Back to Journals » International Medical Case Reports Journal » Volume 10
Corneal deposits associated with topical tosufloxacin following penetrating keratoplasty: a case report
Authors Katahira H, Kumakura S, Hattori T, Goto H
Received 17 January 2017
Accepted for publication 28 March 2017
Published 13 July 2017 Volume 2017:10 Pages 239—241
DOI https://doi.org/10.2147/IMCRJ.S132531
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Haruki Katahira, Shigeto Kumakura, Takaaki Hattori, Hiroshi Goto
Department of Ophthalmology, Tokyo Medical University, Shinjukuku, Tokyo, Japan
Purpose: To report the adverse event of topical tosufloxacin administered after penetrating keratoplasty in one patient.
Case report: A 60-year-old female was referred to our hospital for treatment of vision loss due to corneal opacification, etiology was unknown. Slit lamp examination showed dense opacification in corneal stroma. Penetrating keratoplasty was performed on her left eye. She was treated with topical applications of 1.5% levofloxacin, 0.5% cefmenoxime, 0.1% betamethasone, 0.1% hyaluronate sodium, and 3% aciclovir after penetrating keratoplasty. Delayed epithelialization of the donor graft was observed at day 4 post-transplantation. Because of the concern that levofloxacin induced corneal epithelialization delay, 1.5% levofloxacin was changed to 0.3% tosufloxacin. At day 6 post-transplantation, deposits on the epithelial defect of the donor graft were observed. Tosufloxacin was suspected to be the cause of deposits, and tosufloxacin eye drop was discontinued. The deposits decreased gradually and completely disappeared by 5 months post-transplantation.
Conclusion: Topical tosufloxacin treatment has the risk of precipitation on compromised corneas such as corneal grafts with epithelial defect after penetrating keratoplasty. After discontinuation of therapy, the deposit may resolve spontaneously without surgical removal.
Keywords: corneal deposit, tosufloxacin, penetrating keratoplasty
Introduction
Tosufloxacin, a fourth-generation fluoroquinolone, has been used topically for the treatment of ocular surface infection and prophylaxis after various intraocular surgeries because of its broad-spectrum antibacterial effects against gram-negative and gram-positive pathogens. However, corneal deposits have been reported as an adverse effect of topical application of fluoroquinolones such as ciprofloxacin,1-4 ofloxacin5 and norfloxacin,6,7 only two case reports (three cases) of corneal deposits caused by tosufloxacin have been published.8,9 Moreover, among the three cases, only one was of tosufloxacin precipitation after penetrating keratoplasty.9
We report a case of corneal deposition associated with topical tosufloxacin administered after penetrating keratoplasty.
Case Report
Written informed consent was provided by the patient to have the case details and accompanying images published.
A 60-year-old woman was referred to our hospital due to loss of vision in her left eye due to diffuse corneal opacification. She had been treated for keratitis of unknown etiology in both eyes. Her best corrected visual acuity at presentation to our hospital was 0.7 with +1.25–2.50 ×40 in the right eye and 0.1 with +4.75–4.25 ×100 in the left eye. Slit lamp examination showed stromal opacity in both corneas. Corneal endothelial cell density was 3,247 cells/mm2 in the right eye and 2,551 cells/ mm2 in the left eye. Both the rapid plasma reagin test (RPR) and the Treponema pallidum latex agglutination test (TPLA) performed using serum samples, were negative. The Schirmer test showed decrease in basic tear secretion (9 mm in the right eye, 5 mm in the left eye). After these tests, the cause of corneal opacity was unknown.
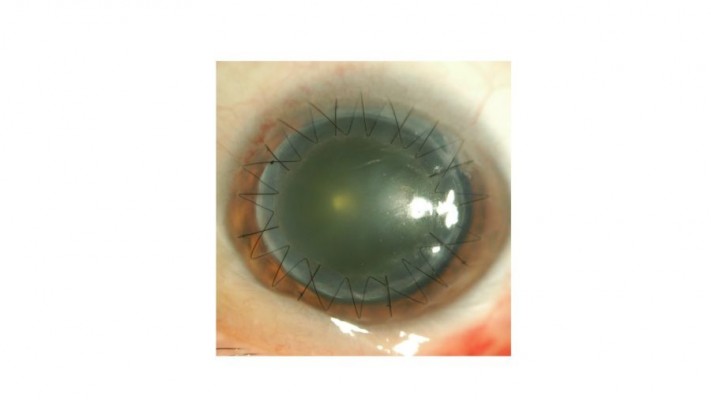
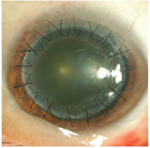
We performed penetrating keratoplasty in her left eye. Though the cause of death of the donor was lung carcinoma, other medical history was unknown. There was no corneal opacity or corneal scarring that indicated prior history of corneal disease or ocular surgery. The graft was stored in the corneal storage medium (Optisol-GS; Bausch & Lomb Incorporated, Bridgewater, NJ, USA) at 4°C for 2 days. After the surgery, the patient was treated with topical applications of levofloxacin (1.5% Cravit®; Santen Pharmaceutical Co., Ltd., Osaka, Japan), cefmenoxime (0.5% Bestron®; Pharmaceutical Co., Ltd., Osaka, Japan), betamethasone (0.1% Rinderon; Shionogi & Co., Ltd., Osaka, Japan), and hyaluronate sodium (0.1% Hyalein; Santen Pharmaceutical Co., Ltd) ophthalmic solutions four times daily, as well as aciclovir eye ointment (3% Zovirax; Santen Pharmaceutical Co., Ltd) two times daily considering the possibility of the hidden history of herpetic keratitis as a cause of the original corneal opacity. At day 4 post-transplantation, delayed epithelialization of the donor graft was observed. Because of the concern that 1.5% levofloxacin and 3% aciclovir may induce corneal epithelialization delay, both 1.5% levofloxacin and 3% aciclovir were discontinued and replaced by 0.3% tosufloxacin. At day 6 post-transplantation, white granular deposits were observed in the epithelial defect on the stroma of the donor graft (Figure 1). The deposits closely resembled those described in two previous reports.8,9
  | Figure 1 White granular deposits were observed in the area of the epithelial defect of the corneal graft at day 6 post-transplantation. |
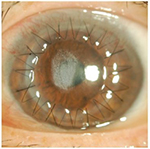
Tosufloxacin was considered the cause of deposition. Therefore, tosufloxacin eyedrops were discontinued. At day 11 post-transplantation, the graft was completely epithelialized. After the re-epithelialization, the epithelium was smooth, transparent, and well adhered. The deposits decreased gradually and had disappeared completely at 5 months post-transplantation (Figure 2). The patient’s final best corrected visual acuity improved to 0.7 in the left eye. No serious complications occurred until 16 months after transplantation.
  | Figure 2 Deposits were completely resolved at 5 months post-transplantation. |
Discussion
We encountered a case of corneal deposits associated with topical tosufloxacin administered after penetrating keratoplasty. Kamiya et al8 performed surgical keratectomy to remove corneal deposits formed after topical tosufloxacin in a patient with poor tear secretion, and analyzed the deposit using infrared spectrophotometry. They reported that the spectroscopic pattern of the deposit was consistent with tosufloxacin. In our case, corneal deposits were resolved spontaneously without surgical removal, and analysis of the deposits was not performed. The clinical appearance of the white granular deposits was very similar to that reported in the previous three cases.8,9 Moreover, deposits were observed shortly after administering topical tosufloxacin and disappeared gradually after discontinuation of tosufloxacin. Although there was an apparent causal relationship between corneal deposition and topical tosufloxacin, the possibility that the deposits were induced by tosufloxacin in combination with other topical medications could not be excluded.
Kim et al9 reported two cases of tosufloxacin-induced corneal deposits limited to areas of large epithelial defects, and suggested that exposed stroma probably has high affinity for tosufloxacin and that the corneal epithelium appears to play an important role as a barrier to prevent contact between the stromal substrate and drug particles. They concluded that tosufloxacin may precipitate on corneas with compromised epithelial integrity. Other risks of corneal deposition include persistent epithelial defect, inflammation, frequent dosing, poor tear secretion, poor tear mixing, and altered pH or poor solubility caused by interaction of other concurrent topical medications.8,9 Older patients are at a higher risk because of a higher prevalence of dry eye and more alkaline ocular surface.2 In the present case, deposits were limited to the area of epithelial defect after penetrating keratoplasty. Thus, the epithelial defect in the graft, reduced basal tear secretion, and interaction with other eye drops probably contributed to corneal deposits in the present case.
Corneal deposits disappeared completely after discontinuation of topical tosufloxacin in two of three previous cases.8,9 Despite immediate discontinuation of tosufloxacin, surgical keratectomy was required in one patient with poor tear secretion.8 In our case, although the donor graft was completely epithelialized while deposits remained beneath the epithelium, the deposits were resolved eventually, by 5 months. Immediate discontinuation of tosufloxacin and long-term careful observation are necessary when tosufloxacin-induced corneal deposits is suspected.
Conclusion
Topical tosufloxacin has a risk of precipitation on compromised corneas such as corneal grafts with epithelial defects after penetrating keratoplasty in eyes with poor tear secretion. The deposits may be resolved without surgical removal.
Disclosure
The authors report no conflicts of interest in this work.
References
Eiferman RA, Snyder JP, Nordquist RE. Ciprofloxacin microprecipitates and macroprecipitates in the human corneal epithelium. J Cataract Refract Surg. 2001;27:1701–1702. | ||
Wilhelmus KR, Abshire RL. Corneal ciprofloxacin precipitation during bacterial keratitis. Am J Ophthalmol. 2003;136(6):1032–1037. | ||
Madhavan HN, Rao SK, Joseph PR, Sulochana KN. Antibacterial activity of the white precipitate formed on the corneal surface after treatment with ciprofloxacin ophthalmic solution. Cornea. 1999;18(5):549–552. | ||
Essepian JP, Rajpal R, O’Brien TP. Tandem scanning confocal microscopic analysis of ciprofloxacin corneal deposits in vivo. Cornea.1995;14(4):402–407 | ||
Sinnaeve BA, Decaestecker TN, Claerhout IJ, Kestelyn P, Remon JP, Van Bocxlaer JF. Confirmation of ofloxacin precipitation in corneal deposits by microbore liquid chromatography-quadrupole time-of-flight tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2003; 785(1):193–196. | ||
Castillo A, Benitez del Castillo JM, Toledano N, Diaz-Valle d, Sayagues O, Garcia-Sanchez J. Deposits of topical norfloxacin in the treatment of bacterial keratitis. Cornea. 1997;16(4):420–423. | ||
Konishi M, Yamada M, Mashima Y. Corneal ulcer associated with deposits of norfloxacin. Am J Ophthalmol. 1998;125(2):258–260. | ||
Kamiya K, Kitahara M, Shimizu K. Corneal deposits after topical tosufloxacin in a patient with poor tear secretion. Cornea. 2009;28(1):114–115. | ||
Kim YD, Kim MK, Wee WR, Choi, HJ. Tosufloxacin deposits in compromised corneas. Optom Vis Sci. 2014;91(9):241–244. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
