Back to Journals » ClinicoEconomics and Outcomes Research » Volume 10
Cost effectiveness of ixekizumab versus secukinumab in the treatment of moderate-to-severe plaque psoriasis in Spain
Authors Johansson E, Nuñez M, Svedbom A, Dilla T, Hartz S
Received 9 May 2018
Accepted for publication 27 July 2018
Published 12 November 2018 Volume 2018:10 Pages 747—759
DOI https://doi.org/10.2147/CEOR.S167727
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Samer Hamidi
Erin Johansson,1 Mercedes Nuñez,2 Axel Svedbom,1 Tatiana Dilla,2 Susanne Hartz3
1Access, Commercialisation & Communications, ICON plc, Stockholm, Sweden; 2Health Outcomes and Real World Evidence, Eli Lilly, Madrid, Spain; 3Global Patient Outcomes and Real World Evidence International, Eli Lilly, Surrey, UK
Background: Currently, several biologic agents are available for the treatment of moderate-to-severe plaque psoriasis, including newer agents with similar mechanisms of action and efficacy; therefore, there is a need to evaluate their efficiency in terms of cost effectiveness.
Objective: This study evaluates the cost effectiveness of recently approved interleukin (IL)-17A antagonists, ixekizumab and secukinumab, for the treatment of moderate-to-severe plaque psoriasis from the perspective of the Spanish National Health System (NHS).
Materials and methods: A Markov model with a lifetime horizon was developed to compare the cost effectiveness of ixekizumab vs. secukinumab in a hypothetical cohort of patients with moderate-to-severe plaque psoriasis. The model used monthly cycles and included four health states: a 12-week induction period, treatment maintenance, best supportive care (BSC), and death. Patients meeting response criteria at the end of the induction period transitioned to maintenance therapy, whereas non-responders transitioned to BSC. It was assumed that, each year, 20% of patients receiving maintenance therapy would discontinue treatment. The model incorporated data from various sources, including published literature, a network meta-analysis, and expert opinion for some variables.
Results: Ixekizumab was dominant over secukinumab in that it gained 0.037 more quality-adjusted life years (QALYs) and saved €1951 in total costs over the lifetime horizon. Probabilistic sensitivity analysis showed a 96.6% likelihood that ixekizumab would be cost effective at a threshold of €30,000 per QALY gained.
Conclusion: For the treatment of moderate-to-severe plaque psoriasis in Spain, ixekizumab provided additional QALYs and potential savings for the Spanish NHS compared with secukinumab. Since the magnitude of the differences in costs and QALYs was modest, other factors such as patient preferences (eg, for number of injections) and long-term safety (eg, related to time on the market) may also be important for guiding clinical decisions.
Keywords: pharmacoeconomics, cost-utility, biologics, IL-17A antagonists
Plain language summary
The treatment of moderate-to-severe plaque psoriasis often includes the use of biologic agents. This cost-effectiveness analysis focused on a comparison of two new biologic agents, ixekizumab and secukinumab, which share a similar mechanism of action. The analysis was conducted from the perspective of the Spanish National Health System (NHS) and a Markov model that incorporated efficacy data from a network meta-analysis was used, as there are currently no head-to-head clinical trials between ixekizumab and secukinumab in moderate-to-severe plaque psoriasis. Ixekizumab provided additional, modest quality-adjusted life years and cost savings compared with secukinumab when modeled over a patient’s lifetime. These findings could translate to savings for the Spanish NHS. Given the limited efficacy and cost-effectiveness data available for the selection of biologics in this area, this study provides information that may help guide treatment decisions, in tandem with factors such as patient preference and long-term safety.
Introduction
Psoriasis is a chronic inflammatory disorder that primarily affects the skin, has a predisposing genetic component, and is associated with reduced health-related quality of life.1,2 In Spain, the prevalence of psoriasis is estimated to be ≈2.3% of the population.3 Plaque psoriasis is the most common form of the disorder and is found in up to 80%–90% of patients with psoriasis.2,4 Moderate-to-severe psoriasis, which affects >10% of the body surface area or involves key areas such as the hands, feet, face, or genitals, occurs in ≈20% of patients with psoriasis.4
Treatment options for psoriasis continue to expand. In addition to conventional therapies that include topical agents, phototherapy, and traditional systemic drugs such as methotrexate and ciclosporin, several biologic therapies are now available.5,6 Although disease severity, patient preferences, cost, reimbursement, and other factors may influence the choice of therapy, biologics are typically used in patients with moderate-to-severe psoriasis. Generally, biologic agents are recommended for patients who have a total Psoriasis Area and Severity Index (PASI) score ≥10 and a Dermatology Life Quality Index (DLQI) score >10 and who have not responded to, are intolerant of, or have contraindications to traditional systemic drugs.6–9 Biologic agents commonly used for patients with moderate-to-severe disease include tumor necrosis factor inhibitors, such as adalimumab, infliximab, and etanercept, and interleukin (IL) antagonists, which include the IL-23 antagonist ustekinumab, the IL-17A antagonist secukinumab, and the most recently introduced agent, ixekizumab, a high-affinity monoclonal antibody that also selectively targets IL-17A, a pro-inflammatory cytokine implicated in the pathogenesis of psoriasis by promoting keratinocyte proliferation and activation.10 Currently, at the national level, neither brodalumab nor guselkumab is reimbursed.
The introduction of new therapies is especially important for the long-term management of patients who initially respond to therapy, but experience marked waning of efficacy and lack of response over time. Various treatment guidelines and consensus documents recommend switching to an alternative biologic if response is inadequate at the end of the induction/trial period or if efficacy is lost over time (or if patients are unable to tolerate the first biologic or if they develop a contraindication).6,11 However, currently, there are no guidelines in Spain regarding the selection of specific initial biologic therapy or recommendations to use biologics in a particular order.7,9 This is similar to the situation elsewhere in Europe, where guidance on the use of sequential biologic therapy is limited and recommendations regarding specific sequences are generally lacking.6,12–16
Cost effectiveness of new biologics needs to be evaluated by measuring the incremental cost per quality-adjusted life year (QALY) gained to better reflect clinical practice and guide decision makers. The present cost-effectiveness analysis, conducted from the perspective of the Spanish National Health System (NHS), compared the two recently approved IL-17A antagonists, ixekizumab and secukinumab, in patients with moderate-to-severe psoriasis.
Materials and methods
Model overview and treatment sequences
A Markov model was developed to compare the cost effectiveness of ixekizumab vs. secukinumab in a hypothetical cohort of patients with moderate-to-severe plaque psoriasis in Spain. A Markov model is appropriate because it is reasonable to assume that transition probabilities for treatment discontinuation beyond the induction period are constant rather than dependent on identifiable events. In this context, psoriasis is a chronic condition that is generally well suited to the memoryless nature of a Markov model framework, which has also been used in other cost-effectiveness analyses with biologics in psoriasis.8,17–19
The model used in the analysis had a framework similar to that of the widely used York model17 but was modified to include age-dependent, gender-weighted mortality risk, the use of a lifetime model horizon (from age 45 to 100 years), and more relevant costing data for best supportive care (BSC) informed by expert opinion to better reflect clinical practice. The model was programmed in Visual Basic for Applications with a user interface in Microsoft® Excel (Microsoft Corporation, Redmond, WA, USA).
The model compared treatment with ixekizumab or secukinumab (Table 1) and used monthly cycles without applying half-cycle correction. Half-cycle corrections are sometimes applied in Markov models because in a Markov cohort, it is assumed that patient transitions occur at the beginning or end of a cycle, whereas patient transition is, in reality, a continuous process that can occur at any time during the cycle. However, we considered the cycle length in our model to be sufficiently short (1 month) to avoid the need for half-cycle correction, which is in agreement with others.20,21 Four health states were included in the model: a treatment induction period that lasted 12 weeks (and reflected the efficacy assessment time point in clinical studies), treatment maintenance (continuous treatment period), BSC, and death. At the end of the induction period with ixekizumab or secukinumab, responders transitioned to maintenance therapy on the same drug. Response was defined as having at least a 75% reduction in baseline PASI (PASI75). PASI75 was a primary endpoint of the UNCOVER trials with ixekizumab in patients with psoriasis and was considered a clinically meaningful response.22 Patients were assumed to maintain this level of response until discontinuation, an assumption supported by long-term data showing sustained response with biologic treatment for psoriasis.23,24 Non-responders (ie, patients not achieving a PASI75 response) and patients who discontinued maintenance therapy transitioned to BSC. An annual all-cause discontinuation rate of 20% was assumed for maintenance therapy, which is the rate used in previous cost-effectiveness analyses based on the York model8,25 and is supported by the results of large long-term registry studies.26,27 This annual rate translated to a 1.84% discontinuation rate per monthly cycle, and the rate of discontinuation was the same regardless of the level of response (ie, PASI75–89, PASI90–99, or PASI100). Patients could transition to death from any health state (Figure 1). Sequential biologic therapy was not considered in the model because of the lack of national guidelines on specific treatment sequencing in Spain.7,9
  | Figure 1 Schematic representation of the model structure. Note: Although not shown in the figure, patients could transition to death from any health state. Abbreviation: BSC, best supportive care. |
Since there are no head-to-head clinical trials comparing the efficacy of ixekizumab and secukinumab, PASI response rates were derived from a network meta-analysis (Table 2) of relevant clinical trials in patients with moderate-to-severe psoriasis identified in a systematic literature review,28–30 which included key studies with ixekizumab and secukinumab. The network meta-analysis focused on trials that used European Medicines Agency (EMA)-approved regimens for the comparator biologics and employed a random-effects Bayesian model for multiarm trials with a multinomial likelihood and probit link. In general, a random-effects model is preferred in a meta-analysis because it weights the average of study effect sizes to address heterogeneity between studies.31 The analysis included PASI response rates (PASI50, PASI75, PASI90, and PASI100) at the end of the induction period because long-term data from well-designed clinical trials were lacking.
Target population and mortality data
The cost-effectiveness analysis was based on a target population of patients with moderate-to-severe plaque psoriasis who had a PASI score ≥10 and a DLQI score >10. Patient characteristics for this hypothetical cohort were the same as those in the Phase III UNCOVER-2 and -3 trials with ixekizumab,22 with a mean age of 45 years and 66.6% being men.
Normal population mortality data for Spain were extracted from the Spanish National Institute of Statistics.32 Mortality functions were subsequently derived from these data and weighted by gender as per the base-case population in the UNCOVER-2 and -3 trials.22
Health-related quality of life: assignment of health utilities
Health-related quality of life was modeled using health utility gains based on the PASI response at the end of the 12-week induction period. Health utility gains were not treatment dependent and thus were the same for ixekizumab and secukinumab for each PASI response level. It was assumed that a response of PASI75–89, PASI90–99, or PASI100 achieved at the end of the induction period was sustained throughout the maintenance period until discontinuation; therefore, patients accrued a fixed health utility gain specific to response. Patients with a response <PASI75 at the end of the induction period did not transition to the maintenance phase or accrue maintenance-related health utility gains. It was assumed that these patients would revert to baseline PASI score at the end of the induction period. For each treatment group, total utility gains were derived as the sum of utilities gained across all patients for each treatment in each arm, ie, ixekizumab→BSC or secukinumab→BSC.
The assigned health utility gains from baseline to the end of the induction period for each PASI response category were estimated using a regression model of EuroQol-5 dimensions-five levels (EQ-5D-5L) data from patients with a DLQI score >10 in the UNCOVER trials,22 applying the Spanish value set33 and adjusting for the baseline EQ-5D-5L (Table 2). A mapping (“cross-walk”) approach was used to derive a value set for the currently available three-level version of the EQ-5D (EQ-5D-3L).34
Health utility gains for BSC were based on the 12-week PASI response data for placebo in the network meta-analysis (ie, the same induction period duration as for ixekizumab and secukinumab, as well as most other biologics included in the network meta-analysis). For the death health state, utility was set to zero.
Resource use and costs
The analysis modeled resources that were relevant to the Spanish NHS and therefore considered only direct medical costs. Health care resource use associated with the biologic agents, and traditional systemic drugs (used for BSC) included nurse and physician visits and various monitoring tests, which were estimated by a panel of four Spanish dermatologists with substantial experience in the treatment of psoriasis (Table 3) and were the same for both biologics.
Drug acquisition costs were derived from the Spanish database BOT Plus 2.035 and included the current public list price of €934.25 per dose for ixekizumab. Costs for biologics were based on the ex-factory price and inclusion of a 7.5% discount, as per Real Decreto-ley 8/2010.36 Unit costs related to the administration and monitoring of treatment were derived from the health care costing database eSalud (Table 4).37
  | Table 4 Unit costs for drug acquisition, administration, and monitoring Notes: aCost of ixekizumab (2016 value) was calculated based on the ex-factory price and inclusion of a 7.5% discount.28 bAnnual cost for BSC calculated from average total cost of methotrexate, ciclosporin, phototherapy, and acitretin (includes treatment, physician visits, and monitoring costs). Abbreviations: BSC, best supportive care; IV, intravenous. |
BSC comprised methotrexate, ciclosporin, phototherapy, and acitretin as proposed by a panel of experts in in the treatment of psoriasis, and Spanish unit costs reflecting current treatment practice were applied. Physician visits and monitoring costs related to BSC were also included.
Table 5 shows total costs for biologic therapy during the 12-week induction period and total annual costs of these agents in the maintenance period. All costs and benefits were discounted at an annual rate of 3.0% as per Spanish guidelines.38
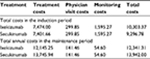  | Table 5 Total costs (€, year 2015 values) for biologic therapy in the 12-week induction period and total annual costs in the maintenance period |
Sensitivity analyses
Sensitivity analyses were conducted to address the inherent uncertainties of economic modeling that relies on the data collected from a variety of sources and includes assumptions or inputs from dermatologists with substantial experience in the treatment of psoriasis where data are lacking. These included a one-way (deterministic) sensitivity analysis and a probabilistic sensitivity analysis. In the one-way sensitivity analysis, one variable was altered at a time to assess the effect on the results, including drug costs, PASI response rates, monitoring frequency, discounting, annual discontinuation rate for maintenance therapy, physician visits, and the annual cost of BSC (Table S1). Where available, upper and lower CIs were used, whereas input parameters were varied by ±20% in most cases where CI values were lacking. Some parameters, such as physician visits and monitoring frequency, were varied by ±1 visit or ±1 test, and discounting was varied from 0% to 5%, as per Spanish guidelines.38
In the probabilistic sensitivity analysis, a Gamma distribution was assigned for parameters that can vary between zero and infinity, such as costs, and a beta distribution was assumed for those that can range between zero and one, such as the annual discontinuation rate for maintenance therapy. Input parameters were sampled from these distributions in 1,000 simulations. The probabilistic sensitivity analysis provides a distribution of incremental cost-effectiveness ratios (ICERs) from which cost-effectiveness acceptability curves can be derived. The input parameters for the probabilistic sensitivity analysis included utilities, annual discontinuation rate for maintenance therapy, health care resource use (eg, physician visits, nurse visits, monitoring, hospital costs), and efficacy.
Results
Base-case analysis
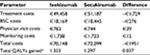
Findings of the base-case analysis showed that ixekizumab was more effective (ie, was associated with more QALYs) and less costly than secukinumab; therefore, ixekizumab dominated secukinumab. Over a lifetime horizon, ixekizumab provided an additional 0.037 QALYs (after rounding; 1.333 vs. 1.297) and reduced total costs by €1951 compared with secukinumab (€70,148 vs. €72,099). For both biologics, treatment costs represented ≈70% of total direct costs and BSC accounted for ≈25% of total direct costs, with the remainder comprising physician visit and monitoring costs (Table 6). In general, differences between treatment arms for costs and QALYs reflected the difference in duration of time in the biologic treatment and BSC health states.
Sensitivity analyses
The model was most sensitive to changes in drug costs for ixekizumab and secukinumab in the one-way sensitivity analysis (Figure 2). When the cost of ixekizumab or secukinumab was modified by ±20% from base-case costs, results ranged from ixekizumab continuing to be less costly and provide more QALYs than secukinumab (ie, negative ICER values in Figure 2 indicate that ixekizumab dominates secukinumab) to an ICER exceeding €220,000 per QALY gained for ixekizumab vs. secukinumab. Results were also sensitive, albeit less so, to changes in PASI75 response rates using 95% CI values and to changes in the number of maintenance doses of secukinumab per year. For example, in the base case, it was assumed that secukinumab maintenance therapy would be administered every 4 weeks (ie, 13 doses annually), as per randomized controlled trials included in the network meta-analysis to derive data on the efficacy of secukinumab. In the one-way sensitivity analysis, administration of secukinumab once monthly as per the current European labeling for maintenance therapy39 (ie, 12 doses annually and assuming the same efficacy) resulted in an ICER of €39,263 per QALY gained (ixekizumab vs. secukinumab). In general, results were robust to changes in other variables.
In the probabilistic sensitivity analysis, 74% of the observations were in the south-east quadrant where ixekizumab is less costly and provides more QALYs than secukinumab (Figure 3). The cost-effectiveness acceptability curve shows an 83.5% likelihood that ixekizumab would be cost effective compared with secukinumab at a willingness-to-pay threshold of €0 per QALY gained and a 96.6% probability of being cost effective at a threshold of €30,000 per QALY gained (Figure 4).
  | Figure 4 Cost-effectiveness acceptability curve for Ixe compared with Sec. Abbreviations: Ixe, ixekizumab; Q2W, every 2 weeks; Sec, secukinumab. |
Discussion
This cost-effectiveness analysis compared two IL-17A antagonists, ixekizumab and secukinumab, in the management of patients with moderate-to-severe plaque psoriasis in Spain. Ixekizumab provided more QALYs at a lower total cost than secukinumab over a lifetime horizon. This is the first cost-effectiveness analysis evaluating these relatively new biologics from the perspective of the Spanish NHS.
In clinical practice, sequential biologic therapy is commonly used for patients with moderate-to-severe plaque psoriasis, typically because of poor response at the end of induction therapy or loss of efficacy over time during maintenance therapy.6,11,40,41 However, the specific treatment sequence for biologic agents is generally at the discretion of the treating dermatologist, as there is no robust evidence to recommend biologics in a particular order at a national level in Spain.7,9 Similarly, European guidelines do not recommend biologics in a specific order.14,15 Therefore, we elected not to include sequential biologic therapy in our cost-effectiveness model.
Our model used a similar framework to that of the well-established York model17 but with important modifications, including age-dependent, gender-weighted mortality risk over a lifetime horizon. Another strength is that our model incorporated utility values directly elicited from patients with moderate-to-severe psoriasis using the EQ-5D-5L instrument in the Phase III UNCOVER trials with ixekizumab. In addition, because of the lack of published real-world data in Spain, we used relatively conservative BSC costs (ie, much lower than published sources from other countries6,42,43) informed by expert opinion, and we consider these costs to be reflective of current treatment practices for psoriasis in Spain. However, varying base-case BSC costs by ±20% had a minimal effect on results. Nevertheless, BSC costs may have a potentially large effect outside of the Spanish context, as BSC may be a key driver of cost effectiveness given its potential impact on total costs and QALYs over the model horizon. For example, in our analysis, BSC costs represented about one-quarter of total costs, and differences between treatment arms for costs and QALYs generally reflected the difference in duration of time in the biologic treatment and BSC health states. Furthermore, these results highlight the importance of modeling based on the evaluation of the relative cost effectiveness of secukinumab and ixekizumab. A more simplistic comparison comprising only medication costs and treatment efficacy would neither account for the impact of effects after discontinuation of the two treatments nor facilitate a quantification of the potential importance of BSC in the decision context. In general, results of the one-way sensitivity analysis were robust to plausible modifications to key input variables. The notable exceptions were changes to the costs of ixekizumab or secukinumab and, to a lesser degree, modifications of PASI75 response rates. The marked variability in results when IL-17A antagonist costs were varied by ±20% highlights a limitation of our analysis in that it used public list prices for these agents, rather than contract prices arranged at the hospital level, other discounted prices, or preferentially priced Patient Access Scheme costs. Probabilistic sensitivity analysis provided a cost-effectiveness acceptability curve showing that the likelihood of ixekizumab being cost effective compared with secukinumab was 83.5%-96.6% at willingness-to-pay thresholds ≤€30,000 per QALY gained. This use of ≤€30,000 per QALY gained is reasonable considering recent estimates of a cost-effectiveness threshold for the Spanish NHS.44 Therefore, although total costs were similar for the two treatments, results may still provide insights useful for decision making, with the proviso that the model did not consider some factors that might drive treatment decisions, such as patient preference for number of injections or long-term safety (eg, related to time on the market).
In our model, treatment responders were assumed to maintain response to biologic therapy at a constant level until discontinuation. For both ixekizumab and secukinumab, responders discontinued therapy at a constant annual rate of 20% during maintenance therapy. This rate of 20% per year has also been used in other analyses8,19,25 based on the York model,17 and real-world evidence from European registry studies26,27 suggests that it is reasonable to assume that it would capture patients who discontinue treatment because of loss of response, adverse events, or other reasons. This assumption is also generally supported by data from clinical practice, but robust evidence is limited or lacking, especially for drug-specific discontinuation rates.
The model did not consider effect modification to account for a potentially reduced efficacy in patients who had previously received biologic therapy; however, it is noteworthy that a prespecified subgroup analysis in the UNCOVER-2 and -3 trials showed similar PASI75 response rates with ixekizumab in biologic-naive and -experienced patients.22 In addition, the analysis did not include costs associated with serious adverse events requiring hospitalization, as data are limited and such events would be expected to make a relatively small contribution to total costs and outcomes.8,45 Long-term safety data on the use of biologics in the treatment of psoriasis indicate a low rate of serious adverse events.46,47 Also of note is that the model did not include real-world published data on health care resource use because these data are lacking in Spain. Instead, we used estimates from dermatologists in Spain with extensive experience in treating patients with psoriasis.
As highlighted in various systematic reviews, numerous cost-effectiveness analyses have been conducted with biologics in the management of psoriasis, although results have been variable,48,49 and until our study, no analyses had been conducted with ixekizumab in Spain. Interestingly, a recent cost-consequence analysis conducted from the Spanish NHS perspective found secukinumab to be the most efficient biologic for achieving clear skin in patients with moderate-to-severe plaque psoriasis; however, the study did not include ixekizumab and had only a 2-year time horizon.50 A UK cost-effectiveness analysis of sequential biologic therapy with ixekizumab vs. secukinumab as first-line therapy in patients with moderate-to-severe psoriasis also showed that ixekizumab was dominant over secukinumab, although again the differences in costs and QALYs were modest.51
Considering the variability in results of economic analyses with biologics in moderate-to-severe plaque psoriasis, including some of the findings in the one-way sensitivity analysis of our own model, alternative frameworks to the Markov-based York model could be used to further explore modeling uncertainty. For example, patient-level simulations could be used if data were available. Further cost-effectiveness analyses comparing ixekizumab and secukinumab could be designed to include real-world registry data on treatment continuation and long-term efficacy, as this could validate the model and may provide further support for the robustness of our findings.
The limited data for selection of biologics based on the efficacy and cost effectiveness from the Spanish NHS perspective in patients with moderate-to-severe plaque psoriasis, together with the lack of national guidelines providing recommendations in this area, especially for the selection of specific sequential biologic regimens, highlight the difficulties that dermatologists face in clinical practice. Nevertheless, this study provides additional information in this area, which may help guide treatment decisions.
Conclusion
This cost-effectiveness analysis showed that, over a lifetime horizon, ixekizumab provided additional QALYs and lower costs than secukinumab in patients with moderate-to-severe plaque psoriasis in Spain. Therefore, our findings could translate to savings for the Spanish NHS, although cost and QALY differences between treatments was modest, and other factors such as patient preferences and long-term safety may also be considered in clinical decision making. Results were most sensitive to changes in costs for these IL-17A antagonists.
Acknowledgments
The authors would like to acknowledge Greg Plosker and Karen Goa (Rx Communications, Mold, UK) for medical writing assistance with the preparation of this manuscript. This study was funded by Eli Lilly and Company.
Disclosure
EJ and AS are employees of Mapi Group, which was commissioned by Eli Lilly and Company to develop the cost-effectiveness model and conduct this research. MN, SH, and TD are employees of Eli Lilly and Company, receive a salary, and own company stock. The authors report no other conflicts of interest in this work.
References
Smith CH, Barker JN. Psoriasis and its management. BMJ. 2006;333(7564):380–384. | ||
Lebwohl M, Psoriasis LM. Psoriasis. Lancet. 2003;361(9364):1197–1204. | ||
Ferrándiz C, Carrascosa JM, Toro M. Prevalence of psoriasis in Spain in the age of biologics. Actas Dermosifiliogr. 2014;105(5):504–509. | ||
Menter A, Gottlieb A, Feldman SR, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: Section 1. Overview of psoriasis and guidelines of care for the treatment of psoriasis with biologics. J Am Acad Dermatol. 2008;58(5):826–850. | ||
Boehncke WH. New biologics in psoriasis – progress and problems. J Eur Acad Dermatol Venereol. 2017;31(10):1577–1578. | ||
National Institute for Health and Care Excellence [webpage on the Internet]. Psoriasis – The Assessment and Management of Psoriasis [CG153]. London: NICE; 2012. Available from: https://www.nice.org.uk/guidance/cg153?unlid=9392308262017125121344. Accessed November 4, 2017. | ||
Daudén E, Puig L, Ferrándiz C, Sánchez-Carazo JL, Hernanz-Hermosa JM; Spanish Psoriasis Group of the Spanish Academy of Dermatology and Venereology. Consensus document on the evaluation and treatment of moderate-to-severe psoriasis: Psoriasis Group of the Spanish Academy of Dermatology and Venereology. J Eur Acad Dermatol Venereol. 2016;30 Suppl 2(S2):1–18. | ||
National Institute for Health and Care Excellence [webpage on the Internet]. Secukinumab for Treating Moderate to Severe Plaque Psoriasis. Technology Appraisal Guidance TA350. London: NICE; 2015. Available from: https://www.nice.org.uk/guidance/ta350. Accessed November 7, 2017. | ||
Puig L, Carrascosa JM, Carretero G, et al. Spanish evidence-based guidelines on the treatment of psoriasis with biologic agents, 2013. Part 1: on efficacy and choice of treatment. Actas Dermosifliogr. 2013;104(8):694–709. | ||
Medicines Agency. Ixekizumab (Taltz®) Summary of Product Characteristics. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/003943/WC500205804.pdf. Accessed November 27, 2017. | ||
Mrowietz U, de Jong EM, Kragballe K, et al. A consensus report on appropriate treatment optimization and transitioning in the management of moderate-to-severe plaque psoriasis. J Eur Acad Dermatol Venereol. 2014;28(4):438–453. | ||
Cohen SN, Baron SE, Archer CB; British Association of Dermatologists and Royal College of General Practitioners. Guidance on the diagnosis and clinical management of psoriasis. Clin Exp Dermatol. 2012;37 Suppl 1:13–18. | ||
Greater Manchester Medicines Management Group (GMMMG), New Therapies Subgroup [webpage on the Internet]. The Sequential Use of Biologic Agents in the Treatment of Chronic or Plaque Psoriasis, for Those Patients, Fulfilling NICE Criteria for a Biologic. 2015. Available from: http://studyres.com/doc/19079427/biologic-agents-sequential-use-in-psoriasis. Accessed February 26, 2018. | ||
Nast A, Gisondi P, Ormerod AD, et al. European S3-Guidelines on the systemic treatment of psoriasis vulgaris--Update 2015--Short version--EDF in cooperation with EADV and IPC. J Eur Acad Dermatol Venereol. 2015;29(12):2277–2294. | ||
Nast A, Spuls PI, van der Kraaij G, Kraaij G, et al. European S3-Guideline on the systemic treatment of psoriasis vulgaris - Update Apremilast and Secukinumab - EDF in cooperation with EADV and IPC. J Eur Acad Dermatol Venereol. 2017;31(12):1951–1963. | ||
Smith CH, Anstey AV, Barker JNWN, et al. British Association of Dermatologists’ guidelines for biologic interventions for psoriasis 2009. Br J Dermatol. 2009;161(5):987–1019. | ||
Woolacott N, Hawkins N, Mason A, et al. Etanercept and efalizumab for the treatment of psoriasis: a systematic review. Health Technol Assess. 2006;10(46):1–233–i–iv. | ||
Zhang W, Islam N, Ma C, Anis AH. Systematic review of cost-effectiveness analyses of treatments for psoriasis. Pharmacoeconomics. 2015;33(4):327–340. | ||
Villacorta R, Hay JW, Messali A. Cost effectiveness of moderate to severe psoriasis therapy with etanercept and ustekinumab in the United States. Pharmacoeconomics. 2013;31(9):823–839. | ||
Siebert U, Alagoz O, Bayoumi AM, et al; ISPOR-SMDM Modeling Good Research Practices Task Force. State-transition modeling: a report of the ISPOR-SMDM Modeling Good Research Practices Task Force – 3. Value Health. 2012;15(6):812–820. | ||
Naimark DM, Kabboul NN, Krahn MD. The half-cycle correction revisited: redemption of a kludge. Med Decis Making. 2013;33(7):961–970. | ||
Griffiths CE, Reich K, Lebwohl M, et al. Comparison of ixekizumab with etanercept or placebo in moderate-to-severe psoriasis (UNCOVER-2 and UNCOVER-3): results from two phase 3 randomised trials. Lancet. 2015;386(9993):541–551. | ||
Bartos S, Hill D, Feldman SR. Review of maintenance of response to psoriasis treatments. J Dermatolog Treat. 2016;27(4):293–297. | ||
Blauvelt A, Gooderham M, Iversen L, et al. Efficacy and safety of ixekizumab for the treatment of moderate-to-severe plaque psoriasis: results through 108weeks of a randomized, controlled phase 3 clinical trial (UNCOVER-3. J Am Acad Dermatol. 2017;77(5):855–862. | ||
National Institute for Health and Care Excellence [webpage on the Internet]. Apremilast for Treating Moderate to Severe Plaque Psoriasis. Final Appraisal Determination. London: NICE; 2015. Available from: https://www.nice.org.uk/guidance/gid-tag469/resources/psoriasis-plaque-moderate-to-severe-apremilast-id679-final-appraisal-determination-document2. Accessed November 4, 2017. | ||
Warren RB, Smith CH, Yiu ZZN, et al. Differential drug survival of biologic therapies for the treatment of psoriasis: a prospective observational cohort study from the British Association of Dermatologists Biologic Interventions Register (BADBIR). J Invest Dermatol. 2015;135(11):2632–2640. | ||
Gniadecki R, Bang B, Bryld LE, Iversen L, Lasthein S, Skov L. Comparison of long-term drug survival and safety of biologic agents in patients with psoriasis vulgaris. Br J Dermatol. 2015;172(1):244–252. | ||
Hartz S, Dutronc Y, Kiri SH, Schacht A, Walzer S, Dakin H. Network meta-analysis to evaluate the efficacy of ixekizumab in the treatment of moderate-to-severe psoriasis. Poster presented at: ISPOR 19th Annual European Congress; 2016 October 29–November 2; Vienna. | ||
National Institute for Health and Care Excellence. Single Technology Appraisal: Ixekizumab for Treating Moderate to Severe Plaque Psoriasis [ID904]. London: Committee Papers NICE; 2017:section 4.9: 137–4.9:174. Available from: https://www.nice.org.uk/guidance/ta442/documents/committee-papers-3. Accessed November 5, 2017. | ||
Nuñez M, Johansson E, Svedbom A, Dilla T, Hartz S. Cost-effectiveness of ixekizumab vs. secukinumab in the treatment of moderate to severe plaque psoriasis in Spain. Poster presented at: 45th Congreso Nacional, Academia Española de Dermatología y Venereología (AEDV); 2017; Madrid, Spain. | ||
Augustin M, Mcbride D, Gilloteau I, et al. Cost-effectiveness of secukinumab as first biologic treatment, compared with other biologics, for moderate to severe psoriasis in Germany. J Eur Acad Dermatol Venereol. In press 2018. | ||
National Statistics Institute Spain [homepage on the Internet]. Mortality Rate (Each 1. 000 Habitants) by Sex and Age: (Mortality Data from 2016 Were Used in the Analysis). Available from: http://www.ine.es. Accessed June 15, 2018. | ||
Badia X, Roset M, Herdman M, Kind P. A comparison of United Kingdom and Spanish general population time trade-off values for EQ-5D health states. Med Decis Making. 2001;21(1):7–16. | ||
van Hout B, Janssen MF, Feng YS, et al. Interim scoring for the EQ-5D-5L: mapping the EQ-5D-5L to EQ-5D-3L value sets. Value Health. 2012;15(5):708–715. | ||
BOT Plus 2.0 (Conejos General de Colegios Oficiales de Farmacéuticos). [General Council Official Associations of Pharmacists]. [homepage on the Internet]. Available from: https://botplusweb.portalfarma.com/. Accessed December 1, 2015. Spanish. | ||
Agencia Estatal Boletín Oficial del Estado [webpage on the Internet]. Real Decreto-ley 8/2010, de 20 de mayo, por el que se adoptan medidas extraordinarias para la reducción del deficit public. [Royal Decree-Law 8/2010, May 20, which adopts extraordinary measures to reduce public deficit.] Available from: https://www.boe.es/diario_boe/txt.php?id=BOE-A-2010-8228. Accessed November 3, 2017. Spanish. | ||
Oblikue [webpage on the Internet]. eSalud-Infomación económica del sector sanitario (Oblikue consulting). [eSalud-Economic information of the health sector (Oblikue consulting)]. Available from: http://www.oblikue.com/bddcostes/. Accessed December 1, 2015. Spanish. | ||
López-Bastida J, Oliva J, Antoñanzas F, et al. Spanish recommendations on economic evaluation of health technologies. Eur J Health Econ. 2010;11(5):513–520. | ||
European Medicines Agency. Secukinumab (Cosentyx®) Summary of Product Characteristics. Available from: https://ec.europa.eu/health/documents/community-register/2015/20150115130444/anx_130444_en.pdf. Accessed November 27, 2017. | ||
Kerdel F, Zaiac M. An evolution in switching therapy for psoriasis patients who fail to meet treatment goals. Dermatol Ther. 2015;28(6):390–403. | ||
Leman J, Burden AD. Sequential use of biologics in the treatment of moderate-to-severe plaque psoriasis. Br J Dermatol. 2012;167 (Suppl 3):12–20. | ||
Sawyer LM, Wonderling D, Jackson K, et al. Biological therapies for the treatment of severe psoriasis in patients with previous exposure to biological therapy: a cost-effectiveness analysis. Pharmacoeconomics. 2015;33(2):163–177. | ||
Fonia A, Jackson K, Lereun C, et al. A retrospective cohort study of the impact of biologic therapy initiation on medical resource use and costs in patients with moderate to severe psoriasis. Br J Dermatol. 2010;163(4):807–816. | ||
Vallejo-Torres L, Garcia-Lorenzo B, Serrano-Aguilar P. Estimating a Cost-Effectiveness Threshold for the Spanish NHS. Available from: http://documentos.fedea.net/pubs/eee/eee2016-22.pdf. Accessed November 6, 2017. | ||
Anis AH, Bansback N, Sizto S, Gupta SR, Willian MK, Feldman SR. Economic evaluation of biologic therapies for the treatment of moderate to severe psoriasis in the United States. J Dermatolog Treat. 2011;22(2):65–74. | ||
Mansouri Y, Goldenberg G. Biologic safety in psoriasis: review of long-term safety data. J Clin Aesthet Dermatol. 2015;8(2):30–42. | ||
Jancin B [webpage on the Internet]. Registry shows no increased cancer risk with biologics for psoriasis. Dermatology News. 2016. Available from: https://www.mdedge.com/edermatologynews/article/106783/psoriasis/registry-shows-no-increased-cancer-risk-biologics. Accessed May 31, 2018. | ||
Gutknecht M, Krensel M, Augustin M. Health economic analyses of psoriasis management: a systematic literature search. Arch Dermatol Res. 2016;308(9):601–616. | ||
Mauskopf J, Samuel M, Mcbride D, Mallya UG, Feldman SR. Treatment sequencing after failure of the first biologic in cost-effectiveness models of psoriasis: a systematic review of published models and clinical practice guidelines. Pharmacoeconomics. 2014;32(4):395–409. | ||
Puig L, Notario J, Jiménez-Morales A, et al. Secukinumab is the most efficient treatment for achieving clear skin in psoriatic patients: a cost-consequence study from the Spanish National Health Service. J Dermatolog Treat. 2017;28(7):623–630. | ||
Johansson EC, Hartz S, Kiri SH, Kumar G, Svedbom A. Cost-effectiveness analysis of sequential biologic therapy with ixekizumab versus secukinumab as first-line treatment of moderate-to-severe psoriasis in the UK. J Med Econ. 2018;21(8):810–820. |
Supplementary material
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.