Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 10 » Issue 1
COPD exacerbation frequency and its association with health care resource utilization and costs
Authors Dhamane A, Moretz C, Zhou Y, Burslem K, Saverno K, Jain G, Renda A , Kaila S
Received 9 June 2015
Accepted for publication 13 October 2015
Published 3 December 2015 Volume 2015:10(1) Pages 2609—2618
DOI https://doi.org/10.2147/COPD.S90148
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Amol D Dhamane,1 Chad Moretz,2 Yunping Zhou,2 Kate Burslem,1 Kim Saverno,2 Gagan Jain,1 Andrew Renda,3 Shuchita Kaila1
1Boehringer Ingelheim Pharmaceuticals Inc., Ridgefield, CT, USA; 2Comprehensive Health Insights Inc., Louisville, KY, USA; 3Humana Inc., Louisville, KY, USA
Background: Chronic obstructive pulmonary disease (COPD) exacerbations account for a substantial proportion of COPD-related costs.
Objective: To describe COPD exacerbation patterns and assess the association between exacerbation frequency and health care resource utilization (HCRU) and costs in patients with COPD in a Medicare population.
Methods: A retrospective cohort study utilizing data from a large US national health plan was conducted including patients with a COPD diagnosis during January 1, 2007 to December 31, 2012, aged 40–89 years and continuously enrolled in a Medicare Advantage Prescription Drug plan. Exacerbation frequency, HCRU, and costs were assessed during a 24-month period following the first COPD diagnosis (follow-up period). Four cohorts were created based on exacerbation frequency (zero, one, two, and ≥three). HCRU and costs were compared among the four cohorts using chi-square tests and analysis of variance, respectively. A trend analysis was performed to assess the association between exacerbation frequency and costs using generalized linear models.
Results: Of the included 52,459 patients, 44.3% had at least one exacerbation; 26.3%, 9.5%, and 8.5% had one, two, and ≥three exacerbations in the 24-month follow-up period, respectively. HCRU was significantly different among cohorts (all P<0.001). In patients with zero, one, two, and ≥three exacerbations, the percentages of patients experiencing all-cause hospitalizations were 49.7%, 66.4%, 69.7%, and 77.8%, respectively, and those experiencing COPD-related hospitalizations were 0%, 40.4%, 48.1%, and 60.5%, respectively. Mean all-cause total costs (medical and pharmacy) were more than twofold greater in patients with ≥three exacerbations compared to patients with zero exacerbations ($27,133 vs $56,033; P<0.001), whereas a greater than sevenfold difference was observed in mean COPD-related total costs ($1,605 vs $12,257; P<0.001).
Conclusion: COPD patients frequently experience exacerbations. Increasing exacerbation frequency is associated with a multiplicative increase in all-cause and COPD-related costs. This underscores the importance of identifying COPD patients at risk of having frequent exacerbations for appropriate disease management.
Keywords: COPD, exacerbations, frequency, costs
Introduction
Chronic obstructive pulmonary disease (COPD) is mostly a preventable and treatable disease characterized by airflow limitation that is not fully reversible.1 Typical symptoms include shortness of breath, cough, and sputum production. An estimated 15 million adults are reported to have been diagnosed with COPD in the US,2 and it is the leading cause of morbidity and mortality. Chronic lower respiratory disease, which primarily includes COPD, is the third leading cause of death in the US.3
One of the important manifestations of COPD is exacerbation – worsening of the typical COPD symptoms.4 The American Thoracic Society (ATS) and the European Respiratory Society (ERS)’s 2004 Guidelines for the Diagnosis and Treatment of COPD define a COPD exacerbation (hereafter referred to as an “exacerbation”) as
An event in the natural course of the disease characterized by a change in the patient’s baseline dyspnea, cough and/or sputum beyond day-to-day variability sufficient to warrant a change in management.1
Exacerbations have been shown to worsen the decline in lung function and quality of life and the decline becomes worse with increasing exacerbation frequency.5–7 Exacerbation frequency is also considered as one of the indicators of COPD disease stage, with higher frequency of exacerbations indicating a more severe disease.8,9 Furthermore, high exacerbation frequency suggests a higher risk of future exacerbations. In fact, according to the Global Initiative for Obstructive Lung Disease (GOLD) guidelines, patients with two or more exacerbations in the past 12 months are at a high risk of future exacerbations compared to low-risk patients with one or no exacerbations in the past 12 months.9
Exacerbations also pose a substantial economic burden, accounting for 50%–75% of the total economic burden due to COPD.1,9 In 2006, there were more than 1.2 million hospitalizations due to acute exacerbations of COPD, with associated costs of approximately $14 billion.10 It is evident that exacerbations that lead to hospitalizations have a significant economic impact; however, use of other health care resources – such as medications, diagnostic and laboratory testing, and outpatient and general practitioner visits – also pose a burden on the health care system.11
A description of health care resource utilization and costs in relation to exacerbation frequency is vital to provide a current real-world characterization of the burden associated with COPD exacerbations. Moreover, since almost half of the COPD patient population is reported to be over 65 years of age,2 assessment of exacerbations is important in this age group. The objective of this study was to describe COPD exacerbation patterns and assess the association between exacerbation frequency and health care resource utilization (HCRU) and costs in patients with COPD in a Medicare population.
Methods
Design and data source
A retrospective cohort study using the Humana® (a large US national health plan) administrative claims database was conducted. This database includes inpatient and outpatient medical, pharmacy, and lab-related claims and enrollment information for over 12 million current and former members enrolled in a commercial plan or a Medicare Advantage Plan with prescription drug benefits (MAPD).
Patient selection
Only data on MAPD members from January 1, 2007 to December 31, 2012 (study period) were used for this study. Patients were eligible for inclusion in the study sample if they had a COPD diagnosis, ie, ≥2 medical claims occurring on separate dates within 90 days with a COPD diagnosis code (International Classification of Diseases, 9th Revision, Clinical Modification [ICD-9-CM] code 491.xx, 492.xx, or 496.xx) in the primary or secondary position during January 1, 2008 to December 31, 2010 (identification period). The index date was defined as the date of the first medical claim with an ICD-9-CM diagnosis code for COPD during the identification period. Patients were also required to be continuously enrolled in an MAPD plan for a 12-month period prior to the index date (preindex period) and a 24-month postindex period (including the index date) and be 40–89 years of age as of index date. Patients were excluded from the study sample if they had ≥1 medical claim with any of the following ICD-9-CM diagnosis codes in any position during the study period: 277.0x (cystic fibrosis), 011.x (pulmonary tuberculosis), or 140.xx–172.xx, 174.xx–209.3x, 209.7x (malignant neoplasms).
Cohort formation
Four cohorts were formed based on the frequency of exacerbations (zero, one, two, and ≥three) experienced during the postindex period. Operational definitions of exacerbations for this study were based on definitions provided by ATS and ERS, and are presented in Table 1.1 These definitions list exacerbations by severity type; however, cohorts were formed based on exacerbation frequency regardless of severity. Only one exacerbation could be attributed to a patient during any 30-day period in the postindex period.12 If the patient met the criteria for >1 exacerbation during the 30-day window, only a single exacerbation of the most severe type was recorded. Once the 30-day period ended, it was possible for another exacerbation to be recorded.
  | Table 1 Exacerbation definition by severity type |
Baseline patient characteristics
Baseline patient characteristics were measured in the preindex period or as stated otherwise. Demographic characteristics included age (as of index date), sex, race/ethnicity, geographic region, and plan characteristics including low-income subsidy and dual eligibility (ie, eligible for Medicaid and Medicare) status. Clinical characteristics included use of oxygen therapy, number of exacerbations, and Deyo–Charlson comorbidity index (DCCI). DCCI uses 17 categories of comorbidity to calculate a score that reflects cumulative likelihood of 12-month mortality.13 Airflow testing status was assessed in preindex or postindex periods. Patients were considered to have had an airflow test if they had ≥1 medical claim with a procedure code for an airflow test (Current Procedural Terminology-4 [CPT-4] Code 94010, 94014, 94015, 94016, 94060, 94070, 94150, 94200, 94240, 94370, 94375, 94620, 94621, 94681, or 94720).
Outcomes
Outcomes included all-cause and COPD-related HCRU and costs in the postindex period. Specifically, all-cause and COPD-related hospitalizations, rehospitalizations, emergency room (ER) visits, and outpatient visits were assessed. Hospitalizations or outpatient visits were considered to be “all-cause” if the associated medical claim(s) had an ICD-9-CM diagnosis code in any position for any condition. Hospitalizations or visits were considered to be “COPD-related” if the associated medical claim(s) had either of the following: 1) COPD diagnosis code in the primary position or 2) respiratory failure diagnosis code (ICD-9-CM code 518.81, 518.83, or 518.84) in the primary position accompanied by a COPD diagnosis code in the secondary position or 3) any of the diagnosis codes indicative of an acute exacerbation (ICD-9-CM codes 466–466.19, 480–486, 487.0, 490, 493.12, 493.22, 493.92, 494.1, 506.0–506.3, 511.0–511.1, or 518.82) in the primary position and a COPD diagnosis code in the secondary position. ER visits leading to hospitalizations were considered as hospitalizations only. Respiratory antibiotic and oral corticosteroid use was identified based on pharmacy claims for these medications.
A COPD-related rehospitalization was defined as a “subsequent” COPD-related hospitalization occurring within 30 days of the initial COPD-related hospitalization. An all-cause rehospitalization was defined as a “subsequent” all-cause hospitalization occurring within 30 days of the initial all-cause hospitalization.
Patient and plan-paid all-cause and COPD-related medical and pharmacy costs were computed. All-cause medical costs were defined as the costs associated with all-cause hospitalizations, ER visits, and outpatient visits. COPD-related medical costs were defined as the costs associated with COPD-related hospitalizations, ER visits, and outpatient visits. All-cause pharmacy costs were defined as costs associated with a pharmacy claim for any medication, whereas COPD-related pharmacy costs were defined as those associated with a pharmacy claim for a COPD-related medication (eg, phosphodiesterase-4 inhibitors, xanthines, anticholinergic bronchodilators, anticholinergic beta-agonist combination agents, inhaled corticosteroids, long-acting beta agonists, long-acting beta agonist/inhaled corticosteroid combinations, oral corticosteroids, short-acting beta2 agonists, asthma and bronchodilator agent combination, mucolytics, oxygen therapy). Total health care costs were defined as the sum of medical and pharmacy costs. Costs were adjusted to 2012 US dollars using the annual medical care component of the Consumer Price Index.14
Statistical analysis
Exacerbation patterns, assessed in terms of exacerbation frequency and exacerbation severity, were presented as number and proportion of patients experiencing exacerbations. Summary statistics including mean and median were also reported.
Baseline patient characteristics and outcomes were assessed for the four cohorts of interest (zero, one, two, and ≥three exacerbations). Summary statistics were used to describe patient characteristics; continuous measures were described using means and standard deviations, whereas categorical measures were reported using numbers and proportions. Univariate statistical comparisons of patient characteristics and outcomes were made between cohorts using analysis of variance (ANOVA) (for continuous measures) and chi-square tests (for categorical measures). A trend analysis was performed to assess if increase in frequency of COPD exacerbations was associated with higher costs (all-cause and COPD-related) using generalized linear models with a gamma distribution and a log link. All data analyses were conducted using SAS version 9.3.
Results
A total of 232,147 patients with a COPD diagnosis were identified during the study period. Approximately, 22.6% (N=52,459) of these patients satisfied all patient selection criteria and were included in the final study sample. A graphical representation of steps taken to select the patients is provided in Figure 1.
Exacerbation patterns
Exacerbation patterns are presented in Table 2. Almost half (44.3%) of the patients in the final study sample had at least one exacerbation during the postindex period. More than 25% of the patients had one exacerbation, 9.5% had two exacerbations, and 8.5% had ≥three exacerbations. The proportions of patients experiencing level I, level II, and level III exacerbations were 24.0%, 15.2%, and 5.1%, respectively. Based on the exacerbation frequency information, four mutually exclusive cohorts were formed: 1) zero exacerbations (n=29,222), 2) one exacerbation (n=13,797), 3) two exacerbations (n=4,982), and 4) ≥three exacerbations (n=4,458).
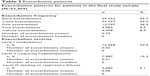  | Table 2 Exacerbation patterns |
Baseline patient characteristics
The patients in the final study sample had a mean age of 71.1 years, 55% were female, 87% were Caucasian, and 68.4% resided in the Southern US (Table 3). Statistically significant (P<0.05) differences were noted in all baseline patient characteristics assessed among the four cohorts of interest. It was observed that 40.9% of patients in the ≥three exacerbations cohort experienced at least one exacerbation in the preindex period. In comparison, only 11.4% of the one exacerbation cohort experienced an exacerbation in the preindex period. Oxygen therapy use ranged from 11.0% in the zero exacerbation cohort to 32% in the ≥three exacerbations cohort. Comorbidity burden measured using DCCI ranged from 1.62 to 1.84 in patients with zero to patients with ≥three exacerbations. Airflow testing was also most common (65%) in the ≥three exacerbations cohort compared to 39.6%, 47.4%, and 55.5% in the zero, one, and two exacerbation cohorts, respectively.
HCRU by exacerbation frequency
HCRU by exacerbation frequency during the postindex period is presented in Table 4. Statistically significant (P<0.001) differences were observed among the four cohorts for all of the HCRU outcomes assessed. All-cause hospitalizations were more common in patients with one, two, and ≥three exacerbations (66.4%, 69.7%, and 77.8% respectively) than in patients with zero exacerbations (49.7%). Furthermore, the proportion of patients with a COPD-related hospitalization was also highest in patients with ≥three exacerbations (60.5%) and the lowest in patients with one exacerbation (40.4%).
Similar patterns were observed for all-cause and COPD-related 30-day rehospitalizations with proportions increasing among patients with higher exacerbation frequency. The proportion of patients with an all-cause rehospitalization was 18.9% and 30.9% in patients with one and ≥three exacerbations, respectively, and the proportion of patients with a COPD-related rehospitalization was 1.7% and 11.2%, respectively. There were statistically significant (P<0.001) differences in respiratory antibiotic and oral corticosteroid use among the four cohorts.
Health care costs by exacerbation frequency
Health care costs by frequency of exacerbations during the postindex period are presented in Figures 2 and 3. Greater frequency of exacerbations was associated with greater all-cause and COPD-related total, medical, and pharmacy costs.
  | Figure 2 COPD-related health care costs by exacerbation frequency. |
  | Figure 3 All-cause health care costs by exacerbation frequency. |
All-cause medical costs increased more than twofold from $21,771 in patients with zero exacerbations to $47,061 in patients with ≥three exacerbations, exhibiting a statistically significant (P<0.001) linear trend. A corresponding 24-fold ($301 to $7,209) increase was noted in COPD-related medical costs. All-cause medical costs contributed the most (>80%) toward the all-cause total costs, whereas the COPD-related medical costs contribution to the COPD-related total costs ranged from 18.7% to 58.8%.
All-cause pharmacy costs increased from $5,362 in patients with zero exacerbations to $8,972 in patients with ≥three exacerbations. An approximately fourfold increase ($1,305 to $5,048) was noted in COPD-related pharmacy costs.
Discussion
This study provides a current real-world characterization of exacerbation patterns and describes the health care utilization and costs in patients with COPD with respect to exacerbation frequency in a Medicare population in the US. Pasquale et al also assessed the impact of exacerbation frequency on HCRU and costs in a Medicare population. However, their study population was restricted to patients on maintenance medications, which, given that many are not on maintenance medications, would produce an unrepresentative sample of COPD patients.15 The results of the current study indicate that exacerbations occurred frequently in patients with COPD. Almost half (44.3%) of the patients in the final sample had at least one exacerbation, 26.3% had one, 9.5% had two, and 8.5% had three or more exacerbations during a 24-month period. In comparison, Pasquale et al reported that 49.8% of COPD patients in the Medicare population had an exacerbation in a 12-month period. The differences may also be attributable to other differences in the study design such as a shorter assessment period and differing exacerbation definitions.
A few other published studies have previously explored the association between COPD exacerbations and HCRU and costs; however, COPD populations represented in these studies were different from the current study and some of the studies were out-of-date given evolving definitions and guidelines.16–18 For example, a study by Punekar et al reported health care resource use and costs in COPD patients using the Clinical Practice Research Datalink (CPRD) database that contains data for patients who are mostly from England and Wales.16 Another study by Dalal et al17 was US-based, but focused on a COPD patient population predominantly enrolled in commercial health plans. The current study focused on a Medicare population in the US.17 Considering that 51% of the medical costs attributable to COPD in the US are paid for by Medicare and almost half of the US COPD population is 65 years or older, evaluation of HCRU and costs in this population segment is particularly important.12,19
The results of the current study suggest that exacerbation frequency had a significant impact on HCRU and costs. Hospitalizations, ER, and outpatient visits occurred more frequently in patients with one, two, and ≥three exacerbations than patients without exacerbations. Both all-cause and COPD-related costs increased significantly with an increase in exacerbation frequency, exhibiting a statistically significant (P<0.001) linear trend. This significant impact of COPD exacerbations on HCRU and costs underscores the importance of preventing exacerbations in patients with COPD. This study provides real-world evidence to suggest increase in COPD exacerbation is associated with a substantial increase in economic burden. It provides more granularity with respect to the exacerbation patterns and the magnitude of differences in HCRU and costs by exacerbation frequency. For example, an eightfold difference was noted in COPD-related health care cost between the zero and three+ exacerbation cohorts. This information may benefit US payers, providers, and employers in order to better understand and manage their COPD patient population.
Exacerbation prevention may be a key strategy in COPD disease management due to its substantial clinical and economic impact. Prior research has demonstrated that a prior exacerbation is the best predictor of future exacerbations.8 The current study also shows an association between greater exacerbation frequency in the assessment period and a greater baseline exacerbation frequency. A comprehensive assessment of predictors of future exacerbations or a tool to enable early identification of such “high-risk” patients could prove useful. Another approach for exacerbation prevention may be appropriate treatment with several US Food and Drug Administration (FDA)-approved medications indicated to reduce exacerbations.20,21
Results of the current study should be interpreted with caution due to certain limitations. First, the study results may not be generalizable to other populations; however, Humana® is a large national health plan in the US with significant representation of the Medicare population. Second, clinical parameters such as lung function test results and COPD symptoms were not available in this database that may have led to incorrect identification of patients with COPD. Third, large sample sizes may have resulted in statistically significant differences across comparison groups that may not be clinically relevant. Lastly, administrative claims data are prone to coding errors of omission and commission and incomplete claims information.
Conclusion
This study demonstrated the economic burden of exacerbations in patients with COPD as related to exacerbation frequency in a Medicare population and underscored the importance of identifying COPD patients at risk of having frequent exacerbations for appropriate disease management. Future research is needed to confirm and validate these findings in other health plan populations.
Disclosure
Amol D Dhamane, Kate Burslem and Shuchita Kaila are employees of Boehringer Ingelheim Pharmaceuticals Inc., Ridgefield, CT, USA. Gagan Jain was an employee of Boehringer Ingelheim Pharmaceuticals Inc., Ridgefield, CT, USA at the time of the study. Chad Moretz, Yunping Zhou and Kim Saverno are employees of Comprehensive Health Insights Inc., Louisville, KY, USA. Andrew Renda is an employee of Humana Inc., Louisville, KY, USA. The authors report no other conflicts of interest in this work.
References
American Thoracic Society, European Respiratory Society. Standards for the Diagnosis and Management of Patients With COPD; 2004. Available from: http://www.thoracic.org/clinical/copd-guidelines/resources/copddoc.pdf. Accessed February 5, 2015. | ||
Centers for Disease Control and Prevention (CDC). Chronic obstructive pulmonary disease among adults – United States. Morb Mort Wkly Rep. 2011;61(46):938–943. | ||
Hoyert DL, Xu J. Deaths: preliminary data for 2011. Natl Vital Stat Rep. 2012;61(6):1–51. Available from: http://www.cdc.gov/nchs/data/nvsr/nvsr61/nvsr61_06.pdf. Accessed February 5, 2015. | ||
American Thoracic Society. What Is an Exacerbation. Available from: http://www.thoracic.org/clinical/copd-guidelines/for-patients/what-is-an-exacerbation.php. Accessed February 5, 2015. | ||
Halpin DM, Decramer M, Celli B, Kesten S, Liu D, Tashkin DP. Exacerbation frequency and course of COPD. Int J Chron Obstruct Pulmon Dis. 2012;7:653–661. | ||
Makris D, Moschandreas J, Damianaki A, et al. Exacerbations and lung function decline in COPD: new insights in current and ex-smokers. Respir Med. 2007;101(6)1305–1312. | ||
Hoogendoorn M, Feenstra TL, Hoogenveen RT, Al M, Molken MR. Association between lung function and exacerbation frequency in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2010;5:435–444. | ||
Hurst JR, Vestbo J, Anzueto A et al; For the Evaluation of COPD Longitudinally to Identify Predictive Surrogate Endpoints (ECLIPSE) Investigators. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N Engl J Med. 2010;363(12):1128–1138. | ||
Global Initiative for Chronic Obstructive Lung Disease, I. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease; 2014. Available from: http://www.goldcopd.org/uploads/users/files/GOLD_Report_2014_Jun11.pdf. Accessed February 5, 2015. | ||
Perera PN, Armstrong EP, Sherrill DL, Skrepnek GH. Acute exacerbations of COPD in the United States: inpatient burden and predictors of costs and mortality. COPD. 2012;9(2):131–141. | ||
Toy EL, Gallagher KF, Stanley EL, Swensen AR, Duh MS. The economic impact of exacerbations of chronic obstructive pulmonary disease and exacerbation definition: a review. COPD. 2010;7(3):214–228. | ||
Centre for Disease Control and Prevention. Chronic obstructive pulmonary disease among adults – United States, 2011. MMWR. 2012;61(46):938–943. | ||
Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol. 1992;45(6):613–619. | ||
U.S. Department of Labor, Bureau of Labor Statistics. Measuring Price Change for Medical Care in the CPI. Washington, DC: BLS; 2010. | ||
Pasquale MK, Sun SX, Song F, Hartnett HJ, Stemkowski SA. Impact of exacerbations on health care cost and resource utilization in chronic obstructive pulmonary disease patients with chronic bronchitis from a predominantly Medicare population. Int J Chron Obstruct Pulmon Dis. 2012;7:757–764. | ||
Punekar YS, Shukla A, Müllerova H. COPD management costs according to the frequency of COPD exacerbations in UK primary care. Int J Chron Obstruct Pulmon Dis. 2014;9:65–73. | ||
Dalal AA, Patel J, D’Souza A, Farrelly E, Nagar S, Shah M. Impact of COPD exacerbation frequency on costs for a managed care population. J Manag Care Spec Pharm. 2015;21(7):575–583. | ||
Andersson F, Borg S, Jansson SA, et al. The costs of exacerbations in chronic obstructive pulmonary disease (COPD). Respir Med. 2002;96(9):700–708. | ||
Ford ES, Murphy LB, Khavjou O, Giles WH, Holt JB, Croft JB. Total and state-specific medical and absenteeism costs of COPD among adults aged ≥18 years in the United States for 2010 and projections through 2020. Chest. 2015;147(1):31–45. | ||
Spiriva® [package insert]. Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT; 2014. Available from: http://bidocs.boehringer-ingelheim.com/BIWebAccess/ViewServlet.ser?docBase=renetnt&folderPath=/Prescribing+Information/PIs/Spiriva/Spiriva.pdf | ||
Advair Diskus® [package insert]. Advair: GlaxoSmithKline, Research Triangle Park, NC; 2014. Available from: https://www.gsksource.com/gskprm/htdocs/documents/ADVAIR-DISKUS-PI-MG.PDF |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



