Back to Journals » International Journal of General Medicine » Volume 14
Comparison of Prediction Models for Acute Kidney Injury Among Patients with Hepatobiliary Malignancies Based on XGBoost and LASSO-Logistic Algorithms
Authors Zhang Y, Wang Y, Xu J, Zhu B, Chen X, Ding X, Li Y
Received 21 January 2021
Accepted for publication 17 March 2021
Published 16 April 2021 Volume 2021:14 Pages 1325—1335
DOI https://doi.org/10.2147/IJGM.S302795
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Yunlu Zhang,1– 3,* Yimei Wang,1– 3,* Jiarui Xu,1– 3 Bowen Zhu,1– 3 Xiaohong Chen,1– 3 Xiaoqiang Ding,1– 3 Yang Li1– 3
1Department of Nephrology, Zhongshan Hospital, Fudan University, Shanghai, People’s Republic of China; 2Shanghai Medical Center of Kidney, Shanghai, People’s Republic of China; 3Shanghai Key Laboratory of Kidney and Blood Purification, Shanghai, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Xiaoqiang Ding; Yang Li Email [email protected]; [email protected]
Background: Based on the admission data, we applied the XGBoost algorithm to create a prediction model to estimate the AKI risk in patients with hepatobiliary malignancies and then compare its prediction capacity with the logistic model.
Methods: We reviewed clinical data of 7968 and 589 liver/gallbladder cancer patients admitted to Zhongshan Hospital during 2014 and 2015. They were randomly divided into the training set and test set. Data were collected from the electronic medical record system. XGBoost and LASSO-logistic were used to develop prediction models, respectively. The performance measures included the classification matrix, the area under the receiver operating characteristic curve (AUC), lift chart and learning curve.
Results: Of 6846 participants in the training set, 792 (11.6%) cases developed AKI. In XGBoost model, the top 3 most important variables for AKI were serum creatinine (SCr), glomerular filtration rate (eGFR) and antitumor treatment in liver cancer patients. Similarly, SCr and eGFR also ranked second and third most important variables in the gallbladder cancer-related AKI model just after phosphorus. In the classification matrix, XGBoost model possessed a comparably better agreement between the actual observations and the predictions than LASSO-logistic model. The Youden’s index of XGBoost model was 47.5% and 59.3%, respectively, which was significantly higher than that of LASSO-logistic model (41.6% and 32.7%). The AUCs of XGBoost model were 0.822 in liver cancer and 0.850 in gallbladder cancer. By comparison, the AUC values of Logistic models were significantly lower as 0.793 and 0.740 (p=0.024 and 0.018). With the accumulation of training samples, XGBoost model maintained greater robustness in the learning curve.
Conclusion: XGBoost model based on admission data has higher accuracy and stronger robustness in predicting AKI. It will benefit AKI risk classification management in clinical practice and take an advanced intervention among patients with hepatobiliary malignancies.
Keywords: hepatobiliary malignancy, acute kidney injury, extreme gradient boosting, LASSO-logistic regression, disease prediction, machine learning
Introduction
Liver and gallbladder malignancies have burdened the healthcare system enormously,1 which correlate with each other because of the adjacent anatomic position and conjoint function in the biliary tree system. According to the global estimation in 2018, liver cancer ranked the 6th most generally diagnosed cancers and the 4th most malignant carcinomas causing death,2 among which 46.6% of cases were located in China.3 Though not featured among the top 10 cancers, gallbladder cancer also had a high annual rate in Chile, Canada and some Asian countries.4 Most of the hepatobiliary cancer patients experienced different antitumor treatments depending on their cancer progression, including surgeries (like hepatectomy, liver transplantation and cholecystectomy), chemotherapy, immunotherapy and palliative care.5 Acute kidney injury (AKI) is one of the most severe complications, and its 1-year risk in liver and gallbladder cancer patients reaches 33.0% and 33.6%.6 Other studies also revealed that 12~20% of patients acquired AKI after partition hepatectomy or liver transplantation within 72 hours.7,8 Apart from the general risk factors, AKI in hepatobiliary cancer settings is also affected by hepatic dysfunction, nephrotoxic damage, electrolyte disturbances, and inflammatory responses.9 It is of significance to identify liver cancer patients at high risk of AKI in the early stage. About one-fifth of AKI cases acquired in hospitals could be prevented if appropriate interventions were taken action in time, such as monitoring electrolytes, identifying risk factors and other treatments.10 If not, AKI would convert to irreversible damage and increase the in–hospital mortality and the risk of chronic kidney disease (CKD).11,12
Gradient boosting machine (GBM) is regarded as an upgraded machine learning algorithm, which can converge the weak learners into a strong learner. During the gradient descent process, GBM improves the performance of the prediction model. As an extension of GBM, XGBoost (extreme gradient boosting) can further increase the calculation accuracy within a shorter model running time.13 It has been widely applied for disease diagnosis and prediction.14–16
An applicable prediction model with high accuracy could help identify patients who would benefit most from intensive monitoring of SCr or AKI biomarkers. It also allows clinicians to intervene early-stage and further increase the survival rate for liver/gallbladder cancer patients. Based on the admission data, we aim to apply the XGBoost algorithm to create a prediction model that estimates the AKI risk in patients diagnosed with hepatobiliary malignancies, and then compare its prediction capacity with the traditional logistic model.
Methods
Study Design and Participants
This study was designed as a retrospective cohort study. All inpatients diagnosed with hepatobiliary malignancies admitted to Zhongshan hospital from October 2014 to September 2015 were involved. Exclusion criteria included: patients who were admitted to the hospital less than 24 hours; patients already on renal replacement therapy (RRT); patients who lacked the sequential serum creatinine (SCr) tests. After review of the admission register, 7968 liver cancer patients and 589 gallbladder cancer patients were selected as potentially eligible. They were further randomly divided into two datasets in a ratio of 4:1. Of them, 6375 liver cancer and 471 gallbladder cancer participants were assigned to the training set for model establishment. The remaining 1593 and 118 participants were assigned to the test set for external validation (Figure S1). This study was approved by the Institutional Committee of Zhongshan Hospital (B2018-175) and was conducted according to the Declaration of Helsinki principles. Informed consent was waived due to the retrospective and non-interventional nature of this study. Patient identity information was replaced as a unique code to ensure the data confidentiality.
Data Collection
Data were extracted from electronic medical records and laboratory databases. All of the biochemical indicators were tested within 24 h after admission. The analyzed variables included demographics [age, gender, and body mass index (BMI)], pre-existing comorbidities [hypertension, diabetes, stroke, coronary heart disease (CHD), chronic obstructive pulmonary disease (COPD), CKD], cancer characteristics [metastatic, emergency admission, antitumor treatments], physical status [systolic blood pressure (SBP), diastolic blood pressure (DBP), and glucose], hepatic function [alanine aminotransferase (ALT), aspartate aminotransferase (AST), and total bilirubin (TBiL)], renal function [blood urea nitrogen (BUN), SCr, estimated glomerular filtration rate (eGFR), and serum uric acid (SUA)], blood routine [album, hemoglobin, and leukocyte], and electrolyte test [serum sodium, potassium, chloride, calcium, magnesium, phosphorus, carbon dioxide and anion gap (AG)].
Disease Definition
AKI diagnosis is based on the 2012 Kidney Disease Improving Global Outcomes (KDIGO) criteria: a maximal increase in SCr by ≥0.3 mg/dL (26.5μmol/L) within 48 hours, or by ≥1.5 times baseline within the previous seven days. Due to the enormous lack of urine volume data, we dropped urine volume changes to diagnose AKI. The diagnosis of liver cancer (C22) and gallbladder cancer (C23) is based on the international classification of diseases (ICD-10).17 The level of eGFR is calculated by using the chronic kidney disease epidemiology collaboration (CKD-EPI) 2009 creatinine equation.18
XGBoost Model
XGBoost is proposed in 2016 by Tianqi Chen.19 It is based on the sparsity-aware algorithm and weighted quantile sketch, which can converge the weak learners step-wisely into the ensemble to form a strong learner. Overfitting also can be reduced largely in XGBoost model through parallel calculation and regularization lifting technology. Meanwhile, XGBoost can customize the cost function and process missing data. It makes learning faster and enables robust model exploration. The objective function of XGBoost is expressed as 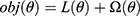 . The cost function is
. The cost function is  .
. 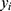 is the true value of sample
is the true value of sample 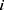 .
. 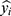 is the predictive value and presented as
is the predictive value and presented as 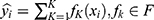 .
. 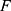 is the set of all candidate CART tree models.
is the set of all candidate CART tree models. 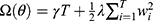 reflects the complexity of tree models, in which
reflects the complexity of tree models, in which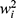 is the weighted score on the
is the weighted score on the 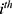 leaf node, T is the number of leaf nodes in the tree. By training and tuning the parameters, the objective function of
leaf node, T is the number of leaf nodes in the tree. By training and tuning the parameters, the objective function of 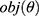 is continuously optimized.
is continuously optimized.
LASSO-Logistic Model
LASSO, the least absolute and selection operator, is proposed in 1996 by Robert Tibshirani.20 The principle of LASSO is to impose a penalty on the model’s coefficient on the basis of the ordinary least-squares estimation (OLS). LASSO-logistic model, derived from the traditional logistic model, can select variables through the tuning parameter λ, and then shrink the estimates of irrelevant variables to zero. The parameter estimation in LASSO-logistic model can be expressed as: 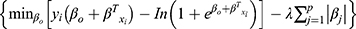 . Ten-fold cross-validation is applied to select the optimal value of
. Ten-fold cross-validation is applied to select the optimal value of 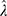 . We defined the fitted model including tuning parameter as
. We defined the fitted model including tuning parameter as 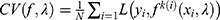 .
. 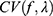 is an error curve that changes with λ. The optimal value of
is an error curve that changes with λ. The optimal value of 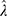 indicates the
indicates the 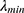 or
or 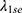 . After that, the LASSO-logistic model is created as
. After that, the LASSO-logistic model is created as 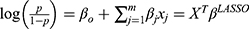 , which only contains variables selected by LASSO process.
, which only contains variables selected by LASSO process.
Statistical Analysis
Statistics were conducted by using R 3.6.1 software (R core team). We used QQ-plot to test the normality of continuous variables (Figure S2). It was run in “car” package. Normal data were presented as mean± standard deviation, and non-normal data were presented as median with inter-quartile range (IQR). Categorical variables were expressed in numbers and percentages (“Hmisc” package). Missing data were found in less than 10% of the records, mainly focusing on the electrolyte tests (calcium, magnesium, phosphorus). We imputed the missing values by the methods of multiple imputations (“mice” package). Homogeneity analyses were performed between the training set and test set. Student’s t-test and Wilcoxon test were applied to compare the distributional difference of continuous variables. Pearson chi-square test and Fisher’s exact test were applied to compare the proportional difference of categorical variables (“gmodels” package).
By using XGBoost and LASSO-logistic, we developed the liver cancer and gallbladder cancer-related AKI prediction models, respectively. In XGBoost model (“xgboost” package), the main tuning parameter was set as follows: eta (learning rate)=0.3; max_depth (maximum depth of a tree)=6; min_child_weight (minimum sum of instance weight)=1; nrounds (max number of boosting iterations)=3; lambda (L2 regularization)=0; alpha (L1 regularization)=0. Model performance was evaluated by the matrix of “error” (binary classification error rate). In LASSO-logistic model, we initially conducted 10-fold cross-validation of LASSO to select candidate variables (“glmnet” package). The main tuning parameter was set as follows: type.measure (loss to use for cross-validation)=“class”, family=“binomial”, nfolds=10. The complexity of LASSO is controlled by 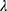 , which penalizes models with more variables with greater force. We defined
, which penalizes models with more variables with greater force. We defined  (largest value of
(largest value of 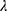 such that error is within 1 standard error of the minimum) as the criteria to acquire the optimal model with the least variables. Then these significant variables were incorporated into the logistic model, and their associations with AKI were evaluated by odds ratios. All statistical tests were two-tailed, and we regarded p<0.05 as the criterion for statistical significance.
such that error is within 1 standard error of the minimum) as the criteria to acquire the optimal model with the least variables. Then these significant variables were incorporated into the logistic model, and their associations with AKI were evaluated by odds ratios. All statistical tests were two-tailed, and we regarded p<0.05 as the criterion for statistical significance.
After that, the trained perdition models were further validated externally with the test dataset. The predictive capacity of XGBoost model and LASSO-logistic model were compared on three aspects. Firstly, we used the area under the receiver operating characteristic (AUROC) curves to quantify the predictive accuracy (“qROC” package). Delong test was used to compare the significance of ROC curves. Secondly, the classification matrix was created to determine whether the predicted value matched the actual value (“caret” package). Indicators for evaluation included sensitivity, specificity, false-positive rate, false-negative rate, accuracy rate, Youden’s index, positive predictive value and negative predictive value. The purpose of this process is to assess the specific classification ability of AKI cases and non-AKI cases. Lastly, lift charts and learning curves were plotted to evaluate the model’s robustness (“ROCR” package). The Lift chart graphically represents the improvement that a mining model provides when compared against a random guess and measures the change in terms of a lift score. The learning curve shows the validation score of an estimator for varying numbers of training samples. It is a tool to determine how much we benefit from adding more training data and whether the estimator suffers more from a variance error or a bias error.
Results
AKI Incidence and Risk Factors Analysis
A total of 7968 liver cancer patients and 589 gallbladder cancer patients were recruited, which were randomly divided into the training set and test set as the ratio of 4:1. The average age was 58.1±11.3 years old, and male patients accounted for 81.7% (n=6990). Table S1 suggested that all variables’ statistical distributions between the training set and test set were comparable (p > 0.05). According to KDIGO classification, 792 (11.6%) patients developed AKI in the training set. Of them, 701 patients were located in AKI Stage-1, and another 55 and 36 cases were located in Stage-2 and Stage-3. About 0.3% of AKI patients underwent renal replacement treatment (n=21).
Table 1 lists the risk factors of AKI. Patients who were elder age, female, lower BMI, or suffered from hypertension, stroke, COPD and CKD were at higher risk of developing AKI. Emergent admission, low blood pressure and high glucose level were positively associated with AKI. Given the fact that patients already had pre-existed hepatic and renal dysfunction, the risk of cancer-related AKI was also increased. Compared to non-AKI patients, AKI patients had lower albumin and hemoglobin levels and higher leukocyte counts. In terms of electrolyte variables, hyponatremia, hypochloremia, hypocalcemia and hypophosphatemia had notable impacts on AKI while higher AG level also increased the risk of AKI.
 |
Table 1 Demographic and Clinical Factors of AKI in Patients with Hepatobiliary Malignancies |
XGBoost Model and LASSO-Logistic Model Establishment
Fifteen candidate variables were screened out via the XGBoost models. The importance matrix plot of variables is shown in Figure 1. In the liver cancer-related AKI model, predictors’ detection ability was ordered from high to low: SCr, eGFR, treatments, sodium, BUN, chloride, AST, SBP, TBiL, CO2, leukocyte, glucose, Magnesium, ALT, and DBP. Of them, the fractional contribution of SCr and eGFR to the model gain was over 20%. Similarly, SCr and eGFR also ranked second and third after phosphorus in the gallbladder cancer-related AKI model. Other factors included potassium, BMI, hemoglobin, glucose, TBiL, leukocyte, age, AST, sodium, treatment, AG and SUA.
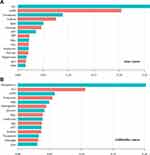 |
Figure 1 (A) Importance matrix plot of AKI predictors in XGBoost model among liver cancer patients. (B) Importance matrix plot of AKI predictors in XGBoost model among gallbladder cancer patients. |
Figure S3 showed the coefficient profile plot of the variable coefficient paths for a fitted LASSO. When 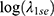 reached −4.537, 17 predictors were selected from the original feature–pool among patients with liver cancer. It included gender, stroke, COPD, emergent condition, antitumor treatment, SBP, TBiL, BUN, SCr, eGFR, SUA, hemoglobin, sodium, potassium, chloride, calcium and AG. Following the similar steps with a
reached −4.537, 17 predictors were selected from the original feature–pool among patients with liver cancer. It included gender, stroke, COPD, emergent condition, antitumor treatment, SBP, TBiL, BUN, SCr, eGFR, SUA, hemoglobin, sodium, potassium, chloride, calcium and AG. Following the similar steps with a 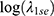 of −3.912, 15 variables were selected in gallbladder cancer patients. It included age, BMI, CKD, metastatic tumors, emergent condition, treatment, AST, BUN, SCr, eGFR, albumin, hemoglobin, sodium, phosphorus and AG. Then, we presented these predictors into multiple logistic regression to quantify the association strength with AKI (Table 2).
of −3.912, 15 variables were selected in gallbladder cancer patients. It included age, BMI, CKD, metastatic tumors, emergent condition, treatment, AST, BUN, SCr, eGFR, albumin, hemoglobin, sodium, phosphorus and AG. Then, we presented these predictors into multiple logistic regression to quantify the association strength with AKI (Table 2).
 |
Table 2 Predictive Variables of AKI Selected by LASSO-Logistic Regression |
Prediction Capacity Comparison in Two Models
We performed the external validation by using the patients’ data from the test set. The classification matrix of AKI is elaborated in Table 3. In liver and gallbladder cancer patients, XGBoost model possessed a comparably better agreement between the actual observations and the predictions. The Youden’s index of XGBoost model was 47.5% and 59.3%, respectively. It was significantly higher than that of LASSO-logistic model (41.6% and 32.7%, all p<0.001). ROC curves of two prediction models are drawn in Figure 2. The AUC value of XGBoost models was 0.822 (95% CI 0.789~0.855) in liver cancer, 0.850 (95% CI 0.775~0.920) in gallbladder cancer. Logistic models, by comparison, shared lower AUC values of 0.793 and 0.740. DeLong’s test further verified such a difference between XGBoost and logistic models was statistically significant (p=0.024 and 0.018). In the lift chart, no notable difference was found between the two models, while the XGBoost model performed slightly better than the logistic model. With the accumulation of training samples, XGBoost models maintained greater robustness in the learning curve. Their AUC levels were also consistently higher than the others.
 |
Table 3 External Validation of AKI Prediction Models Based on XGBoost and LASSO-Logistic Algorithms |
Discussion
In this study, the incidence of AKI was 11.6% in patients with hepatobiliary malignancies. It is consistent with reported literature ranging from 7.6% to 15.0%.21–23 Apart from the common factors, we found that AKI in liver/gallbladder cancer patients was affected by antitumor treatment, haptic-renal dysfunction, and electrolyte disorders. Hepatobiliary surgery, especially the liver partial excision or transplantation, triggers a series of ischemia-reperfusion injuries, inflammatory mediators and complement activation, free radicals production and allograft rejection.24 Iodinated radiocontrast medium used during angiography, such as transarterial chemoembolisation (TACE), also increases the risk of AKI.25 Additionally, we observed that patients with elevated levels of AST, TBiL, SCr and BUN on admission were more susceptible to AKI (OR ranging from 1.00 to 1.04). Xu et al also proved that preoperative high AST/ALT was a potential independent predictor of AKI in patients with hepatocellular carcinoma.23 Liver cirrhosis and dysfunction may lead to dilation of visceral blood vessels and hormonal imbalance, resulting in renal vasoconstriction and impaired renal function.26 If left untreated, patients would progress to hepatorenal syndrome (HRS) in a short time.27 Hyponatremia is a common adverse effect due to emesis metabolic disturbances, volume depletion (diarrhea, vomiting, and drainage of ascites).28 Syndrome of inappropriate antidiuretic hormone (SIADH) also can induce hyponatremia due to ectopic production of arginine vasopressin by the tumor tissue.29 Besides, we found that the serum phosphate level was an AKI predictor in gallbladder cancer patients. It might be caused by improper use of chemotherapy medications, particularly molecular-targeted agents. One study in Japan revealed that the incidence of cancer-related hypophosphatemia (<2.5 mg/dL) was 49.4%, and patients with gallbladder cancer shared a higher risk (OR: 1.12).30 The main pathophysiology of hypophosphatemia works as disturbed electrolytes intracellular shifts, as a result of combined interactions among insulin, catecholamines, hyperalimentation, and respiratory alkalosis, decreased intestinal absorption, and increased urinary losses.31
If patients at high risk of AKI were detected on admission, early nephrology management could benefit their clinical outcomes.32,33 Regular monitoring of urine volume, appropriate dosage adjustment, strict avoidance of nephrotoxic drugs and early initiation of RRT can reduce the incidence and mortality of AKI.34–36 Compared with low osmolar contrast, isosmolar contrast could remarkably reduce the risk of interventional therapy-related AKI, and had higher cost-effectiveness.37 However, AKI is often neglected by non-nephrologists, who usually provide the first or main healthcare for patients with AKI.38 Therefore, it is necessary to develop a predictive model of AKI based on admission data to assist clinicians in identifying more high-risk patients. The implement of electronic automated early warning systems for AKI can remind clinicians of the courteous use of nephrotoxic drugs and facilitate more intensive monitoring for SCr or novel biomarkers of AKI risk.39
We applied the XGBoost algorithm to construct models so as to predict the risk of AKI in patients with hepatobiliary cancers and compared its predictive performance with LASSO–logistic regression model. In the past two decades, many risk prediction scores for AKI have been developed on the basis of Logistic regression.40–43 A study conducted in Mayo Clinic created a general AKI prediction model for non-ICU patients, and the AUC of this Logistic model was 0.75.44 As the classic model, Logistic is easy to implement in SPSS software and makes a certain sense to explore the risk factors of AKI. But under the concept of big medical data, more studies focus on machine learning. Novel algorithms, such as XGBoost and artificial neural networks, were constantly proposed to cope with the complex medical data that traditional statistics cannot handle.45 Logistic regression assumes that each variable should be independent, and the model only exists a linear partition surface. Yet, the association between diseases and exposure factors is usually influenced by other confounders. This nonlinear relationship makes Logistic inference prone to a large deviation in model fitting. In contrast, XGBoost has the following advantages. (1) XGBoost combines the regularization method of L1+L2, which can prevent the over-fitting problem caused by the excessive data dimension. (2) Variables in the XGBoost model are only used for importance order. The outlier data have relatively little impact on modeling parameters. (3) XGBoost has the flexibility to target functions and evaluation index, which makes it applicable to process hybrid data. (4) The operation time of XGBoost model was notably shortened via parallel computing on a kernel cluster. (5) XGBoost combines decision trees with gradient lifting algorithms, which makes it reserve the correlation between variables in the modeling process.46 Despite the many AKI prediction tools in the literature, parallel tools to detect AKI risk in patients with hepatobiliary malignancies are lacking. One notable exception is a recent study by Lei et al3 that compared AKI risk prediction based on different machine learning algorithms in patients after liver cancer resection. The AUCs for the four algorithms were: GBDT (0.772), GBM (0.725), forest (0.662) and Decision tree (0.628). As the upgraded version of GBDT and GBM, XGB model in the present study showed a higher accuracy, better generalizability and stronger robustness. The AUCs of XGBoost reached 0.822 and 0.850. It might be because we enrolled more electrolyte data to manifest patients’ homeostasis. As compared with LASSO-logistic, XGBoost model also shared lower false positive/negative rates and higher predictive values. Specificity superior to sensitivity suggests that XGBoost model could be most effectively used to exclude AKI. For high-risk patients, further testing (SCr or novel biomarkers) is necessary to stratify their AKI risk. Moreover, the learning curves of XGBoost model also performed better than that of Logistic regression model. It indicated that XGBoost maintained a robust prediction capacity even in the small sample size. The importance matrix in XGBoost model reflects the variables’ contribution value to AKI prediction, which is slightly different from the clinical significance of traditional risk factors. The higher the ranking of variables, the more conductive to assist clinical diagnosis. In our study, variables screened by XGBoost and LASSO were not identical, which reflects that the criteria of variable selection in the modeling are quite different. The top variables in XGBoost models were those reflecting kidney function, electrolyte level, and antitumor therapy. From the perspective of pathophysiology, it is more consistent with clinical practice. Due to the inability to handle data collinearity, Logistic model often includes some variables that are difficult to explain or not of strong biological significance.
This study had several limitations to be stated. Firstly, this study was a single-center study, and the validation cohort was derived from the same source population. It is necessary to conduct multicenter external validation in the future. Secondly, the sample size and number of predictive variables in this study were limited. When the sample size and variables increase, the difference between XGBoost and logistic models might be better manifested. Thirdly, the prediction model was based on admission predictors. We did not use the longitudinal data to display their dynamic fluctuations during hospitalization. In future studies, a multicenter prospective cohort should be established to verify the prediction effect of factors found in this study. Time-varying covariates also should be imported into models in a proper way to manifest the wax and wane of AKI during hospitalization.47
Conclusion
XGBoost model can be used to predict AKI in patients with hepatobiliary malignancies and has higher accuracy and stronger robustness compared with the traditional model. It will help perfect the practice of AKI risk classification management and identify patients who would benefit most from intensive monitoring of SCr or AKI biomarkers.
Acknowledgments
The study was supported by the major projects of scientific research and innovation plan of Shanghai education commission, under Grant 2017-01-07-00-07-E00009; Shanghai medical center of kidney, under Grant 2017ZZ01015; Shanghai key laboratory of kidney and blood purification, under Grant 14DZ2260200; Zhongshan Hospital Science Foundation for Youths, under Grant 2019ZSQN19.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bjerregaard JK, Mortensen MB, Pfeiffer P. Trends in cancer of the liver, gall bladder, bile duct, and pancreas in elderly in Denmark, 1980–2012. Acta Oncol. 2016;55(Suppl 1):40–45. doi:10.3109/0284186x.2015.1114675
2. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.21492
3. Lei L, Wang Y, Xue Q, Tong J, Zhou CM, Yang JJ. A comparative study of machine learning algorithms for predicting acute kidney injury after liver cancer resection. PeerJ. 2020;8:e8583. doi:10.7717/peerj.8583
4. Schmidt MA, Marcano-Bonilla L, Roberts LR. Gallbladder cancer: epidemiology and genetic risk associations. Chin Clin Oncol. 2019;8(4):31. doi:10.21037/cco.2019.08.13
5. Anwanwan D, Singh SK, Singh S, Saikam V, Singh R. Challenges in liver cancer and possible treatment approaches. Biochim Biophys Acta Rev Cancer. 2020;1873(1):188314. doi:10.1016/j.bbcan.2019.188314
6. Christiansen CF, Johansen MB, Langeberg WJ, Fryzek JP, Sorensen HT. Incidence of acute kidney injury in cancer patients: a Danish population-based cohort study. Eur J Intern Med. 2011;22(4):399–406. doi:10.1016/j.ejim.2011.05.005
7. Chen X, Ding X, Shen B, et al. Incidence and outcomes of acute kidney injury in patients with hepatocellular carcinoma after liver transplantation. J Cancer Res Clin Oncol. 2017;143(7):1–10. doi:10.1007/s00432-017-2376-8
8. Tomozawa A, Ishikawa S, Shiota N, Cholvisudhi P, Makita K. Perioperative risk factors for acute kidney injury after liver resection surgery: an historical cohort study. Can J Anaesth. 2015;62(7):753–761. doi:10.1007/s12630-015-0397-9
9. Gross JL, Prowle JR. Perioperative acute kidney injury. BJA Educ. 2015;15(4):213–218. doi:10.1093/bjaceaccp/mku030
10. Mayor S. UK report into acute kidney injury deaths urges electrolyte checks in all emergency admissions. BMJ. 2009;338(7708):1407.
11. Fang Y, Ding X, Zhong Y, et al. Acute kidney injury in a Chinese hospitalized population. Blood Purif. 2010;30(2):120–126. doi:10.1159/000319972
12. Basile DP, Anderson MD, Sutton TA. Pathophysiology of acute kidney injury. Compr Physiol. 2012;2(2):1303–1353. doi:10.1002/cphy.c110041
13. Sheridan RP, Wang WM, Liaw A, Ma J, Gifford EM. Extreme gradient boosting as a method for quantitative structure-activity relationships. J Chem Inf Model. 2016;56(12):2353–2360. doi:10.1021/acs.jcim.6b00591
14. Tunc HC, Sakar CO, Apaydin H, et al. Estimation of Parkinson’s disease severity using speech features and extreme gradient boosting. Med Biol Eng Comput. 2020;58(11):2757–2773. doi:10.1007/s11517-020-02250-5
15. Chen T, Li X, Li Y, et al. Prediction and risk stratification of kidney outcomes in IgA nephropathy. Am J Kidney Dis. 2019;74(3):300–309. doi:10.1053/j.ajkd.2019.02.016
16. Yu D, Liu Z, Su C, et al. Copy number variation in plasma as a tool for lung cancer prediction using Extreme Gradient Boosting (XGBoost) classifier. Thorac Cancer. 2020;11(1):95–102. doi:10.1111/1759-7714.13204
17. World Health Orgnization. International statistical classification of diseases and related health problems 10th revision; 2016. Available from: https://icd.who.int/browse10/2016/en.
18. Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604–612. doi:10.7326/0003-4819-150-9-200905050-00006
19. Chen T, Guestrin C. XGBoost: a scalable tree boosting system. In
20. Tibshirani R. Regression shrinkage and selection via the Lasso. J Royal Stat Soc. 1996;58(1):267–288.
21. Lim C, Audureau E, Salloum C, et al. Acute kidney injury following hepatectomy for hepatocellular carcinoma: incidence, risk factors and prognostic value. HPB. 2016;18(6):540–548. doi:10.1016/j.hpb.2016.04.004
22. Cho E, Kim SC, Kim MG, Jo SK, Cho WY, Kim HK. The incidence and risk factors of acute kidney injury after hepatobiliary surgery: a prospective observational study. BMC Nephrol. 2014;15:169. doi:10.1186/1471-2369-15-169
23. Xu J, Xia Y, Li S, et al. A retrospective pilot study to examine the potential of aspartate aminotransferase to alanine aminotransferase ratio as a predictor of postoperative acute kidney injury in patients with hepatocellular carcinoma. Ann Clin Biochem. 2019;56(3):357–366. doi:10.1177/0004563218817797
24. Durand F, Francoz C, Asrani SK, et al. Acute kidney injury after liver transplantation. Transplantation. 2018;102(10):1636–1649. doi:10.1097/tp.0000000000002305
25. Lee BC, Liu KL, Lin CL, Kao CH. Risk of acute kidney injury after transarterial chemoembolisation in hepatocellular carcinoma patients: a nationwide population-based cohort study. Eur Radiol. 2017;27(11):1–8. doi:10.1007/s00330-017-4893-x
26. Amin AA, Alabsawy EI, Jalan R, Davenport A. Epidemiology, pathophysiology, and management of hepatorenal syndrome. Semin Nephrol. 2019;39(1):17–30. doi:10.1016/j.semnephrol.2018.10.002
27. Ginès P, Solà E, Angeli P, Wong F, Nadim MK, Kamath PS. Hepatorenal syndrome. Nat Rev Dis Prim. 2018;4(1):23. doi:10.1038/s41572-018-0022-7
28. Deshpandey RP, Kumar HSA, Mathew S. Dyselectrolytemias and acid–base disorder in acute kidney injury. Clin Queries Nephrol. 2012;1(1):0–75. doi:10.1016/S2211-9477(11)70009-7
29. Lameire N, Van Biesen W, Vanholder R. Electrolyte disturbances and acute kidney injury in patients with cancer. Semin Nephrol. 2010;30(6):534–547. doi:10.1016/j.semnephrol.2010.09.002
30. Yoshida T, Taguchi D, Fukuda K, et al. Incidence of hypophosphatemia in advanced cancer patients: a recent report from a single institution. Int J Clin Oncol. 2017;22(2):244–249. doi:10.1007/s10147-016-1063-0
31. Lameire N, Van Biesen W, Vanholder R. Acute renal problems in the critically ill cancer patient. Curr Opin Crit Care. 2008;14(6):635–646. doi:10.1097/MCC.0b013e32830ef70b
32. Balasubramanian G, Al-Aly Z, Moiz A, et al. Early nephrologist involvement in hospital-acquired acute kidney injury: a pilot study. Am J Kidney Dis. 2011;57(2):228–234. doi:10.1053/j.ajkd.2010.08.026
33. Sykes L, Sinha S, Hegarty J, et al. Reducing acute kidney injury incidence and progression in a large teaching hospital. BMJ Open Qual. 2018;7(4):e000308. doi:10.1136/bmjoq-2017-000308
34. Arias Pou P, Aquerreta Gonzalez I, Idoate García A, Garcia-Fernandez N. Improvement of drug prescribing in acute kidney injury with a nephrotoxic drug alert system. Eur J Hosp Pharm Sci Pract. 2019;26(1):33–38. doi:10.1136/ejhpharm-2017-001300
35. Ehrmann S, Helms J, Joret A, et al. Nephrotoxic drug burden among 1001 critically ill patients: impact on acute kidney injury. Ann Intensive Care. 2019;9(1):106. doi:10.1186/s13613-019-0580-1
36. Zarbock A, Kellum JA, Schmidt C, et al. Effect of early vs delayed initiation of renal replacement therapy on mortality in critically ill patients with acute kidney injury: the ELAIN randomized clinical trial. JAMA. 2016;315(20):2190–2199. doi:10.1001/jama.2016.5828
37. Suh K, Kellum JA, Kane-Gill SL. A systematic review of cost-effectiveness analyses across the acute kidney injury landscape. Expert Rev Pharmacoecon Outcomes Res. 2021;1–8. doi:10.1080/14737167.2021.1882307
38. Yang L, Xing G, Wang L, et al. Acute kidney injury in China: a cross-sectional survey. Lancet (London, England). 2015;386(10002):1465–1471. doi:10.1016/S0140-6736(15)00344-X
39. Vanmassenhove J, Kielstein J, Jörres A, Biesen WV. Management of patients at risk of acute kidney injury. Lancet (London, England). 2017;389(10084):2139–2151.
40. Thakar CV, Arrigain S, Worley S, Yared JP, Paganini EP. A clinical score to predict acute renal failure after cardiac surgery. J Am Soc Nephrol. 2005;16(1):162–168. doi:10.1681/asn.2004040331
41. Palomba H, de Castro I, Neto AL, Lage S, Yu L. Acute kidney injury prediction following elective cardiac surgery: AKICS score. Kidney Int. 2007;72(5):624–631. doi:10.1038/sj.ki.5002419
42. Mehta RH, Grab JD, O’Brien SM, et al. Bedside tool for predicting the risk of postoperative dialysis in patients undergoing cardiac surgery. Circulation. 2006;114(21):
43. Wijeysundera DN, Karkouti K, Dupuis JY, et al. Derivation and validation of a simplified predictive index for renal replacement therapy after cardiac surgery. JAMA. 2007;297(16):1801–1809. doi:10.1001/jama.297.16.1801
44. Safadi S, Hommos MS, Enders FT, Lieske JC, Kashani KB. Risk factors for acute kidney injury in hospitalized non-critically ill patients: a population-based study. Mayo Clin Proc. 2020;95(3):459–467. doi:10.1016/j.mayocp.2019.06.011
45. Deo RC. Machine learning in medicine. Circulation. 2015;132(20):1920–1930. doi:10.1161/circulationaha.115.001593
46. Thomas J, Hepp T, Mayr A, Bischl B. Probing for sparse and fast variable selection with model-based boosting. Comput Math Methods Med. 2017;2017:1421409. doi:10.1155/2017/1421409
47. Kitchlu A, McArthur E, Amir E, et al. Acute kidney injury in patients receiving systemic treatment for cancer: a population-based cohort study. J Natl Cancer Inst. 2018. doi:10.1093/jnci/djy167
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

