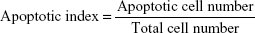Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 12
Decreased levels of irisin, a skeletal muscle cell-derived myokine, are related to emphysema associated with chronic obstructive pulmonary disease
Authors Sugiyama Y, Asai K , Yamada K, Kureya Y, Ijiri N, Watanabe T, Kanazawa H, Hirata K
Received 1 November 2016
Accepted for publication 30 January 2017
Published 2 March 2017 Volume 2017:12 Pages 765—772
DOI https://doi.org/10.2147/COPD.S126233
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Yukari Sugiyama, Kazuhisa Asai, Kazuhiro Yamada, Yuko Kureya, Naoki Ijiri, Tetsuya Watanabe, Hiroshi Kanazawa, Kazuto Hirata
Department of Respiratory Medicine, Graduate School of Medicine, Osaka City University, Osaka, Japan
Background: Cigarette smoking-induced oxidant–antioxidant imbalance is a factor that contributes to the pathogenesis of COPD through epithelial cell apoptosis. Irisin is a skeletal muscle cell-derived myokine associated with physical activity. Irisin is also known to decrease oxidant-induced apoptosis in patients with diabetes mellitus. However, the correlation between irisin and emphysema in COPD and its role in epithelial cell apoptosis remains unknown.
Subjects and methods: Forty patients with COPD were enrolled in this study. Pulmonary function tests and measurements of the percentage of low-attenuation area on high-resolution computed tomography images were performed, and the results were evaluated for correlation with serum irisin levels. The effect of irisin on cigarette-smoke extract-induced A549 cell apoptosis and the expression of Nrf2, a transcription factor for antioxidants, was also examined in vitro.
Results: Serum irisin levels were significantly correlated with lung diffusing capacity for carbon monoxide divided by alveolar volume (r=0.56, P<0.01) and percentage of low-attenuation area (r=–0.79, P<0.01). Moreover, irisin significantly enhanced Nrf2 expression (P<0.05) and reduced cigarette-smoke extract-induced A549 cell apoptosis (P<0.05).
Conclusion: Decreased serum irisin levels are related to emphysema in patients with COPD and involved in epithelial apoptosis, resulting in emphysema. Irisin could be a novel treatment for emphysema in patients with COPD.
Keywords: chronic obstructive pulmonary disease, COPD, emphysema, irisin, Nrf2, cigarette-smoke extract, CSE, apoptosis
Background
COPD is an inflammatory disorder caused by the long-term inhalation of harmful substances, such as cigarette smoke, which results in irreversible respiratory impairment.1 Airflow obstruction usually has a progressive course characterized by chronic cough, sputum production, and shortness of breath, resulting in decreased physical activity. Two hypotheses have been suggested for the pathogenesis of COPD, namely, the oxidant–antioxidant imbalance hypothesis and the protease–antiprotease imbalance hypothesis.2 Cigarette smoke contains various harmful substances, including oxidants (approximately 1017).3 Oxidants activate inflammatory gene expression, mainly through NFκB signaling. The evoked local inflammation promotes the apoptosis of airway epithelial and vascular endothelial cells, resulting in emphysema. This pathological course induces to irreversible disease progression.4,5
Irisin, a skeletal muscle cell-derived hormone, is a member of the myokine family, and has been revealed to be cleaved from FNDC5. A previous report showed that irisin production was closely related to physical activity,6 and we also reported that the serum irisin level in patients with COPD was significantly decreased and correlated with a patient’s physical activity level.7 Irisin plays important roles in adipose-tissue browning and the regulation of energy expenditure. It has a critical role in metabolic diseases, such as diabetes mellitus and hyperlipidemia.8,9 Moreover, studies have demonstrated its involvement in various other conditions, such as inflammation, hippocampal neurogenesis, and aging.10–12 According to recent reports, irisin has a protective effect against hyperglycemia-induced apoptosis in human vascular endothelial cells.11 However, the relationship between irisin and cigarette-smoke extract (CSE)-induced apoptosis resulting in emphysema in COPD remains unknown.
Nrf2 is a master regulator of antioxidant responses.13 We have reported that Nrf2 expression was significantly decreased in bronchial and alveolar epithelial cells in patients with COPD and that Nrf2 has a protective role against CSE-induced apoptosis.14 The impaired Nrf2 expression in patients with COPD might be involved in emphysema in COPD.
We hypothesized that decreased irisin levels would enhance emphysema in COPD and epithelial apoptosis. To address this hypothesis, we evaluated whether irisin level was correlated with emphysema and whether irisin could inhibit CSE-induced bronchial epithelial apoptosis.
Subjects and methods
Study subjects and measurements
We enrolled 40 patients with COPD in this study. All were stable outpatients at Osaka City University Hospital. They had been diagnosed with COPD according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines,15 and had no pulmonary comorbidities. They had no history of malignant diseases, and they had been free of acute exacerbation or pneumonia for at least the preceding 6 months.
All patients underwent pulmonary function testing. Forced vital capacity (FVC), forced expiratory volume in 1 second (FEV1), FEV1% (FEV1/FVC), diffusing capacity of the lungs for carbon monoxide (DLCO), and DLCO divided by the alveolar volume (DLCO/VA), ΔN2, and V50/V25 were measured using spirometry (Chestac-25F; Chest MI, Tokyo, Japan). DLCO/VA is a more sensitive parameter of diffusion capacity than DLCO, as diffusion capacity is corrected by VA. ΔN2 and V50/V25 are parameters of distal airway obstruction. Body-composition analyses, such as body mass index (BMI), muscle mass (MM), fat-free mass (FFM), and FFM index (FFMI), were performed with bioelectrical impedance analysis with the InBody 3.0 system analyzer (InBody, Seoul, South Korea). Chest high-resolution computed tomography (CT) was performed using a 64-slice spiral CT system (Somatom Sensation 64/Cardiac 64; Siemens, Munich, Germany). The scan time was 0.5 seconds, and the image matrix was 512×512 pixels. We reconstructed 1 mm-thick thin-slice CT images for all lung fields, and all images were analyzed using Airway Inspector software (Surgical Planning Laboratory at Brigham and Women’s Hospital, Boston, MA, USA). Areas with attenuation less than −950 Hounsfield units were defined as low-attenuation areas (LAAs) suggesting emphysema, and the ratio of LAA to all lung areas was defined as %LAA. We quantified %LAA for the upper-, middle-, and lower-lung areas separately in three sections with equal volumes, as well as %LAA for the total lung area, as previously described.16 Serum irisin levels were measured using a commercially available enzyme-linked immunosorbent assay (ELISA) kit (EK-067-29; Phoenix Pharmaceuticals, Burlingame, CA, USA) following the manufacturer’s protocol.
This study was approved by the ethics committee of Osaka City University Hospital (approval 3330), and all patients provided written informed consents for their participation. All procedures were performed according to the guidelines of the Declaration of Helsinki.
Effect of irisin on CSE-induced apoptosis and Nrf2 expression
A549 human alveolar epithelial cells (CCL-185; American Type Culture Collection, Manassas, VA, USA) were cultured in Dulbecco’s Modified Eagle’s Medium/F12 (1:1, 11330-032; Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% fetal bovine serum in a humidified 5% CO2 incubator at 37°C. Cells were then subjected to CSE-induced apoptosis analyses via a time-lapse cell-imaging assay using the IncuCyte Zoom system with CellPlayer 96-well kinetic caspase 3/7 reagent (Essen BioScience, Ann Arbor, MI, USA), as previously described.14 The apoptotic index was calculated according to the equation:
|
|
CSE was prepared as previously described.14 Cells were stimulated with 10% or 15% CSE diluted with cell-culture medium and pretreated with 15, 30, or 60 ng/mL irisin (ADI-908-307; Enzo Life Sciences, Farmingdale, NY, USA), referring to the irisin levels of controls and subjects with COPD in our previous study.7
Then, A549 cells were cultured in Dulbecco’s Modified Eagle’s Medium/F12 (1:1) supplemented with 10% fetal bovine serum in a humidified 5% CO2 incubator at 37°C at a density of 0.02×106 cells per well in a 12-well plate. After 24 hours of starvation, cells were treated with 30 or 60 ng/mL irisin and harvested after 12 hours for Nrf2 messenger RNA (mRNA) evaluation and after 24 hours for Nrf2 protein evaluation. Total RNA was extracted by using the RNeasy minikit (Qiagen NV, Venlo, the Netherlands), and complementary DNA was synthesized using a Ready-to-Go T-primed first-strand kit (GE Healthcare, Little Chalfont, UK). Complementary DNA was subjected to quantitative real-time polymerase chain reaction (PCR) with an Applied Biosystems 7500 real-time PCR system (Thermo Fisher Scientific) using TaqMan gene-expression assays (Thermo Fisher Scientific) for Nrf2 (Hs00975961_g1) and glyceraldehyde 3-phosphate dehydrogenase (Hs99999905_m1). Proteins were subjected to Western blotting for Nrf2, as previously described.14
Statistical analysis
Continuous variables are described as mean and standard deviation when normally distributed, and median and range when not normally distributed. Distributions were confirmed by Kolmogorov–Smirnov test. For comparing two groups, parametrically unpaired Student’s t-tests and nonparametric Mann–Whitney U test were performed. Analysis of variance followed by the Tukey–Kramer procedure or Kruskal–Wallis test was performed for comparing more than three groups. Statistical analyses were performed using JMP version 10.0.0 software for Windows (SAS Institute, Cary, NC, USA). In all statistical analyses, P<0.05 was considered significant.
Results
Correlation of irisin levels with body composition, pulmonary function, and emphysema
The characteristics of the 40 patients are shown in Table 1. The mean age of the patients was 73±9.3 years, and 92.5% of them had GOLD stage II–III. All patients were ex-smokers with smoking histories of 47.2±30.6 pack-years on average. Hypertension, hyperlipidemia, and cardiovascular diseases were prevalent in study subjects, and patients were receiving treatment for these conditions (data not shown).
Irisin was detectable in all serum specimens from study subjects. There was significant correlation between physical activity level and serum irisin level (Figure 1A), though there was no correlation between BMI and serum irisin level (data not shown). There were significant positive correlations of serum irisin level with MM, FFM, and FFMI (Figure 1B–D, P<0.05).
No correlation was observed of serum irisin level with %FVC or %FEV1, whereas significant correlations were observed serum irisin level and ΔN2 (r=−0.42, P<0.05) and V50/V25 (r=0.4, P<0.05) (Figure 2A–D). Moreover, a strong correlation was observed between DLCO/VA and serum irisin level (Figure 2E; r=0.56, P<0.01).
Interestingly, a significant negative correlation was observed between serum irisin level and %LAA. The correlation with %LAA was strongest in the upper-lung fields (Figure 3; upper, r=−0.81, P<0.01; middle, r=−0.75, P<0.01; lower, r=−0.63, P<0.01; total, r=−0.79, P<0.01) (Figure 3).
Irisin inhibited CSE-induced apoptosis in A549 cells and enhanced Nrf2 expression
CSE-induced apoptosis increased proportionally over time (Figure 4A). CSE-induced apoptosis was significantly higher in the 15% CSE group than the control group at 12 hours, as observed in our previous study.14 In contrast, apoptosis was significantly lower in the groups pretreated with 30 or 60 ng/mL irisin than in the 15% CSE-only group (Figure 4B; P<0.05 and P<0.01, respectively). Nrf2 mRNA-expression level was significantly higher in the group treated with 60 ng/mL irisin than in the control group (Figure 5A; P<0.05). Similarly, Nrf2 protein-expression level was significantly higher in the group treated with 60 ng/mL irisin than in the control group (Figure 5B and C; P<0.05).
Discussion
The present study newly reveals that decreased serum irisin levels are significantly correlated with emphysema severity measured by %LAA in patients with COPD. To our knowledge, this is the first report illustrating a strong correlation between the myokine irisin and emphysema in patients with COPD. We also evaluated that irisin inhibited CSE-induced apoptosis in A549 alveolar epithelial cells in an in vitro setting. Epithelial apoptosis is one of the main causes of emphysema progression. Our observations suggest a potential protective role for irisin in COPD pathogenesis.
COPD is a complex disease characterized by both emphysema and small-airway inflammation.17 Chronic inflammatory responses caused by noxious gas inhalation, mainly cigarette smoke, can induce parenchymal tissue destruction, thereby resulting in emphysema, and disrupt the normal repair and defense mechanisms of peripheral airways, thereby resulting in small-airway fibrosis. Both emphysema and small-airway inflammation can lead to progressive obstructive airflow impairment.18,19 Therefore, obstructive impairment in pulmonary function testing has been adopted as a diagnostic procedure for COPD. However, no correlation was found between parameters of obstructive impairment, such as FEV1 and %FEV1 and serum irisin levels, as shown in our previous study.7 In contrast, we found significant correlations of serum irisin levels with ΔN2 and V50/V25, which are factors considered to reflect changes to the distal airway. Although FEV1 and %FEV1 are parameters of obstructive impairment, they have been reported to reflect changes to the more proximal airway.20 Moreover, the strongest correlation exists between DLCO/VA and decreased serum irisin level. DLCO/VA is known to be a parameter of diffusion capacity and reflecting parenchymal destruction, rather than distal airway obstruction. Parenchymal destruction causes the loss of alveolar attachment to the distal airway and leads to distal airway obstruction.21 Significant correlations of serum irisin levels with ΔN2 and V50/V25 might be a result of parenchymal destruction.
These results strengthen our hypothesis of a correlation between emphysema and serum irisin levels pathologically. Therefore, an assessment of the correlation between %LAA, an indicator of emphysema, and serum irisin levels was conducted using image analysis. Results revealed a strong correlation between emphysema and decreased serum irisin levels. In general, progressive or developed emphysema is usually found in the upper-lung area in patients with COPD,16,22 and %LAA for the upper-lung area exhibited the strongest correlation with serum irisin levels when lung fields were divided equally into three sections. Previous reports have shown significant correlations between %LAA and BMI23 and body-composition measurements, such as FFMI.24 This suggests the presence of an emphysematous COPD phenotype with muscle wasting. However, there have been no reports of the underlying mechanism in the relationship between muscle wasting and emphysema. In our study populations, there are also significant correlations of %LAA with BMI, FFM, FFMI, and MM. Irisin level was significantly correlated with MM-related body-composition indices, ie, FFM, FFMI, and MM, as well. These results were probably owing to the fact that irisin is secreted from skeletal muscle. On the other hand, irisin level was closely related to %LAA. These clinical results suggested that irisin might be a mediator that connects muscle wasting and emphysema. Emphysema could progress in patients with decreased irisin levels, and irisin might have a role in preventing emphysema progression. However, its mechanism remains unknown.
Irisin is known to prevent oxidative stress-induced apoptosis in human umbilical vein endothelial cells due to hyperglycemia by suppressing PKCβ/NADPH oxidase and the NFκB–iNOS pathway.25 Other reports have also documented the protective effect of irisin against hyperglycemia-induced apoptosis, and uncovered that the effect was mediated by the AMPK- or ERK-signaling pathway.10,11 Emphysema is characterized by enlargement of the distal air spaces, and is caused by destruction of the airway walls. It is postulated that the inflammatory response results in protease–antiprotease imbalance, and this tissue is destroyed in distal airspaces. Protease–antiprotease imbalance could not account well for the loss of epithelial cells in emphysema, while it could account for the loss of extracellular matrix. Segura-Valdez et al proposed an apoptosis hypothesis for emphysema.26 In an animal model, epithelial and endothelial cell apoptosis were increased, and inhibition of apoptosis resulted in a decrease in emphysema lesions.27 A major cause of apoptosis in emphysema is thought to be cellular damage by oxidant stress. Oxidant–antioxidant imbalance is now considered another major hypothesis for COPD.
Irisin might have the potential to reducing oxidative stresses in various biological settings. Cigarette smoke is a major cause of COPD, and contains many oxidants, which are known to induce apoptosis.28 To clarify whether irisin can affect CSE-induced alveolar epithelial apoptosis through its antioxidant effect against cigarette smoking-induced oxidative stress, we conducted in vitro experiments using A549 cells. Nrf2, an antioxidant transcription factor, prevents CSE-induced pulmonary epithelial apoptosis.29 Nrf2 and several other downstream molecules have an essential protective role in the lungs against oxidative stress from environmental pollutants and toxicants, such as cigarette smoke. The Nrf2 pathway may act as a major determinant of susceptibility to cigarette smoke-induced emphysema by upregulating antioxidant defenses and decreasing lung inflammation and alveolar apoptosis.29 Accordingly, we hypothesized that irisin may play a partial role in the inhibition of oxidant stress through an oxidative-restraint pathway involving Nrf2, leading to cell apoptosis, as has been revealed in preceding studies.13 A significant increase in CSE-induced apoptosis was identified in A549 cells treated with 15% CSE at 12 hours. Circulating mediators extravasate and accumulate at the site, at which they exert their biological effects, and their local concentrations are sometimes much higher than those in serum. The effective concentration of irisin was of the order of that previously estimated to be released by sensitized human lung tissue. Even a previous study estimated local concentration to be high.30 We employed higher concentrations to clarify better the function of localized irisin in alveolar epithelial cells, referring to our previous observation as a possible concentration in this study. As a result, CSE-induced apoptosis showed significant dose-dependent reductions in the groups pretreated with 30 or 60 ng/mL irisin than in the control group (15% CSE-only group). In this study, we also evaluated whether the expression of Nrf2 was significantly increased at the mRNA and protein levels upon treatment with 60 ng/mL irisin. However, we could not conclusively prove the existence of a pathway that directly leads to Nrf2 expression. To date, the receptors for irisin or intracellular signaling systems remain unknown and under investigation. Further research is needed to understand the link between irisin and Nrf2. In addition, the progression of emphysema could be mediated by both oxidative stress and various other mechanisms, including protease–antiprotease imbalances. Therefore, it is necessary to clarify the pathways and mechanisms by which irisin affects emphysema. This study had some limitations. First, the study was conducted at a single center with a limited sample size. Second, the irisin levels of patients with COPD in this study were different from those of patients in our previous study, because the ELISA kit used for measuring serum irisin levels in this study was a commercially available improved version of a previous one. The specificity of the antibodies was adjusted, and the reference value of irisin level in healthy subjects was also changed. Therefore, measured irisin values are different, though values measured by the new kit are significantly correlated with those of the previous one. Third, we employed A549 cells derived from adenocarcinomatous human alveolar basal epithelial cells in in vitro experiments for technical reasons. Primary cells from COPD subjects are better for in vitro experiments. We will try to reproduce the effect of irisin for CSE-induced epithelial apoptosis in future study.
Finally, COPD is related to decreased serum irisin levels, which in turn might be a cause of decreased Nrf2 expression in epithelial cells. This decreased Nrf2 expression accelerates alveolar apoptosis and lung parenchymal destruction, resulting in emphysema. In this study, irisin secreted by skeletal muscles may act on the lungs through systemic blood circulation, and it may restrain oxidative stress in part through Nrf2. We reported a novel biological function of irisin, ie, its protective effect against CSE-induced apoptosis, and its relationship with emphysema, the so-called muscle–pulmonary relationship. Nevertheless, further studies are warranted to confirm our findings.
Acknowledgment
This work was supported in part by research grants from the Japan Society for the Promotion of Science (JSPS) Kakenhi grant 15K09185 to KA.
Disclosure
The authors report no conflicts of interest in this work.
References
Hogg JC, Timens W. The pathology of chronic obstructive pulmonary disease. Annu Rev Pathol. 2009;4:435–459. | ||
Hou G, Yin Y, Han D, Wang QY, Kang J. Rosiglitazone attenuates the metalloprotease/anti-metalloprotease imbalance in emphysema induced by cigarette smoke: involvement of extracellular signal-regulated kinase and NFκB signaling. Int J Chron Obstruct Pulmon Dis. 2015;10:715–724. | ||
Church DF, Pryor WA. Free-radical chemistry of cigarette smoke and its toxicological implications. Environ Health Perspect. 1985;64:111–126. | ||
Pryor WA, Dooley MM, Church DF. Mechanisms of cigarette smoke toxicity: the inactivation of human α-1-proteinase inhibitor by nitric oxide/isoprene mixtures in air. Chem Biol Interact. 1985;54(2):171–183. | ||
Aoshiba K, Yokohori N, Nagai A. Alveolar wall apoptosis causes lung destruction and emphysematous changes. Am J Respir Cell Mol Biol. 2003;28(5):555–562. | ||
Boström P, Wu J, Jedrychowski MP, et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481(7382):463–468. | ||
Ijiri N, Kanazawa H, Asai K, Watanabe T, Hirata K. Irisin, a newly discovered myokine, is a novel biomarker associated with physical activity in patients with chronic obstructive pulmonary disease. Respirology. 2015;20(4):612–617. | ||
Moreno-Navarrete JM, Ortega F, Serrano M, et al. Irisin is expressed and produced by human muscle and adipose tissue in association with obesity and insulin resistance. J Clin Endocrinol Metab. 2013;98(4):E769–E778. | ||
Sanchis-Gomar F, Lippi G, Mayero S, Perez-Quilis C, García-Giménez JL. Irisin: a new potential hormonal target for the treatment of obesity and type 2 diabetes. J Diabetes. 2012;4(3):196. | ||
Wu F, Song H, Zhang Y, et al. Irisin induces angiogenesis in human umbilical vein endothelial cells in vitro and in zebrafish embryos in vivo via activation of the ERK signaling pathway. PLoS One. 2015;10(8):e0134662. | ||
Song H, Wu F, Zhang Y, et al. Irisin promotes human umbilical vein endothelial cell proliferation through the ERK signaling pathway and partly suppresses high glucose-induced apoptosis. PLoS One. 2014;9(10):e110273. | ||
Li M, Yang M, Zhou X, et al. Elevated circulating levels of irisin and the effect of metformin treatment in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2015;100(4):1485–1493. | ||
Kensler TW, Wakabayashi N, Biswal S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu Rev Pharmacol Toxicol. 2007;47:89–116. | ||
Yamada K, Asai K, Nagayasu F, et al. Impaired nuclear factor erythroid 2-related factor 2 expression increases apoptosis of airway epithelial cells in patients with chronic obstructive pulmonary disease due to cigarette smoking. BMC Pulm Med. 2016;16:27. | ||
Vestbo J, Hurd SS, Agustí AG, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2013;187(4):347–365. | ||
Xie M, Wang W, Dou S, Cui L, Xiao W. Quantitative computed tomography measurements of emphysema for diagnosing asthma-chronic obstructive pulmonary disease overlap syndrome. Int J Chron Obstruct Pulmon Dis. 2016;11:953–961. | ||
Hogg JC. Lung structure and function in COPD. Int J Tuberc Lung Dis. 2008;12(5):467–479. | ||
Szulakowski P, Mróz RM, Pierzchała W, Chyczewska E, MacNee W. Patogeneza przewlekłej obturacyjnej choroby płuc. Cześć II. Podłoze molekularne. [Pathogenesis of chronic obstructive pulmonary disease: molecular mechanisms (part II)]. Wiad Lek. 2006;59(3–4):250–254. Polish. | ||
Cantin AM. Cellular response to cigarette smoke and oxidants: adapting to survive. Proc Am Thorac Soc. 2010;7(6):368–375. | ||
Burgel PR, Bourdin A, Chanez P, et al. Update on the roles of distal airways in COPD. Eur Respir Rev. 2011;20(119):7–22. | ||
Stewart JI, Criner GJ. The small airways in chronic obstructive pulmonary disease: pathology and effects on disease progression and survival. Curr Opin Pulm Med. 2013;19(2):109–115. | ||
Bastos HN, Neves I, Redondo M, et al. Influence of emphysema distribution on pulmonary function parameters in COPD patients. J Bras Pneumol. 2015;41(6):489–495. | ||
Ogawa E, Nakano Y, Ohara T, et al. Body mass index in male patients with COPD: correlation with low attenuation areas on CT. Thorax. 2009;64(1):20–25. | ||
Rutten EP, Grydeland TB, Pillai SG, et al. Quantitative CT: associations between emphysema, airway wall thickness and body composition in COPD. Pulm Med. 2011;2011:419328. | ||
Zhu D, Wang H, Zhang J, et al. Irisin improves endothelial function in type 2 diabetes through reducing oxidative/nitrative stresses. J Mol Cell Cardiol. 2015;87:138–147. | ||
Segura-Valdez L, Pardo A, Gaxiola M, Uhal BD, Becerril C, Selman M. Upregulation of gelatinases A and B, collagenases 1 and 2, and increased parenchymal cell death in COPD. Chest. 2000;117(3):684–694. | ||
Rangasamy T, Misra V, Zhen L, Tankersley CG, Tuder RM, Biswal S. Cigarette smoke-induced emphysema in A/J mice is associated with pulmonary oxidative stress, apoptosis of lung cells, and global alterations in gene expression. Am J Physiol Lung Cell Mol Physiol. 2009;296(6):L888–L900. | ||
Bodas M, Van Westphal C, Carpenter-Thompson R, Mohanty D, Vij N. Nicotine exposure induces bronchial epithelial cell apoptosis and senescence via ROS mediated autophagy-impairment. Free Radic Biol Med. 2016;97:441–453. | ||
Huang C, Wang JJ, Ma JH, Jin C, Yu Q, Zhang SX. Activation of the UPR protects against cigarette smoke-induced RPE apoptosis through up-regulation of Nrf2. J Biol Chem. 2015;290(9):5367–5380. | ||
Shute JK, Solic N, Shimizu J, McConnell W, Redington AE, Howarth PH. Epithelial expression and release of FGF-2 from heparan sulphate binding sites in bronchial tissue in asthma. Thorax. 2004;59(7):557–562. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.