Back to Journals » Cancer Management and Research » Volume 10
Comparison of the effects of stellate ganglion block and paroxetine on hot flashes and sleep disturbance in breast cancer survivors
Authors Rahimzadeh P, Imani F, Nafissi N , Ebrahimi B, Faiz SHR
Received 8 May 2018
Accepted for publication 27 July 2018
Published 26 October 2018 Volume 2018:10 Pages 4831—4837
DOI https://doi.org/10.2147/CMAR.S173511
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Harikrishna Nakshatri
Poupak Rahimzadeh,1 Farnad Imani,1 Nahid Nafissi,2 Behzad Ebrahimi,3 Seyed Hamid Reza Faiz1
1Pain Research Center, Iran University of Medical Sciences, Tehran, Iran; 2Department of Surgery, Faculty of Medicine, Iran University of Medical Sciences, Tehran, Iran; 3Department of Anesthesiology, Iran University of Medical Sciences, Tehran, Iran
Background: The incidence of menopausal symptoms, including hot flashes and sleep disturbance, caused by drug treatment is a common problem in breast cancer survivors. Considering the limitations of hormone therapy in such patients, several studies have been conducted to find alternative methods. The aim of this study was to investigate and compare the effectiveness of stellate ganglion block (SGB) with that of paroxetine, which was approved by the US Food and Drug Administration (FDA) as a medicine for the treatment of hot flashes and ensuing sleep disturbance.
Patients and methods: A total of 40 patients survived from breast cancer and complaining of these symptoms were equally assigned to two groups of 20 each. In the study group, SGB was performed successfully under sonography guidance using 10 mL of 0.5% bupivacaine, and in the control group (paroxetine), the daily administration of 7.5 mg of paroxetine was conducted for 6 weeks. The frequency and severity of hot flash attacks and sleep quality of patients were evaluated prior to the intervention and after 2, 4 and 6 weeks. The incidence of adverse events during treatment or follow-up was recorded.
Results: A significant decrease in hot flash score and sleep disturbance index (SDI) was observed in both groups. Comparison of the results showed no noticeable difference between the two groups. Two participants in the control group had discontinued medication due to gastrointestinal symptoms, and only one case of mild headache was reported in the study group.
Conclusion: SGB is as much effective as paroxetine in controlling hot flashes and sleep disturbances in breast cancer survivors and is associated with few complications.
Keywords: paroxetine, breast cancer, hot flashes, stellate ganglion block, ultrasound guidance
Introduction
Subsequent to chemotherapy, ovarian suppression and the sudden cessation of hormone therapy, breast cancer survivors often suffer from acute menopausal symptoms such as hot flash attacks that interfere with their daily functioning, quality of life and sleep. Antiestrogens can further aggravate these negative effects, as more than half of the patients taking tamoxifen complained of hot flashes, night sweats and disturbed sleep quality.1 Hot flashes occur in nearly 90% of perimenopausal women, and are more common, severe, longer and problematic especially in breast cancer patients for the abovementioned reasons.2 Ban on the use of hormone replacement therapy (HRT) has led to the administration of various drugs including clonidine, gabapentin, selective serotonin reuptake inhibitors and selective selective norepinephrine reuptake inhibitors so as to alleviate vasomotor symptoms (VMSs) in perimenopausal women. The adverse effects of these treatments have frequently outweighed their advantages and led to the discontinuation of treatment in many patients.3–6 Paroxetine mesylate is the only drug approved by the US Food and Drug Administration (FDA) for the treatment of moderate-to-severe VMSs and for improved sleep quality.7,8 A review of some studies into the effectiveness of paroxetine has shown a significant reduction in the frequency and severity of hot flashes compared to placebo; however, its use was associated with mild-to-moderate symptoms, which were better tolerated following the consumption of low doses (7.5–12.5 mg).9 Stellate ganglion block (SGB) is a well-established and valuable procedure widely used to diagnose and manage the sympathetically mediated pain of head, neck and upper extremities, and its success and safety will increase if the procedure is performed under ultrasound guidance.10,11 In recent years, the procedure has been used in some studies to manage hot flash attacks. Most of these studies have been conducted as a pilot trial and a review of case reports, and randomized clinical trials performed to date are generally limited in number.12–16 The aim of this study was to evaluate the effectiveness of SGB by comparing it with that of paroxetine, which is an approved medicine for the treatment of hot flashes and one of their consequences – night sleep disturbance.
Patients and methods
After obtaining approval from the ethics committee of the Iran University of Medical Sciences with reference number of IR.IUMS.REC.1395.28823 and when the trial was registered online on “Iranian Registry of Clinical Trials” website with registry code of Irct2016100710599N13, sampling was performed among breast cancer survivors, with complaints of hot flashes and the ensuing consequences thereof, including sleep disturbance, who had visited women’s or oncology clinics in Rasoul-e-Akram Teaching Hospital and then had been referred to pain management services. Given confidence coefficient of 0.05 and statistical power of 90%, the sample size was calculated (20 in each group and totally 40).
The inclusion criteria for this study consisted of complaints about symptoms experienced for at least a month, aged 21–45 years and life expectancy ≥6 months. The use of tamoxifen, raloxifene or aromatase inhibitors was allowed. Patients undergoing treatment with chemotherapy, androgens, estrogens or analogs of progesterone or those receiving other known treatments for menopausal symptoms, including vitamin E, antidepressants and gabapentin, patients with coagulation disorders or those taking aspirin and patients with acute infections and heart disease American Society of Anesthesiologists≥III were all excluded from the study. After obtaining voluntary, written informed consent, the patients who were admitted were randomly assigned to two groups: study and control.
In the study group, SGB was performed under ultrasound guidance, wherein the patients were provided with appropriate monitoring in supine position with a slight extension of the neck. Midazolam 1 mg and fentanyl 50 µg were administered intravenously. To do diagnostic imaging, a MicroMaxx ultrasound machine with high-frequency linear probe (Sonosite Bothell, Washington, WA, USA) was used. Ultrasound imaging was performed on axial plane at the level of cricoid cartilage (Figure 1). After identifying the appropriate location, 10 mL of 0.5% bupivacaine (AstraZeneca plc, Stockholm, Sweden) was injected, with in-plane technique, using a 21 G sonovisible nerve block needle (Pajunk, Geisingen, Germany). Incidence of Horner’s syndrome was deemed a symptom of successful block, and the patients were discharged after a recovery period of at least 2 hours.
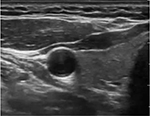  | Figure 1 Sonographic view of drug distribution in the SGB. Abbreviation: SGB, stellate ganglion block. |
In the second group, paroxetine (Brisdelle; Sebela Pharmaceuticals, Roswell, Georgia, GA, USA) was administered orally for 6 weeks at a daily dose of 7.5 mg. The frequency and severity of hot flash attacks and sleep quality of the patients were evaluated before the intervention and then at 2, 4 and 6 weeks after the block or after commencing the medication in both groups. Any possible adverse effects of the administration were recorded. The person filling out the questionnaire was unaware of the patients’ group assignment. The intensity of hot flashes was measured using Sloan’s hot flash scoring scale ranging from mild, moderate, severe to very severe and the frequency of occurrence of each, from 0 to 100. In this method, the number of attacks per 24 hours is multiplied by the severity of them (with a gradation of 1–4), and the scores ranging from 0 to 100 are given to patients. These scores are for 24 hours but are estimated at our review intervals (0, 2, 4 and 6 weeks after the intervention).17 The Pittsburgh Sleep Quality Index (PSQI) was used to assess sleep quality. PSQI is a nine-component questionnaire rating patients on several variables, including sleep quality and duration, delay in sleep onset, habitual sleeping problems, daytime dysfunction and the everyday need for sedation, wherein lower scores indicate better sleep quality.18
At the end of the 6th week, the data were collected and the results were expressed as mean ± SD and the percentage of quantitative and qualitative variables, respectively. The qualitative variables were compared by the Student’s t-test for normally distributed variables or by the Mann–Whitney U test for non-normally distributed variables. The chi-squared test or Fisher’s exact test was performed for comparison of the qualitative variables. The correlation between quantitative variables was examined using the Pearson correlation coefficient and Spearman’s rank correlation coefficient. The data collected were analyzed using the SPSS version 21 (and the SAS version 1.9 software (SAS institute, Cary, North Carolina, NC, UA). The level of significance was set at P<0.05.
Results
Table 1 summarizes the comparison of key demographic characteristics and clinical details of the paroxetine and SGB groups, indicating no significant difference between the two groups.
Of the 20 patients in the SGB group, one patient was withdrawn from the study due to the lack of Horner syndrome, which was considered a sign of successful block, and was replaced with a new patient. No serious side effects were observed in any of the patients in this group. Horner syndrome occurred shortly after the blockage and lasted for about 4–6 hours. Only one patient was reported to have a mild headache for 1 day to 2 days.
In the P group, two patients were withdrawn from the study since they had stopped taking the drug during the initial days because of gastrointestinal side effects – nausea and vomiting – and were replaced with new patients. Meanwhile, of the two patients, one who had taken the medicine for a week declared overall decline in symptoms.
The comparison of the hot flash scores and sleep quality index between the two groups, indicating no significant difference, both before the intervention and over the follow-up period (P>0.05), is given in Table 1.
Figure 2 illustrates the changes in hot flash scores and sleep quality in the two groups. A downward trend in the hot flash scores and sleep quality indexes was observed in both groups (P<0.001). Lower scores indicated better sleep quality and improvement in hot flashes. There was not a noticeable difference in the trend of changes in hot flash scores between the two groups (P=0.778). The trend of changes in sleep quality index in the two groups was alike (P=0.365).
Figure 3 illustrates the CONSORT flowchart of the study design.
  | Figure 3 CONSORT flowchart depicting the process of patient inclusion, randomization and exclusion. Abbreviations: P, paroxetine; SGB, stellate ganglion block. |
Discussion
The results of the present study supported that the SGB method can be as effective as paroxetine in reducing the frequency of hot flash attacks and improving sleep quality in breast cancer survivors, with minimal side effects and acceptable patients’ tolerance.
The use of non-pharmaceutical and nonhormonal methods in managing menopausal symptoms caused by breast cancer treatment has attracted great attention in recent years. Reviewing pilot studies conducted on various medicines and methods particularly used in this regard, Kontos et al3 found the effectiveness of desvenlafaxine and SGB as noticeable; they recommended further studies, however. After studying the existing evidences, Boutet19 in his proposed algorithm introduced venlafaxine, paroxetine and gabapentin as a first-line drug treatment for severe hot flashes and adjourned indications of SGB until further investigations were carried out. Fisher et al20 investigated different pharmaceutical, nutritional, supplemental and behavioral methods and examined any evidence for each.
The effectiveness of different doses of paroxetine has been investigated in several studies. Weitzner et al21 maintained that paroxetine was effective in reducing hot flashes as well as sleep disturbances, fatigue and depression in breast cancer survivors. In another study, Loprinzi et al22 investigated and compared the efficacy of fluoxetine to that of placebo and found that the former was mildly effective in improving hot flash attacks. Compared with placebo, daily doses of paroxetine 7.5–12.5 mg reduced the frequency and intensity of hot flashes and diminished the number of nighttime awakenings caused by VMSs.7–9 Paroxetine was approved by the US FDA in 2013 for the treatment of moderate-to-severe VMSs and for sleep quality improvement. In our study also, paroxetine 7.5 mg proved to be remarkably effective in both reducing hot flash scores and improving sleep quality indexes.
The effect of SGB in reducing hot flash symptoms and improving sleep quality has also been investigated in several studies. In a case study of six patients with severe hot flash attacks, Lipov et al23 tated that the method reduced hot flash attacks for 2–5 weeks following the first time blockade and for 4–18 weeks after the second blockade. In addition, performing SGB in a pilot study of 13 breast cancer survivors, they followed up the patients for 12 weeks and reported that this method reduced hot flashes and sleep disturbances, without or with minimal adverse effects.24
In their study of 34 breast cancer survivors suffering from menopause, Haest et al1 assessed the patients for 24 weeks and observed that the SGB lessens hot flash symptoms and improves sleep quality. They also found that the effect of blockage on hot flashes would diminish over time, but its effect on sleep quality lasted longer. Guttuso12 estimated the reducing effect of the SGB on the symptoms of hot flashes from 28–90%, but since the placebo effect exceeded 50%, the researcher recommended further studies to judge the findings.
In a study of 40 patients in the control group, Walega et al15 injected saline solution into their subcutaneous tissue of neck and concluded that although no significant difference was observed in the overall number of hot flashes between the two groups, the SGB group suffering from moderate-to-severe hot flashes had greater reduction in the number of attacks.
Nearly, all the studies conducted indicated the effectiveness of the SGB in controlling the symptoms with minimal side effects. In addition, the results of our study are in accordance with these findings, although this study lacked a placebo group.
This is the first study that has compared the effectiveness of SGB to that of paroxetine as an FDA-approved drug; the results of which are consistent with the findings obtained from the studies carried out on each method alone. Based on the results of this study, SGB can be an effective way to treat the symptoms of menopause arising from breast cancer. This method is associated with least side effects and is free from restrictions accompanied with gastric intolerance to oral paroxetine.
The limitations of this study include problems with following up the patients as well as single-center study. Further studies with larger sample sizes, different doses, a placebo group and longer follow-up are recommended to be designed and implemented to evaluate the effectiveness of this method.
Conclusion
Stellate ganglion block is as much effective as paroxetine in controlling hot flashes and sleep disturbances in breast cancer survivors and is associated with few complications.
Acknowledgments
The authors would like to express their gratitude to participants and staff of the breast surgery clinic for their contribution. In addition, they wish to thank Rasoul Akram Hospital Clinical Research Development Center, Iran University of Medical Sciences.
Disclosure
The authors report no conflicts of interest in this work.
References
Haest K, Kumar A, van Calster B, et al. Stellate ganglion block for the management of hot flashes and sleep disturbances in breast cancer survivors: an uncontrolled experimental study with 24 weeks of follow-up. Ann Oncol. 2012;23(6):1449–1454. | ||
Othman AH, Zaky AH. Management of hot flushes in breast cancer survivors: comparison between stellate ganglion block and pregabalin. Pain Med. 2014;15(3):410–417. | ||
Kontos M, Agbaje OF, Rymer J, Fentiman IS. What can be done about hot flushes after treatment for breast cancer? Climacteric. 2010;13(1):4–21. | ||
Joffe H, Guthrie KA, Lacroix AZ, et al. Low-dose estradiol and the serotonin-norepinephrine reuptake inhibitor venlafaxine for vasomotor symptoms: a randomized clinical trial. JAMA Intern Med. 2014;174(7):1058–1066. | ||
Krause MS, Nakajima ST. Hormonal and nonhormonal treatment of vasomotor symptoms. Obstet Gynecol Clin North Am. 2015;42(1):163–179. | ||
Tella SH, Gallagher JC. Efficacy of desvenlafaxine succinate for menopausal hot flashes. Expert Opin Pharmacother. 2014;15(16):2407–2418. | ||
Pinkerton JV, Joffe H, Kazempour K, Mekonnen H, Bhaskar S, Lippman J. Low-dose paroxetine (7.5 mg) improves sleep in women with vasomotor symptoms associated with menopause. Menopause. 2015;22(1):50–58. | ||
Kaunitz AM, Manson JE. Management of Menopausal Symptoms. Obstet Gynecol. 2015;126(4):859–876. | ||
Carroll DG, Lisenby KM, Carter TL. Critical appraisal of paroxetine for the treatment of vasomotor symptoms. Int J Womens Health. 2015;7:615–624. | ||
Narouze S. Ultrasound-guided stellate ganglion block: safety and efficacy. Curr Pain Headache Rep. 2014;18(6):424. | ||
Imani F, Hemati K, Rahimzadeh P, Kazemi MR, Hejazian K. Effectiveness of Stellate Ganglion Block Under Fluoroscopy or Ultrasound Guidance in Upper Extremity CRPS. J Clin Diagn Res. 2016;10(1):UC09–12. | ||
Guttuso T. Stellate ganglion block for treating hot flashes: a viable treatment option or sham procedure? Maturitas. 2013;76(3):221–224. | ||
Pachman DR, Barton D, Carns PE, et al. Pilot evaluation of a stellate ganglion block for the treatment of hot flashes. Support Care Cancer. 2011;19(7):941–947. | ||
van Gastel P, Kallewaard JW, van der Zanden M, de Boer H. Stellate-ganglion block as a treatment for severe postmenopausal flushing. Climacteric. 2013;16(1):41–47. | ||
Walega DR, Rubin LH, Banuvar S, Shulman LP, Maki PM. Effects of stellate ganglion block on vasomotor symptoms: findings from a randomized controlled clinical trial in postmenopausal women. Menopause. 2014;21(8):807–814. | ||
Carris N, Kutner S, Reilly-Rogers S. New pharmacological therapies for vasomotor symptom management: focus on bazedoxifene/conjugated estrogens and paroxetine mesylate. Ann Pharmacother. 2014;48(10):1343–1349. | ||
Sloan JA, Loprinzi CL, Novotny PJ, Barton DL, Lavasseur BI, Windschitl H. Methodologic lessons learned from hot flash studies. J Clin Oncol. 2001;19(23):4280–4290. | ||
Buysse DJ, Reynolds CF, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28(2):193–213. | ||
Boutet G. Management of hot flushes for breast cancer survivors. Gynecol Obstet Fertil. 2012;40(4):241–254. | ||
Fisher WI, Johnson AK, Elkins GR, et al. Risk factors, pathophysiology, and treatment of hot flashes in cancer. CA Cancer J Clin. 2013;63(3):167–192. | ||
Weitzner MA, Moncello J, Jacobsen PB, Minton S. A pilot trial of paroxetine for the treatment of hot flashes and associated symptoms in women with breast cancer. J Pain Symptom Manage. 2002;23(4):337–345. | ||
Loprinzi CL, Sloan JA, Perez EA, et al. Phase III evaluation of fluoxetine for treatment of hot flashes. J Clin Oncol. 2002;20(6):1578–1583. | ||
Lipov E, Lipov S, Stark JT. Stellate ganglion blockade provides relief from menopausal hot flashes: a case report series. J Womens Health. 2005;14(8):737–741. | ||
Lipov EG, Joshi JR, Sanders S, et al. Effects of stellate-ganglion block on hot flushes and night awakenings in survivors of breast cancer: a pilot study. Lancet Oncol. 2008;9(6):523–532. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


