Back to Journals » Vascular Health and Risk Management » Volume 10
Consumption of water containing over 3.5 mg of dissolved hydrogen could improve vascular endothelial function
Authors Sakai T, Sato B, Hara K, Hara Y, Naritomi Y, Koyanagi S, Hara H, Nagao T, Ishibashi T
Received 4 June 2014
Accepted for publication 13 August 2014
Published 17 October 2014 Volume 2014:10 Pages 591—597
DOI https://doi.org/10.2147/VHRM.S68844
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Daniel Duprez
Takaaki Sakai,1 Bunpei Sato,2 Koji Hara,3 Yuichi Hara,3 Yuji Naritomi,3 Samon Koyanagi,1 Hiroshi Hara,3 Tetsuhiko Nagao,4 Toru Ishibashi5
1Department of Cardiology, Haradoi Hospital, Fukuoka, Japan; 2MiZ Company Limited, Fujisawa, Kanagawa, Japan; 3Department of Internal Medicine, Haradoi Hospital, Fukuoka, Japan; 4Midorino Clinic, Aoba, Higashi-ku, Fukuoka, Japan; 5Department of Rheumatology and Orthopedic Surgery, Haradoi Hospital, Fukuoka, Japan
Background: The redox imbalance between nitric oxide and superoxide generated in the endothelium is thought to play a pivotal role in the development of endothelial dysfunction. A third reactive oxygen species (ROS), H2O2, is known to have both beneficial and detrimental effects on the vasculature. Nonetheless, the influence of the hydroxyl radical, a byproduct of H2O2 decay, is unclear, and there is no direct evidence that the hydroxyl radical impairs endothelial function in conduit arteries. Molecular hydrogen (H2) neutralizes detrimental ROS, especially the hydroxyl radical.
Objectives: To assess the influence of the hydroxyl radical on the endothelium and to confirm that a gaseous antioxidant, H2, can be a useful modulator of blood vessel function.
Methods: The efficacy of water containing a high concentration of H2 was tested by measuring flow-mediated dilation (FMD) of the brachial artery (BA). The subjects were randomly divided into two groups: the high-H2 group, who drank high-H2 water containing 7 ppm H2 (3.5 mg H2 in 500 mL water); and the placebo group. Endothelial function was evaluated by measuring the FMD of the BA. After measurement of diameter of the BA and FMD at baseline, volunteers drank the high-H2 water or placebo water immediately and with a 30-minute interval; FMD was compared to baseline.
Results: FMD increased in the high-H2 group (eight males; eight females) from 6.80%±1.96% to 7.64%±1.68% (mean ± standard deviation) and decreased from 8.07%±2.41% to 6.87%±2.94% in the placebo group (ten males; eight females). The ratio to the baseline in the changes of FMD showed significant improvement (P<0.05) in the high-H2 group compared to the placebo group.
Conclusion: H2 may protect the vasculature from shear stress-derived detrimental ROS, such as the hydroxyl radical, by maintaining the nitric oxide-mediated vasomotor response.
Keywords: flow-mediated dilation, reactive oxygen species, molecular hydrogen, hydroxyl radical, 5–7 ppm, peroxynitrite
Introduction
The vascular endothelium is a functional tissue that responds to shear stress caused by the varying blood flow and pressure, thereby maintaining the appropriate vasomotor tone.1 Endothelial dysfunction, therefore, causes accumulation of vascular damage and induces chronic inflammation, followed by atherosclerosis and cardiovascular disease.2 Endothelium-derived relaxing factors, including nitric oxide (NO),3,4 prostacyclin,5 and endothelium-derived hyperpolarization factor (EDHF)6–8 dilate the vessels to maintain homeostasis of the vasculature. Among these mechanisms, NO-mediated vasodilation plays a pivotal role.9,10 To assess endothelial function, the flow-mediated dilation (FMD) test has been developed.11,12 FMD in response to occlusion-induced hyperemia has been presumed to be a useful method for the estimation of the bioavailability of endothelium-derived NO,10 especially in a conduit artery like the brachial artery.13,14 Using this noninvasive and rapid measurement technique, the risk factors of endothelial dysfunction, including aging,15 diabetes mellitus,16 smoking,17,18 hypertension,19,20 and postprandial hyperglycemia21 have been validated.
Under all of these conditions, oxidative stress is elevated, and the redox imbalance is thought to cause a serious failure of endothelial function.22
In the vasculature, including endothelium and vascular smooth muscle cells,23 reactive oxygen species (ROS) perform crucial functions in the pathogenesis of vascular disease and serve as a modulator of vasomotor function. One of those ROS, superoxide, generated by nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, xanthine oxidase, and electron leakage from the mitochondrial respiratory chain, is the key factor that attenuates bioavailability of NO by inactivating it through conversion to a detrimental ROS, peroxynitrite.22 NADPH oxidases that enzymatically produce superoxide and H2O2 are grouped in the Nox family of proteins and are thought to be a major source of ROS in the vasculature. Among them, Nox1, Nox2, and Nox4 are thought to perform important functions in the vasculature. These Nox proteins are activated by the vascular shear stress.1,24–26 In particular, Nox1 and Nox2 predominantly produce superoxide through a single electron transfer to molecular hydrogen. Superoxide rapidly reacts with NO and inactivates it. This reaction generates peroxynitrite, which has adverse effects on the NO-modulated vasodilation.27
Furthermore, such oxidative conditions (when peroxynitrite is generated) inhibit activity of the endothelial nitric oxide synthase (eNOS) (ie, NO production). Oxidation of the authentic cofactor of eNOS, tetrahydrobiopterin (BH4), to the inactive form, 7,8-dihydropterin (BH2), induces uncoupling of eNOS, and this process, in turn, produces the antagonistic superoxide.28–30 Such a deleterious feedback cycle seems to promote excessive levels of superoxide, which can accelerate progression of endothelial dysfunction.
Superoxide is detoxified by superoxide dismutase (SOD) and is converted to H2O2, which is a more stable ROS.31 Unlike superoxide, H2O2 functions as an agonist for vasodilation, probably as EDHF in some types of vessels like resistance arteries32 and as an antagonistic ROS.33 It is known that – unlike Nox1 and Nox2 – Nox4 predominantly produces H2O2 via a specific alteration in its E-loop.34,35 The biological properties of H2O2 depend on the size of the vessels or the concentration of H2O2 that the endothelium is exposed to.36,37 On the other hand, in microcirculation like observed in the coronary artery, H2O2 has adverse effects via upregulation of arginase, which degrades the precursor of NO, l-arginine. Although it is still being studied how H2O2 contributes to or impairs endothelial function, it is inevitable that H2O2 is converted to a more reactive ROS, the hydroxyl radical, by the Fenton reaction or Haber–Weiss reaction in the presence of reduced transitional metals.38
Despite the highly detrimental effects of the hydroxyl radical caused by its rapid and indiscriminate reactivity, the mechanism underlying the pathogenicity of ROS in the endothelium is not well understood. Recently, it has been demonstrated that H2 selectively scavenges both hydroxyl radical and peroxynitrite among ROS and does not react with NO.39 To determine the involvement of deleterious ROS, such as the hydroxyl radical and peroxynitrite in the endothelial function, we evaluated the influence of molecular hydrogen (H2) on the vasodilation capability during FMD of the brachial artery (BA) in healthy volunteers. H2 is known as a selective scavenger of peroxynitrite and of the hydroxyl radical and is expected to be a useful antioxidant with a therapeutic potential.40–44 In this study, we observed the favorable influence of molecular hydrogen (H2) on FMD of the BA in the volunteers who drank the high-H2 water. The results here will improve the understanding not only about the toxicity of the hydroxyl radical, but also about the agonistic capabilities of H2 in endothelial vasomotor responses.
Materials and methods
Study design and measurement of FMD using an ultrasound system
Thirty-four volunteers were enrolled in this pilot study (with written informed consent from all the subjects), and this study was approved by the Haradoi Hospital Ethics Committee. The study goals and intended measures were explained individually to the prospective study participants. They did not take any medications or diet supplements or receive any medical treatments for >6 months before this study was performed. Biochemical tests did not show any aberrations, either. The subjects were randomly distributed into two groups – the high-H2 group (n=16) and the placebo group (n=18). All the subjects rested in the supine position after at least 4 hours of fasting before the first scan and remained supine during the evaluation procedures.
Endothelial function was evaluated by measuring FMD of the BA in a core ultrasound laboratory by using a standardized protocol according to the international guidelines and Japanese guidelines of the Vascular Failure Working Group.2,14 We used an ultrasound system equipped with an edge-tracking device for two-dimensional imaging and a pulsed Doppler flow velocimeter for automatic measurement (UNEXEF38G; Unex Co, Ltd, Nagoya, Japan). Regarding the reproducibility of the FMD measurements using this system, the correlation coefficient between two examination sessions was reported to be 0.86 with a coefficient of variance of 11.2%.45
In brief, the diameter of the BA at rest was measured in the cubital region. Subsequently, the cuff was inflated to 50 mmHg above systolic blood pressure for 5 minutes and then deflated. The diameter was manually measured, which usually takes ~2 minutes. The diameter at the same point was monitored continuously. The maximum dilation of the BA was confirmed and measured according to a plateau or till no further increase in the diameter for at least 1 minute after cuff deflation was observed. FMD was calculated as follows:

Preparation of high-H2 water and placebo water
The high-H2 water (hydrogen water 7.0; Ecomo International Co, Ltd, Iizuka-shi, Fukuoka, Japan) was prepared according to previously described methods.40 Briefly, hydrogen gas was produced in an acrylic resin tube in an elastic poly(ethylene terephthalate) (PET) bottle filled with 530 mL of water by mixing 75% of metal aluminum grains with 25% of calcium hydroxide (by weight) and 0.5 mL of water. During the reaction, the hydrogen gas presses against the surface of the water in the standing bottle, which is gradually hardened by the increasing pressure of the emerging hydrogen gas. After the reaction is terminated, the hydrogen gas is dissolved by shaking the bottle with decantation for ~30 seconds. The concentration of hydrogen gas in water at room temperature of the examination room was measured according to the method described previously.46 The placebo water was prepared by filling up a PET bottle (which is usually used for carbonated drinks) containing water with N2 gas under 0.8 MPa to make the placebo bottles as hard as the bottles with the high-H2 water. An acrylic resin tube with a placebo nonwoven fabric (which is used to produce H2 gas in a tube) was also placed in the placebo bottles so that the volunteers could not distinguish between the two types of water.
Statistical analysis
The data are shown as mean ± standard deviation. The significance of differences in variables before and after taking water (high-H2 water or placebo water) in each group was determined by comparing changes in FMD using paired Student’s t-test. The analysis of variance was used to compare changes in variables between the two groups.
Results
The high-H2 water (>5 ppm within an hour40) was prepared more than 12 hours before the volunteers drank it. After the waiting period, the concentration of H2 exceeded 7 ppm, which corresponds to 3.5 mg of H2 molecules in 500 mL of water, as shown in Figure 1. The participants drank it immediately after opening the PET bottle so that they could ingest approximately 3.5 mg of H2.
The baseline demographics of the subjects are presented in Table 1. All subjects enrolled in this study had normal mean levels of baseline biometric parameters and normal results of clinical chemistry and hematology tests. At baseline, there were no significant differences in age, sex, body height, body weight, systolic blood pressure, diastolic blood pressure, and heart rate between the two groups (Table 1).
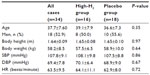  | Table 1 Characteristics of the enrolled volunteers |
The peak concentration of H2 in the bloodstream is thought to be achieved within 10 minutes;40 thus, we performed the FMD test 30 minutes after the subjects drank the high-H2 water so that the endothelium of each subject was exposed to the highest concentration of H2. As shown in Figure 2, in the high-H2 group (eight males; eight females), FMD increased by 19.9%±41.6% after ingestion of 3.5 mg H2; whereas, in the placebo group (placebo water; ten males, eight females), FMD decreased slightly. Although in both groups the change in FMD (%) was not statistically significant, the ratio of the changes in FMD of the high-H2 group was significantly improved when it was compared with the placebo group (P=0.0221).
The differences at baseline between the two groups were assessed regarding vascular parameters, as shown in Table 2. Before ingestion of the high-H2 water or placebo water, no significant differences were observed between the two groups in systolic blood pressure (SBP), diastolic blood pressure (DBP), heart rate (HR), the resting arterial diameter, and FMD%. (These variables might affect FMD).
To evaluate the direct effects of H2 on vasodilation, the changes in diameter of the BA after drinking either high-H2 water or placebo water were measured in three healthy volunteers from each group. In this experiment, the same method for FMD measurements (based on the ultrasound techniques) was used except for the stoppage of the arterial flow. As shown in Table 3, no remarkable changes in the diameter of the BA were observed during the 2-hour measurement in either the high-H2 group or the placebo group.
  | Table 3 Influence of H2 on vasodilation of the brachial artery |
Discussion
ROS including NO, superoxide, peroxynitrite, and H2O2 occupy the center stage in a functional endothelium. The most important biosignal that regulates the level of these reactive oxygen/nitrogen species is shear stress on the vasculature. Flow-mediated shear stress activates NADPH oxidases and eNOS, and they generate superoxide and vasoactive NO, respectively. Once the redox balance between these conflicting bioactive molecules is tipped in favor of the oxidative state, the production of peroxynitrite and H2O2 seems to become excessive, leading to endothelial dysfunction.
Although H2O2 seems to possess agonistic properties for endothelial vasodilation,8,36 it is a precursor of the hydroxyl radical. Despite the strong reactivity of the hydroxyl radical, studies of the influence of this molecule on the endothelium are scarce, and most of them deal with the function of H2O2. In the coronary artery, where H2O2 acts as EDHF,36 the suppression of the production of the hydroxyl radical by chelating ferric ions prevents the adverse effects of H2O2; thus, the hydroxyl radical seems to be responsible for the H2O2-related endothelial dysfunction in the relevant diseases.37
In larger conduit arteries like the BA, it is still unclear whether the hydroxyl radical has antagonistic properties toward endothelial function, where the NO-mediated pathway is predominant. In the present study, we show that a direct scavenger of the hydroxyl radical, H2, significantly improves FMD of the BA. As shown in Tables 1 and 3, the changes in diameter of the BA caused by H2 are dependent on the increased blood flow after the arterial flow is shut off. Therefore, it is possible that the improvement in FMD under the influence of H2 is the result of flow-mediated vasodilation mechanisms accompanied by vascular shear stress. These data are suggestive of the importance of constant removal of a byproduct of H2O2, the hydroxyl radical, to maintain a vasomotor response of a conduit artery and of resistance arteries; both types of artery are frequently exposed to shear stress.
It was demonstrated previously that H2 does not react with NO but scavenges peroxynitrite in vitro.39 Therefore, there is an additional possibility that H2 may improve FMD by eliminating the NO-derived deleterious ROS, peroxynitrite.
Recently, it was demonstrated that H2 has a therapeutic potential against chronic inflammatory diseases, including rheumatoid arthritis (RA).40 In that study, as a result of dissolving the H2 gas at high pressure, the water contained over 5 ppm of H2 (high-H2 water), which is much higher than the saturation concentration of H2 in water, 1.6 ppm. The H2 ingested by drinking the high-H2 water reaches the maximum concentration in the lungs within 10 minutes; these data are indicative of an elevated concentration of H2 in the whole body, including the blood. The concentration of exogenous H2 in the body and its therapeutic efficacy have been studied and discussed elsewhere.41,42 The anti-inflammatory properties of H2 is probably due to the blocking of proinflammatory feedback loops composed of ROS, nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), interleukin-6, and tumor necrosis factor alpha. This mechanism may also explain the preventive potential of H2 in relation to atherosclerosis.44 In addition to the anti-inflammatory potential, the results shown here indicate that by maintaining the NO-mediated vasomotor response, H2 is expected to prevent the vascular shear stress from triggering endothelial dysfunction.
Endothelial dysfunction is considered an initial step in the development of atherosclerosis, which is a chronic inflammatory disease controlled by cytokine-induced immune responses.2,14 To maintain healthy functioning of the endothelium in vasculature, it is therefore important to prevent atherosclerosis, which is the source of cardiovascular disease. Numerous studies have been published about vitamin supplements, such as the antioxidants vitamin C and vitamin E. Nonetheless, a desirable consensus has not been attained, nor is there any evidence that vitamins can reduce the risks of cardiovascular diseases.47,48
Furthermore, there is no evidence that the classical antioxidants can alleviate the chronic inflammation of RA, in contrast to the water containing a high concentration (over 5 ppm) of H2, which considerably reduces the disease activity of RA, according to a recent study.40 The clinical efficacy of H2 against such a classic inflammatory disease raises the hopes of prevention of endothelial dysfunction from daily oxidative injury that precedes atherosclerosis or cardiovascular disease. FMD experiments are also used as a model of ischemia-reperfusion injury. Daily consumption of water containing over 7 ppm H2 may prevent such latent or microscale vascular injury. Although most of the capabilities of H2 are explained by its antioxidant action against the hydroxyl radical, there remains a possibility of other, yet unknown mechanisms triggered by H2.41,44
H2 gas is generated endogenously in human intestines by commensal bacteria, and a small amount of absorbed H2 is detectable in exhaled air.40,41 These observations are indicative of safety of H2 gas. It should be noted that the volume of H2 gas that is produced in the intestines is greater than the amount consumed with the 7-ppm-H2 water in the course of the proposed regimen. It is thought that under atmospheric pressure, most of the intestinal H2 cannot be absorbed by the human body and cells because of the low solubility of H2 in water. Other antioxidant supplements are oxidized after they perform their antioxidant function, and many of these oxidized compounds enhance oxidative stress, which is a major cause of their adverse effects. In contrast, H2 generates H2O after reducing the hydroxyl radical; this is the most important property of H2 that contributes to its safety. Therefore, it is hard to think of any adverse effects of daily intake of H2. No adverse effects have been reported by the deep-sea divers who inhale 70% of H2 under high pressure on a regular basis for more than 10 years. Accordingly, researchers who study H2 think that this gas is an ideal antioxidant. It should be noted that the data presented here are restricted to the acute reactions in the BA within approximately 30 minutes after the administration of H2.
Here, we show only one round of enhanced protection of endothelium by H2 against the FMD-generated shear stress and oxidative stress. Further research is necessary to clarify whether the accumulation of blood flow-mediated oxidative damage can be prevented to some extent by daily consumption of high-H2 water.
Alternatively, we can expect to disrupt the inflammatory cascade composed of ROS, NF-κB, and proinflammatory cytokines by daily consumption of high-H2 water, according to a previous study regarding RA;40 the latter is also linked to atherosclerosis.44 Because of this mechanism, in addition to the protective potential of H2 toward the endothelium in the present study, some other short-term beneficial effects of high-H2 water may be observed.
Recently, the potential efficacy of H2 was tested in type II diabetes49 and metabolic syndrome.50 Both of these medical conditions are linked to endothelial dysfunction,16,19 although the direct influence of H2 on the endothelium has not been proven yet. It is important to study the influence of H2 on FMD under pathological conditions, as well as in health. It should be noted that the number of volunteers enrolled in the present study is limited, and the number of conditions that can influence vasomotor functions is also limited here. Larger-scale studies to observe the influence of H2 on FMD will allow us to estimate the efficacy of H2 on the prevention of age-related metabolic or inflammatory disease.
Conclusion
Our data suggest that the daily consumption of water containing a high concentration of H2 (over 7 ppm or 3.5 mg in 500 mL of water) may aid in maintaining functional vasculature via two complementary mechanisms. One is neutralization of detrimental ROS to preserve NO bioavailability. The other mechanism is suppression of the inflammatory events that cause atherosclerosis. In light of this and other studies, including the anti-inflammatory effects of H2, we believe that larger-scale clinical research – including biochemical tests – should be conducted to confirm whether H2 can serve as a modulator of the vasomotor function of vasculature and/or can maintain health by constitutively suppressing subclinical inflammation of vasculature.
Author contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in either drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published.
Acknowledgments
We thank M Hanada and K Nagao for the technical support. We are grateful to R Kurokawa, S Hirano, T Seo, M Nagao, K Kiyota, and K Fukuoka for their excellent advice on the preparation of placebo water.
Disclosure
The authors report no conflicts of interest in this work.
References
Nigro P, Abe J, Berk BC. Flow shear stress and atherosclerosis: a matter of site specificity. Antioxid Redox Signal. 2011;15(5):1405–1414. | |
Inoue T, Matsuoka H, Higashi Y, et al; Vascular Failure Workshop Group. Flow-mediated vasodilation as a diagnostic modality for vascular failure. Hypertens Res. 2008;31(12):2105–2113. | |
Furchgott RF, Zawadzki JV. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature. 1980;288(5789):373–376. | |
Joannides R, Haefeli WE, Linder L, et al. Nitric oxide is responsible for flow-dependent dilatation of human peripheral conduit arteries in vivo. Circulation. 1995;91(5):1314–1319. | |
Koller A, Kaley G. Prostaglandins mediate arteriolar dilation to increased blood flow velocity in skeletal muscle microcirculation. Circ Res. 1990;67(2):529–534. | |
Taylor SG, Weston AH. Endothelium-derived hyperpolarizing factor: a new endogenous inhibitor from the vascular endothelium. Trends Pharmacol Sci. 1988;9(8):272–274. | |
Garland CJ, Hiley CR, Dora KA. EDHF: spreading the influence of the endothelium. Br J Pharmacol. 2011;164(3):839–852. | |
Stoner L, Erickson ML, Young JM, et al. There’s more to flow-mediated dilation than nitric oxide. J Atheroscler Thromb. 2012;19(7):589–600. | |
Singel DJ, Stamler JS. Chemical physiology of blood flow regulation by red blood cells: the role of nitric oxide and S-nitrosohemoglobin. Annu Rev Physiol. 2005;67:99–145. | |
Green D. Point: Flow-mediated dilation does reflect nitric oxide-mediated endothelial function. J Appl Physiol (1985). 2005;99(3):1233–1234; discussion 1237–1238. | |
Celermajer DS, Sorensen KE, Gooch VM, et al. Non-invasive detection of endothelial dysfunction in children and adults at risk of atherosclerosis. Lancet. 1992;340(8828):1111–1115. | |
Harris RA, Nishiyama SK, Wray DW, Richardson RS. Ultrasound assessment of flow-mediated dilation. Hypertension. 2010;55(5):1075–1085. | |
Bellien J, Iacob M, Gutierrez L, et al. Crucial role of NO and endothelium-derived hyperpolarizing factor in human sustained conduit artery flow-mediated dilatation. Hypertension. 2006;48(6):1088–1094. | |
Corretti MC, Anderson TJ, Benjamin EJ, et al; International Brachial Artery Reactivity Task Force. Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: a report of the International Brachial Artery Reactivity Task Force. J Am Coll Cardiol. 2002;39(2):257–265. | |
Taddei S, Virdis A, Ghiadoni L, et al. Age-related reduction of NO availability and oxidative stress in humans. Hypertension. 2001;38(2):274–279. | |
Kawano H, Motoyama T, Hirashima O, et al. Hyperglycemia rapidly suppresses flow-mediated endothelium-dependent vasodilation of brachial artery. J Am Coll Cardiol. 1999;34(1):146–154. | |
Esen AM, Barutcu I, Acar M, et al. Effect of smoking on endothelial function and wall thickness of brachial artery. Circ J. 2004;68(12):1123–1126. | |
Peluffo G, Calcerrada P, Piacenza L, Pizzano N, Radi R. Superoxide-mediated inactivation of nitric oxide and peroxynitrite formation by tobacco smoke in vascular endothelium: studies in cultured cells and smokers. Am J Physiol Heart Circ Physiol. 2009;296(6):H1781–H1792. | |
Palmieri V, Russo C, Pezzullo S, Di Minno MN, Celentano A. Relation of flow-mediated dilation to global arterial load: impact of hypertension and additional cardiovascular risk factors. Int J Cardiol. 2011;152(2):225–230. | |
Gokce N, Holbrook M, Duffy SJ, et al. Effects of race and hypertension on flow-mediated and nitroglycerin-mediated dilation of the brachial artery. Hypertension. 2001;38(6):1349–1354. | |
Ceriello A, Taboga C, Tonutti L, et al. Evidence for an independent and cumulative effect of postprandial hypertriglyceridemia and hyperglycemia on endothelial dysfunction and oxidative stress generation: effects of short- and long-term simvastatin treatment. Circulation. 2002;106(10):1211–1218. | |
Taniyama Y, Griendling KK. Reactive oxygen species in the vasculature: molecular and cellular mechanisms. Hypertension. 2003; 42(6):1075–1081. | |
Clempus RE, Griendling KK. Reactive oxygen species signaling in vascular smooth muscle cells. Cardiovasc Res. 2006;71(2):216–225. | |
Sorescu GP, Song H, Tressel SL, et al. Bone morphogenic protein 4 produced in endothelial cells by oscillatory shear stress induces monocyte adhesion by stimulating reactive oxygen species production from a nox1-based NADPH oxidase. Circ Res. 2004;95(8):773–779. | |
Lu X, Guo X, Wassall CD, Kemple MD, Unthank JL, Kassab GS. Reactive oxygen species cause endothelial dysfunction in chronic flow overload. J Appl Physiol (1985). 2011;110(2):520–527. | |
Laurindo FR, Pedro Mde A, Barbeiro HV, et al. Vascular free radical release. Ex vivo and in vivo evidence for a flow-dependent endothelial mechanism. Circ Res. 1994;74(4):700–709. | |
Pacher P, Beckman JS, Liaudet L. Nitric oxide and peroxynitrite in health and disease. Physiol Rev. 2007;87(1):315–424. | |
Vásquez-Vivar J, Kalyanaraman B, Martásek P, et al. Superoxide generation by endothelial nitric oxide synthase: the influence of cofactors. Proc Natl Acad Sci U S A. 1998;95(16):9220–9225. | |
Kondo T, Hirose M, Kageyama K. Roles of oxidative stress and redox regulation in atherosclerosis. J Atheroscler Thromb. 2009;16(5):532–538. | |
Sindler AL, Delp MD, Reyes R, Wu G, Muller-Delp JM. Effects of ageing and exercise training on eNOS uncoupling in skeletal muscle resistance arterioles. J Physiol. 2009;587(Pt 15):3885–3897. | |
Hsu JL, Hsieh Y, Tu C, O’Connor D, Nick HS, Silverman DN. Catalytic properties of human manganese superoxide dismutase. J Biol Chem. 1996;271(30):17687–17691. | |
Takaki A, Morikawa K, Tsutsui M, et al. Crucial role of nitric oxide synthases system in endothelium-dependent hyperpolarization in mice. J Exp Med. 2008;205(9):2053–2063. | |
Kuo L, Hein TW. Vasomotor regulation of coronary microcirculation by oxidative stress: role of arginase. Front Immunol. 2013;4:237. | |
Ago T, Kitazono T, Ooboshi H, et al. Nox4 as the major catalytic component of an endothelial NAD(P)H oxidase. Circulation. 2004;109(2):227–233. | |
Takac I, Schröder K, Zhang L, et al. The E-loop is involved in hydrogen peroxide formation by the NADPH oxidase Nox4. J Biol Chem. 2011;286(15):13304–13313. | |
Matoba T, Shimokawa H, Morikawa K, et al. Electron spin resonance detection of hydrogen peroxide as an endothelium-derived hyperpolarizing factor in porcine coronary microvessels. Arterioscler Thromb Vasc Biol. 2003;23(7):1224–1230. | |
Thengchaisri N, Hein TW, Wang W, et al. Upregulation of arginase by H2O2 impairs endothelium-dependent nitric oxide-mediated dilation of coronary arterioles. Arterioscler Thromb Vasc Biol. 2006;26(9):2035–2042. | |
Ingelman-Sundberg M, Johansson I. Mechanisms of hydroxyl radical formation and ethanol oxidation by ethanol-inducible and other forms of rabbit liver microsomal cytochromes P-450. J Biol Chem. 1984;259(10):6447–6458. | |
Ohsawa I, Ishikawa M, Takahashi K, et al. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat Med. 2007;13(6):688–694. | |
Ishibashi T, Sato B, Rikitake M, et al. Consumption of water containing a high concentration of molecular hydrogen reduces oxidative stress and disease activity in patients with rheumatoid arthritis: an open-label pilot study. Med Gas Res. 2012;2(1):27. | |
Ohno K, Ito M, Ichihara M, Ito M. Molecular hydrogen as an emerging therapeutic medical gas for neurodegenerative and other diseases. Oxid Med Cell Longev. 2012;2012:353152. | |
Ono H, Nishijima Y, Adachi N, et al. A basic study on molecular hydrogen (H2) inhalation in acute cerebral ischemia patients for safety check with physiological parameters and measurement of blood H2 level. Med Gas Res. 2012;2(1):21. | |
Gharib B, Hanna S, Abdallahi OM, Lepidi H, Gardette B, De Reggi M. Anti-inflammatory properties of molecular hydrogen: investigation on parasite-induced liver inflammation. C R Acad Sci III. 2001;324(8):719–724. | |
Ishibashi T. Molecular hydrogen: new antioxidant and anti-inflammatory therapy for rheumatoid arthritis and related diseases. Curr Pharm Des. 2013;19(35):6375–6381. | |
Tomiyama H, Matsumoto C, Yamada J, et al. The relationships of cardiovascular disease risk factors to flow-mediated dilatation in Japanese subjects free of cardiovascular disease. Hypertens Res. 2008;31(11):2019–2025. | |
Seo T, Kurokawa R, Sato B. A convenient method for determining the concentration of hydrogen in water: use of methylene blue with colloidal platinum. Med Gas Res. 2012;2:1. | |
Sesso HD, Buring JE, Christen WG, et al. Vitamins E and C in the prevention of cardiovascular disease in men: the Physicians’ Health Study II randomized controlled trial. JAMA. 2008;300(18):2123–2133. | |
Kris-Etherton PM, Lichtenstein AH, Howard BV, Steinberg D, Witztum JL; Nutrition Committee of the American Heart Association Council on Nutrition, Physical Activity, and Metabolism. Antioxidant vitamin supplements and cardiovascular disease. Circulation. 2004;110(5):637–641. | |
Kajiyama S, Hasegawa G, Asano M, et al. Supplementation of hydrogen-rich water improves lipid and glucose metabolism in patients with type 2 diabetes or impaired glucose tolerance. Nutr Res. 2008;28(3):137–143. | |
Nakao A, Toyoda Y, Sharma P, Evans M, Guthrie N. Effectiveness of hydrogen rich water on antioxidant status of subjects with potential metabolic syndrome-an open label pilot study. J Clin Biochem Nutr. 2010;46(2):140–149. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



