Back to Journals » ClinicoEconomics and Outcomes Research » Volume 9
Dalteparin versus vitamin K antagonists for the prevention of recurrent venous thromboembolism in patients with cancer and renal impairment: a Canadian pharmacoeconomic analysis
Authors Dranitsaris G, Shane LG, Crowther M, Feugere G, Woodruff S
Received 2 November 2016
Accepted for publication 2 December 2016
Published 10 January 2017 Volume 2017:9 Pages 65—73
DOI https://doi.org/10.2147/CEOR.S126379
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Giorgio Colombo
George Dranitsaris,1 Lesley G Shane,2 Mark Crowther,3 Guillaume Feugere,4 Seth Woodruff2
1Health Economic and Outcomes Research, Augmentium Pharma Consulting Inc, Toronto, ON, Canada; 2Pfizer Inc, New York, NY, USA; 3McMaster University, Hamilton, ON, 4Pfizer Canada, Montreal, QC, Canada
Background: Patients with cancer are at increased risk of venous thromboembolism (VTE) and the risk is further elevated after a primary VTE. To reduce the risk of recurrent events, extended prophylaxis with vitamin K antagonists (VKA) is available for use. However, in a large randomized trial (Comparison of Low-Molecular-Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer [CLOT]; Lee et al), extended duration dalteparin reduced the relative risk of recurrent VTE by 52% compared to VKA (p=0.002). A recent subgroup analysis of patients with moderate-to-severe renal impairment also revealed lower absolute VTE rates with dalteparin (3% vs. 17%; p=0.011). To measure the economic value of dalteparin as an alternative to VKA, a patient-level cost utility analysis was conducted from a Canadian perspective.
Methods: Resource use data captured during the CLOT trial were extracted and linked to 2015 Canadian unit cost estimates. Health state utilities were then measured using the Time-Trade-Off technique in 24 randomly selected members of the general Canadian public to estimate the gains in quality-adjusted life years (QALYs).
Results: For the entire CLOT trial population (n=676), the dalteparin group had significantly higher mean costs compared to the VKA group ($Can5,771 vs. $Can2,569; p<0.001). However, the utility assessment revealed that 21 of 24 respondents (88%) selected dalteparin over VKA, with an associated gain of 0.14 (95% confidence interval [CI]: 0.10–0.18) QALYs. When the incremental cost of dalteparin was combined with the QALY gain, dalteparin had a cost of $Can23,100 (95% CI: $Can19,200–$Can25,800) per QALY gained. The analysis in patients with renal impairment suggested even better economic value with the cost per QALY gained being <$14,000.
Conclusion: Extended duration dalteparin is a cost-effective alternative to VKA for the prevention of recurrent VTE in patients with cancer, especially in those with renal impairment.
Keywords: venous thromboembolism, cancer, recurrence, low-molecular-weight heparin, dalteparin, vitamin K antagonists
Introduction
Deep vein thrombosis (DVT) and pulmonary embolism (PE) are expressions of venous thromboembolism (VTE). Cancer patients with active disease are at higher than average risk compared to a non-cancer population.1 In one study, cancer patients with solid tumors had a VTE prevalence of 7.8% (95% confidence interval [CI]: 6.2%–9.4%) or 1 event per 12.8 patients.2 Following the occurrence of an initial event, the risk of recurrence remains high.1,3,4 As a result, extended thromboprophylaxis in cancer patients is recommended by the American College of Chest Physicians and other groups in order to prevent recurrent VTE.3–5
For many years, vitamin K antagonists (VKA), such as warfarin, were used for extended thromboprophylaxis in cancer patients. However, current international guidelines recommend low-molecular-weight heparin (LMWH) monotherapy over VKA for the secondary prevention of cancer-associated VTE.4,6,7 The advantages of LMWH over VKA are a more favorable safety profile, fewer interactions with food and other drugs and a more predictable anticoagulation response, which eliminates the need for continuous blood monitoring.4,6,7 The disadvantages associated with LMWH are the need for daily subcutaneous (SC) administration for up to 6 months and the higher drug acquisition cost compared to VKA.
The three LMWH that have been investigated in randomized comparative trials against VKA in the setting of secondary VTE prophylaxis in cancer patients are enoxaparin, tinzaparin, and dalteparin. There were two small trials comparing enoxaparin to VKA and both failed to demonstrate significant improvements over VKA.8,9 Similarly, there were also two tinzaparin trials that failed to reach statistical significance, one being a large well-powered multinational study that enrolled 900 cancer patients.10,11
The only LMWH to have demonstrated a statistically and clinically meaningful benefit in terms of VTE risk reduction has been dalteparin. In the multinational CLOT (Comparison of Low-Molecular-Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer) trial, patients were to receive dalteparin (200 IU/kg SC once daily in the first month, then 150 IU/kg SC once daily from months 2 to 6) or 5–7 days of dalteparin (200 IU/kg SC once daily) overlapped with and followed by an oral VKA for up to 6 months.12 The main inclusion criteria were prior acute symptomatic VTE, and the primary end point was the first objectively documented, symptomatic recurrent DVT, or PE during the 6-month study period. During the course of the study, 676 cancer patients with a newly diagnosed VTE were enrolled. By the end of the study, 27 of 336 (8.0%) patients in the dalteparin group developed a recurrent VTE compared to 53 of 336 (15.8%) in the VKA control group (hazard ratio [HR]=0.48; p=0.002). Safety was comparable between groups with no significant differences in major bleeding events (dalteparin=6% vs. VKA=4%; p=0.27).12 Dalteparin also provides benefits across important patient subgroups. In a recent post hoc analysis of the CLOT trial conducted by Woodruff et al, dalteparin patients with moderate (CrCl 30–59 mL/min) to severe (CrCl 15–29 mL/min) renal impairment at randomization had lower absolute rates of VTE than those in the VKA group (3% vs. 17%; p=0.011), and the frequency of major bleeding was similar between subgroups (2.0% vs. 2.4%; p=0.46).13
The original CLOT trial collected health care resource data, and this was initially used to perform a cost utility analysis from the Canadian health care system perspective.14 In that study, the main conclusion was that dalteparin was economically attractive to VKA with the cost per quality-adjusted life year (QALY) being $Can13,800.14 However in that analysis, health state utilities were estimated from a sample of oncology health care professionals (i.e., patient surrogates), which is not recommended by guidelines for the economic evaluation of drugs.15,16 In addition, the economic value of dalteparin as an alternative to VKA was not assessed in patient subgroups of special clinical interest, such as those with renal impairment.
In this study, an updated pharmacoeconomic analysis was conducted to determine the economic value of dalteparin from the perspective of the publicly funded Canadian health care system. Therefore, the two main questions addressed in the current analysis were as follows: does the use of dalteparin for secondary VTE prophylaxis in cancer patients provide good economic value using 2015 costs of care and more appropriate sources of clinical utility? and what is the economic value of dalteparin when used as secondary prophylaxis in cancer patients with moderate-to-severe renal impairment?
Methods
Patients groups within the CLOT trial have been previously described,12 but they were well balanced in terms of important prognostic factors, such as age, performance status, presence of metastatic disease, history of prior VTE, and baseline renal function. Health care resource use data that were collected from the CLOT trial were extracted and used for a patient-level economic analysis. The extracted data included information on dose and duration of therapy for dalteparin and the VKA, biochemistry and hematology tests, the international normalization ratio (INR) in the case of VKA, patient telephone contact, unscheduled clinical visits, diagnostic tests associated with VTE recurrence, hospital length of stay, blood transfusions, and the occurrence of major bleeding events that were possibly or probably related to the study drugs.
Not all aspects of health care resource use were captured in the CLOT trial. The actual type of intervention for the treatment of patients who developed recurrent VTE or for the management of related side effects was not collected. As a result, Canadian and international literature was reviewed to identify costs for the management of DVT, PE, fatal PE, and for adverse events consisting of heparin-induced thrombocytopenia, hematochezia, hematoma, hematuria, hematemesis, melena, retroperitoneal bleeding, intracranial bleeding, and intraperitoneal bleeding.17–22 In addition, only events of grade ≥III (National Cancer Institute’s Common Terminology Criteria for Adverse Events, NCI CTCAE V4.0) were included in the economic analysis. Unless it was otherwise specified within the CLOT trial database, an assumption was made that all patients who developed DVTs were treated in the outpatient setting.
The unit costs for the individual health care resource items were obtained from the Ontario Ministry of Health and the University Health Network in Toronto. All costs were reported in 2015 Canadian dollars, and cost estimates from previous years were converted into 2015 dollars using the Consumer Price Index as reported by the Bank of Canada. The daily cost of supplies for dalteparin administration and the monthly pharmacy dispensing fee (i.e., $Can11.99) were also included in the analysis. The end product of this process was a comprehensive database consisting of patient demographic, clinical outcomes, and 2015 total cost of care for each patient enrolled into the CLOT trial. The final database was then used to conduct a patient-level univariate economic analysis between patients randomized to receive dalteparin or VKA within the CLOT trial.
Subgroup analysis of patients with moderate-to-severe renal impairment
Cancer patients with renal impairment and acute VTE are a clinically high-risk subgroup. Therefore, the economic analysis was extended to this patient subpopulation. This consisted of a univariate economic analysis on the renally impaired subgroup to determine the magnitude of the cost difference between patients treated with dalteparin relative to those who received VKA within the CLOT trial. This cost difference was then used in the subsequent cost analysis to estimate the incremental cost per VTE avoided and QALY gained with dalteparin within the renally impaired subgroup.
Statistical considerations
For the intention to treat (ITT) population and the renally impaired subgroup, the total cost of therapy between patients randomized into the dalteparin and VKA group was compared using the unpaired t-test and the nonparametric Wilcoxon rank-sum test. The cutoff significance for all of the statistical tests was set at the p<0.05 level and there was no adjustment for multiplicity. All of the statistical analyses were performed using Stata, release 11.0 (Stata Corp., College Station, TX, USA).
Health state utilities
A health state utility is a value between 0 and 1 where the former represents death and the latter a state of optimal quality of life.23 In this study, there were four relevant health states associated with secondary VTE prophylaxis in cancer patients. They consisted of treatment with a VKA or dalteparin for up to 6 months to prevent a recurrent VTE within the general CLOT trial population and then in the patient subgroup with renal impairment. Using the Time-Trade-Off technique, the health states were measured as “healthy months equivalence” for the time spent in each health state.24,25 Gains in healthy months equivalence were then converted into QALYs, dividing by 12 months. To illustrate the process, a 12-month health state may have a healthy months equivalence of 6 months. Therefore, the utility would be 0.5, on a scale between 0 and 1 (6 months of healthy months equivalence/12 months in the given health state).
It has been recommended by the Task Force from the International Society of Pharmacoeconomics and Outcomes Research and the guidelines developed by the Canadian Agency for Drugs and Technology in Health that health state utilities be measured from a randomly selected sample from the general public.16,25 Therefore, within this study, subjects consisted of 24 volunteers from the general Canadian tax paying public that were selected through a multistage, random cluster sampling technique with postal code as the unit of selection.
To be eligible for the survey, individuals had to be ≥18 years of age, have permanent resident status in Canada, be indirectly supporting the health care system through tax contributions, and give informed consent to participate in the interview. Respondents were interviewed face-to-face by a trained field investigator via a door-to-door contact strategy. The participant’s name was not asked at any point in the interview. The only personal information recorded was age, marital status, education, household income, number of children, history of VTE, familiarity with the cost of cancer drugs, and whether they had drug insurance. However, no protected information was collected. Participants were assured that participation was voluntary and they were free to withdraw at any time. Permission and approval to conduct the study was received by the Quorum institutional review board, which is a central ethics review board that is fully compliant with relevant US and Canadian regulations.
After informed consent was obtained, respondents were presented with information about the natural history of VTE in patients with cancer, followed by a description of the VKA and dalteparin administration protocol. This included information on the method of administration, monitoring requirements and the associated risks and benefits. During the final part of the interview, the clinical outcomes from the CLOT trial were presented. This consisted of the risk for recurrent DVT, fatal and nonfatal PE, and major bleeding events associated with each therapy. Respondents were then asked how many months of “optimal health” they considered being equivalent to the time spent in each of the less than optimal health states described. These measures were used to weigh the duration within each health state by the quality of life experienced by a patient living through that time period. The interview process was then repeated with the presentation of data for patients with moderate-to-severe renal impairment.
Pharmacoeconomic evaluation
The two components that comprised the pharmacoeconomic analysis consisted of the incremental cost per VTE avoided and the incremental cost per QALY gained when dalteparin was used as an alternative to VKA to prevent recurrent VTE in cancer patients. These estimates were determined for both the full ITT CLOT population and the renally impaired subgroup. The two economic outcomes were calculated by dividing the difference in cost between dalteparin and VKA therapy (numerator) by the difference in VTE avoided or QALYs gained (denominator).
To test for uncertainly in the point estimates, a deterministic one-way sensitivity analysis was conducted on the base cases results. This consisted of reanalyzing the primary outcomes using the upper and lower 95% CIs of the differences in total treatment costs and utility estimates. All cost-effectiveness ratios were rounded to the nearest hundred.
Results
For the original CLOT trial report, a comparison of baseline patient and clinical variables suggested that patients randomized into the dalteparin and VKA groups were well balanced.12 However in the recent subgroup analysis, there were slightly more patients in the VKA group who had moderate-to-severe renal impairment at baseline (26.0% vs. 21.9%), but the difference failed to reach statistical significance (p=0.21).13
A comparison of health care resource use data collected during the CLOT trial indicated that patients in the experimental group received dalteparin for a mean of 126.3 days compared to 8.0 days in the VKA group (Table 1). After the initial 8 days of therapy with dalteparin, patients in the control group received oral VKA therapy only, which was given for a mean of 116.9 days. A comparison of laboratory and diagnostic test use indicated similar levels of utilization, with the exception of INR measurements, which are required for oral anticoagulation therapy with VKAs. An average of 22 INRs was measured per patient randomized to VKA therapy. The number of blood transfusions, units given and INR measurements associated with the transfusions were also higher in the VKA group (Table 1). The comparative analysis on health care resource use was then extended to patients who had renal impairment at baseline. Increased use of some resources was also noted in this patient subgroup (Table 2).
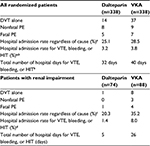
The CLOT trial was powered to detect statistically significant differences in the number of recurrent VTE between treatment groups over the 6-month trial horizon. These consisted of DVTs as well as fatal and nonfatal PEs. A statistically significant reduction in the occurrence of new VTE in patients randomized to the dalteparin group was reported (Table 3). In patients who received therapy in the outpatient setting, there was a hospital admission rate (regardless of cause) of 25.1% in the dalteparin group compared to 28.5% in the control (p=0.33; not statistically significant). When the cause for admission was limited to a recurrent VTE, bleeding or heparin-induced thrombocytopenia, the rates of hospital admission were 3.2% for dalteparin and 3.8% in the oral therapy group (p=0.68). Overall, these rates translated to 32 additional hospital days for dalteparin patients and 40 days in the VKA group (Table 3). Similar benefits in favor of dalteparin were also reported in the renally impaired subgroup (Table 3).
The analysis was continued with the application of Canadian unit costs to all health care resources used by patients from the ITT CLOT population. Dalteparin patients had higher costs for drug therapy and for the treatment of major bleeding events relative to the VKA control group (Table 4). In contrast, the VKA group had increased costs for laboratory monitoring, blood transfusions, and for the management of VTE recurrences. When all of the costs were combined, the total mean cost for all CLOT patients randomized into the dalteparin group was $5,771 (95% CI: $5,324–$6,219) compared to $2,569 (95% CI: $2,328–$2,810) in the VKA group (p<0.001). In the subgroup analysis consisting of renally impaired patients, the differences in cost between the dalteparin and VKA groups remained statistically significant ($5,000 [95% CI: $4,248–$5,569] vs. $2,671 [95% CI: $2,079–$3,264]; p<0.001) but the differential narrowed by $873–$2,329 (Table 4).
Treatment preferences and health state utilities
Treatment preferences and health state utilities for each of the four outcomes were measured from a sample of 24 members of the tax paying general public that were selected through a random multistage sampling strategy. The mean age of respondents was 52 years (range: 32–73) with 11 of 24 (45.8%) being female. Overall, 23 of 24 respondents were married, 15 (62.5%) had postsecondary school education, 11 (45.8%) were working full time, and 12 (57.1%) had annual incomes that exceeded $100,000. The final series of demographic questions focused on respondent’s experience with VTE. The data revealed that only 1 of 24 subjects (4.2%) had previously experienced a VTE, which was treated with warfarin.
When the background information was presented on both drugs, respondents were asked to select their preferred secondary prophylactic intervention from an overall CLOT trial perspective. Overall, 21 of 24 (87.5%) respondents selected dalteparin over a VKA as their drug of choice. Healthy month equivalence scores and health state utilities for each alternative were then estimated from the sample. The higher preferences for dalteparin translated into higher health state utility scores (measured as healthy month equivalence). The utility of the dalteparin health state was almost twofold higher than that seen with treatment with a VKA suggesting improved quality of life (Table 5). The higher utility scores for dalteparin were due to a combination of factors such as improved efficacy, a demonstrated safety profile with extended use and the ability to eliminate continuous INR monitoring. The main positive attribute that respondents indicated about VKA was the oral route of administration. Overall, the gain in healthy month equivalence was ~1.67 months with dalteparin for the full CLOT population, which corresponded to an additional gain of ~0.14 QALYs.
The utility assessment was then extended to patients with moderate-to-severe renal impairment. Under this treatment setting where dalteparin demonstrated an even greater effect size (i.e., number needed to treat=7 vs. 13 for the overall population), 23 of 24 respondents (96%) selected dalteparin as their preferred treatment. This higher preference translated into a healthy month equivalence gain of 2.0 months, which corresponded to an additional gain of ~0.17 QALYs (Table 5).
Pharmacoeconomic analysis
From the results in the ITT CLOT population, dalteparin was associated with an incremental cost of ~$41,200 (95% CI: $34,300–$46,200) per VTE avoided (Table 5). One of the limitations of only reporting the cost per VTE avoided is that it does not incorporate the patient quality of life and utility gain associated with the avoidance of DVT or PE. Such benefits can only be quantified by the QALY. Therefore, the incremental cost of dalteparin (i.e., $3,202) was combined with the 0.14 QALYs gained (a gain of 1.67 health months equivalence corresponds to a gain of 0.14 QALY). The findings revealed an incremental cost of ~$23,100 per QALY gained (95% CI: $19,200–$25,800). The analysis was also done for the renally impaired patient subgroup. When used in patients with moderate-to-severe renal impairment, dalteparin was associated with an incremental cost per VTE avoided and QALY gained of $16,400 and $14,000, respectively (Table 5).
Sensitivity analysis
A series of one-way sensitivity analyses were conducted using the 95% CI of the differences in healthy months equivalent scores (1.18–2.15) and cost ($2,668–$3,588) in the full ITT CLOT population. Under the worst-case scenario for dalteparin where the highest cost difference between the two treatments was used (i.e., $3,588) along with the lowest gain in healthy months equivalence (i.e., 1.18), the cost per QALY gained with dalteparin increased to ~$36,500. Under the best-case scenario for dalteparin where the lowest cost difference (i.e., $2,668) was used along with the highest gain in healthy months equivalence (i.e., 2.15), the cost per QALY gained with dalteparin was reduced to $14,900 in the ITT CLOT population. Repeating the analysis in patients with renal impairment, the cost per VTE avoided and QALY gained changed to $23,500 and $6,800 under the two worst- and best-case scenarios, respectively. These findings imply that the base case cost per VTE avoided and QALY gained estimates were stable.
Discussion
There have been a least six randomized trials comparing extended duration therapy with LMWH to VKA for secondary prophylaxis of VTE in patients with cancer.8–12 From these studies, the only agent to demonstrate a statistically significant reduction in the risk of recurrent VTE has been dalteparin.12 Following the publication of the CLOT trial, a patient-level pharmacoeconomic analysis from a Canadian health care system perspective was initially conducted.14 Deriving health state utilities from a sample of oncology nurses and pharmacists, the study determined that dalteparin as an alternative to VKA was associated with an incremental cost of ~$13,800 (95% CI: $12,400–$15,100) per QALY gained in 2005 Canadian dollars.14 Since that time, there have been changes in the cost of drugs, diagnostics tests, and in managing DVTs and PE.26 In addition, recent data in patients with renal impairment prior to the start of treatment have also become available.13 Hence, a reanalysis of the Canadian economic evaluation of the CLOT trial was undertaken, which included the collection of new health state utility data from members of the general public.
The results from this reanalysis using 2015 costs maintain that secondary dalteparin prophylaxis in cancer patients with prior symptomatic VTE is a cost-effective alternative to VKA therapy, with an incremental cost of $23,100 (95% CI: $19,200–$25,800) per QALY gained. Furthermore, the economic value of dalteparin was especially good in patients with moderate-to-severe renal impairment, with the cost per QALY gained being $14,000. Such low incremental cost-effectiveness ratios are rare in oncology today, as most drugs used in cancer patients have cost per QALY ratios in excess of $100,000.27 Therefore, dalteparin continues to be an economically attractive drug. The main factors responsible for the differences in cost per QALY gained between the original study and the current analysis are changes in the cost of drugs, treatment of major bleeds, and the management of recurrent VTE. However, despite these changes, dalteparin remained a cost-effective therapy.
Limitations
One of the strengths of this study was the availability of patient-level health care resource use data. However, there are some limitations that need to be acknowledged. Because the data source is a randomized trial, some of the hospital resources were mandated by the study protocol and may not completely reflect standard practice. In addition, not all the health care resources needed to manage recurrent VTE were available in the trial database. Therefore, we had to rely on literature reported cost estimates for managing such events in both the hospitalized and outpatient settings. Another limitation was that indirect costs such as lost productivity and care giver costs were not included. The inclusion of indirect costs would probably have improved the economic profile of dalteparin. Finally, members of the general Canadian public instead of cancer patients with VTE were used in the utility assessment. Using general public as a utility source always presents with challenges in terms of ensuring their understanding of the health states under investigation.
Conclusion
Dalteparin used for the secondary prevention of VTE in cancer patients remains cost effective, with an incremental cost of $23,100 per QALY gained. The economic value was further enhanced in cancer patients with moderate-to-severe renal impairment, with the cost per QALY gained being reduced to $14,000. In addition to the practical advantages of dalteparin over VKA, which include less monitoring and improved efficacy, long-term dalteparin therapy remains an economically attractive alternative to VKA for the prevention of recurrent VTE in patients with cancer, especially in those with renal impairment.
Acknowledgments
Funding for this study was provided by Pfizer Inc. through a request for proposal process.
The abstract of this paper was presented at the 21st Annual Meeting of the International Society of Pharmacoeconomics and Outcomes Research, 2016. The poster’s abstract was published in “Poster Abstracts” in Value in Health 2016,19(3):A154–A155. The actual paper, however, has never been published.
Author contributions
GD had full access to the data, designed the study, conducted the analysis, interpreted the results, and had the final responsibility for the decision to submit the paper for peer-reviewed publication. All authors confirm they were involved in study design, data analysis and interpretation, drafting of the paper and final approval of the current version to be published.
Disclosure
LGS, SW, and GF are employees of the sponsor. MC has acted as a consultant to Pfizer Inc. The other authors report no other conflicts of interest in this work.
References
Lee AYY, Levine MN. Venous thromboembolism and cancer: risks and outcomes. Circulation. 2003;107(23 Suppl 1):I17–I21. | ||
Sallah S, Wan JY, Nguyen NP. Venous thrombosis in patients with solid tumors: determination of frequency and characteristics. Thromb Haemost. 2002;87(4):575–579. | ||
Prandoni P, Lensing AW, Piccioli A, et al. Recurrent venous thromboembolism and bleeding complications during anticoagulant treatment in patients with cancer and venous thrombosis. Blood. 2002;100(10):3484–3488. | ||
Lyman GH, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2013;31(17):2189–2204 | ||
Kahn SR, Lim W, Dunn AS, et al. American College of Chest Physicians. Prevention of VTE in nonsurgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-based Clinical Practice Guidelines. Chest. 2012;141(Suppl 2):e195S–e226S. | ||
Farge D, Debourdeau P, Beckers M, et al. International clinical practice guidelines for the treatment and prophylaxis of venous thromboembolism in patients with cancer. J Thromb Haemost. 2013;11(1):56–70. | ||
Mandalà M, Falanga A, Roila F. Management of venous thromboembolism in cancer patients: ESMO clinical practice guidelines. Ann Oncol. 2011;22(Suppl 6):vi85–vi92. | ||
Deitcher SR, Kessler CM, Merli G, Rigas JR, Lyons RM, Fareed J. Secondary prevention of venous thromboembolic events in patients with active cancer: enoxaparin alone versus initial enoxaparin followed by warfarin for a 180-day period. Clin Appl Thromb Hemost. 2006;12(4):389–396. | ||
Meyer G, Marjanovic Z, Valcke J, et al. Comparison of low-molecular-weight heparin and warfarin for the secondary prevention of venous thromboembolism in patients with cancer: a randomized controlled study. Arch Intern Med. 2002;162 (15):1729–1735. | ||
Hull RD, Pineo GF, Brant RF, et al. Long-term low-molecular-weight heparin versus usual care in proximal-vein thrombosis patients with cancer. Am J Med. 2006;119(12):1062–1072. | ||
Lee AY, Kamphuisen PW, Meyer G, et al. Tinzaparin vs. warfarin for treatment of acute venous thromboembolism in patients with active cancer: a randomized clinical trial. JAMA. 2015;314(7):677–686. | ||
Lee AY, Levine MN, Baker RI, et al. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med. 2003;349(2):146–153. | ||
Woodruff S, Feugère G, Abreu P, Jen F. A post-hoc analysis of dalteparin versus oral anticoagulant therapy for the prevention of recurrent venous thromboembolism in patients with cancer and renal impairment. J Thromb Thrombolysis. 2016;42(4):494–504. | ||
Dranitsaris G, Vincent M, Crowther M. Dalteparin vs. warfarin for the prevention of recurrent venous thromboembolic events in cancer patients: a pharmacoeconomic analysis. Pharmacoeconomics. 2006;24(6):593–607. | ||
Canadian Agency for Drugs and Technology in Health. Guidelines for the Economic Evaluation of Health Technologies. 3rd ed. Ottawa, Canada: Canadian Agency for Drugs and Technology in Health; 2006. Available from: http://www.cadth.ca/media/pdf/186_EconomicGuidelines_e.pdf. Accessed August 18, 2015. | ||
Task Force from the International Society of Pharmacoeconomics and Outcomes Research; 2015. Available from: http://www.ispor.org/TaskForces/Presentation-22.pdf. Accessed August 18, 2015. | ||
Gordois A, Posnett J, Borris L, et al. The cost effectiveness of fondaparinux compared with enoxaparin as prophylaxis against thromboembolism following major orthopedic surgery. J Thromb Haemost. 2003;1(10):2167–2174. | ||
Guanella R, Ducruet T, Johri M, et al. Economic burden and cost determinants of deep vein thrombosis during 2 years following diagnosis: a prospective evaluation. J Thromb Haemost. 2011;9(12):2397–2405. | ||
Dasta JF, Pilon D, Mody SH, et al. Daily hospitalization costs in patients with deep vein thrombosis or pulmonary embolism treated with anticoagulant therapy. Thromb Res. 2015;135(2):303–310. | ||
Mickisch G, Gore M, Escudier B, Procopio G, Walzer S, Nuijten M. Costs of managing adverse events in the treatment of first-line metastatic renal cell carcinoma: bevacizumab in combination with interferon-alpha2a compared with sunitinib. Br J Cancer. 2010;102(1):80–86. | ||
Hull RD, Pineo GF, Raskob GE. The economic impact of treating deep vein thrombosis with low molecular weight heparin: outcome of therapy and health economic aspects. Haemostasis. 1998;28(Suppl 3):8–16. | ||
Dranitsaris G, Kahn S, Stumpo C, et al. Pharmacoeconomic analysis of fondaparinux versus enoxaparin for the prevention of thromboembolic events in orthopedic surgery patients. Am J Cardiovasc Drugs. 2004;2004;4(5):325–333. | ||
Drummond MF, Stoddart GL, Torrance GW. Methods for the Economic Evaluation of Health Care Programs. Oxford, England: Oxford University Press; 2005. | ||
Torrance GW. Utility approach to measuring health-related quality of life. J Chron Dis. 1987;40(6):593–600. | ||
Gafni A. Alternatives to the QALY measure for economic evaluations. Support Care Cancer. 1997;5(2):105–111. | ||
Fernandez MM, Hogue S, Preblick R, Kwong JK. Review of the cost of venous thromboembolism. Clinicoecon Outcomes Res. 2015;7:451–462. | ||
Dranitsaris G, Papadopoulos G. Health technology assessment of cancer drugs in Canada, the United kingdom and Australia: should the United States take notice? Appl Health Econ Health Policy. 2015;13(3): |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.