Back to Journals » Open Access Journal of Contraception » Volume 9
Contraceptive use and the risk of sexually transmitted infection: systematic review and current perspectives
Authors Deese J, Pradhan S, Goetz H, Morrison C
Received 16 May 2018
Accepted for publication 28 August 2018
Published 12 November 2018 Volume 2018:9 Pages 91—112
DOI https://doi.org/10.2147/OAJC.S135439
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Igal Wolman
Jennifer Deese, Subarna Pradhan, Hannah Goetz, Charles Morrison
Global Health, Population and Nutrition, FHI 360, Durham, NC, USA
Purpose: Evidence on the association between contraceptive use and risk of sexually transmitted infections (STIs) and bacterial vaginosis (BV) is lacking, with few prospective studies. We systematically reviewed the last 10 years’ evidence on the association between contraception and STI/BV, building on the most recent systematic reviews published in 2006 and 2009.
Methods: We searched the MEDLINE and POPLINE databases for peer-reviewed articles published between January 1, 2008 and January 31, 2018 reporting prospective studies that assessed the association between contraceptive use and incident STI and/or incident or recurrent BV.
Results: We identified 33 articles that evaluated combined oral contraceptives (COC), depot medroxyprogesterone acetate (DMPA), the copper intrauterine device (Cu-IUD), the levonorgestrel intrauterine system (LNG-IUS) and other methods. The strength of the evidence for many specific contraceptive method/STI associations is limited by few prospective studies with comparably defined exposures and outcomes. Available data suggest no association of COCs and Neisseria gonorrhoeae, Trichomonas vaginalis, HSV-2 or syphilis, and mixed evidence on the association with HPV, Chlamydia trachomatis, and BV. For DMPA, none of the studies identified found an association with N. gonorrhoeae or syphilis, and data on C. trachomatis, T. vaginalis, HPV and BV were mixed. Two large studies showed a highly clinically significant increased risk of HSV-2 infection with DMPA use. Data on the effect of Cu-IUD and the LNG-IUS on the acquisition of C. trachomatis, N. gonorrhoeae and T. vaginalis are sparse, and data on HPV and BV are mixed.
Conclusion: Few data are available from prospective studies, including randomized trials, to draw strong conclusions about the relationships between contraceptive methods and specific STIs. The overall evidence on the association between contraceptive use and STI/BV risk is limited by the lack of any randomized trials, few published prospective studies designed to analyze these associations, wide variability in exposure definitions and comparator groups, potential for confounding due to inaccurate sexual behavior data, differential confounder adjustment and differences in study populations and sizes. Despite these limitations, new evidence is supportive of a significantly increased risk of HSV-2 infection among DMPA users which warrants additional research to better understand this association.
Keywords: combined oral contraceptives, depot medroxyprogesterone acetate, intrauterine device, intrauterine system, sexually transmitted infections, chlamydia, gonorrhea, trichomonas, herpes simplex virus, bacterial vaginosis
Introduction
Fifty-seven percent of reproductive aged married or in-union women worldwide use a modern method of family planning including female and male sterilization, oral contraceptive pills, the intrauterine device (IUD), male and female condoms, injectables, implants, vaginal barrier methods and emergency contraception.1 Moreover, in an effort to meet the Sustainable Development Goal of ensuring universal access to sexual and reproductive health services by 2030, Family Planning 2020 aims to enable 120 million more women and girls to use contraceptives by 2020.2 However, women using or in need of contraception are also at risk of sexually transmitted infections (STIs). The World Health Organization estimates that over 357 million new cases of curable STIs, including chlamydia, gonorrhea, syphilis and trichomoniasis, and a similarly high burden of viral STIs (417 million prevalent herpes simplex virus [HSV] and 291 million prevalent human papillomavirus [HPV] infections), not including HIV, occur among individuals aged 15–49 each year.3
Both increasing access to contraceptives, and preventing STIs, are critical to ensuring women’s sexual and reproductive health. However, the evidence base on potential associations between use of specific contraceptive methods and STI risk is lacking, with few prospective studies designed to assess these associations. Moreover, any true association between contraceptive use and STIs could be due to behavioral and/or biological risk factors. Behaviorally, women who use modern methods of contraception may have different condom use patterns, sexual frequency and numbers of sexual partners as compared to women not using modern contraception.4–7 Uncontrolled and/or residual confounding due to inaccurate measurement of sexual behavior is a significant concern in evaluating the results of observational studies on the topic – results may be biased toward or away from the null depending on the type and frequency of misreporting or lack of adjustment for salient variables – and studies which randomize women to contraception are lacking.6 Possible biological mechanisms linking contraceptives and STIs include alteration of the vaginal epithelium, changes in mucus production and in multiple immune responses (eg, production and availability of cytokines, chemokines and relevant cell types in the upper and lower female reproductive tract and alteration of the vaginal microbiota).8–13
Women and providers need the best available information on potential associations between contraceptive use and STI risk. We conducted a systematic review of the recent literature (past 10 years), following published systematic reviews in 2006 and 2009, to summarize the current evidence.14,15
Methods
We report our findings in accordance with the Preferred Items of Reporting for Systematic Reviews and Meta-Analyses (PRISMA).16 We searched the MEDLINE and POPLINE databases for articles published in peer-reviewed journals between January 1, 2008 and January 31, 2018 for prospective studies with data on the association between use of contraceptives and incident protozoan/bacterial (Trichomonas vaginalis, Neisseria gonorrhoeae, Chlamydia trachomatis, and syphillis), and incident and persistent viral (HPV, HSV), STI. We also included studies that evaluated incident and recurrent bacterial vaginosis (BV) due to recent evidence that suggests sexual transmission is an important mechanism of BV recurrence in women, and because BV represents a significant health burden for women.17,18 The search strategy is provided in the Appendix. We identified 1,147 unique articles using the aforementioned search strategy. While we did not decide a priori to exclude articles that were not in English, all 1,147 articles identified were in English. We then systematically reviewed abstracts from the 1,147 articles to identify the study design and to determine the contraceptive methods and STIs evaluated in each study. Abstract review data, including author, title, journal, study design and reason for exclusion were recorded, as well as classification for inclusion in the review (ie, include, exclude, defer to senior author for decision). The specific contraceptive methods and STIs evaluated in each article were abstracted. We included articles which were prospective, in humans, specified the hormone (or at a minimum classified exposure by estrogen-containing vs progesterone-containing) under study if evaluating a hormonal method, and had laboratory results for incident STI and/or incident or recurrent BV outcomes (not including HIV). Additionally, we excluded articles that only evaluated the STI/BV outcome in a subset of participants without defining how the subset was selected. Where these criteria could not be ascertained from the abstract, we reviewed the full manuscript to determine inclusion status. A second reviewer confirmed that all included articles met inclusion criteria. This review resulted in 33 manuscripts which included the following number of studies by contraceptive method: 21 combined oral contraceptives (COC), 17 depot medroxyprogesterone acetate (DMPA), nine levonorgestrel intrauterine system (LNG-IUS), six copper intrauterine device (Cu-IUD), one non-specified IUD type, three contraceptive vaginal ring (CVR), one contraceptive patch, four implant (levonorgestrel or etonogestrel [ENG]), two norethisterone enanthate (NET-EN), one medroxyprogesterone acetate-ethinyl estradiol (MPA-EE) injectable and one that was categorized only by estrogen- or progesterone-containing.
Results
Combined oral contraceptives
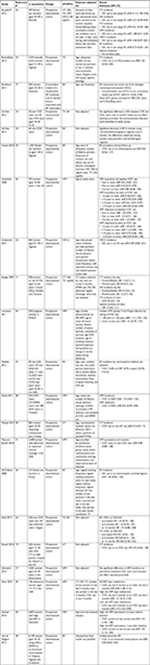
We identified 21 studies which evaluated the association between COC use and chlamydia, gonorrhea, trichomoniasis, HPV, HSV-2, BV and/or syphilis. We provide summary data (author, year, population, sample size, study design, STI/BV outcome, variables adjusted for in analyses and results) in Table 1.
Chlamydia
Four of the included studies evaluated the association between chlamydia and COC use. Russell et al19 evaluated factors associated with incident and ascending C. trachomatis infection among 48 women with current or prior diagnosis of gonorrhea and/or chlamydia. In the analysis of incident C. trachomatis infection, no association with COC use was observed in univariate analysis, therefore COC use was not further analyzed in the final multivariate model. A study of Kenyan women reporting transactional sex, which included over 2,000 person-years of follow-up with monthly follow-up visits, found no association between COC use and incident C. trachomatis infection.20 Similarly, a study conducted among 958 South African, Tanzanian, and Zambian women at risk of STIs and followed monthly for 1 year found no association with baseline oral contraceptive (OC) use and subsequent C. trachomatis infection.21 It is unclear as to whether or not changes in contraceptive methods during follow-up were accounted for in the two aforementioned studies which found no significant association in multivariate analyses. However, a study among 397 Rwandan sex workers, which followed women quarterly for 1–2 years and controlled for time-varying contraceptive use, found a significant association between COC use and C. trachomatis incidence (adjusted odds ratio [aOR] 6.13, 95% CI 1.58–23.8).22
Gonorrhea
Only two of the included studies – the aforementioned studies of 958 South African/Tanzanian/Zambian women and 397 Rwandan sex workers – evaluated the association between COC use and gonorrhea, and found no significant association.21,22
Trichomoniasis
We identified five studies which evaluated the association between COC use and trichomoniasis, none of which identified a significant association. Rezk et al23 enrolled 452 Egyptian women seeking contraception who agreed to COC or LNG-IUS use. Two-hundred and thirty-six women opted to use COCs (194 opted to use the LNG-IUS; 22 participants dropped out and were not included in analyses) and attended follow-up visits at 6 weeks, 6 months and 12 months; T. vaginalis testing was conducted at each follow-up visit by wet mount. The authors found no significant difference in T. vaginalis infection between COC and LNG-IUS users.23 The aforementioned study among Rwandan sex workers also found no association between COC use and trichomoniasis incidence.21,22 A large study among 2,374 Ugandan women participating in the Rakai Community Cohort Study (RCCS), which included annual follow-up with assessment of contraceptive method use, found no significant association between COC use and incident T. vaginalis.24 However, the study of 958 South African/Tanzanian and Zambian women found a reduced risk of T. vaginalis among women using COCs (OR 0.4, [95% CI 0.2–0.7]).21
Human papillomavirus
We identified eight studies which evaluated the association between COC use and incident and/or persistent HPV infection. A Finnish prospective study included 255 HPV-negative pregnant women in their third trimester. Among ever COC users, those who initiated COC use at <20 years of age had a significantly increased risk of incident infection by HPV types 7 and 9 compared to those who initiated COC use at ≥20 years of age (aIRR 1.13, [95% CI 1.02–1.26]).25 Another study among 972 women in Hawaii found that the risk of acquiring any HPV infection varied by baseline COC use as well as by duration of COC use.26 Women using COC at baseline were not at increased risk of incident high-risk HPV (HR-HPV), although past COC users were protected (ever users: adjusted hazard ratio [aHR] 0.81 [95% CI 0.65–1.01], past users: aHR 0.74 [95% CI 0.57–0.97], current user: aHR 0.86 [95% CI 0.68–1.09]). The study also showed significantly decreasing HPV incidence with number of years of past COC use. However, three other studies – all of which incorporated time-varying exposure control in analyses – found no association between COC use and incident HPV infection. Schmeink et al27 followed 2,065 Dutch women over 1 year, classifying women as consistent vs never COC users in analyses; Marks et al28 followed 1,135 women for 18 months, classifying each follow-up interval as non-hormonal contraceptive (NHC) use, DMPA use or COC use; and Borgdorff et al22 followed 397 women for up to 2 years, classifying each follow-up interval as no hormonal contraceptive use, DMPA use, COC use or pregnant.
Among the studies which evaluated HPV, five evaluated persistent infection. A Danish cohort study among 2,874 HR-HPV positive women found significantly increased risk of HPV persistence with current COC use at baseline, as well as with COC use during the first year of follow-up (aOR 1.23 [95% CI 1.02–1.48] and aOR 1.35 [95% CI 1.13–1.63], respectively).29 Similarly, a cohort study among 150 US adolescent women showed a significant association between COC use in the last 3 months and HPV re-detection compared to women who had not used COCs in the last 3 months (aHR 2.0 [95% CI 1.28, 3.15]).30 Another study among 1,135 Thai women found a significant association between COC and reduced risk of HPV clearance compared to NHC use (aHR 0.67 [95% CI 0.49–0.93]).28 However, the aforementioned study by Schmeink, as well as a study among 2,408 women with low-grade or equivocal cytological abnormalities followed for 2 years, found no association between COC use and HPV persistence.27,31
Herpes simplex virus type 2
Two of the included studies, including the aforementioned study of 397 Rwandan sex workers, evaluated the association between COC use and HSV-2 and found no significant association.22,32 The second study was conducted among 682 Ugandan women who were partners of male participants in a randomized trial of male circumcision and were followed up annually, with assessment of time-varying contraceptive method use, over a 2-year follow-up period.
Bacterial vaginosis
We identified nine studies which evaluated the association between COC use and risk of incident and/or recurrent BV; six identified no significant association and three found a significant decrease in BV associated with COC use.
Among 153 US women who were BV-negative at baseline in a sub-study of the CHOICE study,47 no association was observed between COC use and risk of BV over 6 months. Similarly, a study among 60 Italian women, 30 who agreed to COC use and 30 who agreed to CVR use, found no difference in incident BV or intermediate vaginal flora over 6 months between the two groups.33,34 In both of the aforementioned studies, women were analyzed according to chosen/assigned contraceptive method, allowing for discontinuations. Two studies among 151 Kenyan and 1,027 Ugandan female sex workers followed for ~1 to ~2 years respectively, found no significant difference in BV among women using COCs as compared to women using no contraception/(had tubal ligation, Kenya study only) while controlling for time-varying contraceptive use.35,36 Lastly, the aforementioned studies of 194 Egyptian and 397 Rwandan women found no differences in BV risk in COC users relative to LNG-IUS users, or relative to NHC users, respectively.22,23
Three studies observed a significant protective effect of COCs and BV. In a study among 60 COC users, women using a 17β-estradiol/nomegestrol regimen had a significant decrease in BV over time, though no information is provided on whether or not BV treatment was administered for women who were determined to have BV at baseline, or during the follow-up period.34 A sub-study within a BV-treatment trial of 400 women found that current/recent users of estrogen-containing contraceptive (ECC) (n=110, among which 108 were oral contraceptive users and two were NuvaRing users) had significantly lower risk (aHR 0.51 [95% CI 0.33–0.78]) of BV recurrence over 6 months compared to women not reporting current/recent ECC use.37 Lastly, a study among 6,109 women aged 18–35 in Zimbabwe, Uganda and Thailand, which included 15–24 months of follow-up per woman and time-varying control of contraceptive method, also observed a protective association (aHR 0.90, [95% CI 0.84, 0.97]) as compared to women not using hormonal contraception.38
Syphilis
Only two of the included studies – the aforementioned studies of 958 South African/Tanzanian women and 397 Rwandan sex workers – evaluated the association between COC use and T. pallidum and found no association.21,22
COC summary
We found no evidence of any significant association between use of COCs and the risk of trichomoniasis, gonorrhea, HSV-2 or syphilis. There is mixed evidence on COC use and incident chlamydia and HPV infection. One study showed a significant association between COC use and increased incident C. trachomatis as compared to women not using hormonal contraception, whereas three other studies found no association.19–22 Two of eight studies identified modified HPV incidence with COC use. One study showed a significant association between the timing of COC use initiation and HPV incidence, with women initiating COC use before 20 years of age having a slightly higher HPV incidence.25 A second study showed an inverse association between the number of years of COC use and HPV incidence, whereas three other studies found no association with HPV incidence.26 Findings were similarly inconsistent among studies which evaluated HPV persistence/re-detection. There is also conflicting evidence of an association between COC use and BV – six studies found no effect, whereas three studies found reduced risk of BV recurrence associated with COC use.
Depot medroxyprogesterone acetate
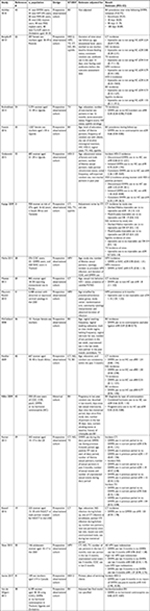
We identified 17 studies which evaluated the association between DMPA use and chlamydia, gonorrhea, trichomoniasis, HPV, HSV-2, BV and/or syphilis. Summary data are provided in Table 2.
Chlamydia
We identified six studies that reported an association between DMPA use and chlamydia, only one of which found a significant association. A study of Kenyan women reporting transactional sex, which included over 2,000 person-years with monthly follow-up visits, found that DMPA was significantly associated with increased incident C. trachomatis infection in adjusted analyses (aHR 1.8 [95% CI 1.1–3.0]), including adjustment for time-varying contraceptive method use when compared to women not using a hormonal contraceptive.20 However, five other studies, also with relatively frequent follow-up assessments, found no significant association. These include: 1) a study of 342 young US women which conducted 3-monthly follow-up visits for a median of 42 months and compared women using DMPA in the current or previous 3-month period to women not reporting DMPA use in those periods; 2) a study among 225 US women with follow-up visits at months 1, 4, 8 and 12 which included 2,058 person-months and compared women reporting DMPA use at any visit to women never reporting DMPA use; 3) a study among 397 Rwandan sex workers that followed women for up to 2 years and compared women reporting DMPA use by follow-up interval to women reporting no hormonal contraceptive use; 4) a study among 958 Tanzanian, Zambian and South African women followed monthly for 1 year; and 5) a study among 567 South African family planning clinic attendees.19,21,22,39,40
Gonorrhea
We identified four studies which evaluated gonorrhea as an outcome, all of which identified no significant association. The aforementioned study of 342 young US women found no association between DMPA use and incident N. gonorrhoeae infection as compared to non-DMPA users.39 Similarly, the studies among Rwandan sex workers, Tanzanian, Zambian, and South African women and South African family planning clinic attendees found no significant association between DMPA use and incident N. gonorrhoeae.21,22,40
Trichomoniasis
We identified five studies which evaluated trichomonas as an outcome. Three of these studies identified no significant association with DMPA use, including the study of 342 young US women, the study of 397 Rwandan sex workers and the study of 567 South African family planning clinic attendees.21,22,39,40 However, the study among 2,374 Ugandan women and the study of 958 Tanzanian, Zambian, and South African women found significantly decreased risk of T. vaginalis incidence among DMPA users (aIRR 0.54, [95% CI 0.30–0.98] and OR 0.6 [95% CI 0.4, 0.8], respectively).24
Human papillomavirus
Four studies analyzed the association between DMPA use and incident HPV infection and/or HPV persistence; three of these studies identified no association and one identified a marginally significant association. One prospective study, which evaluated HPV incidence and persistence among 1,135 Thai COC, DMPA and NHC users followed up every 6 months for 18 months, found that DMPA use, as compared to NHC use, was not significantly associated with incident HPV infection or clearance of prior infection in adjusted analyses.28 Similarly, a smaller study among 146 US young women, followed for an average of 5.8 years, found no association between DMPA use in the prior 3 months and HPV re-detection.30 The study among 397 Rwandan sex workers also found no association between DMPA use and incident HPV.22 However, another study among 2,408 women with baseline HPV infection and cytological abnormalities followed up every 6 months for 2 years found that injectable contraceptive use was marginally significantly associated with HPV persistence relative to no injectable contraceptive use (aOR 1.5 [95% CI 1.01–1.32]).31
Herpes simplex virus type 2
Three of the included studies evaluated the association between DMPA use and HSV-2.22,32,41 The study among Rwandan sex workers found no association, though the total number of DMPA users was small, which may have limited study power. Two larger studies identified significant increases in HSV-2 incidence among DMPA users. A study among 682 women in Uganda found increased risk of HSV-2 acquisition (aHR 2.26, [95% CI 1.09, 4.69]) as compared to women who were not using hormonal contraceptive and were not pregnant. A sensitivity analysis among women with known HSV-2 positive partners strengthened the conclusion with an observed aHR of 6.23 (95% CI 1.49, 26.3) among consistent DMPA users. A second study among 149 Canadian female sex workers with a median follow-up period of 19 months found significantly higher HSV-2 incidence among DMPA users (aHR 4.43 [95% CI 1.90, 10.35]).41
Bacterial vaginosis
Four of seven studies which reported on the association between BV and DMPA found significantly reduced BV detection among women using DMPA. A study among 330 US women attending STI clinics, 105 of which used DMPA at least once during the study period, found DMPA to be protective against BV diagnosis as compared to NHC (aOR 0.42 [95% CI 0.20–0.88]).42 A second large study among 1,027 Ugandan sex workers followed over 24 months, among which 1,309 visits were during reported DMPA use, also found DMPA use to be associated with a reduction in BV (aOR 0.66, [95% CI 0.50, 0.86]).35 Notably, the Ugandan cohort was at high risk of BV as evidenced by high baseline (56%) and follow-up (54%–63%) BV prevalence. Another study among sex workers in Kenya also found DMPA use to be associated with reduced BV prevalence (aHR 0.60, [95% CI 0.48, 0.74]).36 Lastly, the large study among 6,109 women in Zimbabwe, Uganda and Thailand, also observed a protective association (aHR 0.82, [95% CI 0.77, 0.87]) as compared to women not using hormonal contraception.38 Three other studies found no significant change in BV – one which followed 41 new DMPA users for 6 months, another which followed 397 Rwandan sex workers and controlled for time-varying contraceptive use for up to 2 years, and another which followed 567 South African family planning clinic attendees for 1 year.22,40,43
Syphilis
Only two of the included studies – the aforementioned studies of 958 South African/Zambian/Tanzanian women and 397 Rwandan sex workers – evaluated the association between DMPA use and T. pallidum and found no significant association.21,22
DMPA summary
We found no evidence of any association between the use of DMPA and the risk of gonorrhea or syphilis. A single study among Kenyan women did find a significant association between DMPA use and increased C. trachomatis infection, whereas two US-based studies found no association. This may suggest a potential for population-level differences in risk associated with DMPA use. Two studies identified a decreased risk of T. vaginalis infection with DMPA use; while one did not control for potential confounding variables, the study conducted among 2,374 Rwandan women was large and controlled for time-varying contraceptive use as well as other important variables. The available evidence suggests no association between DMPA use and HPV infection, though one study did identify a marginally significant increased risk of HPV persistence among injectable contraceptive users. In regards to HSV-2, one study found no association while two large studies found significantly increased risk of HSV-2 with DMPA use. The large study size and robust sensitivity analyses in the study by Grabowski et al32 provides supporting evidence to a positive association between risk of HSV-2 and DMPA use. Lastly, though there are studies that show both no and negative associations with BV, the weight of the evidence, particularly as evidenced by studies with large sample sizes, suggests that DMPA use is associated with a reduced risk of BV. Notably, several of the studies which identified protective effects were among women at high risk of STI, suggesting that any true protective effect may be population-specific, or may not have been detected in other studies due to overall low risk of BV among the study population.
Levonorgestrel intrauterine systems and copper intrauterine devices
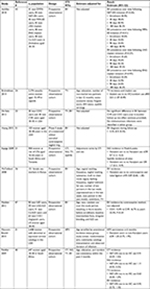
We identified eleven studies which evaluated the association between the LNG-IUS and/or the Cu-IUD and chlamydia, gonorrhea, trichomoniasis, HPV and BV; we did not identify any published studies that met our inclusion criteria which evaluated IUD use and HSV or syphilis. Summary data are provided in Table 3.
Chlamydia
We identified one study that compared chlamydia diagnoses before and 1 year after insertion of an LNG-IUS (n=42) and the Cu-IUD (n=108) in Turkey.44 No chlamydia infections were identified in either group either before or after IUD insertion.
Gonorrhea
The same study conducted in Turkey also evaluated the incidence of gonorrhea among women before and after insertion of an LNG-IUS (n=42) and the Cu-IUD (n=108).44 Again, no gonoccocal infections were identified in either group either before or after IUD insertion.
Trichomoniasis
We identified three studies that evaluated trichomoniasis among LNG-IUS and Cu-IUD users. The aforementioned Turkish study found no trichomoniasis among either group before or after IUD/IUS insertion.44 A second previously described study conducted among 194 LNG-IUS users in Egypt again found no significant difference in T. vaginalis infection between LNG-IUS and COC users.23 Additionally, in a study among 187 new LNG-IUS users in Brazil, no significant alteration in TV was found over 7 years following insertion (P=0.67).50
Human papillomavirus
We identified two studies that considered HPV acquisition, persistence and/or clearance among Cu-IUD and LNG-IUS users. A US study followed 150 new Cu-IUD users and 152 new LNG-IUS users. At a 1-year follow-up there were two (1.7%) new HR-HPV infections in the Cu-IUD group compared with 8 (6.9%) in the LNG-IUS group (P=0.06).45 Clearance of HR-HPV infections was significantly higher in the Cu-IUD group (70% [95% CI 53.6–86.4]) than in the LNG-IUS group (42% [95% CI 25.6–57.8]). A Danish study of type-specific persistence of HR-HPV found that neither LNG-IUS use prior to baseline nor during the 1-year period after baseline resulted in increased HPV persistence.29
Bacterial vaginosis
We identified nine studies that examined LNG-IUS and Cu-IUDs and the prevalence, incidence and/or persistence of bacterial vaginosis; eight of these studies found no significant association. In a study conducted among women in Zimbabwe, BV prevalence significantly increased in women initiating Cu-IUD use from 27% at baseline, to 35% at 30 days, 40% at 90 days, and 49% at 180 days (P=0.005 compared to prevalence at enrollment).43 Another study among 70 Canadian women found 9% BV incidence at 1 month (4/43 women BV negative at enrollment) following IUD placement (either Cu-IUD or LNG-IUS), though no comparisons were done between women using the Cu-IUD or LNG-IUS, and BV history among those women is not reported.46 Among the 153 US women who were BV-negative at baseline in a sub-study of the CHOICE study, no significant association was observed.47 Among 78 new Cu-IUD users and 94 LNG-IUS users, Neale et al48 observed no significant differences in BV at 1 month or 6 months post-insertion between Cu-IUD and LNG-IUS users. Among 252 new LNG-IUS users in Belgium, Donders et al49 found no significant differences in BV pre-insertion to 3-months, 1-year and 5-years after insertion. Similarly, Lessard et al50 found no difference in BV prevalence over 7 years of LNG-IUS use. Rezk et al23 reported no significant difference in BV between LNG-IUS and COC users during follow-up, as did McClelland et al,36 though the type of IUD used, and the number of IUD users during follow-up, was not specified but is presumed to be low based on the small number of IUD users at baseline (n=2). Finally, in the aforementioned Turkish study, there were no significant differences in BV rates before vs 1 year after insertion for 108 new Cu-IUD users and 42 new LNG-IUS users as well as no difference in BV detection at 1 year between the two IUD groups.44
Levonorgestrel IUS and the copper IUD: summary
The available data on the association between Cu-IUD and LNG-IUS use and chlamydia, gonorrhea and trichomoniasis are sparse, but suggest no significant association. The data on IUD use and HPV acquisition and persistence are mixed, with one study suggesting somewhat higher HPV acquisition and persistence among LNG-IUS users than Cu-IUD users, while another study suggested no significant difference in HPV persistence associated with the LNG-IUS compared with non-users, a majority of whom used COCs.29,45 Eight of nine studies which evaluated BV incidence, recurrence and/or persistence found no significant association, with only one study reporting increasing BV over 6 months following Cu-IUD insertion. Additionally, although several studies did not differentiate between Cu-IUD and LNG-IUS users, those that did suggest no difference in BV between the two types of IUDs.
Other contraceptive methods
We identified few studies which evaluated the use of other contraceptive methods and risk of STI; we therefore collapsed the findings by method only below, rather than by STI. Summary data are provided in Table 4.
Contraceptive vaginal ring and patch (combined estrogen + progestin)
We identified three studies that examined the association between combined CVR and STI/BV risk. A phase III study of combined Nestorone and ethinyl estradiol (EE) CVR enrolled 120 US women and found no significant differences in BV or T. vaginalis detection at ring use cycles six or 13 as compared to baseline (prior to CVR insertion).51 Similarly, in the Italian study among 60 new COC and CVR containing desogestrel and EE users, there were no significant differences in incident BV or intermediate vaginal flora over 6 months between the two groups.33 Lastly, in the previously referenced CHOICE sub-study, there were no significant differences in BV incidence among etonogestrel/EE CVR users as compared to women using COCs or the contraceptive patch containing norelgestromin and EE.47
Implant (progestin-only)
We identified five prospective studies that examined the association between progestin-only implant use and STI acquisition. One each evaluated Norplant use and HPV persistence and BV and found no association.31,36 However, a study among Ugandan women found that Norplant use was associated with a significantly higher rate of trichomonas as compared to women not using hormonal contraception or condoms (aIRR 3.01, [95% CI 1.07–8.49]).24 A study conducted in South Africa, Tanzania and Zambia found increased risk of gonorrhea (OR 5.3, [95% CI 1.4–19.8]) but no significant increased risk of syphilis among Norplant users.21 Lastly, a study which evaluated BV prevalence over time among new initiators of the ENG and LNG-IUS implants found no significant associations.43
Other injectables
The study among 567 South African family planning clinic attendees found no significant association between NET-EN and chlamydia, gonorrhea, trichomonas or BV over time when considering only incident infections.40 However, when considering all cases of BV, including positive results followed by a previous positive result, NET-EN showed a protective effect (IRR 0.78 [95% CI 0.64–0.94]). A second study among 266 Zimbabwean women, 44 of whom used NET-EN and 40 who used MPA-EE, found no significant associations.40,43
Other contraceptive methods: summary
The data on other contraceptive methods and STI/BV are sparse. Nevertheless the existing evidence suggests no increased risk of trichomonas or BV among users of the CVR and patch (combined estrogen+progestin). Studies of progestin only implants have generally found no association between implants (primarily Norplant) and BV, HPV or syphilis. However, one study each found increased risks of trichomonas and gonorrhea among implant users. The use of other injectables, primarily NET-EN, are sparse with only one study evaluating chlamydia, gonorrhea and trichomonas, and two evaluating BV with mixed results.
Discussion
Overall quality of the evidence
The overall evidence on the association between contraceptive use and STI/BV risk is highly limited by the lack of any randomized trials, few published prospective studies originally designed to analyze this association, wide variability in exposure definitions and comparator groups, potential mismeasurement of self-reported exposure, potential for confounding by inaccurate sexual behavior data and differential confounder adjustment and differences in study populations and sizes.
A major limitation in the comparability of study findings is wide variability in exposure definitions and comparator/reference groups. Exposure classifications range by method type, mode of delivery, hormones contained within, and timing and duration of exposure. In addition, while some studies limit exposure misclassification by employing statistical methods to allow for time-varying contraceptive use, others risk misclassification by defining exposure at baseline only. Similarly, we identified multiple methods of establishing comparator groups. The most robust method we identified formed discrete time-varying exposure groups by the majority method used during defined and frequent study intervals, whereas other studies used less robust methods, allowing participants to be simultaneously classified in more than one exposure group. This approach results in the formation of comparator groups with different memberships depending on the contraceptive exposure under analysis. Other studies compared women using a specific method to all other women in the analysis, thus allowing the comparator group to consist of a group of women using various methods. Lastly, we excluded several studies due to lumping of contraceptive methods (eg, hormonal vs non-hormonal) which precludes ascertainment of associations between specific contraceptive methods and STI/BVs. Overall, our assessment is that the number of exposure type/timing/duration classification permutations are effectively equivalent to the number of published studies, significantly limiting the comparability of findings and thus the ability to draw robust conclusions from the published evidence base.
In addition to differences in handling of exposure data during the analysis process, concerns about inaccuracy of self-reported hormonal contraceptive (HC) use have recently emerged.52,53 Achilles et al52 found that 27% of participants reporting no contraceptive use at study enrollment had laboratory evidence of recent HC use inconsistent with their self-report, and 36% of study participants had evidence of HC use which they had not reported during follow-up. Only one of the studies we included in this review included objective assessment of contraceptive use (ie, measurement of specific synthetic hormones). Similarly, in an analysis of 1,102 specimens from 664 African women, Pyra et al53 found that 14% of women reporting no hormonal contraceptive use had detectable exogenous hormones in serum, and varying levels of discordance by reported specific contraceptive method use. Thus, the potential for exposure misclassification is of concern in this body of literature, though may be less of an issue for methods such as the IUD and implant which are not subject to daily dosing and/or routine injections.
None of the studies we evaluated randomized women to different contraceptive methods, and users of different contraceptive methods may differ in ways that are directly, or indirectly, related to the risk of STI/BV in terms of number of sexual partners, frequency of sex, sexual networks and condom use. While most studies included in this review reported adjusted risk estimates, with attempted control of sexual behavior variability, multiple studies have demonstrated self-reported sexual behavior data to be partially inaccurate.54–56 Thus, even in the context of robust study design and employment of rigorous analytic approaches, uncontrolled and/or residual confounding may still bias the results due to inaccurate self-reported behavior by study participants.
Lastly, differences in study populations, study size and differential control of confounding variables also reduce the comparability of study results and evidence base. Few studies identified reported a priori power calculations, and many of the studies were of small sample size; these studies may have thus failed to detect true differences in STI/BV risk. Moreover, recent studies have demonstrated important population differences in vaginal mucosal immunologic factors and flora, suggesting that populations may have differential risk of STI/BV, and therefore also potential different risk associated with the use of contraceptives.57,58
Combined oral contraceptives
The evidence for COC use and incident STI remains inconclusive, consistent with the findings reported in prior systematic reviews.14,15 We identified four studies which evaluated risk of chlamydia. Russell et al19 identified no association with incident C. trachomatis infection and COC use. They collected contraceptive use data at each follow-up visit and were therefore able to control for time-varying exposure. Additionally, C. trachomatis nucleic acid amplification testing (NAAT) was conducted at months 1, 4, 8 and 12 following enrollment, providing a robust measure of outcome ascertainment. The study by Kapiga et al21 followed women monthly with high retention rates and found no association; however, it is unclear whether women were analyzed by their baseline contraceptive method or method switching was considered in analyses. Similarly, the study by Masese et al20 followed women monthly and found no association, but again, it is unknown whether contraceptive method switching was taken into account. The study by Borgdorff et al22 reported control of time-varying contraceptive method and found a statistically and clinically significant increased risk of chlamydia with COC use. Given these conflicting results and study designs, we cannot assess whether the differential findings are due to methodological, analytic and/or population differences. The two studies which evaluated COC use and gonorrhea, four which evaluated trichomonas, and two which evaluated HSV-2, suggest no association. We identified more studies (n=8) evaluating COC use and HPV infection; however, the ability to draw definitive conclusions is limited by several factors. While the cohorts in the reported studies are relatively large, many studies have coarse measures of COC use, and there is variability in COC measurement across all included studies. The study by Louvanto et al25 found that women who initiated COC use at less than 20 years of age had 1.3 times the risk of HPV types 7 or 9; however, women initiating contraception early also represent a population that may be initiating sex earlier, and therefore have an inherently higher lifetime risk of HPV. Similarly, the study by Goodman et al found a significantly reduced risk of BV recurrence among COC users.26 Similarly, Goodman et al26 observed decreasing HPV incidence to be associated with years of COC use. The analysis however only adjusted for age at study entry, leaving open the potential for confounding by other important predictors. The results from published studies on BV risk associated with COC use are weighted toward no association (6 of 8 studies). Among those which found no association, sample sizes were notably smaller, and one specifically noted the inability to enroll the target sample size to achieve planned power, whereas Bradshaw et al37 – who identified reduced risk of BV in COC users – had a comparatively larger sample size. Again, as with the evidence on risk of HPV infection among COC users, the comparability of the evidence is highly limited due to differences in comparator groups, variability in outcome definitions, varying intervals between outcome measurements and differential confounder control.
DMPA
Similar to the evidence on COC use, there is minimal new evidence on the association between DMPA use and risk of STI/BV. A single study suggests that DMPA use may result in increased risk of C. trachomatis infection among Kenyan women, but there is no evidence of increased risk in five other studies. Many of these studies included routine laboratory testing among all cohort participants for C. trachomatis infection, and thereby had robust methods for outcome ascertainment. There is no evidence of an association between DMPA use and risk of gonorrhea and syphilis, and mixed evidence suggesting a possible decreased risk of trichomonas among DMPA users. These findings are fairly consistent with prior systematic reviews which concluded a possible association between DMPA and C. trachomatis and no evidence of an association with N. gonorrhoeae or T. vaginalis.14,15 The absence of evidence may thus represent a true null effect, or simply lack of sufficient data. While there were more studies on DMPA use and HPV risk, the inconsistency in endpoint definition – ie, any HPV infection, any HR-HPV infection, and re-detection – in addition to differences in exposure classification groups and confounder control, significantly limits the comparability of results. Nonetheless, the weight of the evidence does not suggest an association between contraceptive use and HPV infection at this time. Two new large studies, both of which accounted for contraceptive method switching in analyses, found statistically and clinically significant increases in HSV-2 incidence among DMPA users.32,41 The study by Grabowski et al32 included a sensitivity analysis among women with known HSV-2 positive partners which supported the primary finding. The strong associations observed, along with other robust evidence of HSV-2 being a risk factor for subsequent HIV infection, warrants concern and further investigation into this potential association. We identified seven new studies which evaluated the DMPA-BV association, among which four observed a decreased risk of BV among DMPA users. These findings are consistent with the results of prior studies on this topic, including a 2013 systematic review which concluded that COCs and DMPA reduce the prevalence and incidence of BV in women, though the overall results among studies remain mixed.38
Intrauterine devices
Overall, there are fewer studies which have evaluated the association between IUD use and STI/BV risk as compared to COCs and DMPA. The available new data on the effect of Cu-IUD and LNG-IUS on the acquisition of chlamydia, gonorrhea and trichomoniasis suggests no increased risk for any STI associated with either device, though the data are sparse. The data related to HPV acquisition and persistence are similarly sparse, with one study suggesting somewhat higher HPV acquisition and persistence among LNG-IUS users than Cu-IUD users, while another study suggested no statistically significant increased HPV persistence between LNG-IUS users compared with non-users.29,45 The majority of the evidence related to the relationship between Cu-IUD and BV suggests no association between BV and Cu-IUD or LNG-IUS use. However, one well-designed study (among nine identified studies) did observe increased BV levels following Cu-IUD use. Additionally, most current studies comparing the two IUDs have found no difference in BV detection rates when comparing users of the different IUD types, and when comparing IUD users (both Cu-IUD and LNG-IUS) to women using combined estrogen-progestin containing contraceptives (COCs, rings, patches).
Other methods
There is no evidence of any association between use of the contraceptive ring or patch and STI/BV, though the data are very sparse. Studies on STI/BV risk among implant and other injectable users are also sparse with mixed results and not sufficiently comparable to draw firm conclusions.
Conclusion
In conclusion, the evidence base for the association between contraceptive use and STI/BV risk remains highly limited. Prospective studies with the primary objective of understanding STI/BV incidence and persistence as a result of hormonal contraceptive are needed. Randomization to contraceptive method would minimize bias associated with method self-selection, though it is unlikely that funding for randomized controlled trials (RCT) will become available. In the absence of an RCT, additional prospective studies with clear exposure measurement are needed. Validation of participant self-reported contraceptive use with systemic exogenous hormonal levels is recommended. Systematic lab-based outcome (STI/BV) testing should be done during follow-up with clear methods to differentiate prevalent vs incident infection. Lastly, measurement of potential confounders (including but not limited to race/ethnicity, location/geography, sexual behavior, other STI/BV diagnoses and concomitant medication use) should be measured during follow-up at multiple timepoints, and appropriate methods for inclusion of confounders in statistical models defined a priori. Such robust studies are needed to provide women and providers accurate information, and thereby facilitate fully informed contraceptive choice by women.
Acknowledgment
Support for drafting this manuscript was provided by the institutional resources of FHI 360.
Author contributions
JD managed the overall writing process; all authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
United Nations Division of Economic and Social Affairs, Population Division. Trends in Contraceptive Use Worldwide 2015; 2015. Available from: http://www.un.org/en/development/desa/population/publications/pdf/family/trendsContraceptiveUse2015Report.pdf. Accessed May 15, 2018. | ||
Family Planning. 2020. Available from: http://www.familyplanning2020.org/microsite/about-us. Accessed April 5, 2018. | ||
World Health Organization. Casting light on old shadows: ending sexually transmitted infection epidemics as public health concerns by 2030: advocacy brief. Geneva. 2017. | ||
Bartz D, Maurer R, Kremen J, Fortin JM, Janiak E, Goldberg AB. High-risk sexual behaviors while on depot medroxyprogesterone acetate as compared to oral contraception. Contracept Reprod Med. 2017;2:8. | ||
Polis CB, Curtis KM, Hannaford PC, et al. Update on hormonal contraceptive methods and risk of HIV acquisition in women. a systematic review of epidemiological evidence. AIDS. 2016;30(17):2665–2683. | ||
Smith JA, Heffron R, Butler AR, Celum C, Baeten JM, Hallett TB. Could misreporting of condom use explain the observed association between injectable hormonal contraceptives and HIV acquisition risk? Contraception. 2017;95(4):424–430. | ||
Bernard C, Zhao Q, Peipert JF. Dual method use among long-acting reversible contraceptive users. Eur J Contracept Reprod Health Care. 2018;23(2):97–104. | ||
Wira CR, Rodriguez-Garcia M, Patel MV. The role of sex hormones in immune protection of the female reproductive tract. Nat Rev Immunol. 2015;15(4):217–230. | ||
Deese J, Masson L, Miller W, et al. Injectable Progestin-Only Contraception is Associated With Increased Levels of Pro-Inflammatory Cytokines in the Female Genital Tract. Am J Reprod Immunol. 2015;74(4):357–367. | ||
Kaushic C, Ashkar AA, Reid LA, Rosenthal KL. Progesterone increases susceptibility and decreases immune responses to genital herpes infection. J Virol. 2003;77(8):4558–4565. | ||
Lee Y, Dizzell SE, Leung V, et al. Effects of Female Sex Hormones on Susceptibility to HSV-2 in Vaginal Cells Grown in Air-Liquid Interface. Viruses. 2016;8(9):241. | ||
Kaushic C, Zhou F, Murdin AD, Wira CR. Effects of estradiol and progesterone on susceptibility and early immune responses to Chlamydia trachomatis infection in the female reproductive tract. Infect Immun. 2000;68(7):4207–4216. | ||
Brooks JP, Edwards DJ, Blithe DL, et al. Effects of combined oral contraceptives, depot medroxyprogesterone acetate and the levonorgestrel-releasing intrauterine system on the vaginal microbiome. Contraception. 2017;95(4):405–413. | ||
Morrison CS, Turner AN, Jones LB. Highly effective contraception and acquisition of HIV and other sexually transmitted infections. Best Pract Res Clin Obstet Gynaecol. 2009;23(2):263–284. | ||
Mohllajee AP, Curtis KM, Martins SL, Peterson HB. Hormonal contraceptive use and risk of sexually transmitted infections: a systematic review. Contraception. 2006;73(2):154–165. | ||
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement. Open Med. 2009;3(3):e123–130. | ||
Unemo M, Bradshaw CS, Hocking JS, et al. Sexually transmitted infections: challenges ahead. Lancet Infect Dis. 2017;17(8):e235–e279. | ||
van de Wijgert J, Jespers V. The global health impact of vaginal dysbiosis. Res Microbiol. 2017;168(9-10):859–864. | ||
Russell AN, Zheng X, O’Connell CM, et al. Analysis of Factors Driving Incident and Ascending Infection and the Role of Serum Antibody in Chlamydia trachomatis Genital Tract Infection. J Infect Dis. 2016;213(4):523–531. | ||
Masese L, Baeten JM, Richardson BA, et al. Incidence and correlates of Chlamydia trachomatis infection in a high-risk cohort of Kenyan women. Sex Transm Dis. 2013;40(3):221–225. | ||
Kapiga S, Kelly C, Weiss S, et al. Risk factors for incidence of sexually transmitted infections among women in South Africa, Tanzania, and Zambia: results from HPTN 055 study. Sex Transm Dis. 2009;36(4):199–206. | ||
Borgdorff H, Verwijs MC, Wit FW, et al. The impact of hormonal contraception and pregnancy on sexually transmitted infections and on cervicovaginal microbiota in african sex workers. Sex Transm Dis. 2015;42(3):143–152. | ||
Rezk M, Sayyed T, Masood A, Dawood R. Risk of bacterial vaginosis, Trichomonas vaginalis and Candida albicans infection among new users of combined hormonal contraception vs LNG-IUS. Eur J Contracept Reprod Health Care. 2017;22(5):344–348. | ||
Brahmbhatt H, Musoke R, Makumbi F, et al. Trichomonas vaginalis Incidence Associated with Hormonal Contraceptive Use and HIV Infection among Women in Rakai, Uganda. J Sex Transm Dis. 2014;2014:916597:1–10. | ||
Louvanto K, Rintala MA, Syrjänen KJ, Grénman SE, Syrjänen SM. Incident cervical infections with high- and low-risk human papillomavirus (HPV) infections among mothers in the prospective Finnish Family HPV Study. BMC Infect Dis. 2011;11:179. | ||
Goodman MT, Shvetsov YB, Mcduffie K, et al. Prevalence, acquisition, and clearance of cervical human papillomavirus infection among women with normal cytology: Hawaii Human Papillomavirus Cohort Study. Cancer Res. 2008;68(21):8813–8824. | ||
Schmeink CE, Massuger LF, Lenselink CH, Quint WG, Melchers WJ, Bekkers RL. Effect of the menstrual cycle and hormonal contraceptives on human papillomavirus detection in young, unscreened women. Obstet Gynecol. 2010;116(1):67–75. | ||
Marks M, Gravitt PE, Gupta SB, et al. Combined oral contraceptive use increases HPV persistence but not new HPV detection in a cohort of women from Thailand. J Infect Dis. 2011;204(10):1505–1513. | ||
Stensen S, Kjaer SK, Jensen SM, et al. Factors associated with type-specific persistence of high-risk human papillomavirus infection: A population-based study. Int J Cancer. 2016;138(2):361–368. | ||
Shew ML, Ermel AC, Tong Y, Tu W, Qadadri B, Brown DR. Episodic detection of human papillomavirus within a longitudinal cohort of young women. J Med Virol. 2015;87(12):2122–2129. | ||
Maucort-Boulch D, Plummer M, Castle PE, et al. Predictors of human papillomavirus persistence among women with equivocal or mildly abnormal cytology. Int J Cancer. 2010;126(3):684–691. | ||
Grabowski MK, Gray RH, Makumbi F, et al. Use of injectable hormonal contraception and women’s risk of herpes simplex virus type 2 acquisition: a prospective study of couples in Rakai, Uganda. Lancet Glob Health. 2015;3(8):e478–e486. | ||
de Seta F, Restaino S, de Santo D, et al. Effects of hormonal contraception on vaginal flora. Contraception. 2012;86(5):526–529. | ||
de Seta F, Restaino S, Banco R, et al. Effects of estroprogestins containing natural estrogen on vaginal flora. Gynecol Endocrinol. 2014;30(11):830–835. | ||
Francis SC, Looker C, Vandepitte J, et al. Bacterial vaginosis among women at high risk for HIV in Uganda: high rate of recurrent diagnosis despite treatment. Sex Transm Infect. 2016;92(2):142–148. | ||
McClelland RS, Richardson BA, Graham SM, et al. A prospective study of risk factors for bacterial vaginosis in HIV-1-seronegative African women. Sex Transm Dis. 2008;35(6):617–623. | ||
Bradshaw CS, Vodstrcil LA, Hocking JS, et al. Recurrence of bacterial vaginosis is significantly associated with posttreatment sexual activities and hormonal contraceptive use. Clin Infect Dis. 2013;56(6):777–786. | ||
van de Wijgert JH, Verwijs MC, Turner AN, Morrison CS. Hormonal contraception decreases bacterial vaginosis but oral contraception may increase candidiasis: implications for HIV transmission. AIDS. 2013;27(13):2141–2153. | ||
Romer A, Shew ML, Ofner S, Gilliam ML, Martins SL, Fortenberry JD. Depot medroxyprogesterone acetate use is not associated with risk of incident sexually transmitted infections among adolescent women. J Adolesc Health. 2013;52(1):83–88. | ||
Pettifor A, Delany S, Kleinschmidt I, Miller WC, Atashili J, Rees H. Use of injectable progestin contraception and risk of STI among South African women. Contraception. 2009;80(6):555–560. | ||
Socias ME, Duff P, Shoveller J, Montaner JSG, Nguyen P. Use of injectable hormonal contraception and HSV-2 acquisition in a cohort of female sex workers in Vancouver, Canada. Sex Transm Infect. 2016:1–6. | ||
Rifkin SB, Smith MR, Brotman RM, Gindi RM, Erbelding EJ. Hormonal contraception and risk of bacterial vaginosis diagnosis in an observational study of women attending STD clinics in Baltimore, MD. Contraception. 2009;80(1):63–67. | ||
Achilles SL, Austin MN, Meyn LA, Mhlanga F, Chirenje ZM, Hillier SL. Impact of contraceptive initiation on vaginal microbiota. Am J Obstet Gynecol. 2018;218(6):622.e1–622. | ||
Erol O, Simavlı S, Derbent AU, Ayrım A, Kafalı H. The impact of copper-containing and levonorgestrel-releasing intrauterine contraceptives on cervicovaginal cytology and microbiological flora: a prospective study. Eur J Contracept Reprod Health Care. 2014;19(3):187–193. | ||
Lekovich JP, Amrane S, Pangasa M, et al. Comparison of human papillomavirus infection and cervical cytology in women using copper-containing and levonorgestrel-containing intrauterine devices. Obstet Gynecol. 2015;125(5):1101–1105. | ||
Alice TND, Kives S, Merovitz L, Nitsch R, Tessler K, Yudin MH. Screening for bacterial vaginosis at the time of intrauterine contraceptive device insertion: is there a role? J Obstet Gynaecol Can. 2012;34(2):179–185. | ||
Madden T, Grentzer JM, Secura GM, Allsworth JE, Peipert JF. Risk of bacterial vaginosis in users of the intrauterine device: a longitudinal study. Sex Transm Dis. 2012;39(3):217–222. | ||
Neale R, Knight I, Keane F. Do users of the intrauterine system (Mirena) have different genital symptoms and vaginal flora than users of the intrauterine contraceptive device? Int J STD AIDS. 2009;20(6):423–424. | ||
Donders GGG, Bellen G, Ruban K, van Bulck B. Short- and long-term influence of the levonorgestrel-releasing intrauterine system (Mirena®) on vaginal microbiota and Candida. J Med Microbiol. 2018;67(3):308–313. | ||
Lessard T, Simões JA, Discacciati MG, Hidalgo M, Bahamondes L. Cytological evaluation and investigation of the vaginal flora of long-term users of the levonorgestrel-releasing intrauterine system (LNG-IUS. Contraception. 2008;77(1):30–33. | ||
Huang Y, Merkatz RB, Hillier SL, et al. Effects of a One Year Reusable Contraceptive Vaginal Ring on Vaginal Microflora and the Risk of Vaginal Infection: An Open-Label Prospective Evaluation. PLoS One. 2015;10(8):e0134460. | ||
Achilles SL, Mhlanga FG, Musara P, Poloyac SM, Chirenje ZM, Hillier SL. Misreporting of contraceptive hormone use in clinical research participants. Contraception. 2018;97(4):346–353. | ||
Pyra M, Lingappa JR, Partners in Prevention HSV/HIV Transmission Study and Partners PrEP Study Teams, et al. Concordance of self-reported hormonal contraceptive use and presence of exogenous hormones in serum among African women. Contraception. 2018;97(4):357–362. | ||
Minnis AM, Steiner MJ, Gallo MF, et al. Biomarker validation of reports of recent sexual activity: results of a randomized controlled study in Zimbabwe. Am J Epidemiol. 2009;170(7):918–924. | ||
Turner AN, Miller WC, Padian NS, et al. Unprotected sex following HIV testing among women in Uganda and Zimbabwe: short- and long-term comparisons with pre-test behaviour. Int J Epidemiol. 2009;38(4):997–1007. | ||
Heffron R, Parikh UM, Partners PrEP Study Team, et al. Objective measurement of inaccurate condom use reporting among women using depot medroxyprogesterone acetate for contraception. AIDS Behav. 2017;21(7):2173–2179. | ||
Cohen CR, Moscicki AB, Scott ME, et al. Increased levels of immune activation in the genital tract of healthy young women from sub-Saharan Africa. AIDS. 2010;24(13):2069–2074. | ||
Fettweis JM, Brooks JP, Serrano MG, et al. Differences in vaginal microbiome in African American women versus women of European ancestry. Microbiology. 2014;160(Pt 10):2272–2282. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.