Back to Journals » OncoTargets and Therapy » Volume 13
Clinical Significance of ZNF711 in Human Breast Cancer
Authors Li X , Tian L, Zhang L , Xu B, Zhang Y, Li Q
Received 28 February 2020
Accepted for publication 28 May 2020
Published 6 July 2020 Volume 2020:13 Pages 6593—6601
DOI https://doi.org/10.2147/OTT.S251702
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Yong Teng
Xiaoyan Li,1 Liu Tian,2 Lina Zhang,1 Baojin Xu,3 Yong Zhang,1 Qiang Li1
1Department of Pathology, Cancer Hospital of China Medical University, Liaoning Cancer Hospital and Institute, Shenyang, Liaoning 110042, People’s Republic of China; 2Psychiatry and Mental Health Center, Shenyang Mental Health Center, Shenyang, Liaoning 110168, People’s Republic of China; 3Departments of Breast Surgery, Cancer Hospital of China Medical University, Liaoning Cancer Hospital and Institute, Shenyang, Liaoning 110042, People’s Republic of China
Correspondence: Qiang Li
Department of Pathology, Cancer Hospital of China Medical University, Liaoning Cancer Hospital and Institute, Dadong District, Shenyang City, Liaoning 110042, People’s Republic of China
Tel/ Fax +86 24-31516152
Email [email protected]
Purpose: To investigate the clinicopathologic and prognostic significance of the zinc-finger protein 711 (ZNF711) in breast cancer (BCa).
Materials and Methods: The relevance of ZNF711 in BCa was analyzed using bioinformatics. The expression of ZNF711 was detected by immunohistochemistry in paraffin blocks of BCa. To evaluate its clinical significance, the correlation between the expression of ZNF711 and BCa clinical indicators, including estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor-2 (HER-2), was analyzed. Finally, the Kaplan–Meier method was applied to explore the prognostic value of ZNF711.
Results: ZNF711 expression was decreased in BCa and was negatively correlated with ER expression (P < 0.05) and positively correlated with HER-2 expression (P < 0.01), but there was no significant correlation between ZNF711 and PR expression. ZNF711 expression was not correlated with age, tumor diameter, or lymph node metastasis; however, ZNF711 expression was correlated with staging in BCa. Survival analysis results showed that the ZNF711-positive group patients had a poorer prognosis compared with the ZNF711-negative group.
Conclusion: The expression of ZNF711 was deceased in BCa and closely related to ER and HER-2 expression. Therefore, ZNF711 could not only serve as a predictor of BCa with poor prognosis but also as a potential biomarker for targeted therapy.
Keywords: breast cancer, zinc-finger protein 711, ER, HER-2
Introduction
Breast cancer (BCa) is the most common malignant tumor in women, accounting for 24.2% of female tumors worldwide, with a mortality rate of up to 15.0%.1 The incidence rate of BCa is gradually increasing, especially in young patients.2,3 The occurrence and development of BCa is a complex biological process involving multiple genes and proteins. Therefore, it is particularly important to identify new biomarkers and therapeutic targets for BCa.
Zinc-finger proteins are transcription factors with a “fingerlike” domain that are widely involved in many biological processes, such as cell differentiation, proliferation, and apoptosis in eukaryotes. It has been shown that zinc-finger proteins could also act as recruiters of chromatin modifiers, as co-factors, or as structural proteins involved in cell migration and invasion. Zinc-finger proteins are correlated with tumorigenesis and the development of multiple types of human cancers and may be useful as prognostic factors.4–9
ZNF711 is a newly discovered zinc-finger protein involved in the development of neurons. Recent studies have shown that ZNF711 is expressed in BCa cell lines, but its function and regulatory mechanisms are unclear.8 In this study, the relevance of ZNF711 expression in breast tumors was analyzed using the UALCAN and kmplot databases, and the expression of ZNF711 in BCa tissue was assessed by immunohistochemical staining. Moreover, we explored the relationship between ZNF711 and clinical pathological factors in BCa.
Materials and Methods
Bioinformatics Analysis
The online database UALCAN (ualcan.path.uab.edu/) was used to obtain the differential expression levels of ZNF711 in BCa and normal breast tissues. The prognostic value of the expression of ZNF711 in BCa was evaluated using Kaplan–Meier survival analysis (kmplot.com/). The interconnection of ZNF711 with other regulatory genes was also explored using the STRING database (https://string-db.org/).
Patients and Tissues Samples
This study was approved by the Liaoning Cancer Hospital and Institute Ethics Review Board (#201601415). Paraffin-embedded specimens from breast surgeries were collected and screened from January 2001 to August 2002 at the Liaoning Cancer Hospital. None of these patients had previously been treated with chemotherapy or radiation therapy. After surgery, all patients received four to six cycles of chemotherapy and HER-2-positive patients received additional treatment with trastuzumab. Seventy-six breast cancer samples with histological types of invasive ductal carcinoma (IDH, 66 cases) and invasive lobular carcinoma (ILH, 10 cases) were included in this study; among them, luminal A (31 cases), luminal B (16 cases), HER-2-positive (17 cases), and basal-like (12 cases) according to the 12th St. Gallen International Breast Cancer Conference (2011).10 These biological subtypes were based on immunohistochemical staining and fluorescence in situ hybridization (FISH). Eighteen cases of normal breast tissues were also collected. The clinical characteristics of the study participants were as follows: all patients were female, 33–79 years old, with an average age of 49 years. The maximum tumor diameter was 0.5–7.0 cm, with an average of 2.1 cm. There were 24 cases with lymph node metastasis and 23 cases were stage I, 50 cases were stage II, and 3 cases were stage III. Follow-up was performed for 150 months in all patients.
Main Reagents
Anti-human ZNF711 rabbit polyclonal antibody was purchased from Abcam (ab254776, 1:400, positive control: human cerebral cortex, cerebellum, and testis tissue, negative control: human liver tissue). Anti-HER-2 rabbit monoclonal antibody, anti-progesterone receptor (PR) rabbit monoclonal antibody, and anti-estrogen receptor (ER) rabbit monoclonal antibody were purchased from Roche Biotechnology Co., Ltd. (Basel, Switzerland). The secondary antibody and supporting staining reagents were purchased from Fuzhou Maixin Biotechnology Co., Ltd. (Fuzhou, China).
Immunohistochemical Staining
Paraffin sections of BCa tissues were baked for 2 h in a 60°C oven, and the slides were placed on a fully automated immunohistochemistry slide frame (Roche Bench Mark GX). The following conditions were used: dewaxing for 15 min, 0.01 mol/L sodium citrate antigen with heat treatment for 30 min, antibody incubation for 28 min, secondary antibody incubation for 8 min, DAB (diaminobenzidine) reaction for 8 min, hematoxylin staining for 12 min, and dye acceleration for 4 min. After the staining was completed, the sealed slides were dried for observation.
FISH Testing
Immunohistochemistry HER-2 (2+) cases were retested using FISH. The FISH analysis was performed on 3- to 5-lm-thick paraffin sections of tumor tissues using Paraffin Pretreatment Reagent KitⅡand PathVysion HER-2 DNA Probe Kit (Gene Tech, Shanghai). Then, detecting the amplification of HER-2 gene followed the manufacturer’s instructions. FISH results were expressed as the ratio between the number of copies of the HER2 gene and the number of copies of chromosome 17 within the nucleus counted in at least 20 cancer cells. The definition of FISH positivity was HER2: chromosome 17 ratio of ≥2.0 (Supplementary Figure 1).
Criteria for Interpretation of Results
In order to ensure the objectivity of the results, two pathologists blinded to the clinical data semi-quantitatively scored the slides by evaluating the staining intensity and percentage of stained cells. The staining intensity was scored on a scale of 1–3 (1, weak; 2, intermediate; 3, strong). The percentage of cells stained was graded on a scale of 1–4 (1, 1–25%; 2, 26–50%; 3, 51–75%; 4, 76–100%). The extent of staining was defined by H-score, the staining intensity score multiplied by the percentage of positive cells. For the interpretation of ZNF711 expression, a total score from 0 to 3 points indicated a negative result, and a score greater than or equal to 4 points indicated a positive result. The interpretation criteria for ER, PR, and HER-2 followed the clinically accepted guidelines of ASCO/CAP breast cancer hormone receptor IHC and the breast cancer HER-2 Detection Guide (2014 Edition).
Statistical Analysis
Data were analyzed using SPSS23.0. P < 0.05 was considered statistically significant. The χ2 test was used to count the data. Spearman correlation analysis was used for hierarchical correlation analysis. The Kaplan–Meier method was used for survival analysis. The Log rank test was used to compare survival rates between the groups.
Results
Expression of ZNF711 in Normal Breast Tissue and BCa
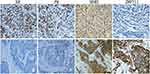
The expression of ZNF711 protein in breast tissue is located in the cell nuclei (proteinatlas.org/). Bioinformatic analysis of 1097 patient samples from the TCGA database via UALCAN indicated that the expression of ZNF711 was decreased in BCa (Figure 1). Immunohistochemical staining demonstrated ZNF711 expression in normal tissues was decreased in BCa (mean score: 8.7 vs. 5.9, P < 0.01) (Figure 2). The negative rate of ZNF711 protein expression in BCa was significantly higher than that in normal tissues (60.5% vs. 0%, P < 0.01). However, 26.3% (20/76) of breast cancers had an immunohistochemical score of 12 for ZNF711 expression, which is higher than that in normal tissues (5.5%, P < 0.01). (Figures 2 and 3)
 |
Figure 1 Expression of ZNF711 mRNA in normal breast tissues (normal) and BCa (primary tumor) based on TCGA database. |
Correlation Between ZNF711 Expression and Clinical Pathological Features in BCa
The expression of ZNF711 was not related to age, tumor diameter, lymph node metastasis, or histological type. However, its level was elevated in advanced breast cancer, as shown in Table 1. ZNF711 expression differed among the different molecular subtypes of BCa. Compared with luminal A type (4/31,13%) and basal-like breast cancer (3/12, 25%), ZNF711 expression was particularly high in luminal B type (9/16, 56%) and HER-2-positive breast cancer (14/17, 82%). ZNF711 expression was significantly higher in HER-2 positive breast cancer tissues (P<0.01).
 |
Table 1 Correlation Between ZNF711 Expression and Clinical Pathological Features in Patients with BCa |
Correlation Between ZNF711 Expression and HER-2, ER, and PR Expression in BCa
To further investigate why ZNF711 is expressed differently in different molecular subtypes of BCa and to explore the correlation with other biological markers of BCa, the correlation between ZNF711 expression and that of ER, PR, and HER-2 was determined in 76 breast cancer cases. Our results showed that ZNF711 was highly expressed in HER-2-positive BCa cells; however, ZNF711 expression was lower in ER-positive cancer tissues (Figures 4 and 5). The correlation analyses are summarized in Table 2. In BCa, ZNF711 expression was positively correlated with HER-2 expression and negatively correlated with ER expression. There was no significant correlation between the expression of ZNF711 and PR (P > 0.05).
 |
Table 2 Correlation of ZNF711 Expression with HER2, ER, and PR in BCa |
 |
Figure 5 Correlation of ZNF711 expression with HER-2, ER, and PR in BCa. The expression of ZNF711 increased in HER-2+ (P < 0.01). In contrast, the expression of ZNF711 decreased in ER+. (P < 0.05). |
Prognostic Significance of ZNF711 in BCa
A total of 76 patients completed the follow-up period, and the mean survival time was 125.04 months in the ZNF711-negative group and 111.87 months in the ZNF711-positive group. Using the Kaplan–Meier method and the Log rank test, the results indicated that high ZNF711 expression in BCa was significantly associated with poorer overall survival (Figure 6, P < 0.05). Multivariate Cox regression analysis was used to examine the relationship between survival time and clinical factors, including ZNF711 expression, which showed that high expression of ZNF711 (HR = 8.798, 95% CI: 2.070–37.224) was an independent prognostic factor for overall survival (Table 3). The KM plotter database also suggested that an increase in ZNF711 expression in BCa positively correlated with poor prognosis (Figure 7).
 |
Table 3 Multivariate Analysis for Overall Survival by Cox Regression Model |
 |
Figure 6 Survival curves for ZNF711-positive and -negative patients with BCa. |
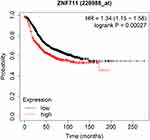 |
Figure 7 Survival curves for ZNF711-positive and -negative patients based on Kmplot database. |
Interconnection of ZNF711 with Other Regulatory Genes
To explore the regulatory mechanism of ZNF in breast cancer, we further explored the interconnection of ZNF711 with other regulatory genes using the STRING database. We found that ZNF711 may directly bind with testis expressed sequence 13B (TEX13B), zinc finger CCCH-type containing 14 (ZC3H14), premature ovarian failure 1B (POF1B), magnesium transporter 1 (MAGT1), tripartite motif-containing 28 (TRIM28), and other genes to play an important role in regulating the progression of breast cancer (Supplementary Figure 2). However, the exact mechanisms of action need to be explored in the future.
Discussion
In 1982, Bogenhagen et al first discovered that the zinc-finger protein transcription factor IIIA (TFIIIA) is involved in the regulation of the 5S RNA gene in Xenopus oocytes, since then the regulatory roles of zinc-finger proteins have been studied in various taxa.11–15 Zinc-finger proteins (ZNFs) are one of the most-abundant groups of proteins and are able to interact with nucleic acids and poly-ADP-ribose (PAR), as well as other proteins. ZNFs are implicated in transcriptional regulation, ubiquitin-mediated protein degradation, signal transduction, actin targeting, DNA repair, cell migration, and other biological processes.8 Previous studies have demonstrated that zinc-finger protein 148 (ZNF148) and zinc-finger E-box-binding protein 1 (ZEB1) are involved in the development and epithelial-to-mesenchymal transition (EMT) of breast cancer.16–20
The ZNF711 gene is located at Xq21.1–q21.2 and encodes a zinc-finger protein with unknown function. The protein contains a potential domain at the amino terminal that may be involved in transcriptional activation, together with 12 Zn-C2H2 domains that are involved in sequence-specific DNA binding. The ZNF711 gene is predominantly expressed in the brain and testis of humans and is an important transcription factor in the development of X chromosome-related intelligence.21 ZNF711 and zinc-finger protein X-linked (ZFX) are known to bind to gene promoter regions in the breast cancer cell line MCF7 and participate in transcriptional regulation.9 However, the specific function and mechanism of ZNF711 in breast cancer cells remain unclear. In this study, the association between ZNF711 expression and clinical characteristics was analyzed in breast cancer patients.
Bioinformatic analysis and immunohistochemical staining were performed to detect the expression of ZNF711 in human normal and cancerous breast tissues. ZNF711 is expressed in normal tissues and decreased in BCa (Figures 1 and 2). ZNF711 expression was reduced in most breast cancers, but was still strongly expressed in 26.3% of breast cancers, which is higher than in normal tissues (Figures 2 and 3). Thus, a paradox emerges: how can ZNF711 in BCa have a polarized mode of expression?
To explore the polarization of ZNF711 expression in BCa, the correlation between ZNF711 expression and clinical pathological features was investigated. The expression of ZNF711 did not correlate with age, tumor size, lymph node metastasis, and histological type (Table 1). ZNF711 expression was significantly higher in advanced breast cancer (stage II–III) than in the early stage (stage I), suggesting that ZNF711 expression is associated with poor prognosis. The expression of ZNF711 was different among different molecular subtypes of BCa, and was significantly higher in HER-2-positive breast cancer tissues and lower in ER-positive cancer tissues (Figures 4 and 5).
The correlation between ZNF711 expression and HER-2, ER, and PR was analyzed in BCa, where expression was positively correlated with HER-2 expression and negatively correlated with ER expression (Table 2). There was no significant correlation between the expression of ZNF711 and PR. Therefore, we hypothesize that the expression of HER-2 and ER may play an important role in ZNF711 gene transcription regulation in BCa.
Although the expression of ZNF711 was decreased in BCa, the survival analysis showed that the ZNF711-positive group patients had a poorer prognosis and a lower survival rate compared with the ZNF711-negative group (P < 0.05). Gene amplification and overexpression of HER-2 have been found in many human cancers, which correlate with the genesis and progress of tumors as well as aggressive biological behaviors. The majority of patients in the ZNF711-positive group (76.7%) were also HER-2 positive (Table 1). Therefore, we believe that this may be the reason for the poor prognosis in the ZNF711-positive group.
Currently, only 20% to 40% of patients with advanced hormone receptor-positive breast cancer respond to first-line endocrine therapy, and about 50% of these patients exhibit recurrence and metastasis due to resistance to endocrine therapy after approximately 14 months.22 Laurentiis et al23 found that endocrine therapy is less effective in patients with HER-2-positive breast cancer, but Ellis et al24 believed that HER-2-positive is not always a marker of endocrine resistance. The specific mechanism of action is still unclear. In this study, the expression of ZNF711 was found to be closely related to the expression of HER-2 and ER; however, whether it participates in the regulatory mechanism between them needs to be studied further.
In summary, the expression of ZNF711 is closely related to the development and prognosis of BCa, and the expression of ZNF711 is significantly correlated with the expression of ER or HER-2. Therefore, ZNF711 may be a novel biomarker for predicting poor prognosis and may serve as a potential therapeutic target for BCa. Furthermore, we also investigated potential ZNF711-binding proteins, which may help to explore the regulation mechanism of ZNF711 in the development of breast cancer.
Abbreviations
BCa, breast cancer; ZNF711, zinc-finger protein 711; ER, estrogen receptor; PR, progesterone receptor; HER-2, erb-b2 receptor tyrosine kinase 2; ZNFs, Zinc-finger proteins; ZFX, zinc-finger protein X-linked; FISH, fluorescence in situ hybridization; IDH, invasive ductal carcinoma; ILH, invasive lobular carcinoma; DAB, diaminobenzidine; TEX13B, testis expressed sequence 13B; ZC3H14, zinc finger CCCH-type containing 14; POF1B, premature ovarian failure 1B; MAGT1, magnesium transporter 1; TRIM28, tripartite motif-containing 28; TFIIIA, transcription factor IIIA; ZNF148, zinc-finger protein 148; ZEB1, zinc-finger E-box-binding protein 1; EMT, epithelial-to-mesenchymal transition.
Highlights
ZNF711 was expressed at lower levels in breast cancer. ZNF711 was highly expressed in breast cancer with high HER-2 expression. In breast cancer, ZNF711 expression was negatively correlated with ER expression. The patients in the ZNF711-positive group had a poor prognosis.
Data Sharing Statement
The datasets generated during the current study are available from the corresponding author upon reasonable request.
Ethics Approval and Consent to Participate
All aspects of this study were approved by the ethics committee of Liaoning Cancer Hospital under protocol 20,170,304 (project approval date 03/16/2017). Ethical practices were followed throughout to cover patient data confidentiality and compliance with the Declaration of Helsinki.
Acknowledgments
The authors would like to thank Liqun Chen and Yue Zhang for their excellent technical support.
Author Contributions
All authors made substantial contributions to the conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
Dr Qiang Li reports grants from the Natural Science Foundation of Liaoning Province during the conduct of the study. The authors declare that they have no other competing interests.
References
1. Bray F, Ferlay J, Soerjomataram I, et al. GLOBOCAN 2018, global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.21492
2. Yan S, Jiao X, Zou H, Li K. Prognostic significance of c-Met in breast cancer: a meta-analysis of 6010 cases. Diagn Pathol. 2015;10:62. doi:10.1186/s13000-015-0296-y
3. O’Brien KM, Fei C, Sandler DP, Nichols HB, DeRoo LA, Weinberg CR. Hormone therapy and young-onset breast cancer. Am J Epidemiol. 2015;181(10):799–807. doi:10.1093/aje/kwu347
4. Kim CJ, Lee J-W, Choi -J-J, et al. High claudin-7 expression is associated with a poor response to platinum-based chemotherapy in epithelial ovarian carcinoma. Eur J Cancer. 2011;47(6):918–925. doi:10.1016/j.ejca.2010.11.007
5. Liu B, Xing X, Li X, et al. ZNF259 promotes breast cancer cells invasion and migration via ERK/GSK3β/snail signaling. Cancer Manag Res. 2018;10:3159–3168. doi:10.2147/CMAR.S174745
6. Bartholow TL, Chandran UR, Becich MJ, Parwani AV. Immunohistochemical profiles of claudin-3 in primary and metastatic prostatic adenocarcinoma. Diagn Pathol. 2011;6:12. doi:10.1186/1746-1596-6-12
7. Yu T, Chen X, Zhang W, et al. Regulation of the potential marker for intestinal cells, Bmi1, by beta-catenin and the zinc finger protein KLF4: implications for colon cancer. J Biol Chem. 2012;287:3760–3768. doi:10.1074/jbc.M111.316349
8. Cassandri M, Smirnov A, Novelli F, et al. Zinc-finger proteins in health and disease. Cell Death Discov. 2017;3(1):17071. doi:10.1038/cddiscovery.2017.71
9. Rhie SK, Yao L, Luo Z, et al. ZFX acts as a transcriptional activator in multiple types of human tumors by binding downstream of transcription start sites at the majority of CpG island promoters. Genome Res. 2018;28(3):310–320. doi:10.1101/gr.228809.117
10. Goldhirsch A, Wood WC, Coates AS, et al. Strategies for subtypes–dealing with the diversity of breast cancer: highlights of the St. Gallen international expert consensus on the primary therapy of early breast cancer 2011. Ann Oncol. 2011;22(8):1736–1747. doi:10.1093/annonc/mdr304
11. Kleine-Kohlbrecher D, Christensen J, Vandamme J, et al. A functional link between the histone demethylase PHF8 and the transcription factor ZNF711 in X-linked mental retardation. Mol Cell. 2010;38(2):165–178. doi:10.1016/j.molcel.2010.03.002
12. Bogenhagen DF, Wormington WM, Brown DD. Stable transcription complexes of Xenopus 5S RNA genes: a means to maintain the differentiated state. Cell. 1982;28(2):413–421. doi:10.1016/0092-8674(82)90359-2
13. Corkins ME, May M, Ehrensberger KM, et al. Zinc finger protein Loz1 is required for zinc-responsive regulation of gene expression in fission yeast. Proc Natl Acad Sci U S A. 2013;110(38):15371–15376. doi:10.1073/pnas.1300853110
14. Gupta SK, Rai AK, Kanwar SS, Sharma TR, Zhang T. Comparative analysis of zinc finger proteins involved in plant disease resistance. PLoS One. 2012;7(8):e42578. doi:10.1371/journal.pone.0042578
15. Zhao XQ, Bai FW. Zinc and yeast stress tolerance: micronutrient plays a big role. J Biotechnol. 2012;158(4):
16. Strasberg Rieber M, Zangemeister-Wittke U, Rieber M. p53-Independent induction of apoptosis in human melanoma cells by a bcl-2/bcl-xL bispecific antisense oligonucleotide. Clin Cancer Res. 2001;7(5):1446–1451.
17. Serova M, Calvo F, Lokiec F, et al. Characterizations of irofulven cytotoxicity in combination with cisplatin and oxaliplatin in human colon, breast, and ovarian cancer cells. Cancer Chemother Pharmacol. 2006;57(4):491–499. doi:10.1007/s00280-005-0063-y
18. Thakur AK, Nigri J, Lac S, et al. TAp73 loss favors Smad-independent TGF-β signaling that drives EMT in pancreatic ductal adenocarcinoma. Cell Death Differ. 2016;23(8):1358–1370. doi:10.1038/cdd.2016.18
19. Heldin CH, Vanlandewijck M, Moustakas A. Regulation of EMT by TGFβ in cancer. FEBS Lett. 2012;586(14):1959–1970. doi:10.1016/j.febslet.2012.02.037
20. Yu P, Shen X, Yang W, Zhang Y, Liu C, Huang T. ZEB1 stimulates breast cancer growth by up-regulating hTERT expression. Biochem Biophys Res Commun. 2018;495(4):2505–2511. doi:10.1016/j.bbrc.2017.12.139
21. Tian C, Xing G, Xie P, et al. KRAB-type zinc-finger protein Apak specifically regulates p53-dependent apoptosis. Nat Cell Biol. 2009;11(5):580–591. doi:10.1038/ncb1864
22. Johnston SR. New strategies in estrogen receptor-positive breast cancer. Clin Cancer Res. 2010;16(7):1979–1987. doi:10.1158/1078-0432.CCR-09-1823
23. De Laurentiis M, Arpino G, Massarelli E, et al. A meta-analysis on the interaction between HER-2 expression and response to endocrine treatment in advanced breast cancer. Clin Cancer Res. 2005;11(13):4741–4748. doi:10.1158/1078-0432.CCR-04-2569
24. Ellis MJ, Coop A, Singh B, et al. Letrozole is more effective neoadjuvant endocrine therapy than tamoxifen for ErbB-1- and/or ErbB-2-positive, estrogen receptor-positive primary breast cancer: evidence from a Phase III randomized trial. J Clin Oncol. 2001;19(18):3808–3816. doi:10.1200/JCO.2001.19.18.3808
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.