Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 17
Correlation Analysis of Early Renal Injury in Elderly Patients with Acute Exacerbations of Chronic Obstructive Pulmonary Disease
Authors Zhang D
Received 10 June 2022
Accepted for publication 17 August 2022
Published 6 September 2022 Volume 2022:17 Pages 2109—2115
DOI https://doi.org/10.2147/COPD.S377847
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Dong Zhang
Department of Internal Medicine, The Second Hospital of Beijing, Beijing, 100031, People’s Republic of China
Correspondence: Dong Zhang, Department of Internal Medicine, The Second Hospital of Beijing, No. 36 of Youfang Hutong, Xicheng District, Beijing, 100031, People’s Republic of China, Tel +86 10 66061122, Email [email protected]
Objective: To investigate the correlation between serum cystatin C (Cys-C) and beta-2 (β 2) microglobulin (β 2-MG) levels and renal injury in elderly patients with acute exacerbations of chronic obstructive pulmonary disease (AECOPD) with a view to detecting renal injury in its early stages.
Methods: A total of 106 patients with AECOPD were enrolled and divided into three groups according to their oxygen partial pressure (PO2) levels: severe hypoxia group, moderate hypoxia group, and mild hypoxia group. Another 60 healthy subjects were selected as the control group. General clinical data were collected from all the study subjects, along with measurements of arterial blood gas, Cys-C, β 2-MG, serum creatinine (Scr), urea nitrogen (BUN), partial pressure of carbon dioxide (PCO2), and high-sensitivity C-reactive protein (hs-CRP).
Results: The levels of hs-CRP, Cys-C, β 2-MG, Scr, and BUN were highest in the severe hypoxia group, followed by the moderate hypoxia group, then the mild hypoxia group, and lowest in the control group. The differences between the groups were statistically significant for these indicators (P < 0.05). Apart from in the cases of Scr and BUN, there were no statistically significant differences between the mild group and the control group (P > 0.05). The levels of Cys-C and β 2-MG were positively correlated with the levels of hs-CRP, PCO2, Scr, and BUN and negatively correlated with PO2 levels. hs-CRP and PO2 were high-risk factors influencing Cys-C levels, and β 2-MG was a risk factor influencing Cys-C levels. The level of PO2 was a high-risk factor influencing β 2-MG levels, and PCO2 and Cys-C were risk factors influencing β 2-MG levels.
Conclusion: Renal injury was found to be present in patients with AECOPD and worsened with increasing degrees of hypoxia. Hypoxia and inflammation might be risk factors for renal injury in patients with AECOPD, Cys-C and β 2-MG could be sensitive indicators for the early detection of renal injury.
Keywords: cystatin C, β 2 microglobulin, acute exacerbations of chronic obstructive pulmonary disease, early renal injury
Introduction
Chronic obstructive pulmonary disease (COPD) is a disease that seriously affects the human respiratory system, but it is preventable and treatable It is usually characterized by persistent airflow limitation caused mainly by an excessive pulmonary inflammatory response to pollutants in the airways, such as cigarettes and smoke.1 Currently, COPD is the fourth leading cause of death worldwide and imposes a heavy economic burden on sufferers.2
The main organ involved in COPD is the pulmonary tissue, and its causes may be attributed to chronic tissue inflammation and long-term hypoxia. The extra-pulmonary comorbidities of COPD have been gaining attention and recent studies have suggested a possible association between COPD and renal insufficiency.3 Acute exacerbations of chronic obstructive pulmonary disease (AECOPD) may be diagnosed in patients with COPD when there is an increase in the frequency and degree of coughing, an increase in sputum volume, or a change in sputum properties, together with an increase in the degree of dyspnea. Long-term ischemia and hypoxia in patients with AECOPD and the impact of inflammation and carbon dioxide retention can eventually result in or aggravate renal injury, and the early detection and treatment of renal injury in patients with COPD has received much attention in recent years. At present, serum creatinine (Scr) is routinely used to detect and evaluate renal injury, but usually there is no abnormal change in Scr in the early stages of renal injury, and when levels rise, it often indicates that the kidney already has a rapidly progressing irreversible injury. Such an injury has a negative effect on the prognosis of patients and increases mortality rates. It is known that changes in Scr levels can be affected by age, gender, diet, muscle quality, drugs, and other factors,4 which means that any change may not accurately reflect a renal function injury. Thus, clinicians need more sensitive and timely biological indicators. One major member of the cysteine protease inhibitor superfamily is serum cystatin C (Cys-C), which is a soluble, non-glycosylated, secreted protein that is continuously produced by nucleated cells in the body and is widely present in body fluids. Moreover, its secretion and excretion are constant and are not affected by many factors. Since Cys-C can freely pass through the glomerular filtration membrane and be completely reabsorbed and catabolized by the renal proximal tubules, and the tubules themselves cannot secrete Cys-C, it is an ideal indicator for the glomerular filtration rate (GFR).5 The production, synthesis, and release of beta-2 (β2) microglobulin (β2-MG) are constant in the normal population; it is filtered through the glomerulus, and 99% is reabsorbed and catabolized in the proximal tubule. It has been used not only to assess tubular function but also to monitor glomerular function. As a new biological marker, the level of β2-MG increases earlier than Scr in acute renal injury and is not affected by muscle metabolism. It has previously been suggested that β2-MG is a better predictor for acute renal injury than Scr.6
The present study aimed to investigate the role of Cys-C and β2-MG as biomarkers for the early identification of renal injury due to hypoxia and inflammatory factors in elderly patients with AECOPD by means of correlation analysis.
Materials and Methods
Study Subjects and Grouping
A total of 106 elderly patients with AECOPD who visited the Internal Medicine Department of the Second Hospital of Beijing between January 2018 and December 2021 were enrolled in the study. All enrolled patients had a definite diagnosis of COPD. This study was conducted in accordance with the declaration of Helsinki and with the approval of the Ethics Committee of the Second Hospital of Beijing. Written informed consent was obtained from all the participants.
The patients were divided into three study groups based on oxygen partial pressure (PO2) levels in the arterial blood gas analysis on admission: 60 mm Hg < PO2 ≤ 80 mm Hg = mild hypoxia group (Group A with 32 cases), 40 mm Hg < PO2 ≤ 60 mm Hg = moderate hypoxia group (Group B with 38 cases), and PO2 ≤ 40 mm Hg = severe hypoxia group (Group C with 36 cases). During the same period, another 60 healthy subjects who visited the hospital for a physical examination were selected as the control group. This group consisted of 36 male patients and 24 female patients, with an average age of (79.44 ± 11.30) years. The differences between the four groups in age and gender were not statistically significant (both P > 0.05), and the data were comparable.
Inclusion and Exclusion Criteria
The inclusion criteria for patients with AECOPD was as follows: (1) the patient met the diagnostic criteria for COPD in the Revised Guidelines for the Diagnosis and Management of Chronic Obstructive Pulmonary Disease issued by the Respiratory Diseases Branch of the Chinese Medical Association;7 (2) the patient was in an acute exacerbation phase at the time of consultation, ie, with complaints of a sudden exacerbation of coughing, sputum, and shortness of breath, and the degree of exacerbation exceeded the range of daily variation to the extent that the original therapeutic plan had to be adjusted; (3) the patient was older than 65 and their medical data record was complete; and (4) the patient or a family member signed an informed consent form.
The exclusion criteria were as follows: (1) patients with multiple organ failures from various causes; (2) patients with primary or secondary kidney disease, such as hypertensive renal injury, chronic glomerulonephritis, systemic lupus erythematosus nephritis, and diabetic renal injury; (3) patients with malignant tumors; (4) patients complicated with hematological and bone marrow diseases; (5) patients with coronary artery disease or heart failure; and (6) patients who had been administered nephrotoxic drugs six weeks or less before admission.
Data Collection and Detection Indicators
General Data
General data, including gender, age, body mass index (BMI), blood pressure, and smoking index were recorded for all enrolled subjects.
Detection Indicators
Each patient’s PO2 and carbon dioxide partial pressure (PCO2) in arterial blood were measured upon admission, and blood specimens were collected under fasting conditions on the morning of the second day after admission. The serum levels of high-sensitivity C-reactive protein (hs-CRP), Scr, urea nitrogen (BUN), Cys-C, and β2-MG were measured uniformly. Levels of Cys-C were measured using the latex-enhanced immunoturbidimetric method, with a kit produced by DiyaLaibo Biotechnology Co., Ltd (China, Jiangsu), while β2-MG was measured using the latex immunoturbidimetric method, with a kit produced by Biosino Biotechnology and Science Inc.(China, Beijing). The Scr and BUN levels were measured using the picric acid urea method, with a kit also produced by Biosino Biotechnology and Science Inc. All indicators were measured using a Beckman AU5800 automatic biochemical analyzer, which was operated in strict accordance with the relevant instructions.
Statistical Analysis
The SPSS 20.0 software package was used for data processing. The measurement data that satisfied the normal distribution were expressed as mean ± standard deviation (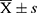 ). An independent-sample t-test was used to compare the means between two groups, one-way analysis of variance was used to compare the means between multiple groups, and a χ2 test was used for countable data. The correlations of the levels of Cys-C and β2-MG with other variables were analyzed using Spearman correlation analysis, and multiple linear regression analysis was constructed based on the results of the correlation analysis. P < 0.05 was considered statistically significant.
). An independent-sample t-test was used to compare the means between two groups, one-way analysis of variance was used to compare the means between multiple groups, and a χ2 test was used for countable data. The correlations of the levels of Cys-C and β2-MG with other variables were analyzed using Spearman correlation analysis, and multiple linear regression analysis was constructed based on the results of the correlation analysis. P < 0.05 was considered statistically significant.
Results
Comparison of the General Characteristics
Of all 106 patients, 55 were male and 51 were female, with an average age of (75.46 ± 8.62) years. The differences between the study groups and the control group in age, gender, BMI, smoking index, systolic blood pressure, and diastolic blood pressure were not statistically significant (P > 0.05). The PO2 levels in the study groups were lower than those in the control group, and the difference was statistically significant (P < 0.05). The levels of hs-CRP, PCO2, Scr, BUN, Cys-C, and β2-MG in the study groups were higher than those in the control group, and the differences were statistically significant (P < 0.05) (see Table 1).
 |
Table 1 Analysis of the General Characteristics Among Different Groups of Subjects |
Comparison of Serum Cystatin C, Beta-2 Microglobulin, Partial Pressure of Carbon Dioxide, Serum Creatinine, and Urea Nitrogen
Compared with the control group, the levels of hs-CRP, Cys-C and β2-MG were significantly higher in the study groups, and the differences were statistically significant (all P < 0.05). The levels of hs-CRP, Cys-C, and β2-MG were highest in Group C, followed by Group B, then Group A, and lowest in the control group, and the differences were statistically significant (all P < 0.05). The levels of Scr and BUN were highest in Group C group, followed by Group B group, then Group A, and lowest in the control group, and the differences between Group A and the control group were not statistically significant (P > 0.05), but the differences between the remaining groups were (all P < 0.05). The PCO2 levels were also highest in Group C, followed by Group B, then Group A, and lowest in the control group, and the differences between Group C and the other groups as well as between Group B and Group A were statistically significant (all P < 0.05). The levels of PO2 were highest in the control group, followed by Group A, then Group B, and lowest in Group C, and the differences were statistically significant (all P < 0.05) (see Table 2).
 |
Table 2 Comparison of the Levels of hs-CRP, Cys-c, β2-MG, Scr, and BUN Among Different Groups |
Correlation Analysis Between Serum Cystatin C and Beta-2 Microglobulin with Each Influencing Factor
The Spearman correlation analysis showed that Cys-C levels were positively correlated with hs-CRP (r = 0.979, P < 0. 001), PCO2 (r = 0.775, P < 0.001), Scr (r = 0.674, P < 0.001), and BUN (r = 0.675, P < 0.001) and negatively correlated with PO2 (r = −0.975, P < 0.001), and there was no correlation with mean age, gender, BMI, smoking index, systolic blood pressure, or diastolic blood pressure. The levels of β2-MG were highly and positively correlated with hs-CRP (r = 0.962, P < 0. 001), PCO2 (r = 0.737, P < 0.001), Scr (r = 0.668, P < 0.001), and BUN (r = 0.688, P < 0.001) and negatively correlated with PO2 (r = −0.965, P < 0.001), and there was no correlation with mean age, gender, BMI, smoking index, systolic blood pressure, or diastolic blood pressure. The level of Cys-C was positively correlated with β2-MG (r = 0.950, P < 0. 001) (see Table 3).
 |
Table 3 Correlation Analysis of the Levels of Cys-C and β2-MG with Each Influencing Factor |
Multivariate Linear Regression Analysis of the Factors Influencing Serum Cystatin C and Beta-2 Microglobulin
Multivariate linear regression analysis was conducted with Cys-C and β2-MG as the dependent variables and hs-CRP, PCO2, PO2, Scr, and BUN as the independent variables. The results showed that hs-CRP (β = 0.139, P < 0.01) and PO2 (β = –0.073, P < 0.01) were high-risk factors influencing Cys-C levels, and β2-MG (β = 0.152, P < 0.05) was a risk factor influencing Cys-C levels. The level of PO2 (β = –0.033, P < 0.01) was a high-risk factor influencing β2-MG levels, and PCO2 (β = 0.048, P < 0.05) and Cys-C (β = 0.317, P < 0.05) were risk factors influencing β2-MG levels.
Discussion
A common, preventable, and treatable respiratory disease, COPD’s main pathological feature is chronic progressive airflow limitation characterized by long-term recurrent chronic inflammation of the airways, pulmonary parenchyma, and blood vessels. The pathological mechanism of COPD is not yet fully understood, but previous studies have shown that patients with COPD are in a state of chronic hypoxia and systemic inflammatory activation, which affects not only the lungs and airways but also the skeletal system, cardiovascular system, and urinary system.8 In a study of 356 elderly patients with COPD, the incidence of renal insufficiency was found to be 43%, compared with 23% in older adults without COPD, and the incidences of acute renal injury and chronic renal injury were significantly higher in patients with hypercapnia or hypoxemia than in the healthy population, and this not only prolonged the length of hospital stays but also led to increased mortality.9 Understanding how to detect and treat renal injury in patients with COPD at an early stage is therefore a pressing issue.
It is known that Scr and BUN levels only change significantly when renal function is already significantly damaged and that they are vulnerable to many factors, such as tumor, muscle content, and high protein food intake. In other words, Scr and BUN levels cannot accurately reflect renal function damage, especially in its early stages.4 Both Cys-C and β2-MG are small molecules with stable levels in the body; they are not affected by the external environment and are mainly absorbed in the renal tubular epithelium and degraded and excreted in urine. In the case of renal tubule injury, the re-absorption of both Cys-C and β2-MG is reduced, and the serum levels in patients in the early stage of injury are increased.10 In the present study, the correlation between Cys-C and β2-MG and renal injury in its early stages was investigated in patients with AECOPD with different degrees of hypoxia.
The results of the present study were the same as those of previous studies,11 in that the levels of Cys-C, β2-MG, hs-CRP, PCO2, Scr, and BUN were significantly increased in the study groups compared with the control group. These results indicate that the presence of renal injury in the study groups, based on chronic hypoxia and hypercapnia, might be related to the glomerular hemodynamic changes caused by chronic hypoxia. Chronic hypoxia is likely to lead to increased glomerular vasoconstriction and decreased blood flow, while ischemia and hypoxia stimulate cytokine expression and a reduction in the GFR, resulting in impaired renal function.12
In the present study, the changes in renal function in patients with AECOPD and different hypoxic conditions were analyzed, and it was concluded that renal injury in AECOPD might increase with the increase in the degree of hypoxia, but the levels of Cys-C and β2-MG were already elevated at the early stage of renal injury. As the degree of hypoxia increased, the levels of Cys-C and β2-MG increased progressively, and the differences were statistically significant in multiple comparisons between the different groups. However, the increase of Scr and BUN levels in the mild hypoxia group was not significant compared with those of the control group, suggesting that the changes in Scr and BUN in patients with mild hypoxia would fail to provide a significant indication. Only when the disease progressed to the moderate or severe stage did changes in Scr and BUN occur, indicating that the increase in Scr and BUN levels was delayed during early renal injury and influenced by a number of factors. Changes in both Scr and BUN levels occurred only when the renal function was significantly impaired, and so relying on them as indicators of renal injury might lead to a delay in the optimal treatment for patients. These findings were consistent with the results of a previous study.13 Therefore, it would appear that monitoring Cys-C and β2-MG levels rather than Scr and BUN levels is more reliable in assessing renal injury early on in patients with AECOPD.
The correlation analysis undertaken in the present study showed that Cys-C levels were positively correlated with hs-CRP, PCO2, Scr, and BUN and negatively correlated with PO2. Levels of β2-MG were positively correlated with hs-CRP, PCO2, Scr, and BUN and negatively correlated with PO2. These results suggest that long-term chronic hypoxia and hypercapnia, together with the stimulation of inflammatory factors, produces a large number of oxygen free radicals, leading to a series of chain reactions like increased intra-renal capillary resistance, reduced renal perfusion, and activation of the renin-angiotensin system, causing a certain degree of injury to renal tissues. Meanwhile, hypoxia can lead to apoptosis or differentiation of renal tubular epithelial cells and mesenchymal cells into fibroblasts, leading to tubular interstitial fibrosis and triggering or aggravating renal injury. All of these effects can lead to renal injury in patients. In addition, acute exacerbation can increase the release of cytokines and inflammatory mediators in large quantities. Numerous studies have found that a variety of inflammatory factors, such as tumor necrosis factor (TNF)-α, interleukin-6, and hs-CRP, are released in patients with COPD and that the levels of inflammatory factors increase significantly as the severity of COPD increases. Both clinical and animal experiments have confirmed that elevated levels of TNF-α and hs-CRP may lead to the development and progression of renal injury by mediating the immune-inflammatory response of the body and causing renal function impairment,14–16 resulting in the onset and development of renal injury.
Conclusion
Renal injury is seen in patients with COPD, and the extent of the renal injury in elderly patients in the acute exacerbation phase might increase with the degree of hypoxia. Compared with Scr and BUN, levels of Cys-C and β2-MG showed changes at the early stage of renal injury, so they may be sensitive indicators for the early detection of renal injury. Furthermore, PO2 and hs-CRP might be high-risk factors for both Cys-C and β2-MG, suggesting that hypoxia and the release of inflammatory factors might be risk factors for renal injury in patients with AECOPD. With the advances in the understanding of Cys-C and β2-MG in renal injury in patients with AECOPD, the early identification of renal injury and effective control of risk factors could be facilitated, potentially improving the long-term survival of patients with AECOPD.
Ethics Approval and Consent to Participate
This study was conducted in accordance with the declaration of Helsinki. This study was conducted with approval from the Ethics Committee of the Second Hospital of Beijing. A written informed consent was obtained from all participants.
Disclosure
The author reports no conflicts of interest in this work.
References
1. Li JS. International clinical practice guideline of Chinese medicine: chronic obstructive pulmonary disease. World J Tradit Chin Med. 2020;6:39–50. doi:10.4103/wjtcm.wjtcm_9_20
2. Iyer AS, Goodrich CA, Dransfield MT, et al. End-of-life spending and healthcare utilization among older adults with chronic obstructive pulmonary disease. Am J Med. 2020;133(7):817–824. doi:10.1016/j.amjmed.2019.11.024
3. Incalzi RA, Corsonello A. Chronic renal failure: a neglected comorbidity of COPD. Chest. 2010;137(4):
4. Yi SB, Hu YF, Lin YH, Chen YX, Pan JY. Comparison on predictive value of blood lactic acid at different time periods for in-hospital mortality of septic patients: based on the medical information mart for intensive care III. Chin J Crit Care Med. 2020;13(1):39–43.
5. Abdelaal N, Shalaby S, Khashana A, et al. Serum cystatin C as an earlier predictor of acute kidney injury than serum creatinine in preterm neonates with respiratory distress syndrome. Saudi J Kidney Dis Transpl. 2017;28(5):1003–1014. doi:10.4103/1319-2442.215148
6. Argyropoulos CP, Shan CS, Yue-Harn N, et al. Rediscovering beta-2 microglobulin as a biomarker across the spectrum of kidney diseases. Front Med. 2017;4(4):73–80. doi:10.3389/fmed.2017.00073
7. Vogelmeier CF, Criner GJ, Martinez FJ, et al.Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease (updated 2015).Am J Respir Crit Care Med. 2017;195(5):557–582.
8. Barnes P, Celli B. Systemic manifestations and comorbidities of COPD. Eur Respir J. 2009;33(5):1165–1185. doi:10.1183/09031936.00128008
9. López-Campos JL, Soler-Cataluña JJ, Miravitlles M. Global strategy for the diagnosis, management, and prevertion of chronic obstructive lung disease 2019 report: future challenges-sciencedirect. Arch Bronconeumol. 2020;56(2):65–67. doi:10.1016/j.arbr.2019.06.014
10. Chen H, Gao ZL. Clinical value of Cys C in early renal damage in patients with chronic kidney disease. J Trop Med. 2017;17(8):
11. John M, Hussain S, Prayle A, Simms R, Cockcroft JR, Bolton CE. Target renal damage: the microvascular associations of increased aortic stiffness in patients with COPD. Respir Res. 2013;14(1):31. doi:10.1186/1465-9921-14-31
12. Hou J, Liu Y, Liu J, Chen X. Correlation between serum biochemical indices and renal pathology in elderly patients with nephropathy. Chin J Gerontol. 2014;34(20):5754–5756.
13. Cai XS, Chi Q, Wu JG, Pan HY. Study on the relationship between renal injury and inflammatory factors in patients with different COPD grades. Chongqing Med. 2020;49(4):614–618.
14. Ghosh N, Choudhury P, Kaushik SR, et al. Metabolomic fingerprinting and systemic inflammatory profiling of asthma COPD overlap(ACO). Respir Res. 2020;21(1):126. doi:10.1186/s12931-020-01390-4
15. Gan WQ, Man SF, Sin DD. The interactions between cigarette smoking and reduced lung function on systemic inflammation. Chest. 2005;127(2):558–564. doi:10.1378/chest.127.2.558
16. Ozgul MA, Cetinkaya E, Cortuk M, et al. Our experience on silicone Y-Stent for severe COPD complicated with expiratory central airway collapse.J. Bronchol Interv Pulmonol. 2017;24(2):104–109. doi:10.1097/LBR.0000000000000346
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
