27 The levels of miR-572, miR-155 and miR-340 were normalized to internal control U6.
Western Blot Analysis
Total proteins were qualified by using a BCA protein assay kit (Thermo Fisher Scientific). Equal amounts of protein (30 μg per lane) were separated on 10% SDS-PAGE and then transferred onto PVDF membranes (Thermo Fisher Scientific). The membranes were blocked with 5% skim milk for 1 hr at room temperature and then incubated with the primary antibody overnight at 4°C. Primary antibodies: anti-NF-κB (Abcam, 1:1000) and anti-GAPDH (Abcam, 1:1000). After that, the membranes were incubated with anti-rabbit IgG secondary antibodies for 50 mins at room temperature. Later on, the blots were visualized by ECL reagent and then captured using the ChemiDoc™XRS+ imaging system (Bio-Rad, Hercules, CA, USA).
Luciferase Reporter Assay
Reporter plasmids of wild-type (WT)-NF-κB and mutant (MT)-NF-κB were synthesized by GenePharma. BV2 cells were cultured to reach 70% confluence. The designed WT-NF-κB or MT-NF-κB and corresponding plasmids (miR-340 mimics or miR-340 inhibitor) were then co-transfected into BV2 cells for 48 hrs using Lipofectamine 2000. The luciferase report activity was measured with the Dual-Luciferase Reporter Assay System (Promega). The rellina luciferase activity was normalized to control group.
Cytokine Analysis By ELISA
BV2 cells (4 ×105 cells per well) were seeded onto 6-well plates overnight at 37°C. The levels of TNF-α, IL-6, IL-1β, IL-2, IL-12, IL-10 and MCP-1 in the supernatant were measured with ELISA kits (KAINOS laboratories, Japan) in accordance with the manufacturer’s instructions.
Phagocytosis Assay
BV2 cells were incubated with FITC-labeled latex beads (Sigma Aldrich, St. Louis, MO, USA) for 1 hr at 37°C. Then, the cells were washed in PBS and analyzed using the Cell Quest Software (BD Biosciences, San Jose, CA) on a FACS Calibur flow cytometer (BD Biosciences).
Statistical Analysis
Each group was performed at least three independent experiments and all data were expressed as the mean ± standard deviation (SD). SPSS v21.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism v5.0 (GraphPad Software, Inc., La Jolla, CA, USA) were used to analyze the experimental data. The comparison between two groups was analyzed by Student’s t-test. The comparisons among multiple groups were made with one-way analysis of variance (ANOVA) followed by Tukey’s test. P<0.05 was considered to indicate a statistically significant difference.
Results
The Effect Of Dex On The Viability Of BV2 Cells
CCK-8 assay was applied to determine the effect of Dex on the viability of BV2 cells. As shown in Figure 1, 25 μg/mL Dex significantly decreased the viability of BV2 cells. Meanwhile, 20 μg/mL Dex had a very limited effect on the proliferation of BV2 cells. Therefore, 20 μg/mL Dex was utilized in the following experiments.
Dex Down-Regulated The Expression Of NF-κB In BV2 Cells
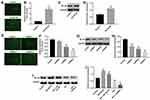
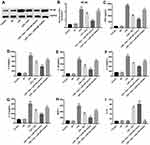
It has been reported that NF-κB plays an important role in neuroinflammation and POCD.28 In order to investigate the role of NF-κB in BV2 cells, cells were infected with lentivirus-NF-κB (lenti-NF-κB) or NF-κB-shRNAs. As illustrated in Figure 2A, BV2 cells were effectively infected with lenti-NF-κB after 48 hrs of incubation. In addition, RT-qPCR and Western blotting indicated that the level of NF-κB was significantly upregulated following transfection with lenti-NF-κB, compared with control group (Figure 2B–D). Moreover, the results indicated that the expression of NF-κB was decreased most significantly in BV2 cells after infection with NF-κB-shRNA3 compared with the control group and other NF-κB-shRNAs (Figure 2E–H). Thus, NF-κB shRNA3 was utilized in the following experiments.
To further investigate the association between the NF-κB gene and Dex in BV2 cells, Western blot was used. The result indicated the level of NF-κB was significantly increased following transfection with lenti-NF-κB, which was markedly reversed by Dex (Figure 2I and J). In addition, Dex notably decreased the expression of NF-κB in cells, which was further decreased following infection with NF-κB-shRNA3 (Figure 2I and J). These results indicated that Dex could down-regulate NF-κB gene expression in BV2 cells.
miR-340 Mimics Enhanced The Anti-Inflammatory Effect Of Dex In BV2 Cells Via Inhibiting NF-κB
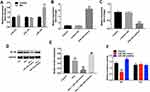
Previous studies indicated that some miRNAs were associated with the occurrence of POCD, such as miR-572 and miRNA-155.29,30 In addition, miR-340 was related to the activity of NF-κB.31,32 Meanwhile, it has been shown that miR-340 could inhibit the progression of glioblastoma cells.33 Therefore, we next explored the association between NF-κB gene and miRNAs in BV2 cells. As demonstrated in Figure 3A, the level of miR-340 was significantly upregulated in Dex-treated BV2 cells. In addition, the level of miR-340 was markedly increased in miR-340 mimics group, while its level was significantly decreased in the miR-340 inhibitor group (Figure 3B and C).
Next, as shown in Figure 3D and E, Dex significantly decreased the expression of NF-κB in BV2 cells, which were further markedly reduced following transfection with miR-340 mimics. However, Dex-induced NF-κB protein decrease in cells was markedly inhibited by miR-340 inhibitor (Figure 3D and E). In addition, the luciferase assay data indicated that the reduced luciferase activity was observed in BV2 cells following transfection with NF-κB-WT and miR-340 mimics, while the increased luciferase activity was observed in BV2 cells following transfection with NF-κB-WT and miR-340 inhibitor (Figure 3F). These data suggested that overexpression of miR-340 could enhance the anti-inflammatory effect of Dex in BV2 cells via inhibiting NF-κB.
Overexpression Of miR-340 Enhanced The Inhibitory Effect Of Dex on NF-κB Pathway-Associated Cytokines In BV2 Cells
Next, ELISA assay was used to detect the levels of NF-κB pathway-associated cytokines in BV2 cells. As shown in Figure 4A–E, NF-κB pathway-associated pro-inflammatory cytokines TNF-α, IL-6, IL-1β, IL-2 and IL-12 were significantly decreased in Dex-treated BV2 cells, which were further reduced following transfection with miR-340 mimics. However, Dex-induced pro-inflammatory cytokine decreases were markedly reversed following transfection with miR-340 inhibitor. These data suggested that overexpression of miR-340 enhanced the inhibitory effect of Dex on NF-κB pathway-associated cytokines in BV2 cells.
Overexpression Of miR-340 Enhanced The Anti-Inflammatory Effect Of Dex In LPS-Stimulated BV2 Cells
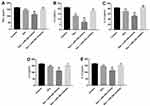
Next, to further investigate the effects of Dex on LPS-induced neuroinflammation, Western blot and ELISA assays were performed. As shown in Figure 5A–H, LPS significantly increased the expression of NF-κB and the production of pro-inflammatory cytokines TNF-α, IL-6, IL-1β, IL-2, IL-12 and MCP-1 in BV2 cells. However, the expression of NF-κB and the levels of pro-inflammatory cytokines were markedly decreased in the presence of Dex, which was further enhanced following transfection with miR-340 mimics. Meanwhile, the production of anti-inflammatory cytokine IL-10 was markedly upregulated in the presence of Dex on LPS-stimulated BV2 cells, which was further increased following transfection with miR-340 mimics (Figure 5I). These data indicated that overexpression of miR-340 could enhance the anti-inflammatory effect of Dex in LPS-stimulated BV2 cells.
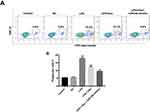
Dex Inhibited LPS-Induced Phagocytosis In BV2 Cells
Further, we examined whether Dex could regulate phagocytosis in BV2 cells. The experiment was performed using fluorescent latex beads in order to induce detectable engulfment by BV2 cells. As shown in Figure 6A and B, the number of phagocytic cells was obviously increased in the LPS group, compared with the control group. However, the number of phagocytic cells was significantly decreased in the presence of Dex, which was further enhanced following transfection with miR-340 mimics (Figure 6A and B). These data indicated that Dex could inhibit LPS-induced phagocytosis in BV2 cells.
Discussion
Previous study demonstrated that surgical could trigger systemic and neuroinflammation, which could induce POCD.34 It has been shown that Dex could alleviate POCD.35 Our results indicated that Dex reduced NF-κB level in LPS-stimulated BV2 cells. In addition, overexpression of miR-340 could enhance the anti-inflammatory effect of Dex in LPS-stimulated BV2 cells via inhibiting NF-κB (Figure 7).
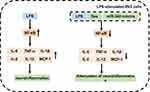 |
Figure 7 MiR-340 mimics exhibited anti-inflammation effect via downregulation of NF-κB pathway. The mechanism by which miR-340 mimics protected BV2 cells against LPS is shown in the figure. |
In neuroinflammatory process, NF-κB plays a key role in neuroinflammation-mediated neurodegeneration.36 Upregulation of NF-κB is closely related to the occurrence of Alzheimer’s disease, and inhibition of NF-κB might be a therapeutic method to treat Alzheimer’s disease.37 It has been indicated that proinflammatory cytokines, such as TNF-α, IL-1β, IL-6 and MCP-1, play an important role in the NF-κB signaling pathway.38 NF-κB could activate the proinflammatory cytokines TNF-α, IL-1β, IL-6 and MCP-1 in cells, which could induce inflammation response.38 Previous studies indicated that the levels of proinflammatory cytokines IL-12 and IL-6 were upregulated, and the level of anti-inflammatory cytokine IL-10 was downregulated in LPS-stimulated BV2 cells.39,40 High levels of pro-inflammatory cytokines in BV2 cells could contribute directly to neuroinflammation.40 In the present study, Dex significantly decreased the expression of NF-κB in LPS-stimulated BV2 cells. Meanwhile, LPS significantly increased the production of TNF-α, IL-6, IL-1β, IL-2, IL-12 and MCP-1 in BV2 cells. Chen et al found that Dex reduces lung injury via inhibiting NF-κB signaling pathway, which was consistent with our results.41 These data indicated that Dex could decrease neuroinflammation in LPS-stimulated BV2 cells via inhibiting NF-κB.
Evidence indicates that miRNAs play an important role in inflammation response, which could mediate gene expression and function.42 Overexpression of miR-146a could alleviate inflammation response in mice with POCD, as evidenced by a decreased level of NF-κB and proinflammatory cytokines (IL-1β and IL-6).43 However, upregulation of miR-30b could induce cognitive dysfunction in AD via increasing the level of NF-κB.44 Previous study indicated that overexpression of miR-340 could alleviate inflammation as well as the levels of TNF-α and IL-6 in the model of chronic constriction injury.45 Meanwhile, miR-340 could inhibit the progression of glioblastoma cells.33 It has been reported that NF-κB was demonstrated to regulate miR-340.32 Currently, no reports indicated the relationship of miR-340 and NF-κB in LPS-stimulated BV2 cells. By analyzing the effects of miR-340 up and down-regulation in BV2 cells, we found that overexpression of miR-340 markedly decreased the level of NF-κB, according to the luciferase assay results. In this study, we found that Dex significantly increased the level of miR-340, which plays an anti-inflammatory role in BV2 cells. In addition, our results showed that the levels of NF-κB and pro-inflammatory cytokines were significantly decreased by Dex in the LPS-stimulated BV2 cells, which were further downregulated following transfection with miR-340 mimics. Our study confirmed that overexpression of miR-340 enhanced the anti-inflammatory effect of Dex in LPS-stimulated BV2 cells by decreasing the level of NF-κB and NF-κB pathway-related proinflammatory cytokines (Figure 7). The protective effect is on the basis of the inhibition of NF-κB pathway.
The limitation in our study is that no behavior tests were performed, and so we do not know whether miR-340 are associated with POCD. In addition, more inflammatory proteins, such as TLR4 and MyD88, are needed to further confirm the roles of Dex in regulating neuroinflammation in LPS-stimulated BV2 cells.
Conclusion
Our data demonstrated that Dex exerts an anti-inflammatory effect in LPS-stimulated BV2 cells. In addition, overexpression of miR-340 could enhance the anti-inflammatory effect of Dex in LPS-stimulated BV2 cells via inhibiting NF-κB pathway. Therefore, Dex may serve as a potential agent for the treatment of neuroinflammation.
Acknowledgments
This study was supported by Shanghai Medical Guidance Project Fund (No. 16411973300) and Shanghai University of Medicine & Health Sciences Fund (SFP-18-20-16-002).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Almahozi A, Radhi M, Alzayer S, Kamal A. Effects of memantine in a mouse model of postoperative cognitive dysfunction. Behav Sci (Basel). 2019;9(3).
2. Monk TG, Weldon BC, Garvan CW, et al. Predictors of cognitive dysfunction after major noncardiac surgery. Anesthesiology. 2008;108(1):18–30. doi:10.1097/01.anes.0000296071.19434.1e
3. Quan C, Chen J, Luo Y, et al. BIS-guided deep anesthesia decreases short-term postoperative cognitive dysfunction and peripheral inflammation in elderly patients undergoing abdominal surgery. Brain Behav;2019. e01238. doi:10.1002/brb3.1238
4. Rundshagen I. Postoperative cognitive dysfunction. Dtsch Arztebl Int. 2014;111(8):119–125. doi:10.3238/arztebl.2014.0119
5. Tang Y, Ouyang W. [Inflammation-relevant mechanisms for postoperative cognitive dysfunction and the preventive strategy]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2017;42(11):1321–1326. doi:10.11817/j.issn.1672-7347.2017.11.013
6. Zhou H, Luo T, Wei C, et al. RAGE antagonism by FPSZM1 attenuates postoperative cognitive dysfunction through inhibition of neuroinflammation in mice. Mol Med Rep. 2017;16(4):4187–4194. doi:10.3892/mmr.2017.7074
7. Huang MY, Tu CE, Wang SC, et al. Corylin inhibits LPS-induced inflammatory response and attenuates the activation of NLRP3 inflammasome in microglia. BMC Complement Altern Med. 2018;18(1):221. doi:10.1186/s12906-018-2317-3
8. Feng N, Huang X, Jia Y. Mycoplasma contamination affects cell characteristics and decreases the sensitivity of BV2 microglia to LPS stimulation. Cytotechnology. 2019;71(2):623–634. doi:10.1007/s10616-019-00311-8
9. Yan N, Wen DS, Zhao YR, Xu SJ. Epimedium sagittatum inhibits TLR4/MD-2 mediated NF-kappaB signaling pathway with anti-inflammatory activity. BMC Complement Altern Med. 2018;18(1):303. doi:10.1186/s12906-018-2317-3
10. Liu X, Ye F, Xiong H, et al. IL-1beta induces IL-6 production in retinal Muller cells predominantly through the activation of p38 MAPK/NF-kappaB signaling pathway. Exp Cell Res. 2015;331(1):223–231. doi:10.1016/j.yexcr.2014.08.040
11. Zhang J, Jiang W, Zuo Z. Pyrrolidine dithiocarbamate attenuates surgery-induced neuroinflammation and cognitive dysfunction possibly via inhibition of nuclear factor kappaB. Neuroscience. 2014;261:1–10. doi:10.1016/j.neuroscience.2013.12.034
12. Li Z, Ni C, Xia C, et al. Calcineurin/nuclear factor-kappaB signaling mediates isoflurane-induced hippocampal neuroinflammation and subsequent cognitive impairment in aged rats. Mol Med Rep. 2017;15(1):201–209. doi:10.3892/mmr.2016.5967
13. Zheng JW, Meng B, Li XY, et al. NF-kappaB/P65 signaling pathway: a potential therapeutic target in postoperative cognitive dysfunction after sevoflurane anesthesia. Eur Rev Med Pharmacol Sci. 2017;21(2):394–407.
14. Tu Y, Liang Y, Xiao Y, et al. Dexmedetomidine attenuates the neurotoxicity of propofol toward primary hippocampal neurons in vitro via Erk1/2/CREB/BDNF signaling pathways. Drug Des Devel Ther. 2019;13:695–706. doi:10.2147/DDDT.S188436
15. Thompson RZ, Gardner BM, Autry EB, Day SB, Krishna AS. Survey of the current use of dexmedetomidine and management of withdrawal symptoms in critically ill children. J Pediatr Pharmacol Ther. 2019;24(1):16–21. doi:10.5863/1551-6776-24.1.16
16. Newman S, Stygall J, Hirani S, Shaefi S, Maze M. Postoperative cognitive dysfunction after noncardiac surgery: a systematic review. Anesthesiology. 2007;106(3):572–590. doi:10.1097/00000542-200703000-00023
17. Mansouri N, Nasrollahi K, Shetabi H. Prevention of cognitive dysfunction after cataract surgery with intravenous administration of midazolam and dexmedetomidine in elderly patients undergoing cataract surgery. Adv Biomed Res. 2019;8:6. doi:10.4103/abr.abr_190_18
18. Zhu YS, Xiong YF, Luo FQ, Min J. Dexmedetomidine protects rats from postoperative cognitive dysfunction via regulating the GABAB R-mediated cAMP-PKA-CREB signaling pathway. Neuropathology. 2019;39(1):30–38. doi:10.1111/neup.12530
19. Li XT, Jiang XM, Zheng ZY, Huang HS. [Effect of dexmedetomidine on inflammatory factors level and cognitive function after femoral head replacement in elderly patients]. Zhongguo Gu Shang. 2018;31(12):1091–1095. doi:10.3969/j.issn.1003-0034.2018.12.003
20. Gong Z, Li J, Zhong Y, Guan X, Huang A, Ma L. Effects of dexmedetomidine on postoperative cognitive function in patients undergoing coronary artery bypass grafting. Exp Ther Med. 2018;16(6):4685–4689. doi:10.3892/etm.2018.6778
21. Tang W, Wang J, Fu GQ, Yuan L. [Effect of dexmedetomidine and midazolam on respiration and circulation functions in patients undergoing open heart surgery under acupuncture-assisted general anesthesia]. Zhen Ci Yan Jiu. 2014;39(3):216–221.
22. Wei C, Luo T, Zou S, et al. Differentially expressed lncRNAs and miRNAs with associated ceRNA networks in aged mice with postoperative cognitive dysfunction. Oncotarget. 2017;8(34):55901–55914. doi:10.18632/oncotarget.18362
23. Stansley B, Post J, Hensley K. A comparative review of cell culture systems for the study of microglial biology in Alzheimer’s disease. J Neuroinflammation. 2012;9:115. doi:10.1186/1742-2094-9-115
24. Zhang D, Li N, Wang Y, et al. Methane ameliorates post-operative cognitive dysfunction by inhibiting microglia NF-kappaB/MAPKs pathway and promoting IL-10 expression in aged mice. Int Immunopharmacol. 2019;71:52–60. doi:10.1016/j.intimp.2019.03.003
25. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25(4):402–408. doi:10.1006/meth.2001.1262
26. Zhang JY, Zhang F, Hong CQ, et al. Critical protein GAPDH and its regulatory mechanisms in cancer cells. Cancer Biol Med. 2015;12(1):10–22. doi:10.7497/j.issn.2095-3941.2014.0019
27. Paeschke N, von Haefen C, Endesfelder S, Sifringer M, Spies CD. Dexmedetomidine prevents lipopolysaccharide-induced MicroRNA expression in the adult rat brain. Int J Mol Sci. 2017;18:9. doi:10.3390/ijms18091830
28. Terrando N, Eriksson LI, Ryu JK, et al. Resolving postoperative neuroinflammation and cognitive decline. Ann Neurol. 2011;70(6):986–995. doi:10.1002/ana.22664
29. Wu C, Wang R, Li X, Chen J. Preoperative serum MicroRNA-155 expression independently predicts postoperative cognitive dysfunction after laparoscopic surgery for colon cancer. Med Sci Monit. 2016;22:4503–4508. doi:10.12659/msm.898397
30. Yu X, Liu S, Li J, et al. MicroRNA-572 improves early post-operative cognitive dysfunction by down-regulating neural cell adhesion molecule 1. PLoS One. 2015;10(2):e0118511. doi:10.1371/journal.pone.0118511
31. Li P, Sun Y, Liu Q. MicroRNA-340 induces apoptosis and inhibits metastasis of ovarian cancer cells by inactivation of NF-x03BA;B1. Cell Physiol Biochem. 2016;38(5):1915–1927. doi:10.1159/000445553
32. Zhou F, Wang W, Xing Y, et al. NF-kappaB target microRNAs and their target genes in TNFalpha-stimulated HeLa cells. Biochim Biophys Acta. 2014;1839(4):344–354. doi:10.1016/j.bbagrm.2014.01.006
33. Li X, Gong X, Chen J, et al. miR-340 inhibits glioblastoma cell proliferation by suppressing CDK6, cyclin-D1 and cyclin-D2. Biochem Biophys Res Commun. 2015;460(3):670–677. doi:10.1016/j.bbrc.2015.03.088
34. Chu JMT, Xiong W, Linghu KG, et al. Siegesbeckia Orientalis L. extract attenuates postoperative cognitive dysfunction, systemic inflammation, and neuroinflammation. Exp Neurobiol. 2018;27(6):564–573. doi:10.5607/en.2018.27.6.564
35. Fang S, Chen Y, Yao P, et al. [Dexmedetomidine alleviates postoperative cognitive dysfunction in aged rats probably via silent information regulator 1 pathway]. Nan Fang Yi Ke Da Xue Xue Bao. 2018;38(9):1071–1075. doi:10.12122/j.issn.1673-4254.2018.09.08
36. Husain I, Akhtar M, Vohora D, et al. Rosuvastatin attenuates high-salt and cholesterol diet induced neuroinflammation and cognitive impairment via preventing nuclear factor KappaB pathway. Neurochem Res. 2017;42(8):2404–2416. doi:10.1007/s11064-017-2264-2
37. Thawkar BS, Kaur G. Inhibitors of NF-kappaB and P2X7/NLRP3/Caspase 1 pathway in microglia: novel therapeutic opportunities in neuroinflammation induced early-stage Alzheimer’s disease. J Neuroimmunol. 2019;326:62–74. doi:10.1016/j.jneuroim.2018.11.010
38. He Z, Zhou Q, Wen K, et al. Huangkui lianchang decoction ameliorates DSS-induced ulcerative colitis in mice by inhibiting the NF-kappaB signaling pathway. Evid Based Complement Alternat Med. 2019;2019:1040847. doi:10.1155/2019/1040847
39. Kim DC, Quang TH, Oh H, Kim YC. Steppogenin isolated from cudrania tricuspidata shows antineuroinflammatory effects via NF-kappaB and MAPK pathways in LPS-stimulated BV2 and primary rat microglial cells. Molecules. 2017;22(12). doi:10.3390/molecules22122130
40. Yang XW, Li YH, Zhang H, et al. Safflower Yellow regulates microglial polarization and inhibits inflammatory response in LPS-stimulated Bv2 cells. Int J Immunopathol Pharmacol. 2016;29(1):54–64. doi:10.1177/0394632015617065
41. Chen H, Sun X, Yang X, et al. Dexmedetomidine reduces ventilator-induced lung injury (VILI) by inhibiting Toll-like receptor 4 (TLR4)/nuclear factor (NF)-kappaB signaling pathway. Bosn J Basic Med Sci. 2018;18(2):162–169. doi:10.17305/bjbms.2018.2400
42. Yu A, Zhang T, Zhong W, et al. miRNA-144 induces microglial autophagy and inflammation following intracerebral hemorrhage. Immunol Lett. 2017;182:18–23. doi:10.1016/j.imlet.2017.01.002
43. Chen L, Dong R, Lu Y, et al. MicroRNA-146a protects against cognitive decline induced by surgical trauma by suppressing hippocampal neuroinflammation in mice. Brain Behav Immun. 2019. doi:10.1016/j.bbi.2019.01.020
44. Song Y, Hu M, Zhang J, Teng ZQ, Chen C. A novel mechanism of synaptic and cognitive impairments mediated via microRNA-30b in Alzheimer’s disease. EBioMedicine. 2019;39:409–421. doi:10.1016/j.ebiom.2018.11.059
45. Gao L, Pu X, Huang Y, Huang J. MicroRNA-340-5p relieved chronic constriction injury-induced neuropathic pain by targeting Rap1A in rat model. Genes Genomics. 2019. doi:10.1007/s13258-019-00802-0
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.