Back to Journals » Drug Design, Development and Therapy » Volume 12
Concomitant food intake does not affect the efficacy of entecavir in chronic hepatitis B patients with virological response: a randomized, multicenter, noninferiority trial
Authors Cho EJ , Yu SJ , Kwon SY , Kim JH , Kim DY , Kim W , Lee JS , Lee JW, Lee YJ , Chae HB , Yoon JH
Received 26 July 2018
Accepted for publication 9 October 2018
Published 2 November 2018 Volume 2018:12 Pages 3767—3774
DOI https://doi.org/10.2147/DDDT.S181561
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sukesh Voruganti
Eun Ju Cho,1 Su Jong Yu,1 So Young Kwon,2 Ji-Hoon Kim,3 Do Young Kim,4 Won Kim,5 June Sung Lee,6 Jin Woo Lee,7 Youn Jae Lee,8 Hee Bok Chae,9 Jung-Hwan Yoon1
1Department of Internal Medicine, Liver Research Institute, Seoul National University College of Medicine, Seoul, South Korea; 2Department of Internal Medicine, Konkuk University School of Medicine, KonKuk University Hospital, Seoul, South Korea; 3Department of Internal Medicine, Korea University Guro Hospital, Korea University College of Medicine, Seoul, South Korea; 4Department of Internal Medicine, Yonsei University College of Medicine, Seoul, South Korea; 5Department of Internal Medicine, Seoul Metropolitan Government Seoul National University Boramae Medical Center, Seoul, South Korea; 6Department of Internal Medicine, Ilsan Paik Hospital, Inje University College of Medicine, Goyang, South Korea; 7Department of Internal Medicine, Inha University School of Medicine, Incheon, Korea; 8Department of Gastroenterology, Inje University Busan Paik Hospital, Busan, South Korea; 9Department of Internal Medicine, Chungbuk National University Hospital, Chungbuk National University College of Medicine, Cheongju, South Korea
Background: Little clinical data are available about the effect of food on the antiviral efficacy of entecavir for chronic hepatitis B virus (HBV) infection. The present study evaluated whether entecavir administration in the fed state had comparable efficacy to the fasted condition for maintenance of viral suppression in HBV-infected patients with virological response on entecavir therapy.
Methods: In this multicenter, randomized, open-label, noninferiority study, patients who were currently receiving entecavir and showed a serum HBV DNA level of <20 IU/mL were randomized to take entecavir either under the fasted or fed condition for 48 weeks.
Results: We randomly assigned 50 patients to the fasted group and 46 patients to the fed group. The full analysis set consisted of 49 patients in the fasted group and 44 patients in the fed group. At week 48, the proportion of patients with HBV DNA <20 IU/mL was not significantly different between the fasted and fed groups (98% vs 100%, P=1.00). The mean log10 HBV DNA changes from baseline were similar between the two groups (-0.004 vs -0.012 log10 IU/mL, P=0.43). There were no significant differences in the proportions of patients with normal alanine aminotransferase (87.8% vs 95.5%, P=0.27) and hepatitis B e-antigen seroconversion (0% vs 6.7%, P=0.47) between the two groups. None of the patients showed viral breakthrough. In pharmacokinetic analysis, the maximum concentration and the area under the concentration–time curve to the last quantifiable concentration decreased by 26.4% and 9.3%, respectively, in the fed group compared with the fasted group. However, the differences between two groups were not statistically significant (P=0.28 and 0.83, respectively).
Conclusion: In patients with virological response under entecavir therapy, concomitant food intake did not affect the antiviral efficacy. For patients with adherence problem, taking entecavir with food may be considered to improve compliance.
Keywords: chronic hepatitis B, entecavir, food effect, efficacy
Introduction
Hepatitis B virus (HBV) represents a major public health concern. More than 250 million people are chronic HBV carriers, and HBV-related end-stage liver disease or hepatocellular carcinoma is responsible for more than 800,000 deaths annually.1,2 The goal of treatment for chronic HBV infection is to suppress viral replication and reduce histological activity before the development of cirrhosis and hepatocellular carcinoma. Oral antiviral agents are widely used for the treatment of chronic HBV infection because of their ease of administration and little adverse effects. However, HBV infection cannot be completely eradicated because of the persistence of covalently closed circular DNA;3–5 therefore, antiviral agents should be administered for long-term periods. However, prolonged use of antiviral agents can increase the risk of poor drug compliance, leading to subsequent treatment failure. Therefore, maintaining satisfactory drug compliance is important for achieving the goal of therapy.
Entecavir is an orally administered guanosine nucleoside analogue that has been approved for the treatment of chronic HBV infection. Because of its potency and high barrier to resistance, various guidelines recommend entecavir as a first-line therapeutic agent for chronic HBV infection.6–12 According to a previous pharmacokinetic study, oral administration of entecavir 0.5 mg with a standard high-fat meal resulted in a minimal delay in absorption (1–1.5 hours in the fed state vs 0.75 hours in the fasted state), a decrease in maximum concentration (Cmax) of 44%–46% and a decrease in area under the concentration–time curve (AUC) of 18%–20%.13 Because the lower Cmax and AUC in the fed condition are not considered to be clinically relevant in treatment-naïve patients, the European Medicines Agency recommends that entecavir can be administered with or without food intake in treatment-naïve patients, except for those with lamivudine-resistant mutation.14 On the contrary, the US Food and Drug Administration and the Korean guideline recommend that entecavir should be administered in a fasted condition (at least 2 hours before or after a meal) because a reduced exposure when taken with food might result in a lower efficacy.7,15 However, this issue has not been clearly evaluated in terms of virological, biochemical, and serological responses.
In this study, we aimed to compare the efficacy of entecavir between administration in the fasted and fed states in patients with chronic HBV infection. Because potential suboptimal efficacy in patients with high viremia levels may increase the risk of hepatic decompensation and liver failure, which would be unethical, and prior exposure to other antiviral agents (especially lamivudine) may increase the risk of entecavir resistance,16,17 this study included only patients who achieved virological response under entecavir therapy. We hypothesized that the efficacy of entecavir when administered in a fed state is not inferior to that when administered in a fasted state.
Methods
Study design
This study was a multicenter, randomized, open-label, noninferiority trial conducted at nine tertiary referral hospitals in South Korea from March 2015 to February 2017. Patients aged 19–75 years who had tested positive for serum hepatitis B surface antigen (HBsAg) for at least 6 months were currently taking entecavir as first-line therapy for ≥48 weeks, and who had serum HBV DNA <20 IU/mL at screening were considered eligible for this study. Patients pretreated with other antiviral agents; those with lamivudine resistance; those with evidence of decompensated liver disease or malignancy; and those with concomitant human immunodeficiency virus infection, hepatitis C or D infection, and decreased renal function (creatinine clearance <50 mL/min, as estimated using the Cockcroft–Gault formula) at the screening visit were excluded.
The study was approved by the Institutional Review Boards of each investigational site, including Seoul National University Hospital (H-1504-083-665), Konkuk University Medical Center (KUH1010675), Korea University Guro Hospital (KUGH15071-002), Seoul Metropolitan Government Seoul National University Boramae Medical Center (20150527/26-2015-67/062), Severance Hospital Yonsei University (4-2015-0486), Inje University Busan Paik Hospital (15-0098), Inha University Hospital (15-049), Chungbuk National University Hospital (CBNUH 2015-06-014-016), and Ilsan Paik Hospital (ISPAIK 2015-09-024-015). All patients provided written informed consent before screening. This study was conducted in accordance with the Declaration of Helsinki.
The patients were randomized in a 1:1 ratio to take entecavir 0.5 mg (Entecavir®; Bukwang Pharmaceutical Co., Seoul, South Korea) once daily either within 30 minutes after meals (fed group) or without meals (>2 hours before or after meals, fasting group) for 48 weeks. Randomization was centralized and stratified by hepatitis B e-antigen (HBeAg) status. Study visits were conducted every 6 weeks until treatment week 24, and at 8-week intervals thereafter. Laboratory assessments included HBeAg and anti-HBe, complete blood count, and liver and renal function tests. At weeks 0, 24, and 48, serum HBV DNA levels were measured at a central laboratory, by using the COBAS TaqMan HBV test (detection limit, 20–1.7×108 IU/mL; Hoffman-La Roche Ltd., Basel, Switzerland).
Study endpoints
The primary endpoint was the proportion of patients with a sustained virological response, defined as an HBV DNA level <20 IU/mL, at week 48.6–8 The secondary endpoints were the proportion of patients with a sustained virological response at week 24, changes in HBV DNA levels from baseline to weeks 24 and 48, the proportion of patients with loss of HBeAg and seroconversion to anti-HBe (HBeAg-positive patients only) at weeks 24 and 48, the proportion of patients with normal alanine aminotransferase (ALT) at weeks 24 and 48, and virological breakthrough (increases in HBV DNA levels ≥1 log10 IU/mL compared with the nadir on two consecutive tests or HBV DNA of ≥100 IU/mL on two consecutive tests) during the treatment period.6–8
Clinical and laboratory adverse events were assessed throughout the 48 weeks. Adherence to medication was assessed using the patients’ drug diaries, by counting the number of pills and empty packets returned at each visit, and through a pharmacokinetic study.
Pharmacokinetic analysis
Pharmacokinetic analysis was performed in three participants from each group who consented to undergo the assessment. Blood was collected for predose pharmacokinetic sampling at weeks 0, 6, 12, and 18. At week 24, sampling was conducted before dose and at 0.25, 0.5, 0.75, 1, 1.5, 2, 3, 4, 5, 6, 8, and 12 hours after dose. Plasma concentrations of entecavir were determined with a liquid chromatography/mass spectroscopy assay as previously described.18 The pharmacokinetic parameters were determined through the noncompartmental method by using Phoenix™ WinNonlin® version 8.0 (Pharsight, St Louis, MO, USA). The following pharmacokinetic parameters were calculated: Cmax, time to reach Cmax (Tmax), AUC to the last quantifiable concentration (AUC0–t), AUC from 0 to infinity (AUC0–inf), and terminal half-life (t1/2).
Statistical analyses
Assuming virological response rates of 90% for both groups and a one-sided noninferior margin of 11.88%,19,20 and considering a dropout rate of 20%, a sample size of 88 subjects (44 per group) was calculated to show that the fed group was noninferior to the fasted group, based on a one-sided 95% CI approach. A total of 96 patients were finally enrolled.
All efficacy analyses were performed in the full analysis set and per-protocol set. The full analysis set was defined as all patients who were randomly assigned and received at least one dose of the study drug and had at least one valid postbaseline efficacy evaluation, and the per-protocol set was defined as a subset of the full analysis population who completed the study without any major protocol violations. Patients with adherence rate for medication below 80% during the study period were excluded from the per-protocol set. Missing data were classified as failures in the efficacy analysis.
Categorical variables were analyzed using the chi-squared test or Fisher’s exact test, and continuous variables were analyzed using the Student’s t-test or Wilcoxon rank-sum test, as appropriate. For results of HBV DNA <20 IU/mL, the values were numerically considered 20 IU/mL. All statistical analyses were performed using SAS version 9.2 (SAS Institute Inc., Cary, NC, USA).
Results
Baseline characteristics
Of the 96 randomized patients, 50 were assigned to the fasted group and 46 to the fed group (Figure 1). Imbalance in number occurred by chance because of block randomization.21 One patient from the fasted group and two from the fed group were excluded from the full analysis set, because of lack of postbaseline efficacy assessment, withdrawal of consent before starting treatment and a violation of eligibility criteria. One patient from the fasted group was lost to follow-up and excluded from the per-protocol analysis. The remaining 92 patients completed the study without notable drug adherence violations.
  | Figure 1 Patient disposition of the whole study population. |
The treatment groups were comparable with regards to baseline characteristics (Table 1). Of 95 participants who received at least one dose of the study drug, 52 patients (54.7%) were male, and the mean age was 52.6 years. Thirty-three (34.7%) patients were HBeAg positive and 90 (94.7%) had HBV DNA level <20 IU/mL at baseline. Fifteen (15.8%) patients had compensated cirrhosis.
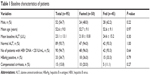  | Table 1 Baseline characteristics of patients |
Efficacy
Virological response
At week 48, there was no significant difference for the primary endpoint between the two groups. The proportion of patients with HBV DNA <20 IU/mL were 98.0% in the fasted group and 100.0% in the fed group (P=1.00, Table 2). At week 24, 98.0% of the patients in the fasted group achieved an HBV DNA <20 IU/mL compared with 95.5% in the fed group (P=0.60). The serum mean log10 HBV DNA changes from baseline were not significantly different between the fasted and fed groups at week 24 (−0.004 vs −0.002 log10 IU/mL, P=0.74) and at week 48 (−0.004 vs −0.012 log10 IU/mL, P=0.43). Up to week 48, no patient showed virological breakthrough. When the results were analyzed in a per-protocol set, similar findings were obtained, showing that entecavir had comparable efficacy between administration in the fasted and fed states (Table S1).
Biochemical and serological responses
The proportion of patients with normal ALT levels was not significantly different between the fasted and fed groups at week 24 (98.0% vs 95.5%, P=0.60) and at week 48 (87.8% vs 95.5%, P=0.27; Table 2). Comparable proportions of patients in both groups showed HBeAg seroconversion at week 48 (0% vs 6.7%, P=0.47). None of the patients achieved HBsAg loss or seroconversion during the study period. Similar findings were observed in the per-protocol analyses (Table S1).
Adverse events
Of 95 patients who took the study drug at least once during the 48-week period, 25 (50.0%) in the fasted group and 18 (40.0%) in the fed group experienced at least one adverse event, although the difference was not statistically significant (P=0.41, Table 3). Severe adverse events were reported in three cases (two in the fasted and one in the fed group); however, none was found to be related to the study drug. There was no grade 3 or 4 adverse event in both groups, and adverse events leading to study drug discontinuation or dose reduction did not occur.
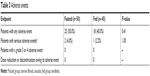  | Table 3 Adverse events |
Pharmacokinetic characteristics
The pharmacokinetic parameters of entecavir at week 24 in the fasted and fed conditions are presented in Table 4. The median Tmax of entecavir under the fed condition was delayed from 0.75 to 1.50 hours compared with that under the fasted condition, but the difference was not statistically significant (P=0.20). The median t1/2 was similar in the fasted and fed groups (8.47 vs 8.59 hours, P=1.00). There were no significant differences in the Cmax and AUC0–t between the fasted and fed groups (median Cmax, 6.30 vs 5.57, P=0.40; median AUC0–t, 1,220.02 vs 1,157.90, P=1.00). These results suggest that entecavir absorption is affected by food intake; however, the effect of food on the bioavailability of entecavir would be insignificant.
Discussion
The present study showed for the first time that entecavir administration in the fed condition had comparable efficacy to the fasted condition in patients achieving virological response during entecavir therapy, in terms of virological, biochemical, and serological responses. In addition, although food delayed the absorption of entecavir, total systemic exposure was similar in both groups in terms of the AUC. Therefore, entecavir might be administered regardless of food intake in some patients with adherence problems.
According to a previous pharmacokinetic study, food reduced Cmax and AUC of entecavir compared to the fasted condition, which led to the recommendation that entecavir should be administered without food. However, subsequent concentration–response analyses revealed that a clear response was shown at a threshold dose of 0.01 mg. The dose of entecavir 0.5 mg for nucleoside-naïve patients was selected based on superiority in viral suppression at week 22 compared with entecavir 0.1 mg.22 The concentration–response study showed a flattening of the curve in the 0.1–0.5 mg range. Furthermore, a simulation study performed in pediatric patients suggested that the exposure of entecavir would be adequate in most patients even with concomitant food intake.23 Similarly, no significant differences in AUC0–t and Cmax were seen between the fed and fasted groups in our study. Furthermore, the proportion of patients who maintained HBV DNA <20 IU/mL at week 48, which was the primary endpoint of our study, was comparable between the two groups. The serum mean log10 HBV DNA changes from baseline, the proportion of patients with normal ALT levels, and HBeAg seroconversion rates at weeks 24 and 48 were also similar between the two groups. In addition, the adverse event profiles were not significantly different between the two groups. Collectively, these findings suggested that the modest decrease in entecavir exposure in the fed condition did not alter the clinical efficacy, and entecavir could be administered without regards to food to patients with stable viral suppression under antiviral therapy.
Adherence to medication is one of the most important factors during antiviral therapy in chronic HBV infection, because it requires long-term and potentially lifelong treatment.24 Recent studies have reported that ~20%–50% of patients with chronic HBV infection showed poor adherence to antiviral therapy.25–27 Although entecavir is generally well tolerated, gastrointestinal symptoms including heartburn, nausea, dyspepsia, and upper abdominal pain are the most common adverse events during entecavir therapy in real-life practice according to a recent nationwide surveillance study.28 Because ingested food could protect the gastrointestinal mucosa, taking entecavir in a fed state might reduce the risk of gastrointestinal adverse events. Therefore, for patients having difficulty in following the prescription instructions or those with gastrointestinal side effects, taking entecavir with food might be considered to improve adherence.
This study has several limitations. First, it was an open-label study, and blinding was not performed due to the nature of intervention. This might have biased the participants in reporting the adherence and adverse events. However, we used objective endpoints and ascertained drug adherence by various methods including the pharmacokinetic study. Second, a small sample size and the noncrossover design in the pharmacokinetic study could be associated with low statistical power. Finally, only patients with virological response to entecavir at baseline were considered in this study, and the study duration was relatively short. Therefore, a study with a longer follow-up performed in a large number of treatment-naïve patients with high viremia levels might be warranted.
Conclusion
We showed that administration of entecavir in a fed state is not inferior to administration in a fasted state in patients with stable suppression of HBV by entecavir, in terms of virological, biochemical, and serological responses. For patients with adherence problems, taking entecavir with food might be considered to improve compliance.
Acknowledgments
This study was sponsored by Bukwang Pharmaceutical Co. Ltd., the manufacturer of Entecavir®. Bukwang Pharmaceutical Co. Ltd. made significant contributions to the design and analysis of the study and assisted in the preparation of research grant applications.
Disclosure
The authors report no conflicts of interest in this work.
References
World Health Organization. Global hepatitis report. 2017. Available from: http://apps.who.int/iris/bitstream/handle/10665/255016/9789241565455-eng.pdf;jsessionid=2ADD1CDC2A6DD09735FA307E671FA498?sequence=1. Accessed July 8, 2018. | ||
The Lancet Global Health. The hidden threat of hepatitis B. Lancet Glob Health. 2016;4(8):e502. | ||
Raimondo G, Allain JP, Brunetto MR, et al. Statements from the Taormina expert meeting on occult hepatitis B virus infection. J Hepatol. 2008;49(4):652–657. | ||
Bréchot C, Thiers V, Kremsdorf D, Nalpas B, Pol S, Paterlini-Bréchot P. Persistent hepatitis B virus infection in subjects without hepatitis B surface antigen: clinically significant or purely “occult”? Hepatology. 2001;34(1):194–203. | ||
Bréchot C, Hadchouel M, Scotto J, et al. State of hepatitis B virus DNA in hepatocytes of patients with hepatitis B surface antigen-positive and -negative liver diseases. Proc Natl Acad Sci U S A. 1981;78(6):3906–3910. | ||
European Association for the Study of the Liver. Electronic address: [email protected]; European Association for the Study of the L. EASL 2017 Clinical Practice Guidelines on the management of hepatitis B virus infection. J Hepatol. 2017;67(2):370–398. | ||
Korean Association for the Study of the Liver. KASL clinical practice guidelines: management of chronic hepatitis B. Clin Mol Hepatol. 2016;22(1):18–75. | ||
Terrault NA, Bzowej NH, Chang KM, et al. AASLD guidelines for treatment of chronic hepatitis B. Hepatology. 2016;63(1):261–283. | ||
Sarin SK, Kumar M, Lau GK, et al. Asian-Pacific clinical practice guidelines on the management of hepatitis B: a 2015 update. Hepatol Int. 2016;10(1):1–98. | ||
Tenney DJ, Rose RE, Baldick CJ, et al. Long-term monitoring shows hepatitis B virus resistance to entecavir in nucleoside-naive patients is rare through 5 years of therapy. Hepatology. 2009;49(5):1503–1514. | ||
Gonzalez SA, Keeffe EB. Entecavir for the long-term treatment of chronic hepatitis B. Expert Rev Anti Infect Ther. 2009;7(9):1053–1062. | ||
Osborn M. Safety and efficacy of entecavir for the treatment of chronic hepatitis B. Infect Drug Resist. 2011;4:55–64. | ||
European Medicines Agency. Baraclude scientific discussion; 2006. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Scientific_Discussion/human/000623/WC500051985.pdf. Accessed July 8, 2018. | ||
European Medicines Agency. EPAR summary for the public; 2014. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Summary_for_the_public/human/000623/WC500051804.pdf. Accessed July 8, 2018. | ||
U. S. Food and Drug Administration. FDA labelling information; 2005. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/021797s018,021798s019lbl.pdf. Accessed July 8, 2018. | ||
Lee JH, Cho Y, Lee DH, et al. Prior exposure to lamivudine increases entecavir resistance risk in chronic hepatitis B Patients without detectable lamivudine resistance. Antimicrob Agents Chemother. 2014;58(3):1730–1737. | ||
Cho EJ, Lee JH, Cho Y, et al. Comparison of the efficacy of entecavir and tenofovir in nucleos(T)ide analogue-experienced chronic hepatitis B patients. PLoS One. 2015;10(6):e0130392. | ||
Yan JH, Bifano M, Olsen S, et al. Entecavir pharmacokinetics, safety, and tolerability after multiple ascending doses in healthy subjects. J Clin Pharmacol. 2006;46(11):1250–1258. | ||
Shin SR, Yoo BC, Choi MS, et al. A comparison of 48-week treatment efficacy between clevudine and entecavir in treatment-naive patients with chronic hepatitis B. Hepatol Int. 2011;5(2):664–670. | ||
Lin Q, Zhang DZ, Zhou Z, et al. Comparison of the efficacy of 48 week – entecavir therapy with that of adefovir therapy for chronic hepatitis B patients. Zhonghua Gan Zang Bing Za Zhi. 2010;18(5):338–341. | ||
Anisimov VV. Effects of unstratified and centre-stratified randomization in multi-centre clinical trials. Pharm Stat. 2011;10(1):50–59. | ||
European Medicines Agency. CHMP variation assessment report; 2011. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Assessment_Report_-_Variation/human/000623/WC500105049.pdf. Accessed July 17, 2018. | ||
Committee for Medicinal Products for Human Use. Assessment report; 2014. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Assessment_Report_-_Variation/human/000623/WC500172948.pdf. Accessed July 17, 2018. | ||
Lee M, Keeffe EB. Study of adherence comes to the treatment of chronic hepatitis B. J Hepatol. 2011;54(1):6–8. | ||
Allard N, Dev A, Dwyer J, Srivatsa G, Thompson A, Cowie B. Factors associated with poor adherence to antiviral treatment for hepatitis B. J Viral Hepat. 2017;24(1):53–58. | ||
Xu K, Liu LM, Farazi PA, et al. Adherence and perceived barriers to oral antiviral therapy for chronic hepatitis B. Glob Health Action. 2018;11(1):1433987. | ||
van Vlerken LG, Arends P, Lieveld FI, et al. Real life adherence of chronic hepatitis B patients to entecavir treatment. Dig Liver Dis. 2015;47(7):577–583. | ||
Kim CW, Kim CS, Kim HY, et al. Large-scale surveillance study of the safety and effectiveness of entecavir in Korean patients with chronic hepatitis B. Korean J Intern Med. 2018;33(1):91–101. |
Supplementary material
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



