Back to Journals » Clinical Ophthalmology » Volume 13
Comparison of visual outcomes after bilateral implantation of two intraocular lenses with distinct diffractive optics
Authors de Medeiros AL, Jones Saraiva F, Iguma CI, Kniggendorf DV, Alves G, Chaves MAPD, Vilar C, Motta AFP, Carricondo PC , Takashi Nakano C, Nosé W, Hida WT
Received 25 January 2019
Accepted for publication 13 June 2019
Published 29 August 2019 Volume 2019:13 Pages 1657—1663
DOI https://doi.org/10.2147/OPTH.S202895
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Video abstract presented by Andre Lins de Medeiros.
Views: 1103
André Lins de Medeiros,1 Flavio Jones Saraiva,2 Camila Ishii Iguma,2 Danilo Varela Kniggendorf,1 Guilherme Alves,2 Mario Augusto Pereira Dias Chaves,1 Cesar Vilar,1 Antonio Francisco Pimenta Motta,1 Pedro Carlos Carricondo,1 Celso Takashi Nakano,1 Walton Nosé,1 Wilson Takashi Hida1,2
1Cataract Sector, Renato Ambrosio Eye Research Center, Brasília, DF, Brazil; 2Cataract Sector, Hospital Oftalmológico De Brasília, Brasília, DF, Brazil
Correspondence: André Lins de Medeiros
Renato Ambrosio Eye Research Center, SGAS 607, Hospital Oftalmológico de Brasilia, Brasília 70200-670, Brazil
Tel +55 5 804 5530
Email [email protected]
Purpose: The aim of this study was to compare the visual outcomes and subjective visual quality between bilateral implantation of an extended depth of focus intraocular lens, J&J Vision Tecnis Symfony® ZXR00 (Group A) and bilateral implantation of a diffractive trifocal intraocular lens, Alcon Acrysof IQ PanOptix® TNFT00 (Group B).
Methods: This prospective, nonrandomized, comparative study of consecutive cases assessed 52 eyes of 26 patients operated on by the same surgeon (WTH) and binocularly implanted with multifocal intraocular lenses between May 2016 and July 2018. Binocular visual acuity for far, intermediate and near was tested in all cases. Ophthalmological evaluation included the measurement of binocular uncorrected distance visual acuity (UDVA), corrected distance visual acuity (CDVA), uncorrected near visual acuity (UNVA) at 40 cm, uncorrected intermediate visual acuity (UIVA) at 70 cm, monocular visual defocus curve and the quality of life (QoL) questionnaire, National Eye Institute Visual Functioning Questionnaire - 25 ( NEI-VFQ 25).
Results: Postoperative UDVA was 0.00 and 0.09 logMAR (P<0.001), UIVA was 0.20 and 0.39 logMAR (P<0.001) and UNVA was 0.16 and −0.01 logMAR (P<0.001) in groups A and B, respectively; postoperative CDVA was −0.05 and 0.06 logMAR (P<0.001) in groups A and B, respectively.
Conclusion: Both groups reported good subjective quality of vision regarding long, intermediate and short distances. Group A had a better performance for binocular UDVA, UIVA at 70 cm and CDVA, while regarding the monocular defocus curve, Group A outperformed Group B for long distances. Furthermore, Group B surpassed it in the short to very short distances, between the range of ≥2.00 D to 5.00 D of vergence. While Group A had a better performance regarding the vergences between 0.00 and 1.00 D (P<0.05) and at the vergence of +2.50 D (P=0.007). Group B outran Group A for UNVA at 40 cm.
Keywords: cataract, surgery, phacoemulsification, optics, chromatic aberration, visual performance
Introduction
Phacoemulsification, a procedure that consists in splitting the crystalline lens into smaller fragments with ultrasound energy, is currently the most commonly performed surgery in humans for its reproducibility and satisfactory postoperative outcomes achieved through a micro-coaxial incision and the implantation of an intraocular lens that will impersonate the natural crystalline lens power; this practice has been incorporated by ophthalmic surgeons since Charles Kelman described it in 1967.1,2
Monofocal spherical intraocular lenses are the most implanted on account of its more affordable cost; however, it has a single focal point and, as a result, provides good visual outcome for long and short distance vision depending on the biometric-calculated target. Utterly, the vast majority of those patients need spectacles to correct the loss of ability to see either intermediate and short or long distances. The continuous improvement of phacoemulsification fluidics and machines, handpiece tips, the addition of femtosecond laser to cataract procedures and its continuous evolution and the use of optimized constants for optical biometry have provided surgeons with tools to enhance their capabilities and to be granted with postoperative emmetropia.1–9
Multifocal intraocular lenses were designed since the late 1980s but were well accepted since the last decade to provide adequate vision for far and near. Those first universally accepted intraocular lenses had a higher addition and were bifocal. Moreover, since the introduction of trifocal intraocular lenses in this decade, surgeons can also address intermediate vision likewise.10–12 Nevertheless, despite the continuous evolution of those implants, patients might present visual disphotopsia which may be a cause of discomfort, such as halos, glare, starburst, reduced contrast sensitivity and unsatisfactory uncorrected distance visual acuity (UDVA); accordingly, accurate patient election must be taken to accomplish acceptable postoperative outcomes.13–17
The Acrysof IQ PanOptix® IOL (Alcon Laboratories, Inc., Fort Worth, TX, USA) has a unique quadrafocal IOL design; however, in terms of function, it acts as a trifocal intraocular lens. It is a single-piece hydrophobic acrylic IOL and has a diffractive kinoform profile. It comprises three step heights, creating a +2.17 D for intermediate vision, a +3.25 for near vision, and another larger step in terms of width that generates +1.085 D, which is the conjunction of 2 steps of +2.17 D with one of +3.25 D in the middle, configuring the third step. This step harness light diffracted to supply the far vision as this focus is approximately at the vergence of 110 cm.11,15,16,18–21 This technology is called Enlighten® Optical Technology by the manufacturer (Alcon Laboratories, Inc.) and there has been a great acceptance and patient satisfaction with this intraocular lens as the intermediate vision is very comfortable and sharp.11,15,16,19–30 The diffractive zone is also in the central portion and occupies 4.5 mm of the optical zone. It adds a negative spherical aberration of −0.1 μm on the anterior face of the lens to compensate the positive spherical aberration generated by the average human cornea.18
The most used EDOF IOL currently is the Tecnis Symfony® (J&J Vision, Inc., Santa Ana, CA, USA)), which is also a single-piece, hydrophobic acrylate-folding IOL with a new design that promotes an extended range of focus. It also has a posterior diffractive surface (kinoform) and an anterior aspherical surface adding −0.27 μm to the cornea aberration. The EDOF concept generated by this IOL can be explained by the splitting of light energy into an elongated focus which could reduce the overlapping of near and far images caused by the traditional diffractive multifocal intraocular lenses, generating less visual disturbances. It also uses a proprietary achromatic diffractive echelette design to correct chromatic aberration, also enhancing CS and has exhibited satisfactory results for near, intermediate and far vision in diverse previous studies.15,20,21,28,30–50
Methods
This study was conducted in accordance with good clinical practices and the 1964 Declaration of Helsinki and its amendments or comparable ethical standards respecting clinical ethical standards of the institutional and national research committee and was approved by the Institutional Review Board of the Medical Staff of Hospital Oftalmológico de Brasília, Brazil.51,52 Patients signed a written informed consent in two copies.
This was a prospective, nonrandomized, comparative study of consecutive cases. All patients underwent an uneventful phacoemulsification with IOL implantation by the same surgeon (WTH) between May 2016 and July 2018. Informed consent was obtained from patients prior to data collection, when the procedures that would be performed during the study were explained to the patients. Analysis and comparison of visual outcomes were performed between the extended depth of focus (EDOF) and trifocal groups, Groups A and B, respectively. Ocular dominance was determined by the Dolman method (hole in the card). Exclusion criteria were analphabetism, presence of any corneal, retina or optic nerve disease, previous refractive surgery, high axial myopia, expected postoperative corneal astigmatism >1.00 D and intraoperative or postoperative complications. Inclusion criteria were phacoemulsification with implantation of either lenses comprised by this study, age above 50 years old and capacity to understand and cooperate with the examination.
The study consisted of a complete eye exam preoperatively and a postoperative visit ranging from 180 to 360 days after surgery for both groups. All patients underwent complete ophthalmological examination, including biomicroscopy, mesopic pupillometry assessed by the OPD-Scan III (Nidek, Gamagori, Japan), tonometry, retinoscopy, fundoscopy, near, intermediate and far visual acuity and defocus curve.
The IOL power was chosen preoperatively based on optical biometry provided by the IOL Master 700 (Carl Zeiss AG, Oberkochen, Germany); the lenses were calculated based on the Barrett Universal II formula; the first negative results and the first positive results were targeted for Groups A and B, respectively.1,53 Uncorrected near visual acuity (UNVA) at 40 cm, uncorrected intermediate visual acuity (UIVA) at 70 cm, distance at 4 m (UDVA) and corrected distance visual acuity (CDVA) at 4 m binocular visual acuity were measured using the reading table model of the Early Treatment Diabetic Retinopathy Study charts (ETDRS; Precision Vision, Woodstock, IL, USA). Preoperative visual acuity data were collected from electronic chart records. Visual monocular defocus curves were obtained in long-distance visual acuity condition, corrected using the same ETDRS charts at a distance of 4 m, at intervals of 0.50 spherical diopters from −5.00 to +2.50 D, with the measurement of luminance with Gossen starlite 2 (Gossen, Nürnberg, Germany); photopic conditions were defined as ~85 cd/m2 and ambient luminance was defined as inferior to 21.25 lux. Data collected were available only to the authors of the study and the examination charts were stored in a safe, as a safety procedure to guarantee the anonymity of the subjects. The Hospital Oftalmologico de Brasília funded all the expenses regarding this paper. Significance was tested using the Tukey, Wilcoxon, Kruskal–Wallis, analysis of variance and chi-square tests by adjusting to a level of significance of 5% (P<0.05) and using software R version 3.3.2 (Foundation for Statistical Computing, Vienna, Austria).
Results
This study comprised 52 eyes of 26 patients, 14 women (53.85%) and 12 men (46.15%). There was homogeneity in the group distribution of lenses regarding age, gender, preoperative CDVA. Postoperative UDVA was better in Group A (0.00±0.05 vs 0.09±0.10 logMAR, P<0.001). CDVA was better in the EDOF group (−0.02±0.05 vs 0.06±0.12, P=0.001). UIVA and UNVA had statistical significance as well: 0.20±0.04 vs 0.39±0.09, P<0.001 and 0.16±0.08 vs −0.01±0.09, P<0.001, respectively. The postoperative data of spherical equivalent (SE) comparison also had statistical significance; nevertheless, axial length (AL) and pupil diameter (PD) had no statistical significance (Table 1).
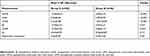 |
Table 1 Descriptive measures for postoperative spherical equivalent, postoperative visual acuities and mesopic pupillometry in Groups A and B |
Regarding the defocus curve (Figure 1), there was statistical significance in the vast majority of vergences assessed between the intraocular lenses. The EDOF group exhibited a plateau of outperformance over the trifocal group ranging from 0.00 (infinite) to −1.0 D (corresponding to 1 m), with an average visual acuity of −0.02 logMAR at 0 diopters (D) or 4 m, −0.04 logMAR at −0.50 D or 2 m and −0.01 logMAR at −1 D or 1 m, respectively. At the vergence of 0.00 D, the average visual acuity in the trifocal group was 0.06 logMAR (P<0.001); 0.02 logMAR at −0.50 D (P=0.02) and 0.11 logMAR at −1 D (P=0.006).
 |
Figure 1 Monocular defocus curve of Groups A and B with distance correction.Notes: Group A: blue - EDOF group. Group B: red - Trifocal Group. *Statistical significance. |
The line corresponding to the trifocal group established a plateau, ranging from −2.0 D to −5.0 D of vergence with a correspondence of vergence from 50 cm to 20 cm, with visual acuities of −0.01 vs 0.08 logMAR (P=0.004) at −2 D or 50 cm; −0.01 vs 0.16 logMAR at −2.50 D or 40 cm (P<0.001); 0.08 vs 0.25 logMAR at −3 D or 33 cm (P<0.001); 0.24 vs 0.41 logMAR at −4 D or 25 cm (P=0.001); 0.36 vs 0.50 logMAR at −4.5 D or 22.2 cm (P=0.005) and 0.47 vs 0.63 logMAR (P=0.002). The EDOF group had a near distance deflection vision regarding the defocus curve. The trifocal group had a deflection of long distance vision at 0.00 D; −0.50D and −1.0 D when compared to the EDOF group that maintained a plateau for these vergences. No intraocular lens was superior regarding myopic tolerance evaluated at the range of +0.50 D to +2.00 D. The EDOF group outperformed the trifocal group regarding myopic tolerance at the vergence of +2.50 D: 0.44 vs 0.54 logMAR (P=0.007), respectively.
Both groups had satisfactory results on the NEI-VFQ 25 questionnaire, with no statistical significance.
Discussion
The demographic analysis distribution of the sample demonstrates homogeneity and enables comparisons between groups, indicating its suitability in comparison with other publications.24,25 Both groups provided acceptable visual outcomes after cataract surgery with good for short, intermediate and far distances, in accordance with previous studies.24,25
Previous studies demonstrated that the EDOF IOL had spectacle independence with functional vision to far and intermediate with some restraint regarding near vision; this paper corroborates on that path.31,32,43 To assess that matter, a subanalysis of 411 patients from the Concerto study, regarding the influence of different levels of monovision on the clinical outcomes achieved with the EDOF intraocular lens, was conducted by Cochener; it was concluded that the magnitude of induced myopia for optimization of the visual outcome with patient satisfaction and spectacle independence was with a micro-monovision of around −0.75D with less rates of photic phenomena when compared with multifocal intraocular lenses.13,39,54–56
The trifocal lenses have surpassed several problems associated with traditional bifocal lenses as they reckon a third focal point that improves intermediate vision and maintain a satisfactory performance at distance and near distances. The EDOF is a newer class of IOLs which targets an improvement of the intermediate vision without compromising distance vision.
As predicted, clinical findings showed that intermediate and far vision was achieved in both groups, with a better performance of the trifocal intraocular lens at shorter distances regarding the defocus curve, which is in accordance with other previous studies with EDOF and trifocal intraocular lenses. The binocular UNVA suffered a deflection regarding the EDOF lens which was surpassed by the trifocal group at 40 cm, as expected. The performance regarding UIVA and UNVA achieved with the trifocal lens is in agreement with previous papers. Surprisingly, the binocular UIVA at 70 cm achieved with the EDOF intraocular lens outperformed the trifocal group in this study.11,23,25,28,31,40
Cochener has performed the same comparison of this current study and concluded that Tecnis Symfony® group had the fewest patients contemplating enhancement with excimer laser, despite a certain level of micro-monovision purposively targeted. This means a greater tolerance of refractive error with this EDOF compared to the diffractive multifocal IOLs, which are more sensitive to achieving emmetropia.25
Alió et al analyzed visual acuity and visual acuity in 52 eyes of 26 patients implanted with the PanOptix® intraocular lens exhibiting good visual performance for all measured distances. A slight deflect in the defocus curve after the vergence of −0.50 D was evidencedmaintaining a plateau until the vergence of −3.00 D as was also demonstrated in this study, although with a more acute deflection at the vergences of −0.5 D and −1.0 D followed by a readjustment with a better visual acuity, reaching a plateau of acceptable vision ranging from −1.5 to −3.0 D.23
In the present study, fewer than 1% of each group referred to positive dysphotopsias, such as nocturnal halos, glare or starburst. Patients affirmed that visual disturbances had little or no impact on their daily functioning. No patient considered excimer laser enhancement in either group.
The study has various limitations: intermediate vision was only tested at 70 cm. Acrysof IQ PanOptix® has an enhanced focal range from 60 to 40 cm. Furthermore, the EDOF lens was targeted for micro-monovision or emmetropia (first negative result), which may have confounded the near results. Future studies should approach those matters. It is necessary to have further studies with longer follow-up times to address the occurrence of posterior capsule opacification and the stability of outcomes.
Overall, both groups endorsed the good postoperative subjective quality of vision regarding long, intermediate and short distances after implantation of these new diffractive intraocular lenses. Group A promoted a better performance for binocular UDVA, UIVA at 70 cm and CDVA, in the monocular defocus curve; Group A outperformed Group B for long distances; nevertheless, Group B surpassed in the short to very short distances, between the range of ≥−2.00 to −5.00 D of vergence. While Group B outran Group A regarding the vergences between 0.00 and −1.00 D (P<0.05) and at the vergence of +2.50 D (P=0.007) and UNVA at 40 cm.
Acknowledgments
This study was funded by Hospital Oftalmológico de Brasília (HOB) and Renato Ambrosio Eye Research Center (CEORA). The facilities of both institutions were used during the conduction of this study. It was approved by the Institutional Review Board of the Hospital Oftalmológico de Brasília, Brasília, DF, Brazil.
Disclosure
The authorshave no conflicts of interest to disclose.
References
1. Shajari M, Kolb CM, Petermann K, et al. Comparison of 9 modern intraocular lens power calculation formulas for a quadrifocal intraocular lens. J Cataract Refract Surg. 2018;44(8):942–948. doi:10.1016/j.jcrs.2018.05.021
2. Shajari M, Rusev V, Mayer W, Diakonis V, Petermann K, Kohnen T. Impact of lens density and lens thickness on cumulative dissipated energy in femtosecond laser-assisted cataract surgery. Lasers Med Sci. 2019. doi:10.1007/s10103-019-02715-6
3. Nishi T, Taketani F, Ueda T, Ogata N. Comparisons of amplitude of pseudoaccommodation with aspheric yellow, spheric yellow, and spheric clear monofocal intraocular lenses. Clin Ophthalmol Auckl NZ. 2013;7:2159–2164. doi:10.2147/OPTH.S52771
4. Liang J-L, Tian F, Zhang H, Teng H. Combination of Toric and multifocal intraocular lens implantation in bilateral cataract patients with unilateral astigmatism. Int J Ophthalmol. 2016;9(12):1766–1771.
5. Bartol-Puyal F, de Talavero AP, Giménez G, et al. Reading and quality of life differences between Tecnis ZCB00 monofocal and Tecnis ZMB00 multifocal intraocular lenses. Eur J Ophthalmol. 2017;27(4):443-453. doi:10.5301/ejo.5000925
6. Gundersen KG, Potvin R. Comparative visual performance with monofocal and multifocal intraocular lenses. Clin Ophthalmol Auckl NZ. 2013;7:1979–1985.
7. Yamauchi T, Tabuchi H, Takase K, Ohsugi H, Ohara Z, Kiuchi Y. Comparison of visual performance of multifocal intraocular lenses with same material monofocal intraocular lenses. PLoS One. 2013;8(6):e68236. doi:10.1371/journal.pone.0068236
8. Ye -P-P, Li X, Yao K. Visual outcome and optical quality after bilateral implantation of aspheric diffractive multifocal, aspheric monofocal and spherical monofocal intraocular lenses: a prospective comparison. Int J Ophthalmol. 2013;6(3):300–306.
9. Hida WT, de Medeiros AL, de Araújo Rolim AG, et al. Prospective randomized comparative study between venturi and peristaltic pumps in WhiteStar Signature® phacoemulsification machine. Clin Ophthalmol Auckl NZ. 2019;13:49–52. doi:10.2147/OPTH.S177978
10. Gatinel D, Pagnoulle C, Houbrechts Y, Gobin L. Design and qualification of a diffractive trifocal optical profile for intraocular lenses. J Cataract Refract Surg. 2011;37(11):2060–2067. doi:10.1016/j.jcrs.2011.05.047
11. Kohnen T, Herzog M, Hemkeppler E, et al. Visual performance of a quadrifocal (trifocal) intraocular lens following removal of the crystalline lens. Am J Ophthalmol. 2017. doi:10.1016/j.ajo.2017.09.016
12. Davison JA, Simpson MJ. History and development of the apodized diffractive intraocular lens. J Cataract Refract Surg. 2006;32(5):849–858. doi:10.1016/j.jcrs.2006.02.006
13. De Vries NE, Webers CAB, Touwslager WRH, et al. Dissatisfaction after implantation of multifocal intraocular lenses. J Cataract Refract Surg. 2011;37(5):859–865. doi:10.1016/j.jcrs.2010.11.032
14. Gundersen KG, Potvin R. Comparison of visual outcomes and subjective visual quality after bilateral implantation of a diffractive trifocal intraocular lens and blended implantation of apodized diffractive bifocal intraocular lenses. Clin Ophthalmol Auckl NZ. 2016;10:805–811.
15. De Medeiros AL, de Araújo Rolim AG, Motta AFP, et al. Comparison of visual outcomes after bilateral implantation of a diffractive trifocal intraocular lens and blended implantation of an extended depth of focus intraocular lens with a diffractive bifocal intraocular lens. Clin Ophthalmol Auckl NZ. 2017;11:1911–1916. doi:10.2147/OPTH.S145945
16. Vilar C, Hida WT, de Medeiros AL, et al. Comparison between bilateral implantation of a trifocal intraocular lens and blended implantation of two bifocal intraocular lenses. Clin Ophthalmol Auckl NZ. 2017;11:1393–1397. doi:10.2147/OPTH.S139909
17. Chaves MAPD, Hida WT, Tzeliks PF, et al. Comparative study on optical performance and visual outcomes between two diffractive multifocal lenses: AMO Tecnis ® ZMB00 and AcrySof ® IQ ReSTOR® Multifocal IOL SN6AD1. Arq Bras Oftalmol. 2016;79(3):171–176. doi:10.5935/0004-2749.20160050
18. Carson D, Xu Z, Alexander E, Choi M, Zhao Z, Hong X. Optical bench performance of 3 trifocal intraocular lenses. J Cataract Refract Surg. 2016;42(9):1361–1367. doi:10.1016/j.jcrs.2016.06.036
19. Gundersen KG, Potvin R. Trifocal intraocular lenses: a comparison of the visual performance and quality of vision provided by two different lens designs. Clin Ophthalmol Auckl NZ. 2017;11:1081–1087. doi:10.2147/OPTH.S136164
20. Monaco G, Gari M, Di Censo F, Poscia A, Ruggi G, Scialdone A. Visual performance after bilateral implantation of 2 new presbyopia-correcting intraocular lenses: trifocal versus extended range of vision. J Cataract Refract Surg. 2017;43(6):737–747. doi:10.1016/j.jcrs.2017.03.037
21. Gatinel D, Loicq J. Clinically relevant optical properties of bifocal, trifocal, and extended depth of focus intraocular lenses. J Refract Surg Thorofare NJ 1995. 2016;32(4):273–280.
22. Akman A, Asena L, Ozturk C, Gür Güngör S. Evaluation of quality of life after implantation of a new trifocal intraocular lens. J Cataract Refract Surg. 2019. doi:10.1016/j.jcrs.2018.12.003
23. Alió JL, Plaza-Puche AB, Alió Del Barrio JL, et al. Clinical outcomes with a diffractive trifocal intraocular lens. Eur J Ophthalmol. 2018;28(4):419–424. doi:10.1177/1120672118762231
24. Böhm M, Hemkeppler E, Herzog M, et al. Comparison of a panfocal and trifocal diffractive intraocular lens after femtosecond laser-assisted lens surgery. J Cataract Refract Surg. 2018;44(12):1454–1462. doi:10.1016/j.jcrs.2018.07.060
25. Cochener B, Boutillier G, Lamard M, Auberger-Zagnoli C. A comparative evaluation of a new generation of diffractive trifocal and extended depth of focus intraocular lenses. J Refract Surg Thorofare NJ 1995. 2018;34(8):507–514.
26. Kretz FTA, Müller M, Gerl M, Gerl RH, Auffarth GU. Binocular function to increase visual outcome in patients implanted with a diffractive trifocal intraocular lens. BMC Ophthalmol. 2015;21(15):110. doi:10.1186/s12886-015-0089-9
27. Madrid-Costa D, Ruiz-Alcocer J, Ferrer-Blasco T, García-Lázaro S, Montés-Micó R. Optical quality differences between three multifocal intraocular lenses: bifocal low add, bifocal moderate add, and trifocal. J Refract Surg Thorofare NJ 1995. 2013;29(11):749–754.
28. Mencucci R, Favuzza E, Caporossi O, Savastano A, Rizzo S. Comparative analysis of visual outcomes, reading skills, contrast sensitivity, and patient satisfaction with two models of trifocal diffractive intraocular lenses and an extended range of vision intraocular lens. Graefes Arch Clin Exp Ophthalmol. 2018;256(10):1913–1922. doi:10.1007/s00417-018-4052-3
29. Mojzis P, Majerova K, Hrckova L, Piñero DP. Implantation of a diffractive trifocal intraocular lens: one-year follow-up. J Cataract Refract Surg. 2015;41(8):1623–1630. doi:10.1016/j.jcrs.2014.11.050
30. Ruiz-Mesa R, Abengózar-Vela A, Aramburu A, Ruiz-Santos M. Comparison of visual outcomes after bilateral implantation of extended range of vision and trifocal intraocular lenses. Eur J Ophthalmol. 2017;27(4):460–465. doi:10.5301/ejo.5000935
31. Cochener B; Concerto Study Group. Clinical outcomes of a new extended range of vision intraocular lens: international multicenter concerto study. J Cataract Refract Surg. 2016;42(9):1268–1275. doi:10.1016/j.jcrs.2016.06.033
32. Pedrotti E, Bruni E, Bonacci E, Badalamenti R, Mastropasqua R, Marchini G. Comparative analysis of the clinical outcomes with a monofocal and an extended range of vision intraocular lens. J Refract Surg Thorofare NJ 1995. 2016;32(7):436–442.
33. Black S. Successful restoration of visual acuity with an extended range of vision intraocular lens after multifocal laser ablation. Case Rep Ophthalmol. 2016;7(3):193–197. doi:10.1159/000450675
34. Black S. A clinical assessment of visual performance of combining the TECNIS® symfony extended range of vision IOL (ZXR00) with the +3.25 D TECNIS Multifocal 1-piece IOL (ZLB00) in subjects undergoing bilateral cataract extraction. Clin Ophthalmol Auckl NZ. 2018;12:2129–2136. doi:10.2147/OPTH.S175901
35. Attia MSA, Khoramnia R, Auffarth GU, Kirchner M, Holzer MP. Near and intermediate visual and reading performance of patients with a multifocal apodized diffractive intraocular lens using an electronic reading desk. J Cataract Refract Surg. 2016;42(4):582–590. doi:10.1016/j.jcrs.2015.11.047
36. Schallhorn SC, Teenan D, Venter JA, et al. Monovision LASIK versus presbyopia-correcting iols: comparison of clinical and patient-reported outcomes. J Refract Surg Thorofare NJ 1995. 2017;33(11):749–758.
37. Millán MS, Vega F. Extended depth of focus intraocular lens: chromatic performance. Biomed Opt Express. 2017;8(9):4294–4309. doi:10.1364/BOE.8.004294
38. Ruiz-Mesa R, Abengózar-Vela A, Ruiz-Santos M. A comparative study of the visual outcomes between a new trifocal and an extended depth of focus intraocular lens. Eur J Ophthalmol. 2017;8;0. doi:10.1016/0006-2952(75)90029-5
39. Gil MA, Varón C, Cardona G, Vega F, Buil JA. Comparison of far and near contrast sensitivity in patients symmetrically implanted with multifocal and monofocal IOLs. Eur J Ophthalmol. 2014;24(1):44–52. doi:10.5301/ejo.5000335
40. Akella SS, Juthani VV. Extended depth of focus intraocular lenses for presbyopia. Curr Opin Ophthalmol. 2018;29(4):318–322. doi:10.1097/ICU.0000000000000490
41. Cochener B. Influence of the level of monovision on visual outcome with an extended range of vision intraocular lens. Clin Ophthalmol Auckl NZ. 2018;12:2305–2312. doi:10.2147/OPTH.S184712
42. Ganesh S, Brar S, Pawar A, Relekar KJ. Visual and refractive outcomes following bilateral implantation of extended range of vision intraocular lens with micromonovision. J Ophthalmol. 2018;2018:7321794. doi:10.1155/2018/7321794
43. Kaymak H, Höhn F, Breyer DRH, et al. [Functional results 3 months after implantation of an “Extended range of vision” intraocular lens]. Klin Monbl Augenheilkd. 2016;233(8):923–927.
44. Palomino-Bautista C, Carmona-González D, Sánchez-Jean R, et al. Refractive predictability and visual outcomes of an extended range of vision intraocular lens in eyes with previous myopic laser in situ keratomileusis. Eur J Ophthalmol. 2018. 1120672118804950. doi:10.1177/1120672118804950
45. Pandit RT. Monocular clinical outcomes and range of near vision following cataract surgery with implantation of an extended depth of focus intraocular lens. J Ophthalmol. 2018;2018:8205824.
46. Pedrotti E, Carones F, Aiello F, et al. Comparative analysis of visual outcomes with 4 intraocular lenses: monofocal, multifocal, and extended range of vision. J Cataract Refract Surg. 2018;44(2):156–167. doi:10.1016/j.jcrs.2017.11.011
47. Pilger D, Homburg D, Brockmann T, Torun N, Bertelmann E, von Sonnleithner C. Clinical outcome and higher order aberrations after bilateral implantation of an extended depth of focus intraocular lens. Eur J Ophthalmol. 2018;28(4):425–432. doi:10.1177/1120672118766809
48. Power B, Murphy R, Leccisotti A, Moore T, Power W, O’Brien P. Maximising refractive outcomes with an extended depth of focus IOL. Open Ophthalmol J. 2018;12:273–280. doi:10.2174/1874364101812010273
49. Sachdev GS, Ramamurthy S, Sharma U, Dandapani R. Visual outcomes of patients bilaterally implanted with the extended range of vision intraocular lens: a prospective study. Indian J Ophthalmol. 2018;66(3):407–410. doi:10.4103/ijo.IJO_286_18
50. Weeber HA, Meijer ST, Piers PA. Extending the range of vision using diffractive intraocular lens technology. J Cataract Refract Surg. 2015;41(12):2746–2754. doi:10.1016/j.jcrs.2015.07.034
51. Wiggins RE, Etz R. Assessment of the American Board of Ophthalmology’s maintenance of certification part 4 (Improvement in Medical Practice). JAMA Ophthalmol. 2016;134(9):967–974. doi:10.1001/jamaophthalmol.2016.1848
52. Rickham PP. Human experimentation. Code of ethics of the World Medical Association. Declaration of Helsinki. Br Med J. 1964;2(5402):177. doi:10.1136/bmj.2.5402.177
53. Moshirfar M, Buckner B, Ronquillo YC, Hofstedt D. Biometry in cataract surgery: a review of the current literature. Curr Opin Ophthalmol. 2019;30(1):9–12.
54. Law EM, Aggarwal RK, Kasaby H. Clinical outcomes with a new trifocal intraocular lens. Eur J Ophthalmol. 2014;24(4):501–508. doi:10.5301/ejo.5000407
55. Mendicute J, Kapp A, Lévy P, et al. Evaluation of visual outcomes and patient satisfaction after implantation of a diffractive trifocal intraocular lens. J Cataract Refract Surg. 2016;42(2):203–210. doi:10.1016/j.jcrs.2015.11.037
56. Frieling-Reuss EH. Comparative analysis of the visual and refractive outcomes of an aspheric diffractive intraocular lens with and without toricity. J Cataract Refract Surg. 2013;39(10):1485–1493. doi:10.1016/j.jcrs.2013.04.034
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
