Back to Journals » International Journal of Women's Health » Volume 12
Description of Maternal Morbidities Amongst 1000 Women During Pregnancy in Ambanja, Madagascar – Opportunities and Challenges of Using an mHealth System
Authors Garcia Vilaplana E , Petignat P , Benski AC, Soaroby A, Sormani J, Vassilakos P, Schmidt NC
Received 18 May 2020
Accepted for publication 8 August 2020
Published 12 October 2020 Volume 2020:12 Pages 823—833
DOI https://doi.org/10.2147/IJWH.S260894
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Everett Magann
Elisabeth Garcia Vilaplana,1,2 Patrick Petignat,2 Anne-Caroline Benski,2– 4 Adelia Soaroby,4 Jessica Sormani,2 Pierre Vassilakos,2,5 Nicole C Schmidt2,6
1Faculty of Medicine, University of Geneva, Geneva, Switzerland; 2Department of Paediatrics, Gynaecology and Obstetrics, University Hospitals of Geneva, Geneva, Switzerland; 3Harvard T.H. Chan School of Public Health, Boston, Massachusetts, USA; 4Centre Medical St Damien, Ambanja, Madagascar; 5Geneva Foundation for Medical Education and Research, Geneva, Switzerland; 6Catholic University of Applied Science, Munich, Germany
Correspondence: Nicole C Schmidt
Catholic University of Applied Science, Preysingstraße 95, Munich 81667, Germany
Tel +49 89 48092-8499
Email [email protected]
Background: The estimated burden of maternal morbidities in lower-income countries, such as Madagascar, is high. However, there is still a lack of data on maternal morbidities, in part due to an absence of standardized assessment tools. This cross-sectional study aims to report maternal morbidities among 1015 women in the district of Ambanja, Madagascar, and to describe the advantages and limitations of a mHealth system.
Methods: Data were collected using the PANDA (pregnancy and newborn diagnosis assessment) system, an mHealth device that incorporates the WHO recommendations for antenatal care (ANC). Data, including personal and medical information, but also clinical data such as hypertension, anemia or HIV were collected from more than 1000 women attending ANC.
Results: A total of 1015 pregnant women were recruited from January 2015 to August 2018. The average age was 24.6 years old, and most women were married (82.3%). The majority lived in urban areas and were unemployed. Prevalence of hypertension and gestational diabetes was relatively low (4% vs 2.2%). Malaria infection was diagnosed in 2.2% and HIV was diagnosed in 1.2% of pregnant women. The most common morbidity was anemia (68.4%) and the only significant factor associated was being single compared to being married (OR 1.68, 95% CI 1.05– 2.70, p-value 0.032).
Discussion: The prevalence of anemia in our study population was much higher than previously reported in Sub-Saharan Africa. This finding highlights the need for regular iron supplementation during pregnancy, especially in vulnerable (eg, single) women. The PANDA mHealth system provides unique opportunities due to its standardization of ANC and creation of a digital database accessible from a distance. However, one of the main challenges was that even a mHealth system such as the PANDA remains dependent on the local procurement chain. Therefore, future studies need to access opportunities of mHealth systems to support health service delivery.
Trial Registration: Registered on ISRCTN on 14th October 2015, number ISRCTN18270380. Retrospectively registered.
Keywords: maternal health, maternal deaths, anemia, antenatal care
Plain Language Summary
There is a lack of data on maternal morbidities in low-income countries such as Madagascar. In this study, a previously tested standardized, digital, mobile tool (mHealth system) was used to record prenatal data amongst 1015 women in Ambanja, Madagascar. The main aims of the study were to describe a) the maternal morbidities and b) to discuss advantages and limitations of the mobile Health system.
The data recorded included personal and medical information, as well as screening data on for example hypertension, anemia, HIV, syphilis or malaria. Data used for the study were taken from women’s first antenatal visit. All pregnant women, regardless of age or stage of pregnancy, were eligible to participate, as long as they were able to understand and act accordingly.
The average age of women was 24.6 years and the majority were married, unemployed and living in urban areas. The most common morbidity was anemia (68.4%) and the only significant associated factor was being single compared to being married. Other maternal morbidities such as hypertension, malarial infection and gestational diabetes had a prevalence of less than five percent.
In conclusion, the study showed that even more than two-thirds of the study population were anemic. As one of the likely causes of anemia is iron deficiency, health campaigns should focus on regular iron supplementation and nutritional support during pregnancy in this population, especially in vulnerable (eg, single) women.
Furthermore, the study demonstrated that a mHealth device such as the PANDA system provides unique opportunities due to its standardization of ANC and the possibility to follow-up patients from a distance but also to its creation of clinical and digital records. However, one of the main challenges was that even a mHealth system such as the PANDA remains dependent on the local procurement chain. Therefore, future studies need to access opportunities of mHealth systems to support health service delivery.
Introduction
Maternal mortality is one of the main indicators of maternal health, and its reduction has been highlighted since the development of the Millennium Development Goals.1 According to the World Health Organization (WHO), 340 000 women died worldwide in 2017 and the risk of a woman dying from a maternal-related cause was 40 times higher in a developing country than in a developed country.1 In Madagascar, maternal mortality continues to be high, with a maternal mortality rate of 353 per 100,000 live births in 2015.2 This condition has been attributed in part to the inadequate access to assisted deliveries but also to the insufficient quality of antenatal care (ANC).3 In 2018, the latest Demographic and Health Surveys for Madagascar reported that only 51% of women attended four or more ANC consultations.4 This is especially important as WHO recommended in 2016 a minimum of eight antenatal care contacts to reduce perinatal mortality and improve women’s experience of care.5
In recent years, more attention has been paid to maternal morbidities. One reason for this is that for each maternal death, an estimated 20–30 women will suffer from associated morbidities. Therefore, WHO suggested recently to focus more on standardizing and measuring non-life-threatening maternal morbidities. The WHO Maternal Morbidity Working Group (MMWG) provided a more expansive definition of maternal morbidities as “any health condition attributed to and/or aggravated by pregnancy and childbirth that has a negative impact on the woman’s wellbeing and/or functioning”.6,7 This expansion includes besides the previous used life-threatening (severe) conditions also the non-life-threatening (non-severe) maternal morbidities.6 To facilitated measuring of the non-severe maternal morbidities a comprehensive list of 121 direct and indirect conditions were suggested.8 As several of those conditions such as anemia, diabetes or infectious diseases (such as syphilis) can be detected, prevented or treated during ANC, measuring those non-severe morbidities can serve as an indicator for the quality of obstetric care.9,10
Therefore, objectives of the study were:
1) To estimate the prevalence of non-severe maternal morbidities in Ambanja, Madagascar, in more than 1000 women attending ANC consultations. Furthermore, to measure associations of indirect maternal conditions and demographic factors or obstetric history.
2) To describe the advantages and limitations of using a mHealth system called PANDA (Pregnancy and New-born Diagnosis Assessment) in the provision of a comprehensive antenatal care consultation and in collecting data on maternal morbidity conditions during the antepartum period.
Methods
A cross-sectional study was conducted following the principles of the Declaration of Helsinki from January 2015 to August 2018 in the district of Ambanja, Madagascar. The local health authorities in Ambanja and the Ethical Cantonal Board of Geneva, Switzerland (CER 14–217; project number CCER PB_2017-00641_Amend n°1) approved the study.
Study Setting and Collaboration
Ambanja is located approximately 500 km from Antananarivo, the capital of Madagascar. The district is subdivided into 21 municipalities, with an estimated 200,000 inhabitants, and is economically dependent on farming. The study was based at the CMC Saint Damien (Centre Médico-Chirurgical Saint Damien), a private non-profit clinic founded in 1988 that collaborates with the Health Ministry. The CMC provides ANC for the urban population at the main clinic and at 18 dispensaries in Ambanja District, which are located in a radius of 250 km in the rural areas.
The collaboration between the Geneva University Hospitals and the CMC Saint Damien Health Centre was established in 2010. This collaboration began with offering cervical cancer screening services at the hospital and the surrounding dispensaries. The collaboration has expanded to offering ANC with the acceptance and inauguration of the PANDA (Pregnancy and New-born Diagnostic Assessment) mHealth application in 2015.
Recruitment of Study Participants
ANC consultations were offered once or twice a week in the maternal health department and all pregnant women, regardless of age or stage of pregnancy, were eligible to participate in the study. The only exclusion criteria were inability to understand or act, as described in a previous publication explaining the acceptability and feasibility of the PANDA mHealth device.11 As the ANC using the PANDA mHealth system was offered to all women consulting for the routine ANC care, the sample is considered to be representative. More than 98% of invited women accepted to participate in the ANC using the PANDA mHealth device. Reasons for refusal were mainly related to time concerns as the ANC using the PANDA mHealth device requires more time than the routine ANC.
Data Collection
Data were collected by health providers using the PANDA mHealth system, a telemedicine device based on mobile technology, that ensures high-quality ANC according to WHO recommendations.5,11 The PANDA mHealth system, which has been described in detail in a previous publication11 includes a smartphone with an Android icon-based application that enables health-care personnel to collect patient information starting from the first ANC at the CMC St. Damian or affiliated facilities.11 The collection of patient data does not only allow a better clinical follow-up but it also permits to collect data on a population level. Especially, the latter has been highlighted to be important for LMIC such as Madagascar which fail to collect routinely reliable data.12,13
The interviews were conducted on a one-on-one basis by health professionals, mainly in the local language Sakalava, more rarely in French. Each interview contained four parts: (a) Personal data collection; b) Medical, surgical and obstetric history; c) Screening to detect obesity/malnutrition, hypertension/eclampsia, anemia, HIV, syphilis, malaria, diabetes, infections other condition d) Health education with a focus on birth preparedness.
Additional to the smartphone with the Android icon-based application, the PANDA includes a solar backpack with photovoltaic power that contains diagnostic devices to screen for blood pressure, fever, or to take measurements such as maternal height and weight as well as uterine fundal height. Furthermore, rapid tests were used to screen for Diabetes (ForaDuo ®), anemia (Bi-Trumed®), HIV, syphilis or malaria (SD Bioline ®). In case of a positive screening result, patients were transferred to the hospital laboratory for blood testing (such as hemagglutination for syphilis and thick blood smears for malaria) in alignment with the Ministry of Health guidelines. The diagnosis criteria for anemia and diabetes were used according to WHO reference values. Lastly, the PANDA mHealth system collects standardized data and allows monitoring from distance using an electronic database.
Data Analysis
The collected data were examined by the principal investigator for completeness, and corrective measures were taken if necessary. Only data from each woman’s first ANC were used in the analysis. Data were coded, entered, cleaned and analyzed using Stata Data Analysis, Statistical Software Version 13 (Stata Corporation, College Station, TX, USA).
First, descriptive data analysis was done reporting frequencies (percentages) and median (interquartile range), but also missing data. WHO definitions were used as references for grouping quantitative data such as Hemoglobin levels into anemia categories.14–16 Secondly, univariable and multivariable logistic regression were carried out in case of outcomes differing from previous literature in Madagascar or Sub-Saharan-Africa, to identify associated factors. Independent variables were socio-demographic data such as age, education or marital status, but also obstetric factors, eg, parity, weeks of pregnancy, infection status or previous mode of delivery. Data were compared either as discrete variables using contingency table chi-squared tests or as continuous variables using two-sample t-test. All univariable and multivariable analyses were adjusted for trimester of pregnancy. Statistical significance was considered as p-value <0.05.
Results
Demographic Information of the Participants
During the study period from January 2015 to August 2018, a total of 1015 eligible women attending their first ANC visit using the PANDA mHealth system were included.
The average age of the participants was 24.6 years. The majority of women (74.7%) lived in urban areas, 82.3% were married and more than half were unemployed (57.6%). Educational status was low; over 50% had only started their primary education, and only 8.8% attended university. Table 1 summarizes further sociodemographic characteristics, including missing data.
 |
Table 1 First-Visit Sociodemographic Characteristics of Pregnant Women Followed in Ambanja District – Madagascar Between January 13, 2015, and August 16, 2018 |
Maternal Morbidity During Pregnancy
Participants coming for their first ANC using the PANDA mHealth system were, on average, in their 2nd trimester of pregnancy and most women were expecting in median their 2nd child (Table 2). More than three-quarters of the participants (79.2%) were diagnosed with at least one maternal morbidity. Details are illustrated in Table 3.
 |
Table 2 Obstetric Details of Pregnant Women from Their First Visit |
 |
Table 3 Screening of Pregnant Women at Their First Visit |
The prevalence of hypertension, defined as a systolic blood pressure ≥140 and/or a diastolic blood pressure ≥90, was four percent, and only four women (0.4%) were diagnosed with severe high blood pressure (≥160 mmHg systolic and/or ≥110 mmHg diastolic blood pressure).14 The prevalence of gestational diabetes, defined by a fasting blood glucose value of ≥126 mg/dl,15 was 2.2%.
Screening at the first ANC with the previously mentioned rapid tests revealed in twenty-five women (2.5%) a positive result for syphilis, 22 women (2.2%) were screened positive for an acute malarial infection and 12 women (1.2%) had an HIV positive rapid test.
According to the WHO’s expanded definition of morbidities, exposure to violence and self-reported substance abuse was questioned.6 Only 24 out of the 1015 (2.4%) women reported abuse of nicotine, alcohol or other substances. When women were asked “have you been subject to any violence at home”, 62 women (6.1%) reported having experienced some form of violence from their current partner or someone else.
The most common maternal morbidity among the 1015 women was anemia (68.4%, N= 849). The WHO criteria define anemia during pregnancy as a hemoglobin value <11 g/dl, with mild anemia from 10- <11 g/dl, moderate from 7-<10 g/dl, and severe from <7 g/dl.16 46.3% of the anemic women were diagnosed with mild anemia, 52.5% were diagnosed with moderate anemia and 1.2% presented with severe anemia (Table 3, Figure 1).
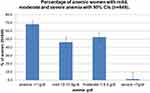 |
Figure 1 Percentage of anemic women, showing mild, moderate and severe anemia with 95% CIs (n=849). A total of 68.4% of women were anemic (the majority presented with moderate anemia). |
The overall prevalence of anemia in the study population was much higher than the prevalence of 38% previously described in Sub-Saharan Africa.17 Therefore, univariable and multivariable analyses were carried out to measure potential associations with demographic and obstetric factors and anemia. All univariable and multivariable analyses were adjusted for trimester of pregnancy, in order to adjust for a potential confounding effect of trimester of pregnancy on anemia.16
However, in the univariable and multivariable analyses, the only variable significantly associated with anemia during pregnancy was marital status (Table 4). Single women had a significantly higher risk of being anemic than married women (OR 1.68, 95% CI 1.08–2.60, p-value 0.020).
 |
Table 4 Risk Factors for Anemia During Pregnancy (No Anemia versus Mild, Moderate or Severe Anemia) (Univariable Analyses and Multivariable Model) |
Discussion
As previously mentioned by WHO, a sufficient number of antenatal care contacts can reduce perinatal mortality, detect maternal morbidities and improve women’s experience of care.5,18
The overall objective of the study was to describe maternal morbidities identified in the 1015 women attending ANC in Ambanja, Madagascar. Surprisingly, nearly 80% of pregnant women in our study presented with at least one maternal morbidity and 43% were diagnosed even with two maternal morbidities (Table 3). This figure is much higher compared to a previous study conducted among a total of 750 women attending ANC visits in Kenya, Jamaica and Malawi, in which only 38.4% of women were diagnosed with at least one morbidity.6
In the following, we will first focus on the most important non-severe maternal morbidities detected in our study. Secondly, we will describe the advantages and limitations of a mHealth system such as the PANDA device to register maternal morbidities.
Important Maternal Morbidities in the Study Population
While in contrast to previous studies the prevalence of hypertensive disorders and infectious diseases (such as syphilis) were relatively low (Table 3), the most important maternal morbidity was anemia, which was detected in 68.4% of pregnant women.3,6,19,20 This is much higher than the prevalence rates reported by previous studies among pregnant women in Madagascar (33%) or Sub-Saharan Africa (57%).19,21 More than half of the women were diagnosed with moderate anemia, while 1.2% presented even with severe anemia. This is in contrast to previous data, in which the majority of pregnant, anemic women in Madagascar, had been diagnosed with mild anaemia.3
According to WHO the most common form of anemia worldwide is iron deficiency anaemia,22 even if macronutrient deficiencies, hemoglobinopathy and infections also play a role. But, as the prevalence of infections was low in our population, this cause seems unlikely.
However, as nearly two-thirds of women were diagnosed with anemia, the access to and utilization of ANC is of great importance. At the time of women’s first antenatal care visit using the PANDA system 61.8% of women (Table 1) had not yet taken any iron supplementation. Furthermore, nearly 90% had their first ANC visit using the PANDA mHealth system only in their second or third trimester of pregnancy (57.7% in their 2nd trimester vs 30.3% in their 3rd trimester) (Table 2). Previous studies have reported that women especially in low- and middle-income countries do not meet the recommended iron intake before pregnancy due to inadequate diet.19 Therefore, as the first ANC visit often occurred only in the second trimester, and the majority had not taken iron supplementation, anemia had probably worsened.23 Therefore, the late start of iron supplementation seems the most likely explanation for the high prevalence of anemia. In consequence, incentives for women to start ANC consultations earlier in pregnancy could help to prevent anemia throughout the pregnancy. Additionally, women and families should receive nutritional education prior to and during pregnancy. This could also be helpful to address barriers related to unscientific beliefs against consuming medications during pregnancy, which have been published by Casey et al.24
Furthermore, as previous studies have reported a positive association of education with a reduced risk for anemia in pregnancy,19 multivariable analysis for a possible correlation between anemia and education were conducted. However, in our study, no significant association could be found; possibly because the level of education was generally low. But, importantly, multivariable analysis showed a significant association with anemia and marital status. Women who reported being single were 1.68 times more likely to be anemic (p=0.032, CI 1.05–2.70). Anecdotal reports from health-care providers in Ambanja stated that single pregnant women have a lower income and have a higher risk of receiving less support from family members. This is in accordance with international studies which found that unmarried pregnant women were significantly less likely to receive high-quality healthcare. The main reason was that women feared stigma or shame from their support network in respect to their current pregnancy.25
Hence, incentives to access ANC visits early in pregnancy should especially target the group of single women, which has been identified by WHO as vulnerable to suffer from health inequalities.
Advantages and Limitations of an mHealth System to Collect Maternal Morbidities
One of the first advantages of the system is that it supports health-care personal to provide standardized high-quality ANC visits in accordance with WHO recommendations. This is especially important, as countries, such as Madagascar, suffer an important lack of health professionals. Standardized mHealth systems support less skilled health personal or health professionals from other health disciplines.11,26
Second, the system allows recording and creating an electronic record for each patient, facilitating follow-up visits. Furthermore, large-scale databases are generated, giving the opportunity to follow-up maternal morbidities not only on the individual but also on the population level. The routine collection of data has been highlighted as important by previous studies, but several LMIC have difficulties to collect those data routinely. A study conducted by Mbondji and colleagues in 2014 in 14 Sub-Saharan countries of the WHO African region showed that even if data are crucial to improve health, several countries had low-performance levels especially in data management (eg, the ability to collect, store, analyze and distribute data). Data management systems such as the PANDA device support this collection of routine clinical data, including results of screening-tests (eg, for syphilis, malaria and HIV) to follow up health trends over time (eg, considering the endemic seasonal characteristics of malaria in this north-eastern area of Madagascar27).
Lastly, this telemedicine application gives the opportunity to follow and analyze at a distance, so decisions for improvement and prevention of maternal morbidities can be made.
Besides the advantages, one of the main challenges is that also mHealth systems such as the PANDA remain dependent on the local procurement chain. In our study prevalence rates for syphilis and HIV were different from previous studies. One reason is that an important number of women were not screened for HIV or syphilis due to stock-outs and logistical constraints. This holds true for the women consulting with the mHealth PANDA system, but also for the routine ANC. Stock-outs have been reported frequently in low- and middle-income countries, compromising individual health and resulting in relapse of disease.28,29
Therefore, more recently mHealth devices have been suggested as a communication solution to support delivery systems.30 For example, a study, conducted in 87 public health facilities in Kenya in 2017, showed a significant reduction in stock-outs of malaria treatment and diagnostic tests by implementing of a weekly reporting of facility stock-counts via structured short messages (SMS) from health-care workers to a web-based system accessed by managers.31
In conclusion, a possible option could be to implement a weekly stock-count with a digital reminder to order rapid tests at a defined stock.
Another challenge relates to the analyzed clinical data. Further information needs to be included to improve the understanding of associations such as details about nutrition or measuring the mid-upper arm circumference. Also, the follow-up of women during delivery and the postpartum period is important to assess associations such as preterm risk factors and neonatal outcomes. As Barreix and colleagues have mentioned, this contributes to the “targets envisioned by the Global Strategy for Women’s, Children’s and Adolescents’ Health” 2016–2030ʹ and the Sustainable Development Goals,6 which outline a “routine post-partum examination” and ‘postnatal contact with an appropriately skilled health-care provider, at home or in the health facility.32
Lastly, the presented study has some limitations in respect to its study design. First, a possible selection bias should be considered. Even if all women consulting for routine ANC were invited for ANC visits using the PANDA system, ANC visits using the device were not available daily and took longer than routine ANC. However, as our data for age of pregnancy at the first ANC or malaria prevalence were comparable to national data, the sample seems to be representative and selection bias is considered to be rather small.3
In addition, only the first ANC visit was included in the analysis. This could have affected the ability to capture the prevalence of some morbidities, such as underestimating hypertension, which often develops throughout pregnancy.
Another limitation of this cross-sectional study is its inability to distinguish between conditions that existed prior to pregnancy or developed during pregnancy. Therefore, a cohort study should be considered in the future.
Conclusion
In summary, the study showed that even if the prevalence of some maternal morbidities (such as hypertension or malaria) were relatively low, more than two-thirds of the study population were anemic. As anemia is most likely due to iron deficiency, improved access and utilization of iron supplementation, as well as nutritional support, especially targeting vulnerable women early in pregnancy should be highlighted.
Furthermore, the study demonstrated that a mHealth device such as the PANDA mHealth system provides unique opportunities due to it is a) standardization of ANC, b) the possibility to follow-up patients by creating a clinical record and c) the creation of a database allowing monitoring of individual clinical, but also population data from a distance.
However, improvements need to be considered to react even better to the local conditions and to respond more appropriately to the needs of mothers and newborns.
Abbreviations
ANC, antenatal care; CI, confidence intervals; CMC, Centre Médico Chirurgical; mHealth, mobile health; OR, odds ratio; PANDA, Pregnancy And New-born Diagnostic-Assessment; WHO, World Health Organization.
Data Sharing Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Ethical Approval and Informed Consent
The local health authorities in Ambanja, Madagascar and the Ethical Cantonal Board of Geneva, Switzerland (CER 14-217; project number CCER PB_2017-00641_Amend n°1) approved the study. All women included in the study gave their informed consent to participate.
Acknowledgments
The authors would like to acknowledge the health-care providers and staff of the Centre Saint Damien for their support, interest and collaboration throughout the duration of the study. We would especially like to thank Adelia, Rossina and Lucie for their assistance in recruiting and conducting the ANC visits. The authors would also like to thank Giovanna Stancanelli and the PANDA team for the opportunity to analyze the data collected in the mHealth intervention.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content, gave final approval of the version to be published; and agree to be accountable of all aspects of the work.
Funding
The statistical analysis performed in the study was supported by a grant received from the Latin branch of the Swiss Society of Gynecology and Obstetrics (GRSSGO). The funding body had no role in study design, data collection, analysis, interpretation, manuscript writing, or the decision to submit the manuscript for publication.
Disclosure
Nicole C Schmidt reports grants from Swiss Society of Gynecology and Obstetrics (GRSSGO), during the conduct of the study. The authors declare that they have no other potential competing interests for this work.
References
1. World Bank Group and the United Nations Population Division, World Health Organization. Trends in maternal mortality 2000 to 2017: estimates by WHO, UNICEF, UNFPA. Geneva; 2019. Available from: http://documents.worldbank.org/curated/en/793971568908763231/pdf/Trends-in-maternal-mortality-2000-to-2017-Estimates-by-WHO-UNICEF-UNFPA-World-Bank-Group-and-the-United-Nations-Population-Division.pdf.
2. WHO, UNICEf, Group WB, UNPD. Maternal mortality in 1990–2015: Madagascar; 2016. Available from: https://www.who.int/gho/maternal_health/countries/mdg.pdf?ua=1.
3. Sharp M, Kruse I. Health, nutrition, and population in Madagascar 2000–09. World Bank Working Paper no.2016. 1st ed; 2011. Available from: http://documents.worldbank.org/curated/en/684481468271245643/pdf/632550PUB0Heal0ID0185380BOX361512B0.pdf.
4. MICS 6, Madagascar 2018: santé maternelle et néonatale; 2018. Available from: https://www.unicef.org/madagascar/sites/unicef.org.madagascar/files/201910/MICS6Madagascar_Snapshot_Health_MaternalNewborn%2026072019%20-%20HQ_INSTAT.pdf.
5. World Health Organization. WHO Recommendations on Antenatal care for positive pregnancy experience; 2016. Available from: http://apps.who.int/iris/bitstream/10665/250796/1/9789241549912-eng.pdf.
6. Barreix M, Barbour K, McCaw-Binns A, et al. Standardizing the measurement of maternal morbidity: pilot study results. Int J Gynecol Obstet. 2018;141:10–19. doi:10.1002/ijgo.12464
7. Maternal Morbidity Working Group (MMWG),World Health Organization Department of Reproductive Health and Research. Maternal WOICE Tool: antenatal care. Pilot version A.18.07.A; 2018. Available from: http://origin.who.int/reproductivehealth/MM_ANC_questionnaire.pdf.
8. Chou D, Tunçalp Ö, Firoz T, et al. Constructing maternal morbidity – towards a standard tool to measure and monitor maternal health beyond mortality. BMC Pregnancy Childbirth. 2016;16(1).
9. Say L, Souza JP, Pattinson RC. Maternal near miss - towards a standard tool for monitoring quality of maternal health care. Best Pract Res Clin Obstet Gynaecol. 2009;23(3):287–296. doi:10.1016/j.bpobgyn.2009.01.007
10. Say L, Barreix M, Chou D, et al. Maternal morbidity measurement tool pilot: study protocol. Reprod Health. 2016;13(1):1–6. doi:10.1186/s12978-016-0164-6
11. Benski AC, Stancanelli G, Scaringella S, et al. Usability and feasibility of a mobile health system to provide comprehensive antenatal care in low-income countries: PANDA mHealth pilot study in Madagascar. J Telemed Telecare. 2016;23(5):536–543. doi:10.1177/1357633X16653540
12. Shaffer J, Doumbia S, Ndiaye D, et al. Development of a data collection and management system in West Africa: challenges and sustainability. Infect Dis Poverty. 2018;7(1). doi:10.1186/s40249-018-0494-4.
13. Mbondji P, Kebede D, Soumbey-Alley E, Zielinski C, Kouvividila W, Lusamba-Dikassa P. Resources, indicators, data management, dissemination and use in health information systems in sub-Saharan Africa: results of a questionnaire-based survey. J R Soc Med. 2016;107(1_suppl).
14. Magee LA, Pels A, Helewa M, et al. Diagnosis, evaluation, and management of the hypertensive disorders of pregnancy: executive summary. J Obstet Gynaecol Can. 2014;36(5):416–438. doi:10.1016/S1701-2163(15)30588-0
15. World Health Organization. Diagnostic criteria and classification of hyperglycaemia first detected in pregnancy. 2016. Available from: https://extranet.who.int/rhl/topics/preconception-pregnancy-childbirth-and-postpartum-care/antenatal-care/who-recommendation-diagnosis-gestational-diabetes-pregnancy-0.
16. World Health Organization. Haemoglobin concentrations for the diagnosis of anaemia and assessment of severity. Vitamin and Mineral Nutrition Information System. 2011. Available from: https://apps.who.int/iris/bitstream/handle/10665/85839/WHO_NMH_NHD_MNM_11.1_eng.pdf?ua=1.
17. Filippi V, Chou D, Ronsmans C, et al. Levels and causes of maternal mortality and morbidity. Black RE, Laxminarayan R, Temmerman M, et al. editors. Reproductive, Maternal, Newborn, and Child Health: Disease Control Priorities.
18. Geller SE, Koch AR, Garland CE, MacDonald EJ, Storey F, Lawton B. A global view of severe maternal morbidity: moving beyond maternal mortality. Reprod Health. 2018;15(Suppl 1). doi:10.1186/s12978-018-0527-2
19. Stephen G, Mgongo M, Hashim TH, Katanga J, Stray-pedersen B, Msuya SE. Anaemia in pregnancy: prevalence, risk factors, and adverse perinatal outcomes in Northern Tanzania. Anemia. 2018;2018:1–9. doi:10.1155/2018/1846280
20. Fokom-Domgue J, Noubiap JJN. Diagnosis of hypertensive disorders of pregnancy in sub-Saharan Africa: a poorly assessed but increasingly important issue. J Clin Hypertens. 2015;17(1):70–73. doi:10.1111/jch.12429
21. Wang W, Alva S, Wang S, Fort A Levels and trends in the use of maternal health services, DHS comparative reports 26. USAID; 2011. Available from: https://dhsprogram.com/pubs/pdf/CR26/CR26.pdf.
22. World Health Organization. Nutritional anaemias: tools for effective prevention and control. World Health Organization; 2017. Available from: https://apps.who.int/iris/handle/10665/259425.
23. Ononge S, Campbell O, Mirembe F. Haemoglobin status and predictors of anemia among pregnant women in Mpigi, Uganda. BMC Res Notes. 2014;7(1):1–8. doi:10.1186/1756-0500-7-712
24. Mbhenyane X, Cherane M. Compliance with the consumption of iron and folate supplements by pregnant women in Mafikeng local municipality, North West Province, South Africa. Afr Health Sci. 2017;17(3):657–670. doi:10.4314/ahs.v17i3.8
25. Smith-Greenaway E. Premarital childbearing in sub-Saharan Africa: can investing in women’s education offset disadvantages for children? SSM Popul Health. 2016;2:164–174. doi:10.1016/j.ssmph.2016.02.001
26. USAID. Global health | Madagascar | U.S. Agency for International Development. Usaid.gov; 2019. Available from: https://www.usaid.gov/madagascar/global-health.
27. Ihantamalala FA, Rakotoarimanana FMJ, Ramiadantsoa T, et al. Spatial and temporal dynamics of malaria in Madagascar. Malar J. 2018;17(1):1–13. doi:10.1186/s12936-018-2206-8
28. Ikoh MU, Udo AU, Charles AO, Charles JO. The influence of ‘’stock out” on health-seeking behaviour of low income women in Uyo urban, Akwa Ibom State, Nigeria. Int Q Community Health Educ. 2008–2009;29(3):257–273. doi:10.2190/IQ.29.3.e
29. Wagenaar B, Gimbel S, Hoek R, et al. Stock-outs of essential health products in Mozambique - longitudinal analyses from 2011 to 2013. Trop Med Int Health. 2014;19(7):791–801.
30. Mikkelsen-Lopez I, Shango W, Barrington J, Ziegler R, Smith T, Desavigny D. The challenge to avoid anti-malarial medicine stock-outs in an era of funding partners: the case of Tanzania. Malar J. 2014;13(1):1–9. doi:10.1186/1475-2875-13-181
31. Githinji S, Kigen S, Memusi D, et al. Reducing stock-outs of life saving malaria commodities using mobile phone text-messaging: SMS for life study in Kenya. PLoS One. 2013;8(1):1–8. doi:10.1371/journal.pone.0054066
32. World Health Organization. The Global Strategies for Women’s, Children’s and Adolescent’s Health (2016–2030). 2016. Available from: https://www.who.int/lifecourse/partners/globalstrategy/globalstrategyreport2016-2030-lowres.pdf.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
