Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Comorbid Premenstrual Dysphoric Disorder in Women with Bipolar Disorder: Management Challenges
Authors Sepede G , Brunetti M, Di Giannantonio M
Received 29 November 2019
Accepted for publication 30 January 2020
Published 10 February 2020 Volume 2020:16 Pages 415—426
DOI https://doi.org/10.2147/NDT.S202881
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Roger Pinder
Gianna Sepede,1 Marcella Brunetti,1 Massimo Di Giannantonio1,2
1Department of Neuroscience, Imaging, and Clinical Sciences, University “G. d’Annunzio”, Chieti, Italy; 2Department of Mental Health - Chieti, National Health Trust, Chieti, Italy
Correspondence: Gianna Sepede
Department of Neuroscience, Imaging, and Clinical Sciences, University “G. d’Annunzio”, Via Dei Vestini 33, Chieti, Chieti Scalo (CH) 66013, Italy
Tel +39 0871 3556901
Fax +39 0871 3556930
Email [email protected]
Abstract: Bipolar disorder (BD) and premenstrual dysphoric disorder (PMDD) are two cyclic mood illnesses, sometimes presenting together. Their comorbidity appears to be linked to common biological mechanisms and usually results in more severity of mood symptoms and a poorer long-term outcome. Nevertheless, the management of comorbid PMDD/BD has been scarcely studied. Therefore, the aim of the present paper was to review the published literature on the treatment of comorbid PMDD/BD and to provide point-by-point hypotheses to address these complex clinical cases. We searched PubMed to identify the studies focused on the treatment and management of comorbid PMDD/BD using the following search words, alone and in combination: premenstrual dysphoric disorder, bipolar disorder, comorbid, treatment, management, pharmacotherapy, psychotherapy. The search was conducted on the 1st of June 2019 and yielded 55 records. Four papers met our inclusion/exclusion criteria and were therefore included in our qualitative synthesis. Integrating the few data pertaining to the treatment of comorbid PMDD/BD with the large amount of published data on the two conditions separately, we can suggest that the management of comorbid PMDD/BD needs as a first step to stabilize the bipolar symptoms by means of optimal dosages of mood stabilizers. Then, in euthymic BD patients, the PMDD symptoms could be treated with estroprogestins (first-line treatment). On the contrary, during acute phases of BD, antidepressants (for major depressive episodes) and atypical antipsychotics/hormonal modulators (for manic episodes) could be considered as promising add-on treatments to mood stabilizers. In case of resistant PMDD/BD symptoms, combined strategies should be taken into account, as well as alternative treatments, such as lifestyle changes. In conclusion, RCTs on comorbid PMDD/BD are still lacking. The management of this complex condition is therefore challenging and it requires a tailored treatment.
Keywords: womens’ health, comorbid mood disorders, combined intervention, treatment challenges
Introduction
Premenstrual dysphoric disorder (PMDD), recently included in the DSM-5 Mood Disorder Section, is characterized by psychological (and sometimes physical) symptoms, such as mood swings, depression, irritability, dysphoria, that begin during the luteal phase and recede after the menses.1 PMDD has an estimated prevalence of 2–7% in women during their reproductive age.2–4 In a recent study on a Brasilian community sample, the prevalence of PMDD is over 15% in young adult women, probably due to cultural differences and a wider age span.5 PMDD is often diagnosed many years after its first onset, thus resulting in a long period of untreated symptoms,6 with a huge impact on functioning and quality of life.7,8 Although PMDD can be observed in women without other psychiatric disorders,9 comorbid conditions are reported in up to 70% of cases, especially anxiety disorder, post-traumatic stress disorder, major depression and bipolar disorder (BD).10,11
BD is a serious psychiatric illness, defined by recurrent depressive, manic and mixed episodes, separated by periods of clinical remission, with an estimated lifetime prevalence of 3–7%.12,13 In a large community sample, the prevalence of BD in women with PMDD was about 10%, seven-times higher than what observed in those without PMDD.14 A systematic review conducted in 2014 reported that 15–27% of women with BD met an adjunctive diagnosis of PMDD, supporting the hypothesis that a subgroup of BD may have a hormonal sensitivity.15 On the other hand, retrospective ratings of PMDD are often inaccurate, with a high percentage of false-positive reports.16,17 Moreover, beliefs about premenstrual syndrome were found to strongly influence the diagnosis,18–20 thus suggesting the use of prospective ratings to correctly assess the prevalence of PMDD in women with and without comorbid conditions.21
BD and PMDD are two cyclic mood illnesses and their comorbidity appears to be linked to common biological mechanisms, such as polygenic risk factors. In particular, the Brain-derived neurotrophic factor (BDNF) and catechol-O-methyltransferase (COMT) polymorphisms, have been found in both conditions.22,23
However, other studies have failed to demonstrate similar pathophysiologic pathways of cyclical symptom change in PMDD and premenstrual exacerbation of chronic mood disorders. For example, PMDD-specific treatments, such as drospirenone-containing combined oral contraceptives, GnRH analogues and isoallopregnanolone, were not effective for patients with premenstrual exacerbation of depression.24–26
Recent evidence suggests a crucial role of estradiol and progesterone in neuroregulation and the cyclic changes of their levels may significantly affect mood and behavior in susceptible women. In particular, reproductive steroids regulate the synthesis of important neurotransmitters such as dopamine, serotonin, noradrenaline, GABA and glutamate,27 thus resulting in significant changes in the activation of limbic and prefrontal brain regions involved in attention,28 reward29 and emotional processing.30 The rapid changes of progesterone levels during the different phases of the menstrual cycle and the estrogen influence on serotonin may result in premenstrual symptoms even in case of normal ovarian function.31–33
Hormonal fluctuations during the menstrual cycle may influence the clinical course of BD: women with BD and premenstrual exacerbation of mood symptoms have a poorer outcome and shorter time to relapse.34 In patients affected by BD, low levels of estrogen were found during episodes of post-partum psychosis.35
Patients with comorbid BD and PMDD showed an earlier age of BD onset, more frequent acute mood episodes and a higher rate of rapid cycling.36 Two different comorbidity studies pointed out that PMDD seems to be more frequent in BD type II (BD-II), with respect to BD type I (BD-I) patients.37,38 On the other hand, the possible confounding effect of a higher medication load, which might reduce the comorbidity rate suppressing the PMDD symptoms in BD-I, should be taken into account.39 A recent paper by Ogasawara et al40 criticizes the APA decision attributing PMDD to depressive disorders, suggesting instead to consider it an illness with several bipolar features, such as mood lability, agitation and dysphoria.
In those subjects affected by PMDD without psychiatric comorbid condition, selective serotonin reuptake inhibitor antidepressants and low doses of combined contraceptives (oral estroprogestins) are considered first-line pharmacological treatments.9,41,42 On the other hand, the presence of a comorbid BD brings out an important safety consideration regarding the use of SSRI and the consequence risk of mood switches.43,44
Aim of the present study was therefore to provide a review of the relevant literature on comorbid BD-PMDD and a set of hypotheses to test going forward in RCT studies, in order to manage these complex cases in the most complete and personalized manner.
Methods
We searched PubMed to identify the studies pertaining to the treatment and management of comorbid PMDD and BD. The following search words were used, alone and in combination: premenstrual dysphoric disorder, bipolar disorder, comorbid, treatment, management, pharmacotherapy, psychotherapy.
The search was conducted on the 1st of June 2019 and yielded 55 records. Inclusion criteria were the following: original articles, written in English, including women affected by comorbid BD and PMDD. Animal studies, in vitro experiments, reviews and commentaries were excluded.
By reading title and abstract, we excluded 14 records. By reading the full texts of the 41 remaining articles, we found four papers which met our inclusion/exclusion criteria and were therefore included in the qualitative synthesis (Figure 1).
 |
Figure 1 Flow chart of the systematic review. |
Results
We included in our qualitative synthesis a total number of four papers pertaining to the treatment of comorbid PMDD in women affected by BD. All the included articles were case report/case series. A total of six patients were included.
The treatments suggested were the following:
- Psychopharmacological treatments:
Mood stabilizers: a single case report, finding a significant efficacy of lamotrigine in reducing the follicular phase depressive and luteal phase mood elevation symptoms in a woman affected by a treatment-resistant rapid cycling BD-II was published.45
We cannot find any published studies reporting the treatment of comorbid PMDD/BD with antidepressant or atypical antipsychotics.
In their study, Frey and Minuzzi46 reported three cases of women affected by comorbid BD/PMDD that, after stabilizing the BD symptoms with a combination of mood stabilizers, antipsychotics and antidepressants (lamotrigine + aripiprazole + bupropion, lithium + quetiapine and fluoxetine + olanzapine, respectively) showed a good response after adding oral (drosperidon/ethinylestradiol) or transdermal (norelgestromin/ethinylestradiol) estroprogestins. In two cases, estroprogestins were well tolerated, whereas in the third one they had to be discontinued due to weight gain and headaches. A young woman affected by BD-I and a comorbid diagnosis of PMDD, after achieving euthymia by means of aripiprazole, also reported a reduction of PMDD symptoms adding an oral contraceptive (estradiol plus levonorgestrel).47
We could not find any paper using psychotherapies to treat comorbid BD/PMDD. For what regard alternative treatments (wake therapy, lifestyle modification, supplements, herbal medicine, light therapy), we found two case reports.47,48
In the above mentioned case of BD-I with comorbid PMDD,47 over hormonal and pharmacological therapy, psychoeducation and lifestyle changes are recommended by the authors, together with vitamin supplementation with 600 mg of elemental calcium twice daily and 100 mg/d of vitamin B6 in order to alleviate premenstrual symptoms. Also, based on PMDD treatments literature, the authors recommended cognitive behavioral therapies (CBT) to reduce affective PMDD symptoms.
A recent case report provides encouraging results obtained with nonpharmacological approach with a 45-year-old woman female with a history of diagnosed BD with psychotic features, PMDD, panic disorder, and repeated suicide attempts.48 She reported scarce results through conventional (ie, pharmacological) treatment. Alternative treatment was based on lifestyle interventions (11 mins of daily practice of a meditation called Kirtan Kriya), daily dry-skin brushing to stimulate lymph drainage and coffee enemas to support liver function, together with dietary changes (inclusion of organic/pastured meat, wild fish, pastured eggs, organic vegetables, fruit, nuts, and seeds). Outcomes indicate significant clinical remission after cessation of medication treatment and engagement of alternative, mind-body techniques.
Discussion
Therapeutic Options for Comorbid BD/PMDD
Pharmacologic Interventions
We could not find any published data focused on the treatment of comorbid BD/PMDD with ADs and it’s almost surprising, due to the large use of this class of drugs in the treatment of both psychiatric conditions.
Several studies reported a gender difference in treatment response to antidepressants in mood disorders.49 In particular, there is some evidence that women respond better to SSRI with respect to men50,51 (probably due to the interactions between estrogen and serotonin in the brain).52 When considering PMDD, The Korean Medication Algorithm project for Depressive Disorder, second revision, reported AD monotherapy as the treatment of choice.53 A recent review reported that the efficacy of SSRI was equivalent for continuous versus intermittent (only during the luteal phase) dosing.54
On the other hand, ADs should be used with caution in subjects affected by BD, for the possible occurrence of a manic switch.43 A treatment-emergent mania (TEM) was found in 35–50% of BD patients during AD monotherapy,55,56 especially during the first month of treatment.57 A recent meta-analysis assessing the efficacy and safety of long-term antidepressant treatment for bipolar disorders reported a good efficacy of ADs in reducing depressive symptoms and a low risk of TEM when ADs were administered in combination with mood stabilizers (MSs).58 The risk profiles for the development of an AD-related TEM in BD differ significantly according to gender, with higher risk in females with a family history of BD-I and comorbid thyroid disorders, and in males with a history of suicide attempts and alcohol abuse.59
So, even with the lack of published data, we can speculate that ADs should be used in comorbid BD/PMDD only during the acute depressive episodes of BD, in addiction to MS. Their use should be avoided during the manic states of BD and limited to refractory cases of PMDD during euthymic states.
MSs are a key treatment in BD. Lithium is an old drug with high efficacy in manic and depressive episodes and in reducing relapses in euthymic bipolar patients.60 In case of manic or mixed episode, lithium is often administered in combination with other MSs, such as valproate and carbamazepine,61,62 whereas in the depressive phase of the illness, lamotrigine is usually preferred.
MSs have been rarely used in women affected by PMDD. An old study conducted in 1993 reported a good efficacy of low doses valproate in only 38% of subjects with premenstrual mood changes.63
In a small pilot study including seven patients with PMDD, a promising effect of levetiracetam in reducing the symptoms was observed in 85% of the cases, without severe side effects.64 In a woman affected by a severe form of PMDD, partially responding to SSRI and with a contraindication for the use of estroprogestins, adding lamotrigine resulted in a significant improvement in premenstrual symptoms.65 In an observational study by Karadag and colleagues,66 34 euthymic women with treatment-responsive BD on mood-stabilizers (lithium and/or valproate) for at least 6 months and 35 normal comparisons with regular menstrual cycles were prospectively followed up for at least two consecutive menstrual cycles using the Daily Record of Severity of Problems-Short Form (DRSP).67 Seven BD women (20.8%) and 31 controls (88.6%) reported 30% or more increase in symptom severity during the premenstrual phase when compared with the postmenstrual phase within a cycle for 2 consecutive months. To explain the significant lower rate of premenstrual mood changes in the group of BD, when compared to normal women, the authors suggested a prophylactic effect of lithium and valproate against premenstrual symptom changes.
The only published study focused on comorbid BD/PMDD was a case report: in a woman affected by a treatment-resistant rapid cycling BD-II with follicular phase depressive and luteal phase mood elevation symptoms, lamotrigine significantly reduced the severity and frequency of menstrual-related mood changes.45
Even though the published data involving patients with comorbid BD/PMDD are very few, the large amount of data supporting the efficacy of MS in both acute (manic/depressive episodes) and euthymic states of BD suggest to use them as first-line maintenance treatment in comorbid BD/PMDD, in order to stabilize the BD and then focus in ameliorating the PMDD symptoms.
AAPs, alone or in combination with mood stabilizers, are considered first-line treatments during acute manic episodes and maintenance in BD-I.43,68 Conversely, there is only a double-blind, placebo-controlled study reporting a good efficacy of quetiapine as an add-on therapy in women affected by SSRI-resistant PMDD.69
No studies were found pertaining to the treatment of comorbid BD-PMDD with AAPs, even though few cases of premenstrual manic symptoms successfully treated with sulpiride (continuously or only during the premenstrual period) were published.70 In the rare occurrence of premenstrual psychosis, olanzapine was found to reduce the symptomatology in a young woman,71 whereas none of the administered AAPs (oral or injectable aripiprazole and risperidone) were efficacious in reducing the auditory hallucination and persecutory delusion in another case of cyclic psychosis.72 In a case of BD-I with severe premenstrual mood instability, confusion, and psychosis, high doses of lithium and lurasidone were required to achieve remission.73
We can therefore suggest the use of AAPs to treat PMDD in women with BD-I and a story of premenstrual exacerbation of mood instability and psychosis.
Hormonal Therapies
To date, oral contraceptives with shortened to no hormone-free interval are considered one of the most efficacious therapeutic options for PMDD without psychiatric comorbidities,1,9,47 especially the combination ethinyl estradiol/drospirenone (DROS/EE).74 On the contrary, the evidence supporting the effectiveness of non-contraceptive estrogen-containing preparations was lower.75
We found only anecdotal reports on the use of hormonal agents in the treatment of comorbid BD/PMDD.
A total of four patients with comorbid BD/PMDD responded well to oral or transdermal contraceptives as an add-on therapy to mood stabilizers and antipsychotics.46,47
The selective estrogen receptor modulator tamoxifen was found to have promising anti-manic effects in women with BD.54 Tamoxifen has different mechanisms of action, such as anti-estrogenic properties and luteotropic effect, increasing progesterone levels during the luteal phase in normal women,76 but to date, it has not yet used in patients with comorbid BD/PMDD.
GABAergic neuroactive steroids have significant mood-stabilizing effects and are today considered a new frontier in the treatment of BD, especially for those patients showing menstrual exacerbation of mood symptoms.77 Pregnenolone, a precursor of allopregnanolone and progesterone, reduced the clinical symptoms in acutely depressed BD patients.78 Another endogenous steroid, allopregnanolone (a metabolite from progesterone), may induce depressive symptoms in sensitive women during the luteal phase, via its effects on GABAA system.79 In a recent randomized, double-blind, placebo-controlled multicentre study, Sepranolone (UC1010), a GABAA modulating allopregnanolone antagonist, was found to significantly reduce negative mood and improve quality of life in women affected by PMDD.80 However, studies pertaining to the treatment of comorbid BD/PMDD with GABAergic steroid modulators are still lacking.
In summary, we can consider oral estroprogestins as the treatment of choice, in addition to MSs, in euthymic BD women with comorbid PMDD. Other hormonal treatments, such as estrogen receptor modulators and GABAA modulating steroids, seem to be promising strategies in manic and depressive states, respectively.
Psychotherapy and Alternative Treatments
Several researches have investigated the effectiveness of psychotherapy in women with PMDD. The efficacy of cognitive therapy may be due to the modification of negative thoughts and increasing coping strategies.81 The effect of Cognitive behavior therapy (CBT) alone and combined with pharmacological treatment has been tested in a Randomized Controlled Trials (RCT) on one hundred and eight women with PMDD.82 After 6 months of treatment, a more rapid improvement in association with Fluoxetine was observed. Furthermore, follow-up 1-year post-treatment revealed a better maintenance of treatment effect with CBT than with Fluoxetine. No additional benefit of combining the two treatments resulted. Actually, as analyzed in a systematic review published in 2009 on empirical studies that investigated the use of CBT for PMDD, the research by Hunter and colleagues was the only RCT study carried out up to 2009 with proper intent-to-treat Repeated Measures ANOVA assessment of CBT effectiveness, which was non-significant.83 Based on the collected data, the authors concluded that more than psychotherapies based on thought and behavior modification, other kind of treatment may be considered for women with PMS/PMDD. Specifically, Acceptance and Commitment Therapy (ACT), a treatment focused on thoughts acceptance and techniques to allow patients to recognize and live according to their values or personal commitments.84 Subsequent meta-analysis and reviews considering both pharmacological and CBT efficacy in PMDD treatment do not find convincing effectiveness proof deriving from CBT empirical studies, mainly due to the small sample sizes, variable methodologies and results.81,85 Kleinstauber and colleagues indicate an individually tailored approach to face the PMDD broad spectrum of symptoms. In line with this suggestion, a case report on the treatment course of a fourteen-year-old young woman diagnosed with PMDD provides encouraging evidences of combined approach.86 The authors have chosen to combine Fluoxetine and CBT taking into account patient and family preference, and the need to conservatively use medications with young patients. Furthermore, the effectiveness of CBT as nonpharmacological treatment for PMDD patients has been considered in two recent reviews that included a large sample of studies.9,87
Interestingly, a recent randomized clinical trial evaluated the effect of Internet-based CBT (iCBT) to reduce the burden of PMDD.88 ICBT is a cognitive behavioral therapy provided through a computer or a mobile device. A total of 174 PMDD patients, randomized to treatment group or waitlist control group participated. Treatment group received an 8-week therapist-guided iCBT. Statistical analysis indicated interaction effects on functional impairment and psychological impairment, impact on everyday life, symptom intensity, and symptom disability in favor of the group under treatment. These data suggested a good efficacy of iCBT treatment for PMDD symptoms reduction, and the importance to address coping styles and stress management in the treatment.
CBT has been also proposed as an efficacious add-on therapy in patients affected by BD. Specifically, CBT improved quality of life and compliance, also reducing the frequency and duration of acute mood episodes.89 A recent review explores Internet-based cognitive behavioral therapy (ICBT) cost-effectiveness and its impact in rural areas.90 The authors investigation reveals that ICBT is used to manage various psychiatric disorders as, among others, bipolar disorder. Its employ seems to be encouraged in order to avoid some disadvantages of traditional therapies, including cost inefficiency and a lack of follow-up.
In the same field, a systematic review evaluated the efficacy of adjunctive psychological interventions for BD in middle-income countries, starting from evidences that combining psychological and pharmacological approaches has better efficacy in preventing or delaying relapse and improving outcomes compared with pharmacotherapy alone.91 A total of 18 adjunctive studies were included, with following treatments: psychoeducation, family intervention, group cognitive–behavioural therapy (CBT) and group mindfulness-based cognitive therapy (MBCT). Results suggested that psychoeducation and family psychoeducation improved treatment adherence, knowledge of and attitudes towards BD and quality of life, together with reduction of relapse rates and hospital admissions. CBT and MBCT reduced depressive-manic and emotional dysregulation, respectively. In general, psychological treatments alone are not validated to treat BD, but they are currently administered in combination with pharmacological treatments.
Another recent review provides further evidences on the efficacy of psychological treatments. Specifically, Interpersonal and social rhythm therapy (IPSRT – a modified version of interpersonal therapy based on the hypothesis that disturbance of social rhythms associated with life events causes relapse) was shown to increase the time until a new episode in BD. Furthermore, CBT with sessions of 90 min or more was demonstrated to decrease relapse rate, severity of depression and mania, and to improve psychosocial function, with more enhanced efficacy for BD I than BD II.92 The authors also strongly suggest combined CBT with neurofeedback treatments. In the same field, a recent systematic review and meta-analysis aiming to address the efficacy of CBT as an adjuvant to pharmacotherapy in BD patients included 19 RCTs, for a total number of 1384 patients with BD-I and BD-II. Results indicate that CBT could lower the relapse rate and improve manic and depressive symptoms, and psychosocial functioning, with a mild-to-moderate effect size.93
Even though we could not find any paper focused on the treatment of comorbid PMDD/BD by means of psychotherapies, we can hypothesize their effectiveness as adjunctive treatments, based on their good proven efficacy on BD and PMDD separately, in accordance with Smith and Frey’s recommendations.47
Several research papers explored the effect of other treatments (ie, neither pharmacological nor psychotherapeutic approaches) on PMDD. Among these approaches, we found wake therapy, lifestyle modification, supplements, herbal medicine, light therapy.
Specifically, a research tested the hypothesis that wake therapy exerts antidepressant effects in PMDD women by altering the relationship between the circadian rhythms of sleep and melatonin secretion. Based on this hypothesis, sleep has been experimentally shifted with respect to melatonin in 19 women with PMDD compared with 18 controls.94 Results suggested that decreasing morning melatonin secretion is not necessary for the therapeutic effects of wake therapy in PMDD. Light therapy and acupuncture have also been described as alternative treatments, but in studies with several methodological issues (see Pearlstein 2012 for a review).81 In this field, a recent study has investigated the pattern of use of Light Therapy by office-based physicians, in order to understand if this treatment was considered appropriate for different disorders. They obtained 13.5% of affirmative answers for PMDD.95
A research study also tested the efficacy of massage therapy in 24 women randomly assigned to a massage group or relaxation control group. The authors observed in massage group women a reduction in affective PMDD symptoms immediately after the first and the last massage sessions, thus suggesting that massage treatment could be considered as an adjunct therapy for treating severe premenstrual symptoms.96
Lifestyle modifications (ie, regular aerobic exercise, avoiding stressful events, etc.) are considered as generally correct for health but their efficacy alone was not experimentally demonstrated (see Kelderhouse & Taylor 2013 for a review).97 Dietary modification as reducing the consumption of sugar and eating small, frequent meals, as well as increasing daily intake of complex carbohydrates has been considered to alleviate mild symptoms.98 Elsewhere, cessation of smoking, limited alcohol, and regular sleep, as well as stress management, anger management, self-help support groups, and light therapy are considered beneficial.99
Among supplements, vitamins B6 and E, calcium carbonate, magnesium, and tryptophan have demonstrated short-term benefits,87,99,100 together with increased daily vitamin D intake.101 These data were confirmed by a recent review, but the systematic evaluation of their efficacy is still absent.102
Chasteberry (Vitex agnus castus – VAC) has been reported to be efficacious for decreasing premenstrual emotional and physical symptoms compared with placebo, with an effectiveness equivalent to fluoxetine and vitamin B6.101,103 Other described herbal remedies are Hypericum perforatum, Ginkgo biloba and Crocus sativus/saffron (see Pearlstein 2012; Sepede et al 2016 and Appleton 2018 for a review).9,81,102 Interestingly, an interdisciplinary expert meeting of general gynecologists, gynecological endocrinologists, psychiatrists and psychologists from Switzerland was held in 2016 to provide an algorithm on PMD management. Among different nonpharmacologic treatments, only chasteberry is approved for PMD treatment in Switzerland.104 Nevertheless, in a simultaneous review and meta-analysis considering PMDD treatment studies with Vitex agnus castus preparation, the authors conclude that there is no convincing and conclusive evidence that VAC effectively reduces symptoms of PMDD.105 Their results on 17 randomized controlled trials indicated that VAC extracts are not superior in efficacy compared to SSRIs or oral contraceptives, but are potentially effective compared to placebo, dietary supplements and other herbal preparations.
For what regards alternative treatments for BD, a systematic review verified the efficacy and tolerability on of different treatments for mood disorders which act on the biological clock (affective chronotherapeutics) – bright light therapy (LT), dark therapy (DT), treatments utilizing sleep deprivation (SD), melatonergic agonists (MA), interpersonal social rhythm therapy (IPSRT) and cognitive behavioral therapy adapted for BD (CBTI-BP). Results indicate that chronotherapies were generally safe and well-tolerated.106 Finally, given the paucity of literature on comorbidity of Bipolar Disorder and Premenstrual Dysphoric Disorder, exhaustive nonpharmacological treatments indications on this issue are missing, and other studies are required to better address the use of those therapeutic options.
Conclusion
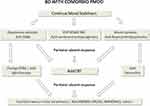
How to Manage Women Affected by Comorbid PMDD/BD (Figure 2)?
Even though BD and PMDD could present together, with a significant impact on the course of both mood conditions, only anecdotal data have been published regarding the treatment of women affected by comorbid BD/PMDD. RCTs of various treatments in this specific comorbid population should be conducted soon.
However, based on the large amount of data pertaining to BD and PMDD separately, we can hypothesize that the management of comorbid PMDD/BD needs as a first step to stabilize the bipolar symptoms by means of optimal dosages of mood stabilizers. Then, in euthymic BD patients, the PMDD symptoms could be treated with estroprogestins (first-line treatment). The ADs should be used with caution due to the risk of inducing mood switches and preferably reserved the treatment of acutely depressed patients. In case of partial or absent response, changing ADs or adding light therapy should be considered. Instead, when facing manic episodes of BD, a good strategy seems to be adding antipsychotic agents and hormonal modulators, in order to reduce both manic and premenstrual dysphoric symptoms.
For what regard psychotherapeutic approaches, CBT seems to be a good second-line treatment in addition to the above mentioned pharmacological treatments in both euthymic and acute states of BD.
In case of resistant PMDD symptoms in women affected by BD, combined strategies should be taken into account, as well as alternative treatments, such as lifestyle changes, herbal drugs and supplements.
Due to the broad spectrum of symptoms experienced by women with comorbid PMDD/BD, tailoring the therapy to the individual needs of the patients should be a mandatory point, in order to significantly improve their symptoms and their quality of life.
Disclosure
The author reports no conflicts of interest in this work.
References
1. Yonkers KA, Simoni MK. Premenstrual disorders. Am J Obstet Gynecol. 2018;218(1):68–74. doi:10.1016/j.ajog.2017.05.045
2. Qiao M, Zhang H, Liu H, et al. Prevalence of premenstrual syndrome and premenstrual dysphoric disorder in a population-based sample in China. Eur J Obstet Gynecol Reprod Biol. 2012;162(1):83–86. doi:10.1016/j.ejogrb.2012.01.017
3. Ogebe O, Mbbs JA, Psych F, et al. Original study a comparison of the prevalence of premenstrual dysphoric disorder and comorbidities among adolescents in the United States of America and Nigeria. J Pediatr Adolesc Gynecol. 2011;24(6):397–403. doi:10.1016/j.jpag.2011.07.009
4. Tschudin S, Bertea PC, Zemp E. Prevalence and predictors of premenstrual syndrome and premenstrual dysphoric disorder in a population-based sample. Arch Womens Ment Health. 2010;13(6):485–494. doi:10.1007/s00737-010-0165-3
5. de Carvalho AB, Cardoso TDA, Mondin TC, et al. Prevalence and factors associated with premenstrual dysphoric disorder: a community sample of young adult women. Psychiatry Res. 2018;268:42–45. doi:10.1016/j.psychres.2018.06.005
6. Baller EB, Ross DA, King F. Clinical commentary premenstrual dysphoric disorder: from plato to petri dishes. Biol Psychiatry. 2019;85(12):e63–e65. doi:10.1016/j.biopsych.2019.04.018
7. Balik G, Hocaoglu C, Kagitci M, Guvenda Guven ES. Comparison of the effects of PMDD and pre-menstrual syndrome on mood disorders and quality of life: a cross-sectional study. J Obstet Gynaecol. 2015;35(6):616–620. doi:10.3109/01443615.2014.991283
8. Yamada K, Kamagata E. Reduction of quality-adjusted life years (QALYs) in patients with premenstrual dysphoric disorder (PMDD). Qual Life Res. 2017;26(11):3069–3073. doi:10.1007/s11136-017-1642-1
9. Sepede G, Sarchione F, Matarazzo I, Di Giannantonio M, Salerno RM. Premenstrual dysphoric disorder without comorbid psychiatric conditions: a systematic review of therapeutic options. Clin Neuropharmacol. 2016;39(5):241–261. doi:10.1097/WNF.0000000000000173
10. Pilver CE, Levy BR, Libby DJ, Desai RA. Posttraumatic stress disorder and trauma characteristics are correlates of premenstrual dysphoric disorder. Arch Womens Ment Health. 2011;14(5):383–393. doi:10.1007/s00737-011-0232-4
11. Alexander JL, Dennerstein L, Kotz K, Richardson G. Women, anxiety and mood: a review of nomenclature, comorbidity and epidemiology. Expert Rev Neurother. 2007;7(11 Suppl):S45–S58. doi:10.1586/14737175.7.11s.S45
12. Clemente AS, Diniz BS, Nicolato R, et al. Bipolar disorder prevalence: a systematic review and meta-analysis of the literature. Rev Bras Psiquiatr. 2015;37(2):155–161. doi:10.1590/1516-4446-2012-1693
13. Bobo WV. The diagnosis and management of bipolar I and II disorders: clinical practice update. Mayo Clin Proc. 2017;92(10):1532–1551. doi:10.1016/j.mayocp.2017.06.022
14. Wittchen H-U, Becker E, Lieb R, Krause P. Prevalence, incidence and stability of premenstrual dysphoric disorder in the community. Psychol Med. 2002;32(1):119–132. doi:10.1017/s0033291701004925
15. Teatero ML, Mazmanian D, Sharma V. Effects of the menstrual cycle on bipolar disorder. Bipolar Disord. 2014;16(1):22–36. doi:10.1111/bdi.12138
16. Rubinow DR, Roy-Byrne P, Hoban MC, Gold PW, Post RM. Prospective assessment of menstrually related mood disorders. Am J Psychiatry. 1984;141(5):684–686. doi:10.1176/ajp.141.5.684
17. Eisenlohr-Moul TA, Girdler SS, Schmalenberger KM, et al. Toward the reliable diagnosis of DSM-5 premenstrual dysphoric disorder: the Carolina Premenstrual Assessment Scoring System (C-PASS). Am J Psychiatry. 2017;174(1):51–59. doi:10.1176/appi.ajp.2016.15121510
18. Marvan ML, Cortes-Iniestra S. Women’s beliefs about the prevalence of premenstrual syndrome and biases in recall of premenstrual changes. Health Psychol. 2001;20(4):276–280. doi:10.1037//0278-6133.20.4.276
19. Hart WG, Coleman GJ, Russell JW. Assessment of premenstrual symptomatology: a re-evaluation of the predictive validity of self-report. J Psychosom Res. 1987;31(2):185–190. doi:10.1016/0022-3999(87)90075-4
20. Cohen LS, Soares CN, Otto MW, Sweeney BH, Liberman RF, Harlow BL. Prevalence and predictors of premenstrual dysphoric disorder (PMDD) in older premenopausal women. The Harvard Study of Moods and Cycles. J Affect Disord. 2002;70(2):125–132. doi:10.1016/s0165-0327(01)00458-x
21. Epperson CN, Hantsoo LV. Making strides to simplify diagnosis of premenstrual dysphoric disorder. Am J Psychiatry. 2017;174(1):6–7. doi:10.1176/appi.ajp.2016.16101144
22. Craddock N, Sklar P. Genetics of bipolar disorder: successful start to a long journey. Trends Genet. 2009;25(2):99–105. doi:10.1016/j.tig.2008.12.002
23. McEvoy K, Osborne LM, Nanavati J, Payne JL. Reproductive affective disorders: a review of the genetic evidence for premenstrual dysphoric disorder and postpartum depression. Curr Psychiatry Rep. 2017;19(12):94. doi:10.1007/s11920-017-0852-0
24. Peters W, Freeman MP, Kim S, Cohen LS, Joffe H. Treatment of premenstrual breakthrough of depression with adjunctive oral contraceptive pills compared with placebo. J Clin Psychopharmacol. 2017;37(5):609–614. doi:10.1097/JCP.0000000000000761
25. Freeman EW, Sondheimer SJ, Rickels K. Gonadotropin-releasing hormone agonist in the treatment of premenstrual symptoms with and without ongoing dysphoria: a controlled study. Psychopharmacol Bull. 1997;33(2):303–309.
26. Freeman EW, Sondheimer SJ, Rickels K, Albert J. Gonadotropin-releasing hormone agonist in treatment of premenstrual symptoms with and without comorbidity of depression: a pilot study. J Clin Psychiatry. 1993;54(5):192–195.
27. Schiller CE, Johnson SL, Abate AC, Schmidt PJ, Rubinow DR. Reproductive steroid regulation of mood and behavior. Compr Physiol. 2016;6(3):1135–1160. doi:10.1002/cphy.c150014
28. Thimm M, Weis S, Hausmann M, Sturm W. Menstrual cycle effects on selective attention and its underlying cortical networks. Neuroscience. 2014;258:307–317. doi:10.1016/j.neuroscience.2013.11.010
29. Diekhof EK, Ratnayake M. Menstrual cycle phase modulates reward sensitivity and performance monitoring in young women: preliminary fMRI evidence. Neuropsychologia. 2016;84:70–80. doi:10.1016/j.neuropsychologia.2015.10.016
30. Amin Z, Epperson CN, Constable RT, Canli T. Effects of estrogen variation on neural correlates of emotional response inhibition. Neuroimage. 2006;32(1):457–464. doi:10.1016/j.neuroimage.2006.03.013
31. Goodnick PJ, Chaudry T, Artadi J, Arcey S. Women’s issues in mood disorders. Expert Opin Pharmacother. 2000;1(5):903–916. doi:10.1517/14656566.1.5.903
32. Schmidt PJ, Nieman LK, Grover GN, Muller KL, Merriam GR, Rubinow DR. Lack of effect of induced menses on symptoms in women with premenstrual syndrome. N Engl J Med. 1991;324(17):1174–1179. doi:10.1056/NEJM199104253241705
33. Schmidt PJ, Martinez PE, Nieman LK, et al. Premenstrual dysphoric disorder symptoms following ovarian suppression: triggered by change in ovarian steroid levels but not continuous stable levels. Am J Psychiatry. 2017;174(10):980–989. doi:10.1176/appi.ajp.2017.16101113
34. Dias RS, Lafer B, Russo C, et al. Longitudinal follow-up of bipolar disorder in women with premenstrual exacerbation: findings from STEP-BD. Am J Psychiatry. 2011;168(4):386–394. doi:10.1176/appi.ajp.2010.09121816
35. Meinhard N, Kessing LV, Vinberg M. The role of estrogen in bipolar disorder, a review. Nord J Psychiatry. 2014;68(2):81–87. doi:10.3109/08039488.2013.775341
36. Slyepchenko A, Frey BN, Lafer B, Nierenberg AA, Sachs GS, Dias RS. Increased illness burden in women with comorbid bipolar and premenstrual dysphoric disorder: data from 1 099 women from STEP-BD study. Acta Psychiatr Scand. 2017;136(5):473–482. doi:10.1111/acps.12797
37. Choi J, Baek JH, Noh J, et al. Association of seasonality and premenstrual symptoms in bipolar I and bipolar II disorders. J Affect Disord. 2011;129(1–3):313–316. doi:10.1016/j.jad.2010.07.030
38. Fornaro M, Perugi G. The impact of premenstrual dysphoric disorder among 92 bipolar patients. Eur Psychiatry. 2010;25(8):450–454. doi:10.1016/j.eurpsy.2009.11.010
39. Cirillo PC, Passos RBF, Bevilaqua MCDN, Lopez JRRA, Nardi AE. Bipolar disorder and premenstrual syndrome or premenstrual dysphoric disorder comorbidity: a systematic review. Rev Bras Psiquiatr. 2012;34(4):467–479. doi:10.1016/j.rbp.2012.04.010
40. Ogasawara K, Nakamura Y, Kimura H, Aleksic B, Ozaki N. Issues on the diagnosis and etiopathogenesis of mood disorders: reconsidering DSM-5. J Neural Transm. 2018;125(2):211–222. doi:10.1007/s00702-017-1828-2
41. Jarvis CI, Lynch AM, Morin AK. Management strategies for premenstrual syndrome/premenstrual dysphoric disorder. Ann Pharmacother. 2008;42(7):967–978. doi:10.1345/aph.1K673
42. Reid RL, Soares CN. Premenstrual dysphoric disorder: contemporary diagnosis and management. J Obstet Gynaecol Can. 2018;40(2):215–223. doi:10.1016/j.jogc.2017.05.018
43. Yatham LN, Kennedy SH, S V P, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disord. 2018;20(2):97–170. doi:10.1111/bdi.12609
44. Gitlin MJ. Antidepressants in bipolar depression: an enduring controversy. Int J Bipolar Disord. 2018;6(1):25. doi:10.1186/s40345-018-0133-9
45. O V B, Rasgon NL, Marsh WK, Glenn T, Ketter TA. Lamotrigine therapy in treatment-resistant menstrually-related rapid cycling bipolar disorder: a case report. Bipolar Disord. 2004;6(5):435–439. doi:10.1111/j.1399-5618.2004.00146.x
46. Frey BN, Minuzzi L. Comorbid bipolar disorder and premenstrual dysphoric disorder: real patients, unanswered questions. Arch Women’s Mental Health. 2013:79–81. doi:10.1007/s00737-012-0313-z
47. Smith M, Frey BN. Treating comorbid premenstrual dysphoric disorder in women with bipolar disorder. J Psychiatry Neurosci. 2016;41(2):E22–3. doi:10.1503/jpn.150073
48. Brogan K. Resolution of refractory bipolar disorder with psychotic features and suicidality through lifestyle interventions: a case report. Adv Mind Body Med. 2017;31(2):4–11.
49. Herzog DP, Wegener G, Lieb K, Muller MB, Treccani G. Decoding the mechanism of action of rapid-acting antidepressant treatment strategies: does gender matter? Int J Mol Sci. 2019;20(4). doi:10.3390/ijms20040949
50. Gorman JM. Gender differences in depression and response to psychotropic medication. Gend Med. 2006;3(2):93–109. doi:10.1016/S1550-8579(06)80199-3
51. Kornstein SG, Schatzberg AF, Thase ME, et al. Gender differences in treatment response to sertraline versus imipramine in chronic depression. Am J Psychiatry. 2000;157(9):1445–1452. doi:10.1176/appi.ajp.157.9.1445
52. Sramek JJ, Murphy MF, Cutler NR. Sex differences in the psychopharmacological treatment of depression. Dialogues Clin Neurosci. 2016;18(4):447–457.
53. Seok Seo J, Rim Song H, Bin Lee H, et al. The Korean medication algorithm for depressive disorder: second revision. J Affect Disord. 2014;167:312–321. doi:10.1016/j.jad.2014.05.031
54. Lanza Di Scalea T, Pearlstein T. Premenstrual dysphoric disorder. Med Clin North Am. 2019;103(4):613–628. doi:10.1016/j.mcna.2019.02.007
55. Fela-Thomas AL, Olotu OS, Esan O. Risk of manic switch with antidepressants use in patients with bipolar disorder in a Nigerian neuropsychiatric hospital. S Afr J Psychiatr. 2018;24:1215. doi:10.4102/sajpsychiatry.v24i0.1215
56. Viktorin A, Lichtenstein P, Thase ME, et al. The risk of switch to mania in patients with bipolar disorder during treatment with an antidepressant alone and in combination with a mood stabilizer. Am J Psychiatry. 2014;171(10):1067–1073. doi:10.1176/appi.ajp.2014.13111501
57. Berkol TD, Balcioglu YH, Kirlioglu SS, Ozarslan Z, Islam S, Ozyildirim I. Clinical characteristics of antidepressant use and related manic switch in bipolar disorder. Neurosciences (Riyadh). 2019;24(1):45–52. doi:10.17712/nsj.2019.1.20180008
58. Liu B, Zhang Y, Fang H, Liu J, Liu T, Li L. Efficacy and safety of long-term antidepressant treatment for bipolar disorders - A meta-analysis of randomized controlled trials. J Affect Disord. 2017;223:41–48. doi:10.1016/j.jad.2017.07.023
59. Scott J, Brichant-Petitjean C, Etain B, et al. A re-examination of antidepressant treatment-emergent mania in bipolar disorders: evidence of gender differences. Acta Psychiatr Scand. 2017;135(5):479–488. doi:10.1111/acps.12728
60. Won E, Kim Y-K. An oldie but goodie: lithium in the treatment of bipolar disorder through neuroprotective and neurotrophic mechanisms. Int J Mol Sci. 2017;18:12. doi:10.3390/ijms18122679
61. Jochim J, Rifkin-Zybutz RP, Geddes J, Cipriani A. Valproate for acute mania. Cochrane Database Syst Rev. 2019;10:CD004052. doi:10.1002/14651858.CD004052.pub2
62. Musetti L, Tundo A, Benedetti A, et al. Lithium, valproate, and carbamazepine prescribing patterns for long-term treatment of bipolar I and II disorders: a prospective study. Hum Psychopharmacol. 2018;33(6):e2676. doi:10.1002/hup.2676
63. Jacobsen FM. Low-dose valproate: a new treatment for cyclothymia, mild rapid cycling disorders, and premenstrual syndrome. J Clin Psychiatry. 1993;54(6):229–234.
64. Kayatekin ZE, Sabo AN, Halbreich U. Levetiracetam for treatment of premenstrual dysphoric disorder: a pilot, open-label study. Arch Womens Ment Health. 2008;11(3):207–211. doi:10.1007/s00737-008-0014-9
65. Sepede G, Martinotti G, Gambi F, Salerno RM, Di Giannantonio M. Lamotrigine augmentation in premenstrual dysphoric disorder: a case report. Clin Neuropharmacol. 2013;36(1):31–33. doi:10.1097/WNF.0b013e318279ee1f
66. Karadag F, Akdeniz F, Erten E, et al. Menstrually related symptom changes in women with treatment-responsive bipolar disorder. Bipolar Disord. 2004;6(3):253–259. doi:10.1111/j.1399-5618.2004.00112.x
67. Endicott J, Nee J, Harrison W. Daily Record of Severity of Problems (DRSP): reliability and validity. Arch Womens Ment Health. 2006;9(1):41–49. doi:10.1007/s00737-005-0103-y
68. Joshi A, Bow A, Agius M. Pharmacological therapies in bipolar disorder: a review of current treatment options. Psychiatr Danub. 2019;31(Suppl 3):595–603.
69. Jackson C, Pearson B, Girdler S, et al. Double-blind, placebo-controlled pilot study of adjunctive quetiapine SR in the treatment of PMS/PMDD. Hum Psychopharmacol. 2015;30(6):425–434. doi:10.1002/hup.2494
70. Hsiao M, Liu C. Unusual manifestations of premenstrual syndrome. Psychiatry Clin Neurosci 2007:120–123. DOI:10.1111/j.1440-1819.2007.01620.x
71. Hu L-Y, Chen P-M. Olanzapine treatment of premenstrual onset psychosis: a case report. Gen Hosp Psychiatry. 2013;35(4):
72. Lee Y-T, Chou Y-H. Lack of efficacy of antipsychotics on premenstrual psychosis: a case report. Psychopharmacol Bull. 2012;45(1):31–34.
73. Susser LC, Hermann AD. Protection against hormone-mediated mood symptoms. Arch Womens Ment Health. 2017;20(2):355–356. doi:10.1007/s00737-016-0702-9
74. Lopez LM, Kaptein AA, Helmerhorst FM. Oral contraceptives containing drospirenone for premenstrual syndrome. Cochrane Database Syst Rev. 2012;(2):CD006586. doi:10.1002/14651858.CD006586.pub4
75. Naheed B, Kuiper JH, Uthman OA, O’Mahony F, O’Brien PMS. Non-contraceptive oestrogen-containing preparations for controlling symptoms of premenstrual syndrome. Cochrane Database Syst Rev. 2017;3:CD010503. doi:10.1002/14651858.CD010503.pub2
76. Steinberg S. The treatment of late luteal phase dysphoric disorder. Life Sci. 1991;49(11):767–802. doi:10.1016/0024-3205(91)90244-6
77. Carta MG, Bhat KM, Preti A. GABAergic neuroactive steroids: a new frontier in bipolar disorders? Behav Brain Funct. 2012;8:61. doi:10.1186/1744-9081-8-61
78. Brown ES, Park J, Marx CE, et al. A randomized, double-blind, placebo-controlled trial of pregnenolone for bipolar depression. Neuropsychopharmacology. 2014;39(12):2867–2873. doi:10.1038/npp.2014.138
79. Andreen L, Nyberg S, Turkmen S, van Wingen G, Fernandez G, Backstrom T. Sex steroid induced negative mood may be explained by the paradoxical effect mediated by GABAA modulators. Psychoneuroendocrinology. 2009;34(8):1121–1132. doi:10.1016/j.psyneuen.2009.02.003
80. Bixo M, Ekberg K, Poromaa IS, et al. Treatment of premenstrual dysphoric disorder with the GABAA receptor modulating steroid antagonist Sepranolone (UC1010)-A randomized controlled trial. Psychoneuroendocrinology. 2017;80:46–55. doi:10.1016/j.psyneuen.2017.02.031
81. Pearlstein T. Psychotropic medications and other non-hormonal treatments for premenstrual disorders. Menopause Int. 2012;18(2):60–64. doi:10.1258/mi.2012.012010
82. Hunter MS, Ussher JM, Browne SJ, Cariss M, Jelley R, Katz M. A randomized comparison of psychological (cognitive behavior therapy), medical (fluoxetine) and combined treatment for women with premenstrual dysphoric disorder. J Psychosom Obstet Gynaecol. 2002;23(3):193–199. doi:10.3109/01674820209074672
83. Lustyk MKB, Gerrish WG, Shaver S, Keys SL. Cognitive-behavioral therapy for premenstrual syndrome and premenstrual dysphoric disorder: a systematic review. Arch Womens Ment Health. 2009;12(2):85–96. doi:10.1007/s00737-009-0052-y
84. Hayes SC. Acceptance and commitment therapy, relational frame theory, and the third wave of behavioral and cognitive therapies - republished article. Behav Ther. 2016;47(6):869–885. doi:10.1016/j.beth.2016.11.006
85. Kleinstauber M, Witthoft M, Hiller W. Cognitive-behavioral and pharmacological interventions for premenstrual syndrome or premenstrual dysphoric disorder: a meta-analysis. J Clin Psychol Med Settings. 2012;19(3):308–319. doi:10.1007/s10880-012-9299-y
86. Adler Nevo GW, Nefsky C. Mind over PMDD: a glimpse into the process of pharmacotherapy-psychotherapy combination treatment. J Can Acad Child Adolesc Psychiatry. 2014;23(2):146–150.
87. Ryu A, Kim T-H. Premenstrual syndrome: a mini review. Maturitas. 2015;82(4):436–440. doi:10.1016/j.maturitas.2015.08.010
88. Weise C, Kaiser G, Janda C, et al. Internet-based cognitive-behavioural intervention for women with premenstrual dysphoric disorder: a randomized controlled trial. Psychother Psychosom. 2019;88(1):16–29. doi:10.1159/000496237
89. da Costa RT, Range BP, Malagris LEN, Sardinha A, de Carvalho MR, Nardi AE. Cognitive-behavioral therapy for bipolar disorder. Expert Rev Neurother. 2010;10(7):1089–1099. doi:10.1586/ern.10.75
90. Kumar V, Sattar Y, Bseiso A, Khan S, Rutkofsky IH. The effectiveness of internet-based cognitive behavioral therapy in treatment of psychiatric disorders. Cureus. 2017;9(8):e1626. doi:10.7759/cureus.1626
91. Demissie M, Hanlon C, Birhane R, Ng L, Medhin G, Fekadu A. Psychological interventions for bipolar disorder in low- and middle-income countries: systematic review. BJPsych Open. 2018;4(5):375–384. doi:10.1192/bjo.2018.46
92. Kato T. Current understanding of bipolar disorder: toward integration of biological basis and treatment strategies. Psychiatry Clin Neurosci. 2019;73(9):526–540. doi:10.1111/pcn.12852
93. Chiang K-J, Tsai J-C, Liu D, Lin C-H, Chiu H-L, Chou K-R. Efficacy of cognitive-behavioral therapy in patients with bipolar disorder: a meta-analysis of randomized controlled trials. PLoS One. 2017;12(5):e0176849. doi:10.1371/journal.pone.0176849
94. Parry BL, Meliska CJ, Martinez LF, et al. Late, but not early, wake therapy reduces morning plasma melatonin: relationship to mood in Premenstrual Dysphoric Disorder. Psychiatry Res. 2008;161(1):76–86. doi:10.1016/j.psychres.2007.11.017
95. Winkler-Pjrek E, Spies M, Baldinger-Melich P, Perkmann L, Kasper S, Winkler D. Use of light therapy by office-based physicians. Neuropsychobiology. 2016;74(4):182–187. doi:10.1159/000477094
96. Hernandez-Reif M, Martinez A, Field T, Quintero O, Hart S, Burman I. Premenstrual symptoms are relieved by massage therapy. J Psychosom Obstet Gynaecol. 2000;21(1):9–15. doi:10.3109/01674820009075603
97. Kelderhouse K, Taylor JS. A review of treatment and management modalities for premenstrual dysphoric disorder. Nurs Womens Health. 2013;17(4):294–305. doi:10.1111/1751-486X.12048
98. Cunningham J, Yonkers KA, O’Brien S, Eriksson E. Update on research and treatment of premenstrual dysphoric disorder. Harv Rev Psychiatry. 2009;17(2):120–137. doi:10.1080/10673220902891836
99. Pinkerton JV, Guico-Pabia CJ, Taylor HS. Menstrual cycle-related exacerbation of disease. Am J Obstet Gynecol. 2010;202(3):221–231. doi:10.1016/j.ajog.2009.07.061
100. Hofmeister S, Bodden S. Premenstrual syndrome and premenstrual dysphoric disorder. Am Fam Physician. 2016;94(3):236–240.
101. Biggs WS, Demuth RH. Premenstrual syndrome and premenstrual dysphoric disorder. Am Fam Physician. 2011;84(8):918–924.
102. Appleton SM. Premenstrual syndrome: evidence-based evaluation and treatment. Clin Obstet Gynecol. 2018;61(1):52–61. doi:10.1097/GRF.0000000000000339
103. Cerqueira RO, Frey BN, Leclerc E, Brietzke E. Vitex agnus castus for premenstrual syndrome and premenstrual dysphoric disorder: a systematic review. Arch Womens Ment Health. 2017;20(6):713–719. doi:10.1007/s00737-017-0791-0
104. Stute P, Bodmer C, Ehlert U, et al. Interdisciplinary consensus on management of premenstrual disorders in Switzerland. Gynecol Endocrinol. 2017;33(5):342–348. doi:10.1080/09513590.2017.1284788
105. Verkaik S, Kamperman AM, van Westrhenen R, Schulte PFJ. The treatment of premenstrual syndrome with preparations of Vitex agnus castus: a systematic review and meta-analysis. Am J Obstet Gynecol. 2017;217(2):150–166. doi:10.1016/j.ajog.2017.02.028
106. Gottlieb JF, Benedetti F, Geoffroy PA, et al. The chronotherapeutic treatment of bipolar disorders: a systematic review and practice recommendations from the ISBD task force on chronotherapy and chronobiology. Bipolar Disord. 2019. doi:10.1111/bdi.12847
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.